Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select the correct order of mobility in aqueous medium. |
|
Answer» `[Li(H_(2)O)_(x)]^(+)gt[LE(H_(2)O)_(y)]^(+2)` `rarr+ve` CHARGE DENSITY increases. `rarr` Zeft increases. `rarr` Size of `Be^(+2)` decreaes `rarr` Surrounding `H_(2)O` molecule increases. `rarr` BULKY nature increaes. `rarr` Mobility DECREASES. |
|
| 2. |
Select the correct order of periodic properties of species : |
|
Answer» `Fe^(2+) LT Fe^(3+)`: IONIC radii |
|
| 3. |
Select the correct order of electron gain enthalpy for F, CI, Br, I. |
|
Answer» I GT BR gt CI gt F |
|
| 4. |
Select the correct order of following property . |
|
Answer» % s -character : `sp^(3) gt sp^(2) gt sp` |
|
| 5. |
Select the correct order of CFSE (Delta) for the ions given below |
|
Answer» `V^(2+) lt MN^(2+) lt Fe^(2+) lt CO^(2+) lt NI^(2+)` |
|
| 6. |
Select the correct order of C-O bond length : |
|
Answer» `CO gt FE(CO)_(5)` |
|
| 7. |
Select the correct order of acidity |
|
Answer» `HI GT HBr gt HCl gt HF` |
|
| 8. |
Select the correct order of acidity : |
|
Answer» `HI gt HBr gt HCl gt HF` |
|
| 9. |
The increasing order of acidity : |
|
Answer» HI `gt` HBr `gt `HCl` gt ` HF |
|
| 10. |
Select the correct order of Acidic strength :- |
|
Answer» HF gt HCl gt HBR gt HI |
|
| 11. |
Select the correct order for the wavelength of absorption for the given complexes : |
|
Answer» `[Cr(CN)_(6)]^(3-)=[CrCl_(6)]^(3-)=[Cr(NH_(3))_(6)]^(3+)=[Cr(H_(2)O)_(6)]^(3+)` `Delta_(0) prop (1)/(lambda)` |
|
| 12. |
Select the correct order |
|
Answer» HOClgtHOBrgtHOI - Acid strength |
|
| 13. |
Select the correct order for the given properties - (I) Thermal stability: BaSO_(4) gt SrSO_(4) gt CaSO_(4) gt MgSO_(4) (II) Basic Nature : ZnO gt BeO gt MgO gt CaO (III) Solubility in water : LiOH gt NaOH gt KOH gt RbOH (IV) Melting point : NaCl gt KCl gt RbCl gt LiCl |
|
Answer» I, IV |
|
| 14. |
Select the correct order. |
|
Answer» Graphite = fullerene lt diamond (no. of CARBON atoms connected PER carbon atom with covalent bond only) while in diamond = 1 (b) Ionic mobility of `Rb^(+)gtK^(+)gtNa^(+)` Ionic mobility of `Br^(-)gtCl^(-)gtF^(-)` `L.E.prop(q_(+)*q_(-))/(r^(+)+r^(-))` between charge and size, charge dominates. |
|
| 15. |
Select the correct order for the strength of bases given below: |
|
Answer» `C_2H_5^rarrNH_2^rarrC_2H_3^rarrOH^rarrC_2H^-` |
|
| 16. |
Select the correct options for the following statements. 1. Cl_(2)O and ClO_(2) are used as bleaching agents. 2. OC l^(-) salts are used as detergent.. 3. OC l^(-) disproportionates in alkaline medium. 4. BrO_(3)^(-) is oxidized in acidic medium. |
|
Answer» 1, 2, 3 correct |
|
| 17. |
Select the correct option( s) : |
|
Answer» PRESSURE in container-I is 3ATM before OPENING the VALVE. |
|
| 18. |
Select the correct options : |
|
Answer» Boiling point , `(P gt Q)` |
|
| 19. |
Select the correct option :- The Grand Alliance of opposition of 1971 |
|
Answer» Got a combined tally of seats that was LESS than 40. |
|
| 20. |
Select the correct option for the leader and the country that helped to reach the Tashkent agreement between India and Pakistan : |
|
Answer» INDIA, Nehru |
|
| 21. |
Select the correct option for the given processes. (P) Process of heating stell to redness and then cooling it very slowly. (Q) Process of heating steel in presence of NH_(3) and producing hard coasting of iron nitrate on the surface of steel. (R ) Process of heating steel to redness and then cooling it suddenly by plunging it into water or oil. (S) Process of heating quencheed steel toa temperature well below redness and then cooling it slowly. |
|
Answer» TEMPERING , Nitriding, Annealing and Quenching RESPECTIVELY |
|
| 22. |
Select the correct option regarding above paragraph: |
|
Answer» A is good absrobent of carbondioxide. `D=Al E=Naoverset(+3)AIO_(2)` |
|
| 23. |
Select the correct option among following at 298 K temperature |
|
Answer» In neutral AQ. SOLUTION, `{[H^+] = SQRT(K_w)}` |
|
| 24. |
Select the correct option. |
|
Answer» `Delta H_(F) [H (g)]` is EQUAL to `Delta H_("atomisaation")` of `H_(2) (g)` |
|
| 25. |
Select the correct option ? |
|
Answer» |
|
| 26. |
Select the correct option : |
|
Answer» `(A)` is `SiF_(4)` 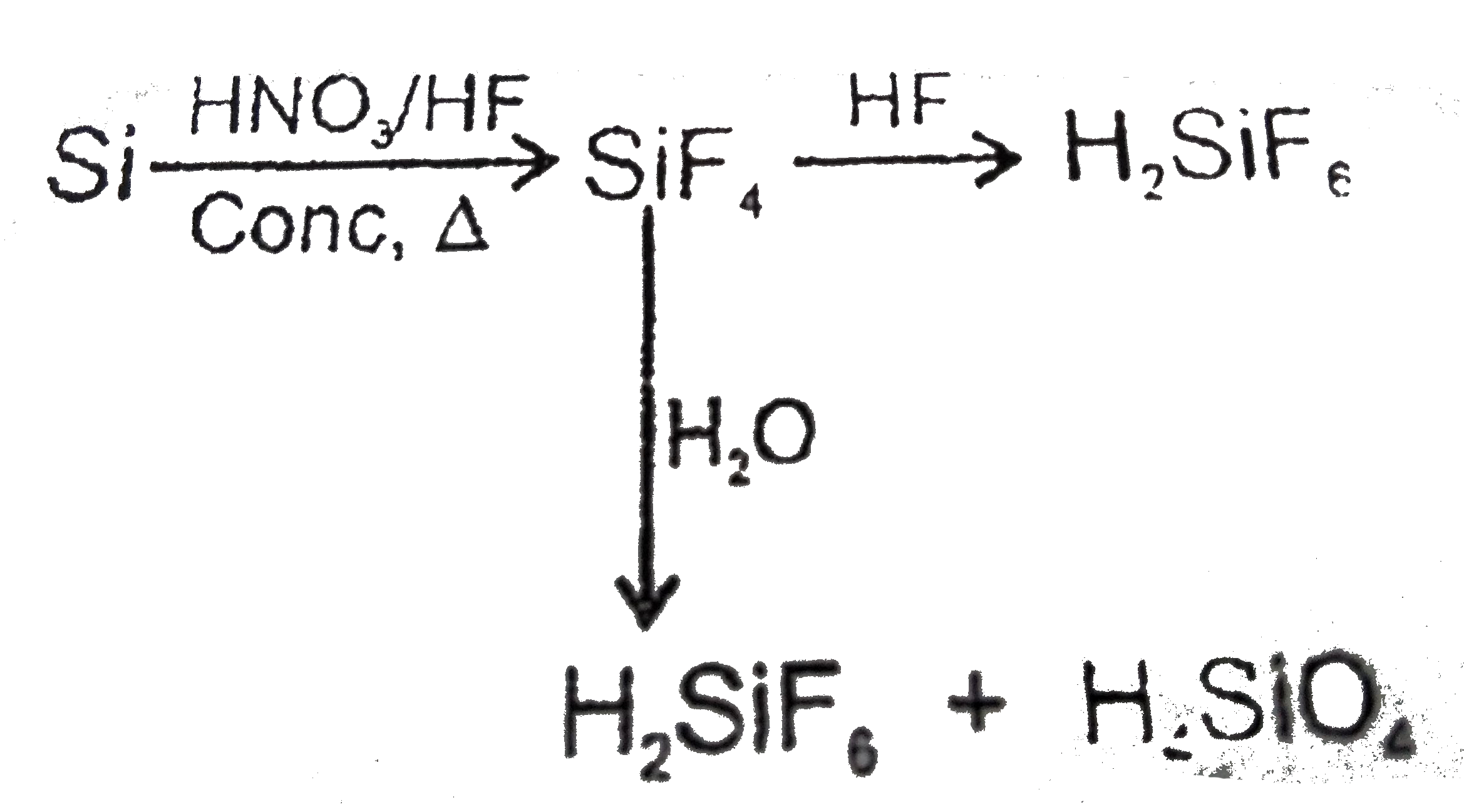
|
|
| 27. |
Select the correct option : |
|
Answer» Isoelectric point is the pH at which an amino acid exists PRIMARILY in its neutral form. |
|
| 28. |
Select the correct match here |
|
Answer» `[Co(o x)(H_2O)(NH_3)]Br` OPTICAL ISOMERISM |
|
| 30. |
Select the correct graph for given property for chalcogen family - |
|
Answer»
|
|
| 31. |
Select the correct limitations of III law of thermodynamics: |
|
Answer» Glassy SOLIDS at zero KELVIN has entropy greater than zero |
|
| 32. |
Select the correct IUPAC name from the following. |
|
Answer» `H[AuCl_(4)]` : Hydrogen tetrachloridoaurate(III) |
|
| 33. |
Select the correct group(s) of reagent(s) used in the following conversions: |
|
Answer» a.`I implies DIL. HNO_(3)`, `II implies Na_(2)Cr_(2)O_(7)//H_(2)SO_(4)` (II) `implies` EWG, e.g., `(-NO_(2))` group makes the benzene ring stable, so either acidic `KMnO_(4)` or acidic `K_(2)Cr_(2)O_(7)` can be used to oxidise the `(Me-)` group to `(-COOH)` group. III `implies (-I)` effect of `Cl` makes the benxene ring stable, so oxidising agent such as ALKALINE `KMnO_(4)` or acidic `K_(2)Cr_(2)O_(7)` can be used to oxidise the `(Me-)` group to `(COOH)`. IV `implies EDG (+R)` effect of `(-OH)` group makes the benzene ring unstable, so either `(-OH)` has to be PROTECTED by tosylation using `TsCl` and then oxidation of `(Me-)` group to `(-COOH)` group by acidic `KMnO_(4)` is carried out FOLLOWED by hydrolysis or by using a very mild O.A. such as `PbO_(2)//overset()(O)H, H_(3)O^(o+)`. So the answers are `(a)` and `(b)`. |
|
| 34. |
Select the correct expression regarding the relation between K_(P) and K_(C) for the reaction aX_((g))+bY_((g))hArrbZ_((g))+aW_((g))- |
|
Answer» <P>`K_(p)=K_(C)(RT)^(a+b)` |
|
| 35. |
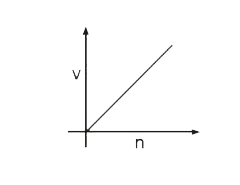
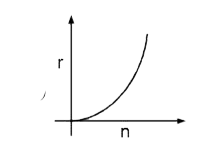
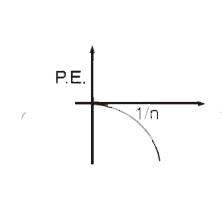
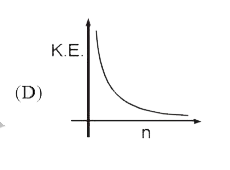
Select the correct curve(s) : If v = velocity of electron in Bohr’s orbit. r = Radius of electron in Bohr’s orbit. P.E. = Potential energy of electron in Bohr’s orbit. K.E. = Kinetic energy of electron in Bohr’s orbit. |
|
Answer»
|
|
| 36. |
Select the correct code regarding total number of space isomers for the following compounds: (I) [Ma_(3)b_(2)c]^(n+-) (II) [M(AB)_(3)]^(n+-) (III) [Ma_(2)b_(2)c_(2)]^(n+-) |
|
Answer» (I)-4,(II)-4,(III)-6 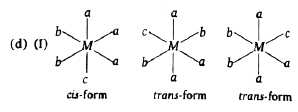 `IMPLIES`All are OPTICALLY inactive, No. of stereoisomers=3 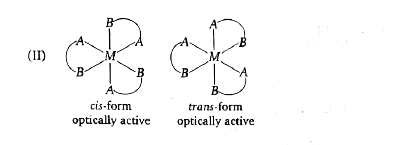 `implies ` Both are optically active, No of stereoisomers=4 (III) 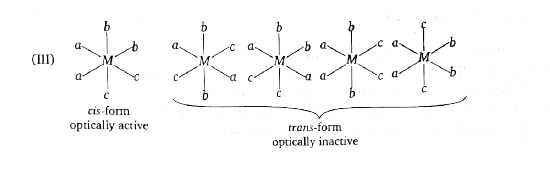 `implies `No. of stereoisomers=6 |
|
| 37. |
Select the correct combination of reactions. |
|
Answer» `CH_(3)-OVERSET(O)overset(||)C-CH_(3) underset(HCl)overset(Zn-Hg)to`, Wolf-Kishner reduction |
|
| 38. |
Select the correct code about complex [Cr(NO_(2))(NH_(3))_(5)][ZnCl_(4)]: (I) IUPAC anem of compound is pentaamminenitrito-N-chromium (III) tetrachlorozincate (II) (II) it shows geometrical isomerism (III) It shows linkage isomerism (IV) it shows coordination isomerism |
|
Answer» III, IV (II) It does not exhibit geometrical isomerism. (III) it SHOW linkage isomerism due to presence of AMBIDENTATE ligand `NO_(2)^(-)` (IV) its coordination ISOMERS are: `[CrCl(NH_(3))_(5)][ZnCl_(3)(NO_(2))],[CrCl_(2)(NH_(3))_(4)][ZnCl_(2)(NO_(2))NH_(3)]` `[Zn(NO_(2))(NH_(3))_(3)][CrCl_(4)(NH_(3))_(2)]` |
|
| 40. |
Select the correct code of TRUE and FALSE for given statements: (a) Peroxide ion as well as dioxygen molecule both are paramagnetic species (b) In set of isomers, [Cr(H_(2)O)_(6)]Cl_(3) and [CrCl(H_(2)O)_(5)]Cl_(2)*H_(2)O, both compounds can easily loose water molecule on treatment with conc. H_(2)SO_(4) (c) During transformation NO to NO^(+), bond length and magnetic behaviour decreases (d) An ether is more volatile than alcohol both having same molecular formula |
|
Answer» FFTT Conc. `H_(2)SO_(4)` can DEHYDRATE water of crystallization from `[CRCL(H_(2)O)_(5)]Cl_(2)*H_(2)O` but it can not remove those water molecules which are working as ligands. |
|
| 41. |
Select the compounds where iron is in +2 oxidation state, |
|
Answer» `K_(3)[Fe(CN)_(6))]` |
|
| 42. |
select correct absorption isobars for chemisoption and physisoption respectivly ,(where x/m= extent of adsorption , T = temperature ) |
|
Answer»
|
|
| 43. |
Select the compound which on treatment with nitrous acid liberates nitrogen. |
|
Answer» Nitroethane `C_(2)H_(5)NH_(2) + HO-N = O overset("COLD") to C_(2)H_(5) OH + H_(2)O + N_(2) uarr` |
|
| 44. |
Select the compounds having metal in zero oxidation state |
|
Answer» `Ni(CO)_4` |
|
| 45. |
Select the compound in which the oxidation number of oxygen is –1:- |
|
Answer» `HClO_(4)` |
|
| 46. |
Select the compound in which chlorine shows highest oxidation state : |
|
Answer» `HClO_(4)` |
|
| 47. |
Select the compound from the following which can undergo S_(N)2, but cannot undergo( or gives minor product), elimination on heating with CH_(3)O^(-). |
|
Answer» 2-bromo-3-methylpentane<BR>1-bromo-2,3-dimethylbutane (primary alkyl halide has more tendency to undergo SUBSTITUTION) `underset(""CH_(3))underset("|")overset(""CH_(3))overset("|")(CH_(3)-CH_(2)-C-CH_(2)Br` ( primary alkyl halide without `alpha`-hydrogen ) |
|
| 48. |
Select the compound from the following which dissolves in water |
|
Answer» `C Cl_(4)` |
|
| 49. |
Select the complex which show both structural and stereo isomerism : |
|
Answer» `[Pt(NH_(3))_(4)Cl_(2)]l` |
|