Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Show how each of the following compounds can be converted to benzoic acid : (i) Ethylbenzene (ii) Acetophenone (iii) Bromobenzene (iv) Phenylethene (Styrene). |
Answer» SOLUTION :
|
|
| 2. |
Show how each of the following compounds can be converted to benzoic acid. (i) Ethylbenzene |
|
Answer» |
|
| 3. |
Show how an aldohexose can be used to synthesize 2-ketohexose. |
Answer» Solution : Here aldohexosereacts with one molecule of phenylhdrazine which CONDENSES with the aldehyde group to give phenylhydrazone. When warmed with axcess of phenyl hydrazine, the secondary alcoholic group ADJACENT to the aldehyde group is oxidised by another molecule of phenylhydrazine, to a ketonic group. With this ketonic group, the third molecule of phenylhydrazine condenses to GIVEN OSAZONE. The phenylhydrazinyl group is transferred from osazone to `C_6H_5CHO` giving `C_6H_5CH=N.NHC_6H_5` and a dicarbonyl compound called an osone. The more reactive aldehyde group of the osone is reduced, not the less reactive keto group and it give the 2-ketohexose. |
|
| 4. |
Show how an acylium ion could be formed from acetic anhydride in the presence of AlCl_3. |
Answer» Solution : We RECOGNIZE that `AlCl_3`is a Lewis acid and that an acid ANHYDRIDE, because it has multiple unshared electron pairs, is a Lewis BASE. A reasonable mechanism starts with a Lewis acid-base reaction and proceeds to FORM an acylium ion in the following way.
|
|
| 5. |
Show how are the following alcohols prepared by the reaction of a suitable Grignard reagent on methanal? (i) CH_3-underset(CH_3) underset(|)CH-CH_2OH(ii) |
Answer» SOLUTION :(i) `CH_3- UNDERSET(CH_3)underset(|)(CH) - CH_2OH` : 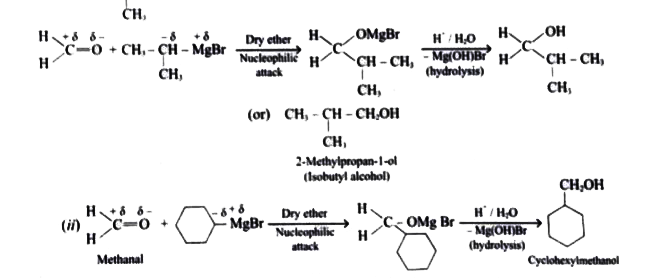
|
|
| 6. |
Show how are the following alcohols are prepared by the reaction of a suitable Grignard reagent on methanal? I) (CH_3)_2CH CH_2OH"" II) |
|
Answer» SOLUTION :The equations showing the preparation of ALCOHOLS from FORMALDEHYDE are: I) `HCHOunderset((II)H_2O)overset((i)(CH_3)_2CHMgBr)(RARR)(CH_3)CHCH_2OH` II) 
|
|
| 7. |
Show graphically that the freezing point of a liquid will be depressed when a non-volatile solute is dissolved in it. The freezing point of a solution containing 0-3 g of acetic acid in 30-0 g of benzene is lowered by 0-45^@. Calculate the van't Hoff factor. (K for benzene = 5-12 K kg mol""^(-1)). |
|
Answer» Solution :Graphical representation of freezing point depression is given in Fig. . Freezing point depression, `Delta T_f =T_(f_f^(@) -T_f` Let us FIRST calculate OBSERVED molar mass. `M_B=(K_fxxW_Bxx1000 )/( Delta T_f XX W_A)` `=(5.12 xx 0.3 xx 1000)/(=5.12 xx 0.3 xx 1000)/(0.45 xx 30.0 )` `113.8` 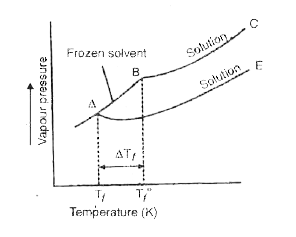 NORMAL molar mass of CH3COOH = 60 Van.t HOFF factor, i = `("Normal molar mass")/("Observed molar mass ")` `=(60)/(113.8)= 0.527` |
|
| 8. |
Show by using rate law, how much rate of the reaction, 2"NO"+"O"_(2)to2"NO"_(2), will change if the volume of the reaction vessel is reduced to one third of its initial value ? |
|
Answer» Whenvolume is reduced to one-third, the concentration of each reactant will BECOME THREE times. New rate `=k(3" a")^(2)(3" b")=27" k a"^(2)b.` |
|
| 9. |
Show graphically how the amount of gas adsorbed on the surface of solid in physical adsorption very with (i) pressure (ii) temperature? |
| Answer» | |
| 10. |
Show by chemical equations only , how would you prepare the following from the indicated starting meterials. Specify the reagents in each step of the synthesis. Hexachloroethane (C_(2)Cl_(6)) from calcium carbide |
|
Answer» Solution :`CaC_(2) + H_(2)O to C_(2)H_(2) + Ca(OH)_(2) and C_(2)H_(2) overset(Ni//H_(2))to CH_(3)- CH_(3)` `CH_(3) - CH_(3) overset(Cl_(2)(EXCESS), light)to UNDERSET("hexachloroethane")(Cl_(3)C -"CCL"_(3))` |
|
| 11. |
Show by chemical equations only , how would you prepare the following from the indicated starting meterials. Specify the reagents in each step of the synthesis. Chloroform from carbon disulphide |
|
Answer» Solution :`CS_(2)+Cl_(2)to "CCl"_(4) + S_(3)Cl_(2) or CS_(2) + 2S_(2)Cl_(2) to "CCl"_(4) + 6S` `"CCl"_(4) + 2[H] overset(Fe//HCL)tounderset("chloroform")CHCl_(3) + HCl` |
|
| 12. |
show by chemical equactions only how would you prepare the following from the indicated startingmaterials. Specifythe reagentsin each step of synthesis of: i. Chloroform from carbondisulphide. ii. Hexachlorothane(C_(2) Cl_(6)) from calcium carbide. |
|
Answer» Solution :i. `CS_(2) + Cl_(2) rarr CCl_(4) + S_(2) Cl_(2)` `CS_(2) + 2SCl_(4) + 6S` `C Cl_(4) + 2 [H] overset(Fe//H_(2)O)(rarr) underset("Chloroform")(CHCl_(3)) + HCl` ii. `CaC_(2) + H_(2) O rarr C_(2) H_(2) + Ca(OH)_(2)` `C_(2) H_(2) overset(Ni//H_(2))(rarr) CH_(3) - CH_(3)` `CH_(3) - CH_(3) underset("prolonged TREATMENT") overset(Cl_(2) "(excess).LIGHT")(rarr) underset("Hexachloroethane")(Cl_(3)C - C Cl_(3))` |
|
| 13. |
Show by a graphic diagram how at a constant pressure a rise in temperature will influence adsorption of a gas on a solid when (a) no compound formation occurs.(b) chemisorption takes place. |
|
Answer» Solution :No compound formation MEANS physical adsorption. In such a case, the extent of adsorption decreases with increase of temperature. (b) In chemisorption, the extent of adsorption first INCREASES, it REACHES a maximum and then decreases afterwards. The graphs SHOWING the VARIATIONS are given in the following diagrams. 
|
|
| 14. |
Show by a graphic diagram how at a constant pressure a rise ihn temperature will influence adsorption of a gas on solid when : (i) No compound formation occurs. (ii) Chemisorption takes place. |
| Answer» | |
| 15. |
Shortest carbon-carbon single bond distance is present in |
|
Answer» CH `equiv` C - C `equiv` CH sp - sp, C - C bond has shortest distance. |
|
| 16. |
Shortest C-C bond length is present in |
|
Answer» `CH_3-CH_2-CH_3` C-C bond length = 1.54 Å C=C bond length =1.33 Å `C-=C` bond length =1.22 Å |
|
| 17. |
Shorter the radioactive half life |
|
Answer» GREATER is the number of ATOMS DISINTEGRATING per second |
|
| 18. |
Shellac' secreated by lac insect is |
|
Answer» NATURAL plastic |
|
| 19. |
Shifting of electrons of a multiple bond under the influence of a reagent is called: |
|
Answer» I-effect |
|
| 20. |
Shawn went to a meat shop to but meat from the butcher. While buying the meat, he observed that the butcher was highely upset. When he asked him the reason, he told that he was suffering a heavy loss as his meat gets spoiled very soon. Shawan suggested him that he should apply common salt on the meat to save it from spoilage. After reading the above passage, answer the following questions : (a) What values are expressed by Shawn? |
| Answer» SOLUTION :Shawn SHOWED social VALUES of helping a POOR MAN who was illiterate. | |
| 21. |
Shawn went to a meat shop to but meat from the butcher. While buying the meat, he observed that the butcher was highely upset. When he asked him the reason, he told that he was suffering a heavy loss as his meat gets spoiled very soon. Shawan suggested him that he should apply common salt on the meat to save it from spoilage. After reading the above passage, answer the following questions : (b) Why did Shawn suggest him to apply salt on the meat? How does it work? |
| Answer» SOLUTION :In the salted meat,the bacterium which is RESPONSIBLE for spoilage loses WATER due to osmosis. As a result, it shrivals and dies. | |
| 22. |
SHE is _________. |
|
Answer» Standard HELIUM Electrode |
|
| 23. |
Shaving soaps give extensive lather due to |
|
Answer» SODIUM rosinate |
|
| 24. |
Shapte of IF_(7) molecules is |
|
Answer» Pyramid No of bond pirs =7, no of lone paris =0 Hybridisation `=SP^(3)d^(3)` SHAPE pentagonal bipyramidal |
|
| 25. |
Shape selective catalysis is a reaction catalysed by :Zeolites,Enzymes,Platinum,Zeigler-Natta catalyst. |
|
Answer» Zeolites |
|
| 26. |
Shape selective catalysis are known by this name because |
|
Answer» DUE of the shape of catalyst |
|
| 27. |
Shape of XeOF_(4) is |
|
Answer» OCTAHEDRAL |
|
| 29. |
Shape of XeOF_4 is : |
|
Answer» octahedral |
|
| 30. |
Shape of XeF_(6) reacts with 2.5 M NaOH gives, ……………… . |
|
Answer» OCTAHEDRON |
|
| 31. |
Shape of XeF_6 molecule is_____. |
| Answer» SOLUTION :DISTORTED PENTAGONAL bipyramidal | |
| 32. |
Shape of ozone molecule isieibi |
|
Answer» V-shpaed |
|
| 33. |
Shape of ozone, …………………. . |
|
Answer» V - SHAPE |
|
| 34. |
Shape of NH_(3) is very similar to : |
|
Answer» `CH_(4)` |
|
| 35. |
Shape of molecule having 4-bond pair and one lone pair is :- |
|
Answer» TRIGONAL bipyramidal 
|
|
| 36. |
Shape of H_3PO_4 is |
|
Answer» Pyramidal |
|
| 37. |
Shape of molecules is decided by : |
|
Answer» `SIGMA-bond` |
|
| 38. |
Shape of Fe(CO)_(5) is , |
|
Answer» PENTAGONAL bipyramidal |
|
| 39. |
Shape of ClO_(3)^(-) ion is |
|
Answer» PYRAMIDAL |
|
| 40. |
Shape of ClF_(3) is ………………. . |
| Answer» Solution :T - shape | |
| 41. |
Shape of ammonia is ………………. . |
| Answer» Solution :Pyramidal | |
| 42. |
Shape of anion A will be : |
|
Answer» Tetrahedral |
|
| 43. |
Shanti , a domesticf helper of Mrs. Anuradha , fainted while mopping the floor. Mrs. Anuradha immediately took her to the nearby hospital where she was diagnosed to be severly anaemic. The doctor prescribed an iron rich diet and multivitamins supplement to her . Mrs. Anuradha supported her financially to get the medicines. After a month , Shanti was diagnosed to be normal. After reading the above passage, answer the following questions : (i) What values are displayed by Anuradha ? (ii) Name the vitamin whose deficiency causes 'pernichious anaemia'. (iii) Give an example of a water soluble vitamin. |
|
Answer» Solution :Anuradha displayed feelings of humanity and sympathy towards people who are POOR and need your help to SUSTAIN their LIVES. (i) DEFICIENCY of vitamin `B_12` causes pernicious anaemia. (ii) Vitamin C is SOLUBLE in water. |
|
| 44. |
Shape and bond angle in ClO_(4)^(-) ion is |
|
Answer» Planar TRIGONAL , `109^(0)28^(1)` |
|
| 45. |
SF_6 is known but SCI_6 is not. Why? |
| Answer» Solution :Due to small SIZE of fluorine, six F IONS can be accommodated around sulphur. CHLORIDE ion being LARGER in size, six of these ions cannot be accommodated around sulphur. Therefore `SCI_6` is not formed. | |
| 46. |
SF_(6) is known but SH_(6) is not known. Explain. |
| Answer» Solution :Fluorine being the strongest oxidising agent OXIDISES sulphur to its MAXIMUM oxidation state of +6 and THUS FORMS `SF_(6)`. In contrast, `H_(2)` being a very weak oxidising agent cannot oxidise S to its maximum oxidation state of +6 and hence does not form `SH_(6)`. | |
| 47. |
SF_6 is known but SCl_6 is not. Why ? |
|
Answer» SOLUTION :This is due to (i) Small size of fluorine (ii) HIGH electronegativity of fluorine. Fluorine being highly electronegative can oxidize sulphur to (+6) oxidation STATE and can be accomodated easily due to its small size. Chlorine on the CONTRARY, cannot be accomodated due to its LARGE size. Thus, `SCl_6` does not exist. |
|
| 48. |
SF_(6) is known but SCl_(6) is not. Why? |
| Answer» Solution :Fluorine atom is smaller in size so six `F^(-)` IONS can surround a sulphur atom. The CASE is not so with chlorine atom due to its large size. So, `SF_(6)` is known but `SCl_(6)` is not known due to interionic repulsion between larger `Cl^(-)` ions. | |
| 49. |
SF_(4) is obtained by treating sulphur with |
|
Answer» `F_(2)` |
|
| 50. |
SF_4 has_____geometry. |
|
Answer» Octahedral |
|