Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
SF_(2), SF_(4) and SF_(6)have hybridisations at sulphur atom respectively as |
|
Answer» `SP^(2), sp^(3), sp^(3)d^(2)` 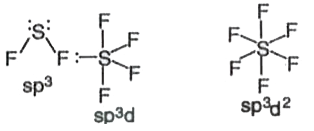
|
|
| 2. |
Sewage water can be purified for recycling with the action of |
|
Answer» AQUATIC plants |
|
| 3. |
Several short-lived radioactive species have been used to determine the age of wood or animal fossils. One of the most intresting substance is ""_(6)C""^(14)(half-life fossils, etc.). Carbon-14 is produced by the bombardment of nitrogen atoms present in the upper atmosphere with neutrons (from cosmic rays). ""_(7)N""^(14)+""_(0)n""^(1)to""_(6)C""^(14)+""_(1)H""_(1) Thus carbon-14 is oxidised, to CO""_(2) and eventually lingested by land animals. The death of plants or animals put an end to the intake of C""_(14) from the atmosphere. After this the amount of C""_(14) in the dead tissues starts decreasing due to its disintegration as per the following reaction : ""_(6)C""^(14)to""_(7)N""^(14)+""_(-1)beta""^(0) The C""^(14) isotope enters the biosphjere when carbon dioxide is taken up in plant photosynthesis. Plants are eaten by animals, which exhale C""^(14) as CO""_(2). Eventually, C""^(14) participates in many aspects of carbon cycle. The C""^(14) lost by radioactive decay-replenishment process, a dynamic rquillibrium is established whereby the ratio of C""^(14) to C""^(12) remains constant in living matter. But when an individual plant or an animal dies, the C""^(14) isotpe inn it is no longer replenished, so the ratio decreases as C""^(14) decays. So, ther number C""^(14) nuclei after time t (after the death of living matter) would be less than in a livingthe following formula, t""_(1//2)=0.693/? The intensity of the cosmic rays have remain the same for 30,000 years. But since some years changes in this are observed due to excessive burning of fossil fuel and nuclear test? A nuclear explosion has taken place leading to increase in concentration of C""^(14) in nearby areas. C""^(14) concentration is C""_(1) in nearly areas and C""_(2) in areas far away. If the age of the fossil is determined to be T""_(1) and T""_(2) at the respective places then |
|
Answer» The age of the FOSSIL will inrease at the where EXPLOSION has taken place and |
|
| 4. |
Sewagecontainingorganicwasteshouldbedisposedin waterbodiesbecauseit causesmajorwater pollution . Fishessucha pollutedwaterdie because of |
|
Answer» Larger NUMBEROF MOSQUITOES. |
|
| 5. |
Several short-lived radioactive species have been used to determine the age of wood or animal fossils. One of the most intresting substance is ""_(6)C""^(14)(half-life fossils, etc.). Carbon-14 is produced by the bombardment of nitrogen atoms present in the upper atmosphere with neutrons (from cosmic rays). ""_(7)N""^(14)+""_(0)n""^(1)to""_(6)C""^(14)+""_(1)H""_(1) Thus carbon-14 is oxidised, to CO""_(2) and eventually lingested by land animals. The death of plants or animals put an end to the intake of C""_(14) from the atmosphere. After this the amount of C""_(14) in the dead tissues starts decreasing due to its disintegration as per the following reaction : ""_(6)C""^(14)to""_(7)N""^(14)+""_(-1)beta""^(0) The C""^(14) isotope enters the biosphjere when carbon dioxide is taken up in plant photosynthesis. Plants are eaten by animals, which exhale C""^(14) as CO""_(2). Eventually, C""^(14) participates in many aspects of carbon cycle. The C""^(14) lost by radioactive decay-replenishment process, a dynamic rquillibrium is established whereby the ratio of C""^(14) to C""^(12) remains constant in living matter. But when an individual plant or an animal dies, the C""^(14) isotpe inn it is no longer replenished, so the ratio decreases as C""^(14) decays. So, ther number C""^(14) nuclei after time t (after the death of living matter) would be less than in a livingthe following formula, t""_(1//2)=0.693/? The intensity of the cosmic rays have remain the same for 30,000 years. But since some years changes in this are observed due to excessive burning of fossil fuel and nuclear test? What should be the age of the fossil meaningful determination of its age? |
|
Answer» 6 years |
|
| 6. |
Several isomeric amines are possible with the molecular formula C_(8)H_(11)N. On the basis of the reaction given by each of the isomers, identify the structure of the compound. a) A(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to p-Nitroso compound b) B(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to Diazonium salt formed c) C(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to N-Nitroso compound formed The structure of 'C' is |
|
Answer»
|
|
| 7. |
Several isomeric amines are possible with the molecular formula C_(8)H_(11)N. On the basis of the reaction given by each of the isomers, identify the structure of the compound. a) A(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to p-Nitroso compound b) B(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to Diazonium salt formed c) C(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to N-Nitroso compound formed The structure of 'B' is |
|
Answer»
|
|
| 8. |
Several isomeric amines are possible with the molecular formula C_(8)H_(11)N. On the basis of the reaction given by each of the isomers, identify the structure of the compound. a) A(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to p-Nitroso compound b) B(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to Diazonium salt formed c) C(C_(8)H_(11)N)overset(NaNO_(2)-HCl)underset(0-5^(@)C)to N-Nitroso compound formed The structure of 'A' is likely to be |
|
Answer»
|
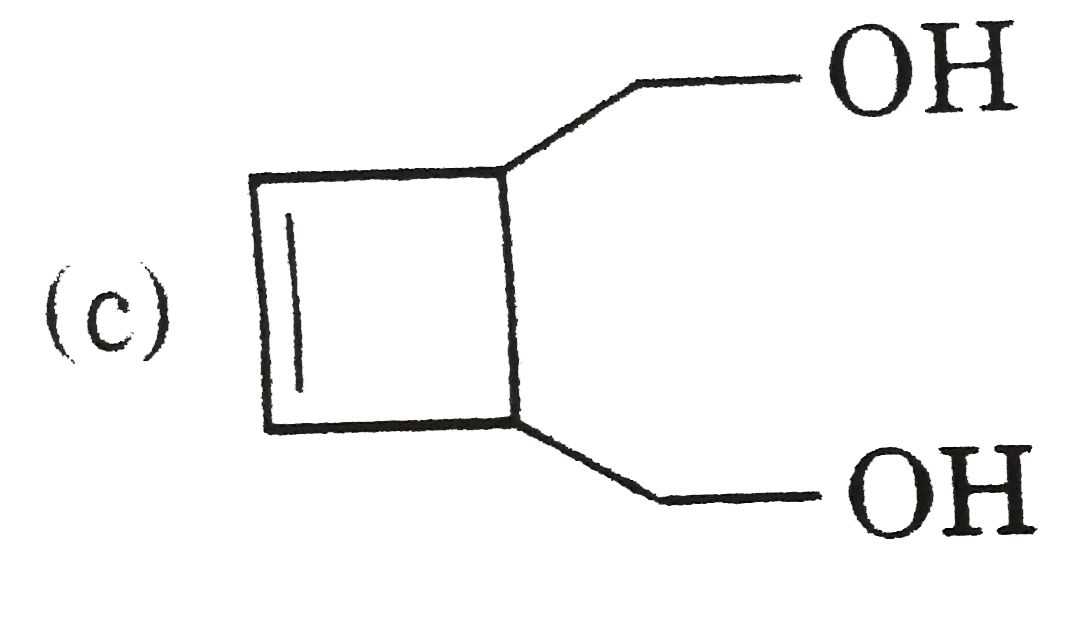
|
| 9. |
Several alkaloids are extracted from the extracts of the plants called marijuana. Marijuana owes its activity to tetrahydro cannabinol, which contains 70% as many as carbon atoms as hydrogen atoms and 15 times as many hydrogen atoms as oxygen atoms. One gram of tetrahydro cannabinol is 0.00318. Percentage composition of carbon in the compound is: |
|
Answer» 0.6046 |
|
| 10. |
Several alkaloids are extracted from the extracts of the plants called marijuana. Marijuana owes its activity to tetrahydro cannabinol, which contains 70% as many as carbon atoms as hydrogen atoms and 15 times as many hydrogen atoms as oxygen atoms. One gram of tetrahydro cannabinol is 0.00318. Molecular formula of the compound is: |
|
Answer» `C_(21)H_(30)O_(2)` |
|
| 11. |
Several alkaloids are extracted from the extracts of the plants called marijuana. Marijuana owes its activity to tetrahydro cannabinol, which contains 70% as many as carbon atoms as hydrogen atoms and 15 times as many hydrogen atoms as oxygen atoms. One gram of tetrahydro cannabinol is 0.00318. Number of oxygen atoms in 1 mol of the tetrahydro cannabinol is: |
|
Answer» `2N_(A)` |
|
| 12. |
Several alkaloids are extracted from the extracts of the plants called marijuana. Marijuana owes its activity to tetrahydro cannabinol, which contains 70% as many as carbon atoms as hydrogen atoms and 15 times as many hydrogen atoms as oxygen atoms. One gram of tetrahydro cannabinol is 0.00318. Molecular mass of the compound is: |
|
Answer» 413amu |
|
| 13. |
Setting of plaster of paris involves: |
|
Answer» OXIDATION with ATMOSPHERIC oxygen |
|
| 14. |
Set up Nernst equation for the standard dry cell. Using this eqation show that the voltage of a dry cell has to decrease with use. |
|
Answer» Solution :The voltage of an electrochemical cell is related to the concentration of the solutions involved in the cell. A change in concentration of cell solutions results in a change in voltage that can be quantitatively described by the Nernst equation. The voltage of an electrochemical cell involves an oxidation reaction and a reduction reaction. In the case of a copper-zinc cell the two half reactions are WRITTEN as REDUCTIONS , and their STANDARD cell potentials are : `Cu^(2+)(aq) + 2e to Cu(s)` reduction =`+0.34V` `Zn^(2+)(aq) + 2e to Zn`(s) reduction =-0.76 V Copper will be reduction species in the copper-zinc cell due it's larger standard reduction potential. The voltage then for a Zn, `Zn^(2+)"||"Cu^(2+) + Zn^(2+)(aq)` Equation 1 : ex. `Cu^(2+)(aq) + 2e to Cu(s) E^(@)` reduction =+0.34 V `Zn(s) + Zn^(2+)(aq) to2e^(-)E^(@)` oxidation =-0.76 V `E_("cell") =E_("reduction")-E_("oxidation")` `+1.10 V =+0.34 V-(-0.76 V)` The voltage fo any electrochemical cell is a function of the molar concentrations of the compounds involved in the cell, as described by the Nernst equation : `aA(s) + bB^(+) (aq) to cC(s)+ DD^(+)(aq)` Equation 2:`E_("cell")=E^(@)-(0.06//n)log{D^(+]d)//[B^(+)]^(b)}` Wquation 3: Using equation 1 as the balanced chemical equation for the `Zn, Zn^(2+)||Cu^(2+) , Cu` cell. Equation 3 becomes : `E_("cell") =E^(@)-(*0.03)log{Zn^(2+]//[Cu^(2+)]` As the `Cu^(2+)` concentration decreases the coltage fo the cell changes . The `Cu^(2+)` concentration can change due to dilution, or due to the formation of a complex ion, `Cu(NH_(3))_(4)^(2+)` , upon addition of `NH_(3)` |
|
| 15. |
Setting of cement is an: |
|
Answer» EXOTHERMIC process |
|
| 16. |
Set of d-orbitals which is used by central metal during formation of MnO_(4)^(-)? |
|
Answer» `d_(x^(2)-y^(2)),d_(x^(2)),d_(XY)` 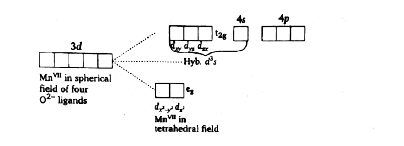
|
|
| 17. |
Set of elements known as chalcogens is : |
| Answer» Answer :A | |
| 18. |
Set of elements known as chalcogens is : 1) O, S, Se, 2) CI, Br, I 3) N, P, S 4) C, Si, Ge |
| Answer» Answer :A | |
| 19. |
Set -I (without catalyst) {:(,"Reaction",,"Temperature",,"E(activation)",,"k"),(,ArarrB,,T_(1)K,,Ea_(1),,k_(1)),(,A rarr B,,T_(2)K,,Ea_(2),,k_(2)):} Set-II (with catalyst) (consider +ve catalyst only) {:(,"Reaction",,"Temperature",,"E(activation)",,"k"),(,A rarr B,,T_(1)K,,Ea_(3),,k_(3)),(,A rarr B,,T_(2)K,,Ea_(4),,k_(4)):} For the Set-I: |
|
Answer» `Ea_(1)gt Ea_(2)if T_(1) gt T_(2)` |
|
| 20. |
Set -I (without catalyst) {:(,"Reaction",,"Temperature",,"E(activation)",,"k"),(,ArarrB,,T_(1)K,,Ea_(1),,k_(1)),(,A rarr B,,T_(2)K,,Ea_(2),,k_(2)):} Set-II (with catalyst) (consider +ve catalyst only) {:(,"Reaction",,"Temperature",,"E(activation)",,"k"),(,A rarr B,,T_(1)K,,Ea_(3),,k_(3)),(,A rarr B,,T_(2)K,,Ea_(4),,k_(4)):} Comparing Set-I and II: |
|
Answer» `k_(4) gt k_(3) and k_(2)k_(1), if T_(2) gt T_(1)` (endothermic) |
|
| 21. |
Set -I (without catalyst) {:(,"Reaction",,"Temperature",,"E(activation)",,"k"),(,ArarrB,,T_(1)K,,Ea_(1),,k_(1)),(,A rarr B,,T_(2)K,,Ea_(2),,k_(2)):} Set-II (with catalyst) (consider +ve catalyst only) {:(,"Reaction",,"Temperature",,"E(activation)",,"k"),(,A rarr B,,T_(1)K,,Ea_(3),,k_(3)),(,A rarr B,,T_(2)K,,Ea_(4),,k_(4)):} For the Set-I: |
|
Answer» If `T_(1) gt T_(2),k_(1)gt k_(2)` always |
|
| 22. |
{:("Set-I",, "Set-II") ,((A) "Noradrenaline" ,, (1) "Stable mental process" ),( (B) "Dopamine",, (2) "Regulation of control of movement "),( (C) "Serotonin",, (3) "Mood changes"),((D) "Histamine ",, (4) "Mild aches & pains "),((-),, (5) "Secretion of HCl "):} |
|
Answer» `{:(A,B,C,D) ,(1,2,3,5):}` |
|
| 23. |
Set containing isoelectronic species is/are |
|
Answer» `C_(2)^(2-), NO^(+), CN^(-), O_(2)^(2+)` |
|
| 24. |
{:("SET-1","SET-2"),((1)"Ostwald-walker",(A) "Osmotic pressure"),((2)"Cotrell's method",(B)"Depression of F.P"),((3)"Rast's camphor method","(C) Elevation of B.P"),((4)"Berkeley and Hartley's method",(D)"Lowering of vapour pressure" ):} |
|
Answer» `{:(A,B,C,D),(4,3,2,1):}` |
|
| 25. |
{:("List - I","List - II"),("(1) Steriod hormone","(a) Cytokinins"),("(2) None steroid hormone","(b) Estrogens"),("(3) Plant hormone","(c ) Auxins"),("(4) Peptide hormone","(d) Insulin"):}The correct match is |
|
Answer» `1 - B, 2- b, 3 - C, 4 - d` |
|
| 26. |
{:("SET-1","SET-2"),((i)"RBC in 0.5% NaCl solution",(A)"Swells"),((ii)"RBC in 1% NaCl solution",(B)"Shrinks"),((iii)"egg (outer shell removed in water)",.),((iv)"egg(outer shell removed in NaCl solution)",.):} |
|
Answer» i-A |
|
| 27. |
Sesquoxides of alkali metals may be represented by the formula : |
|
Answer» `M_(2)O_(5)` |
|
| 29. |
Serpeck's process is used for the extraction of : |
|
Answer» Mg |
|
| 30. |
Serpeck's process is used for bauxite which has following main impurity : |
|
Answer» `SiO_(2)` |
|
| 31. |
Serpeck process may be used to prepare |
|
Answer» CYANIDE |
|
| 32. |
Sequence of acidic character is |
|
Answer» `SO_2 GT CO_2 gt CO gt N_2O_5` |
|
| 33. |
Separation of two layers are seen when Lucas reagent is treated with |
|
Answer» `CH_3OH` |
|
| 34. |
Separation of organic compounds by column chromatography is due to: |
|
Answer» SELECTIVE ADSORPTION |
|
| 35. |
Separationof basic radicals is based on (a) ____ and (b) ____. |
|
Answer» |
|
| 36. |
Separating of d and l enantiomorphs from a racemic mixture iscalled |
|
Answer» RESOLUTION |
|
| 37. |
Separating of d and l enantiorphs from a racemic mixture is called |
|
Answer» Resolution |
|
| 38. |
Semiconductors are solids with conductivities in the range of _________________. |
| Answer» Solution :`10^(-6)" to "10^(4)"OHM"^(-1)m^(-1)` | |
| 39. |
Semiconductors are purified by ______ method. |
|
Answer» ZONE refining |
|
| 40. |
Semi conservative method of DNA duplication means |
|
Answer» Newly synthesized DNA is CONSERVED only in ONE CELL cycle |
|
| 41. |
Semicarbzide is : |
|
Answer» `NH_2CONH_2` |
|
| 42. |
Self-ionisation of liquid ammonia occurs as, 2NH_(3) rarr NH_(4)^(+) + NH_(2)^(-), K = 10^(-10). In this solvent, an acid might be |
|
Answer» `NH_(4)^(+)` |
|
| 43. |
Selet correct statement. |
|
Answer» Saccharine is artificial SWEETENER. |
|
| 44. |
Self-condensation of two moles of ethyl acetate in presence of sodium ethoxide yields. |
|
Answer» ETHYL propionate |
|
| 45. |
Selectr true statement about this reaction : |
|
Answer» Degree of unsaturation is 10 for major product 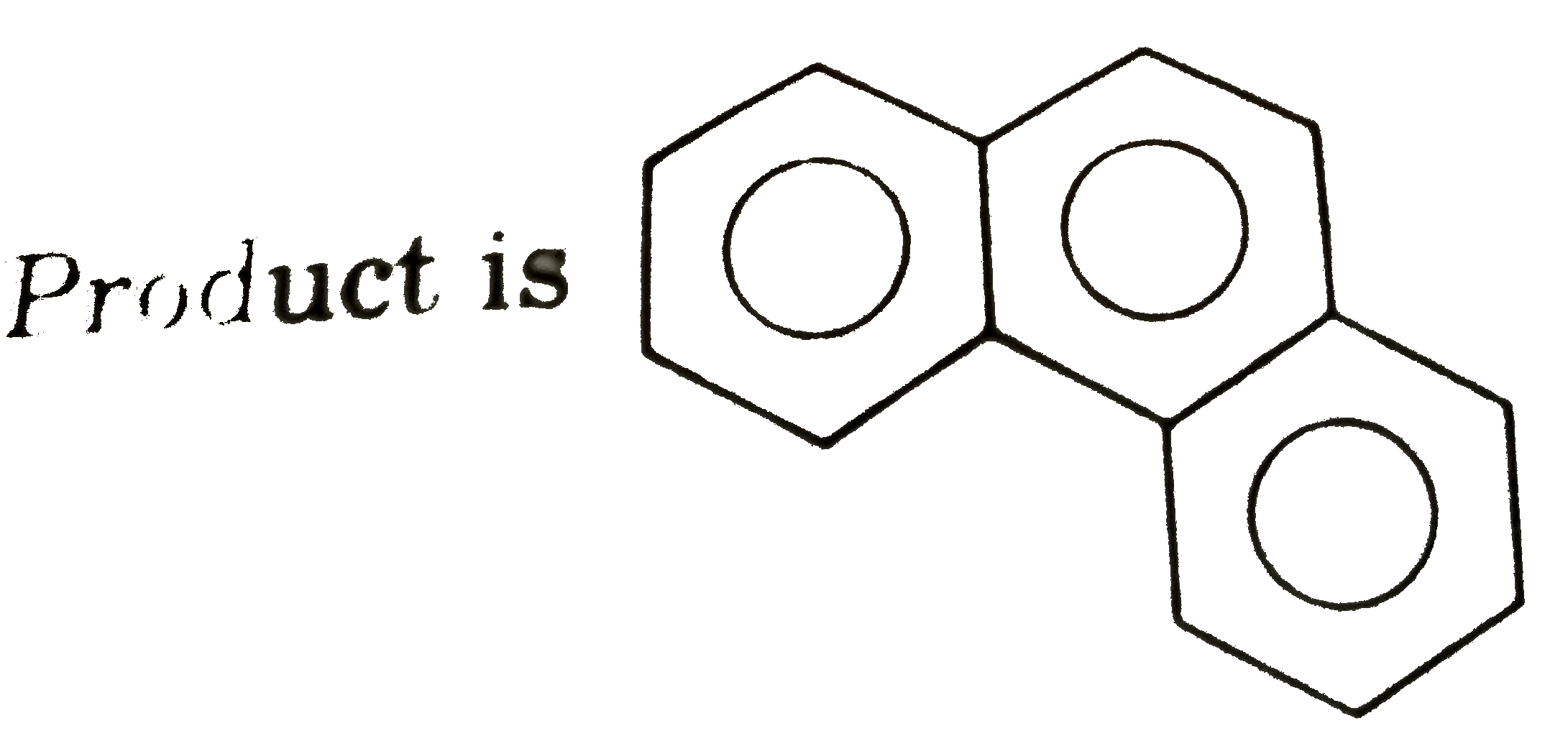
|
|
| 46. |
Selecting the correct statement if any from the following |
|
Answer» during zone rofining of silicon the metal is more soluble in the melt than the impurity Metal like Fe, Zn, Hg are extracted by pyro metallurgy but not .Ag. White bauxite is PURIFIED by Serpeck.s process . Doilamite, Magnesite and graphite are used as REFRACTORY materials in furnace. |
|
| 47. |
Selected the correct option : |
|
Answer»
|
|
| 48. |
Selecthe correct statement(s): |
|
Answer» PHYSICAL chemistry |
|
| 49. |
Selected incorrect satment. |
|
Answer» p can turn blue LITMUS red 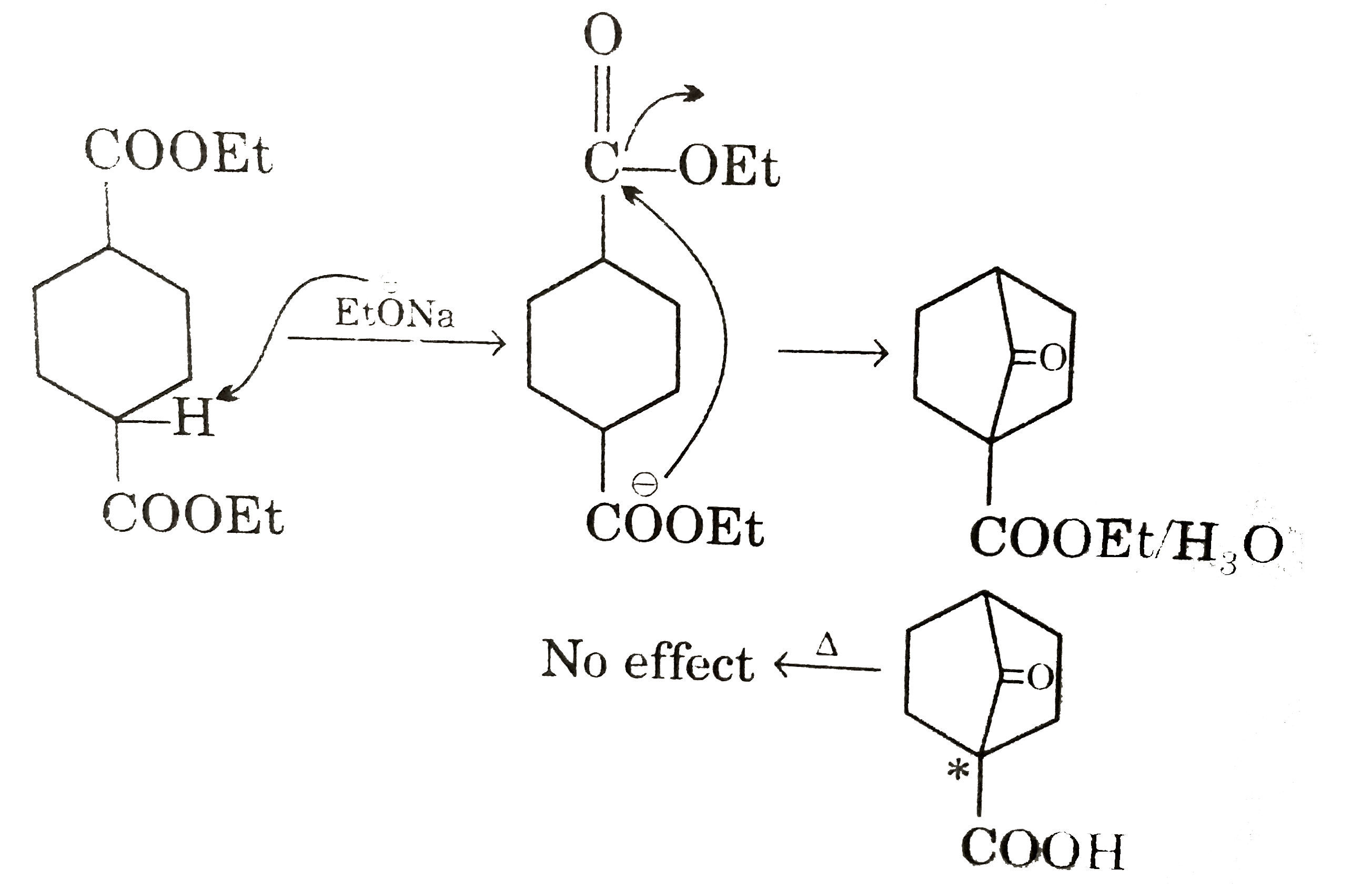
|
|










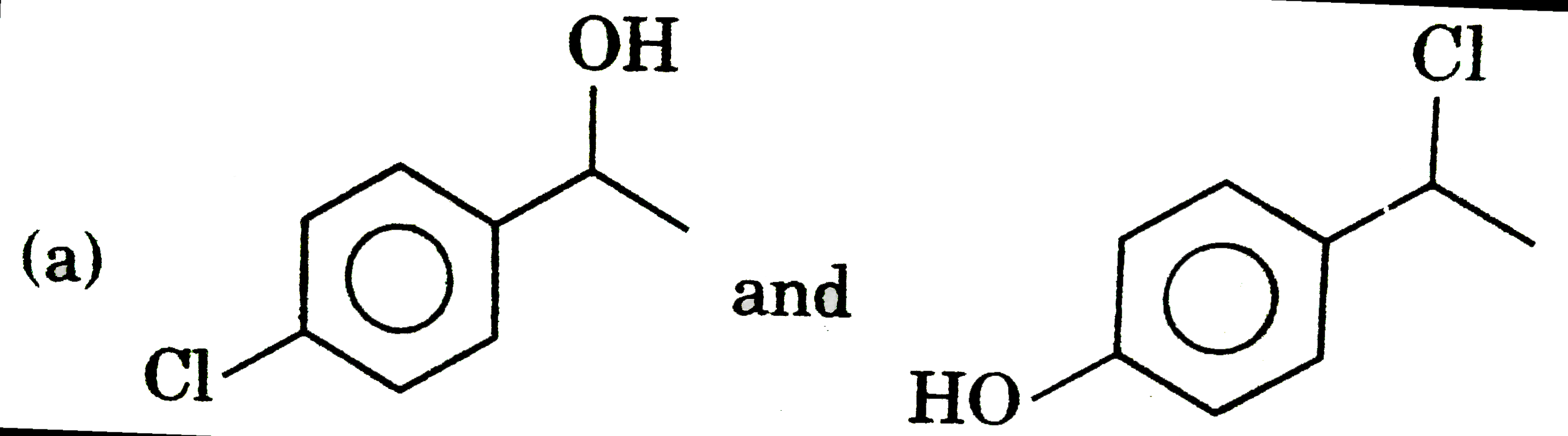
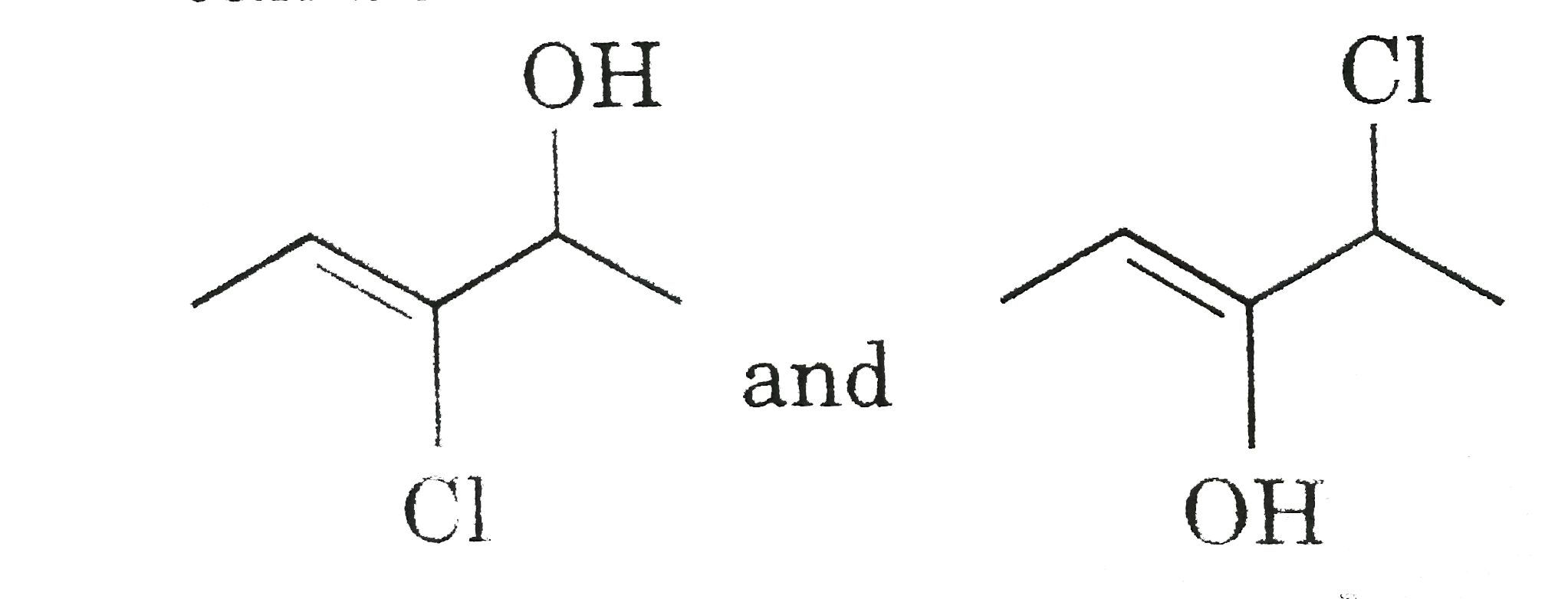 can be differentiated by Bielstien test.
can be differentiated by Bielstien test.