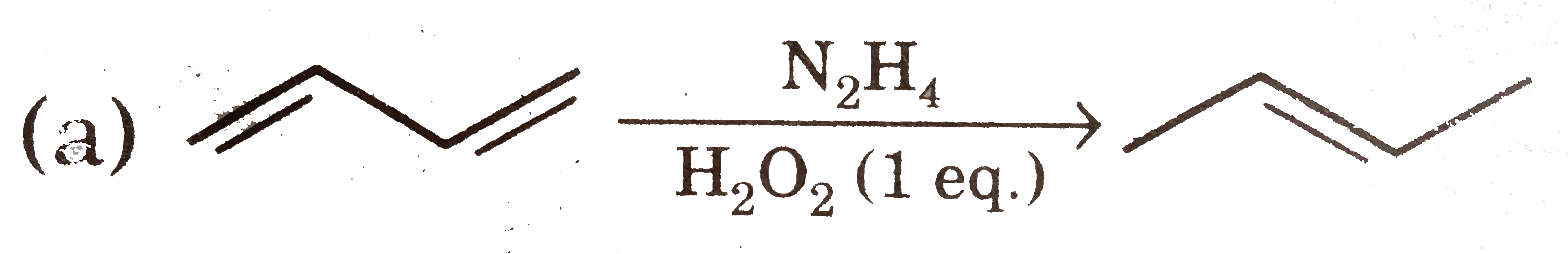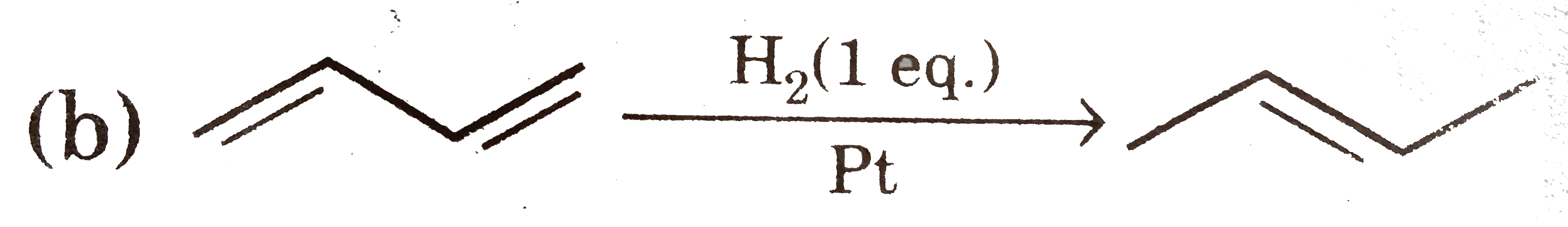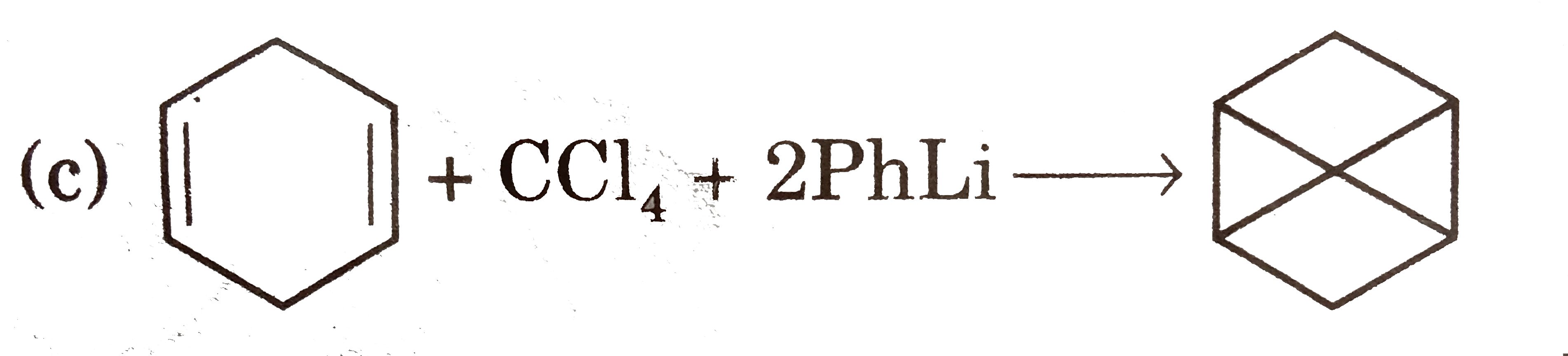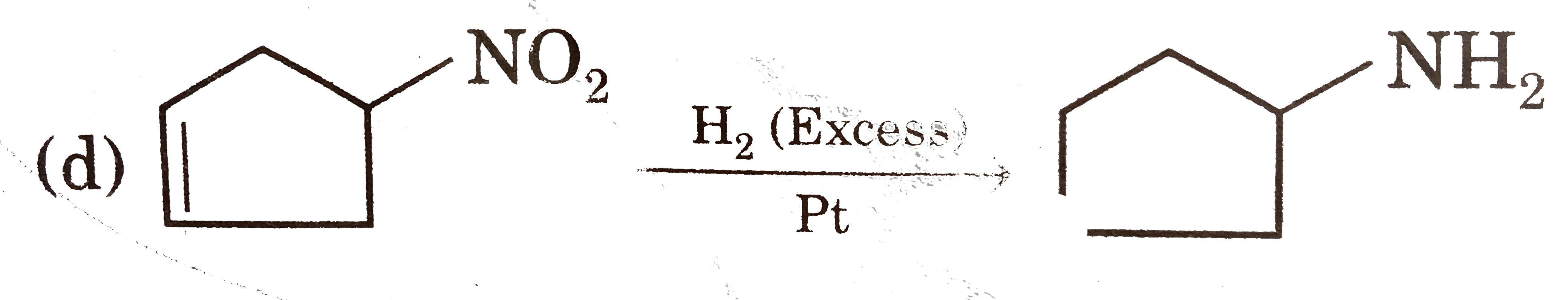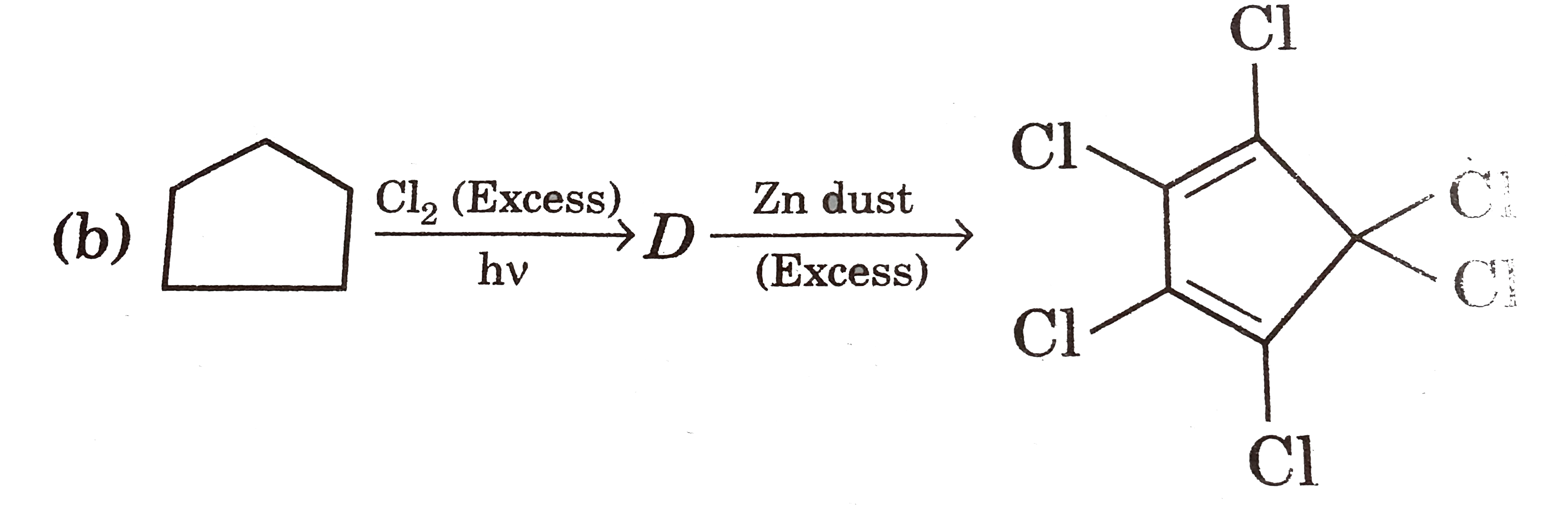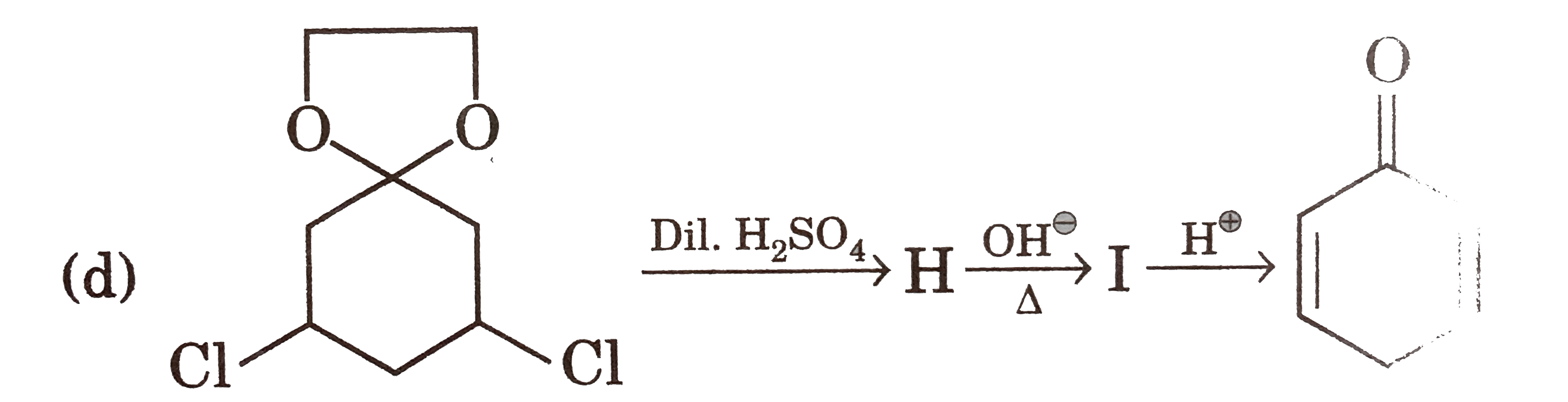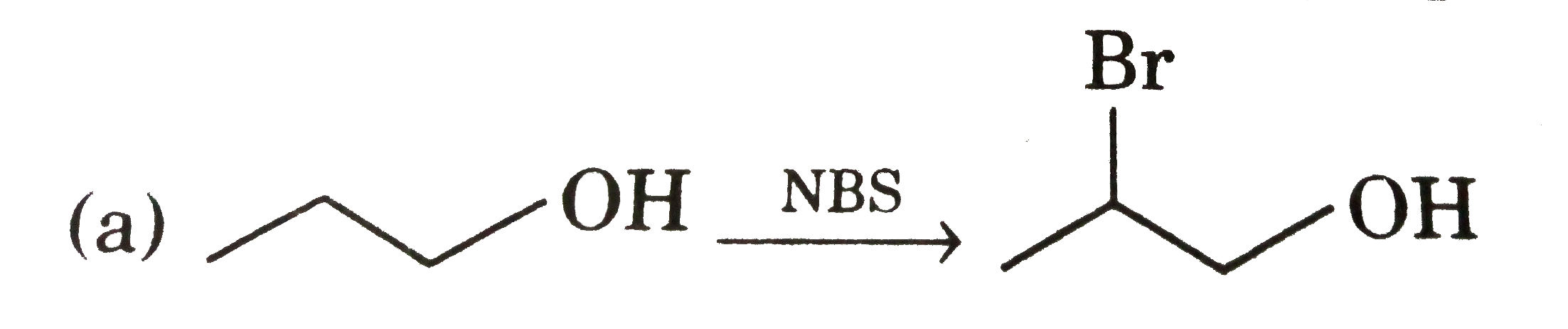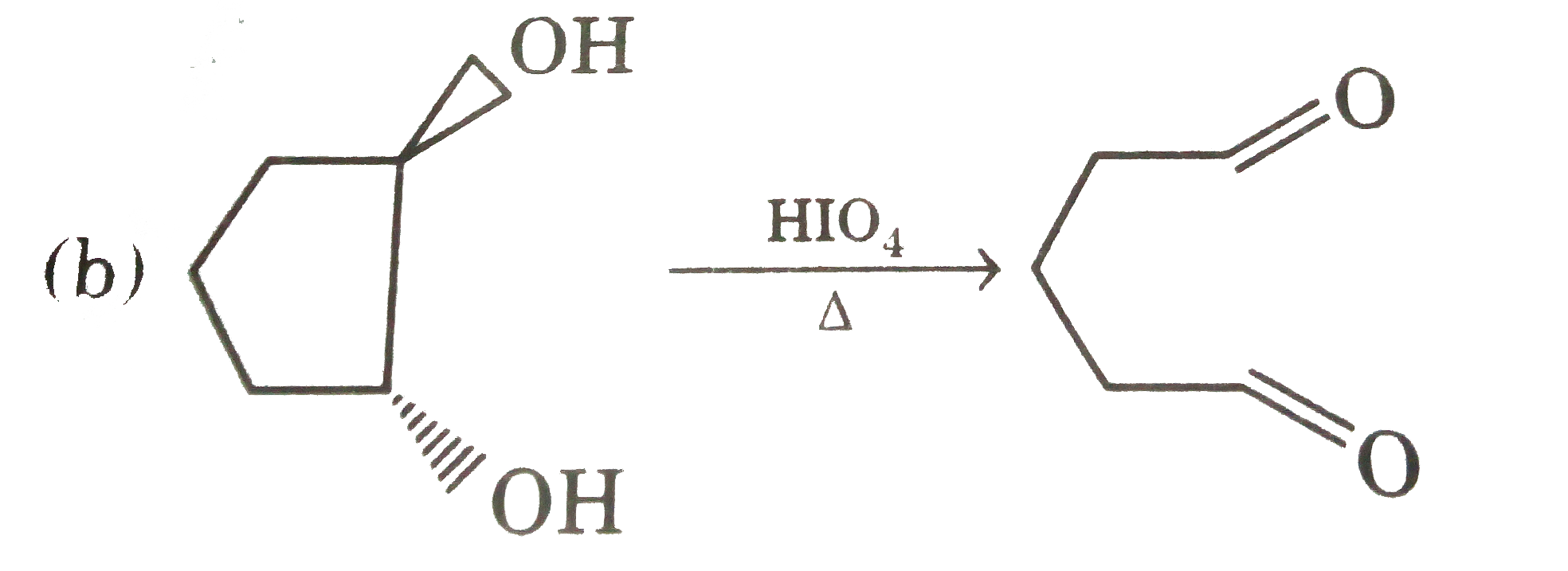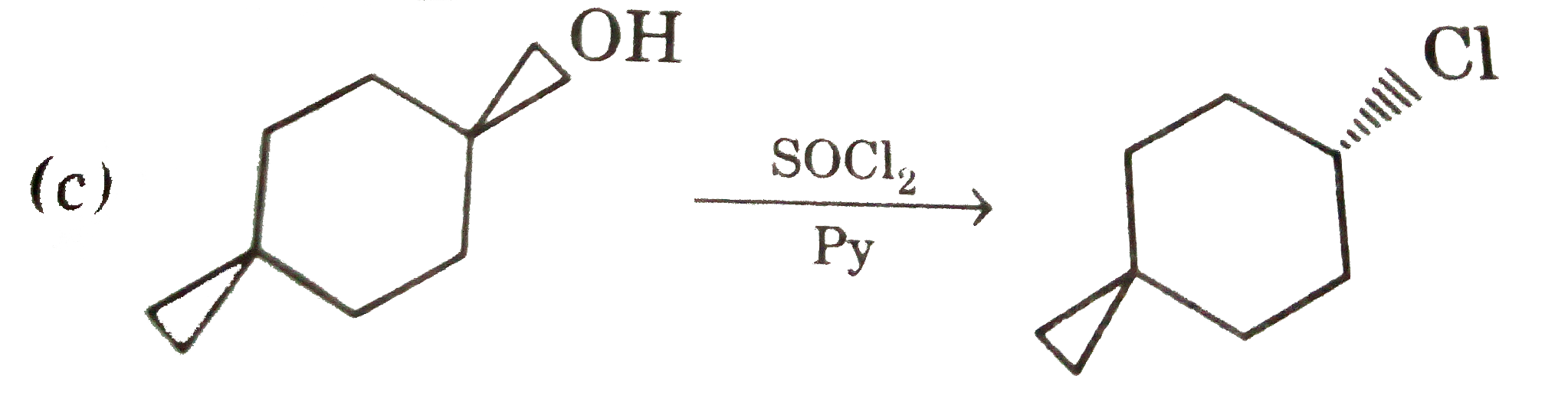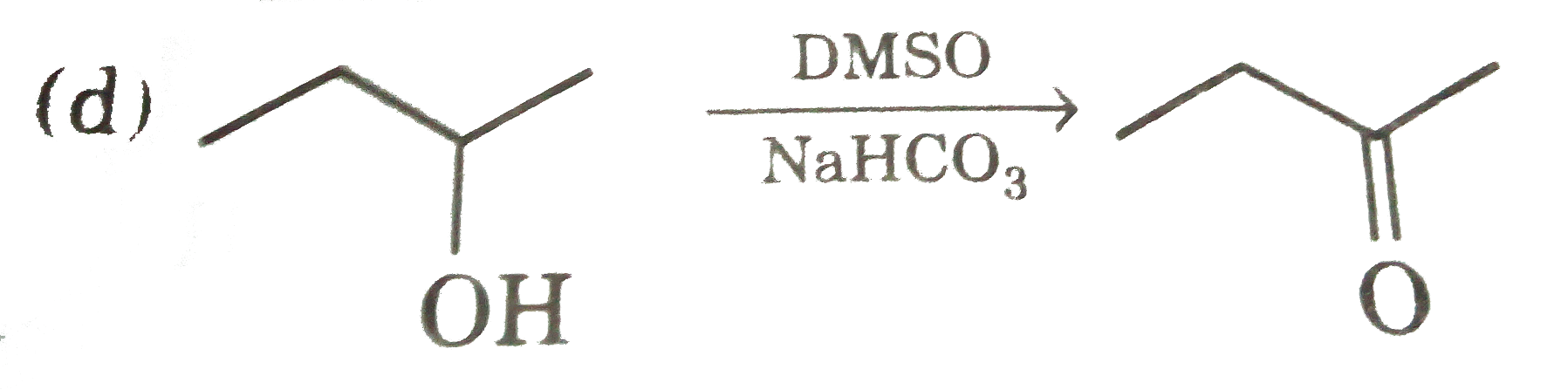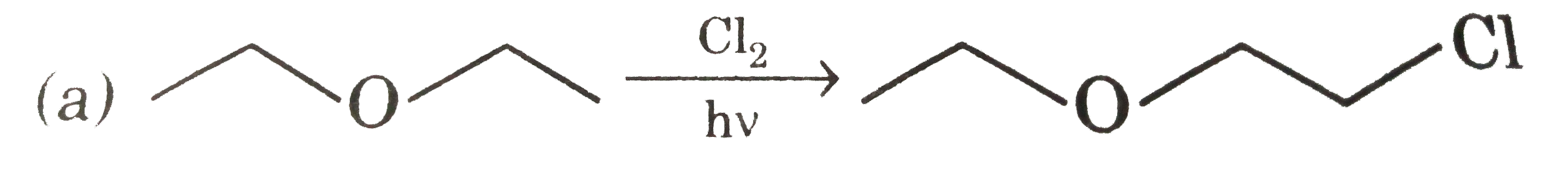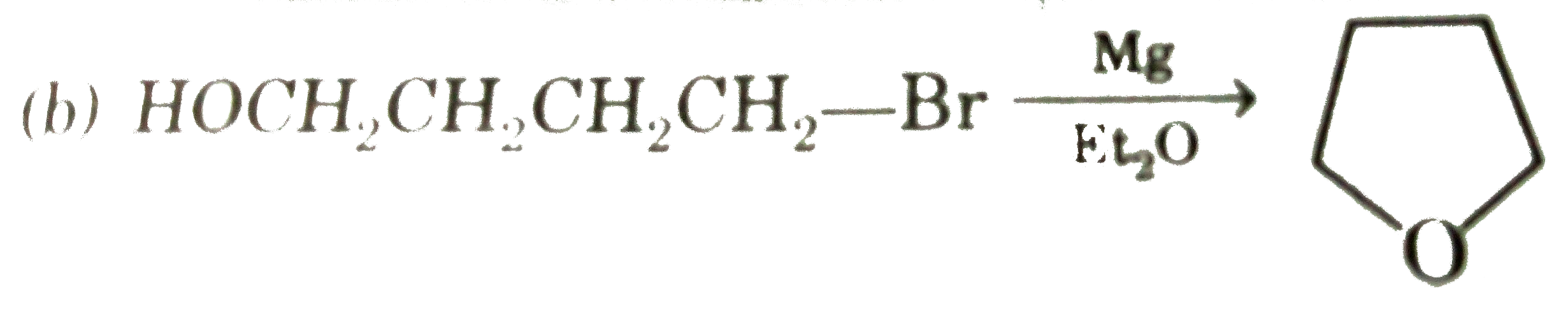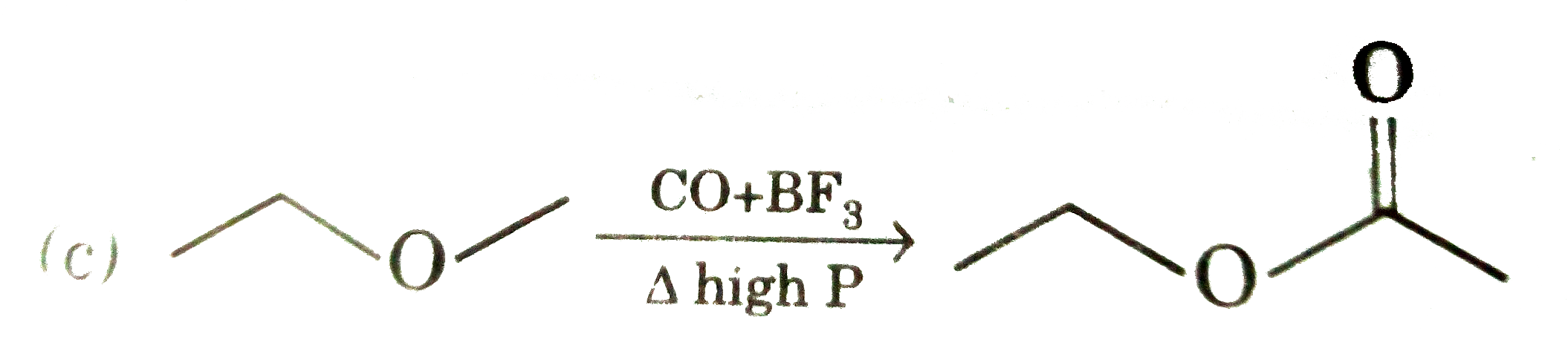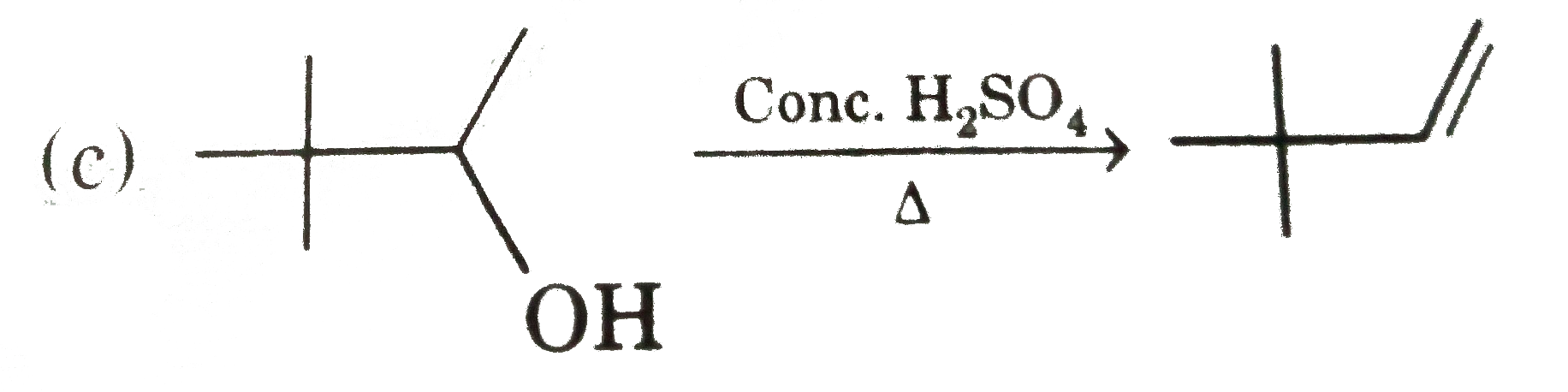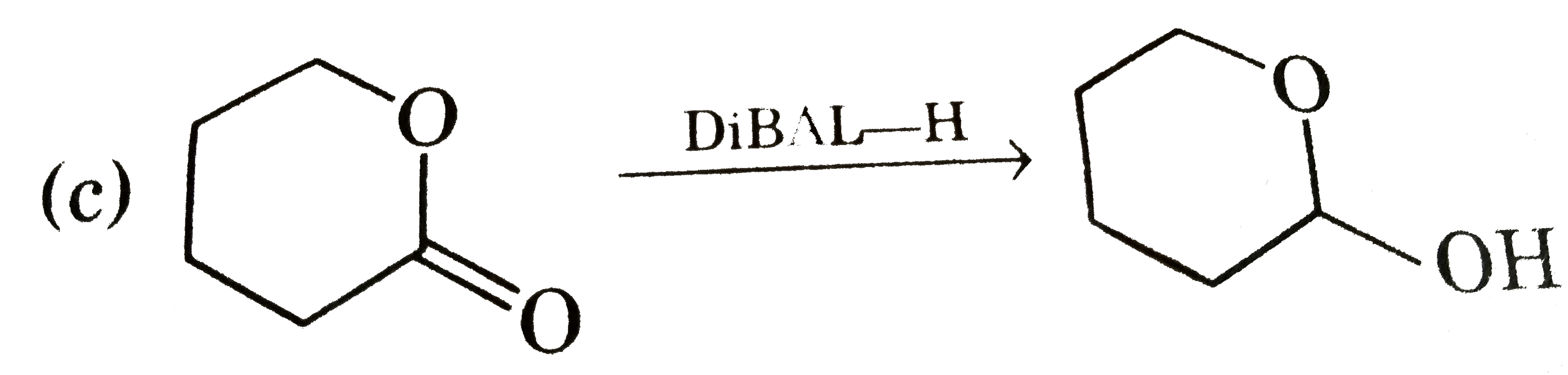Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select the best reagent to carry out the followingtransformations |
|
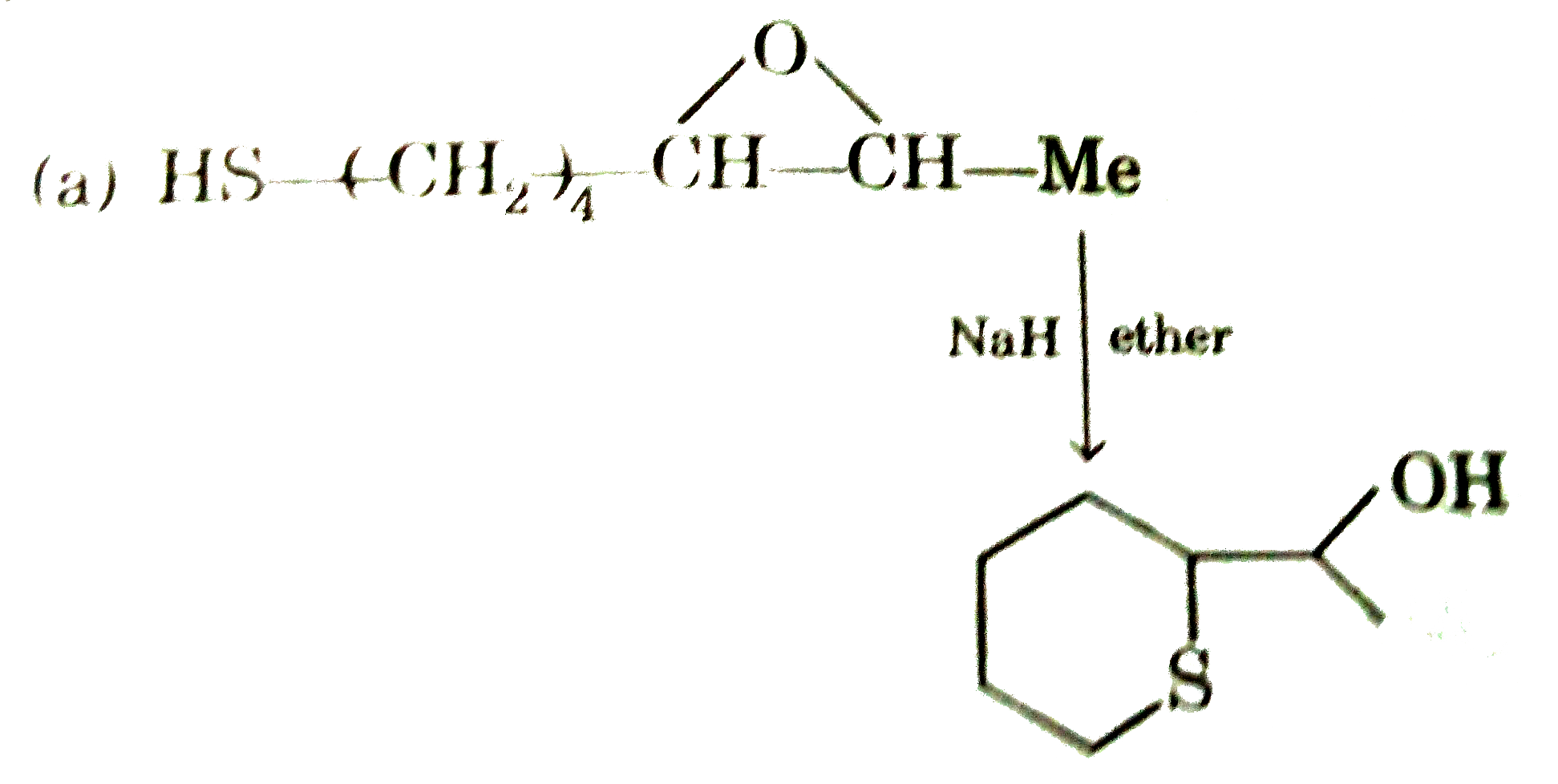
Answer» `{:("(I)","(II)","(III)"),("Acetylene",,),(NaNH_(2),H_(2)//Pd,OsO_(4)//NaHSO_(3)):}` 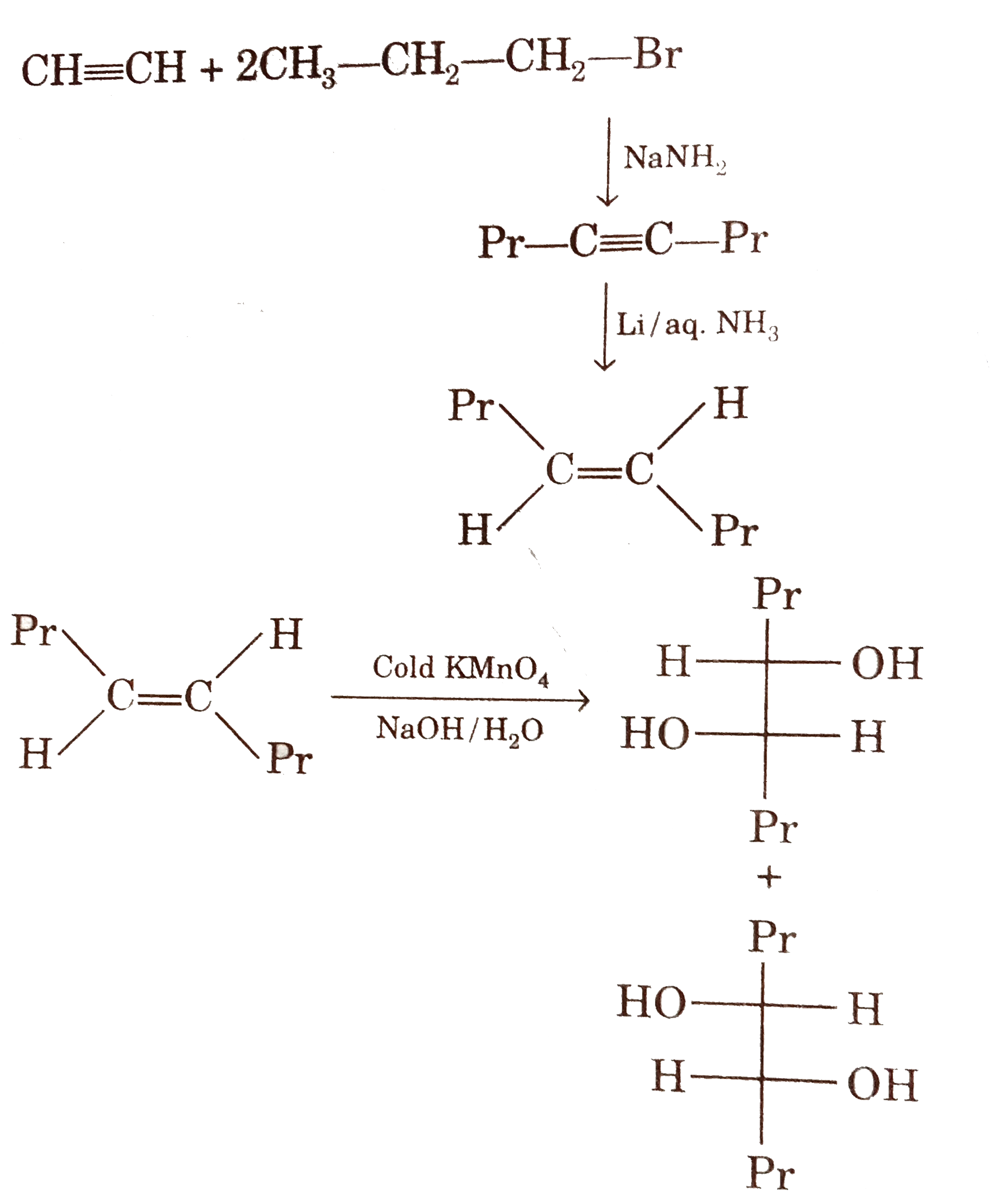
|
|
| 2. |
Select the appropriate combination which gives glycol [undersetunderset(OH)(|)CH_2-undersetunderset(OH)(|)CH_2] as a product. |
|
Answer» <P>(IV)(ii)(S) |
|
| 3. |
Select the amphoteric substance in the following: |
| Answer» Answer :D | |
| 4. |
Select the basic dye from the following |
|
Answer» METHYL red 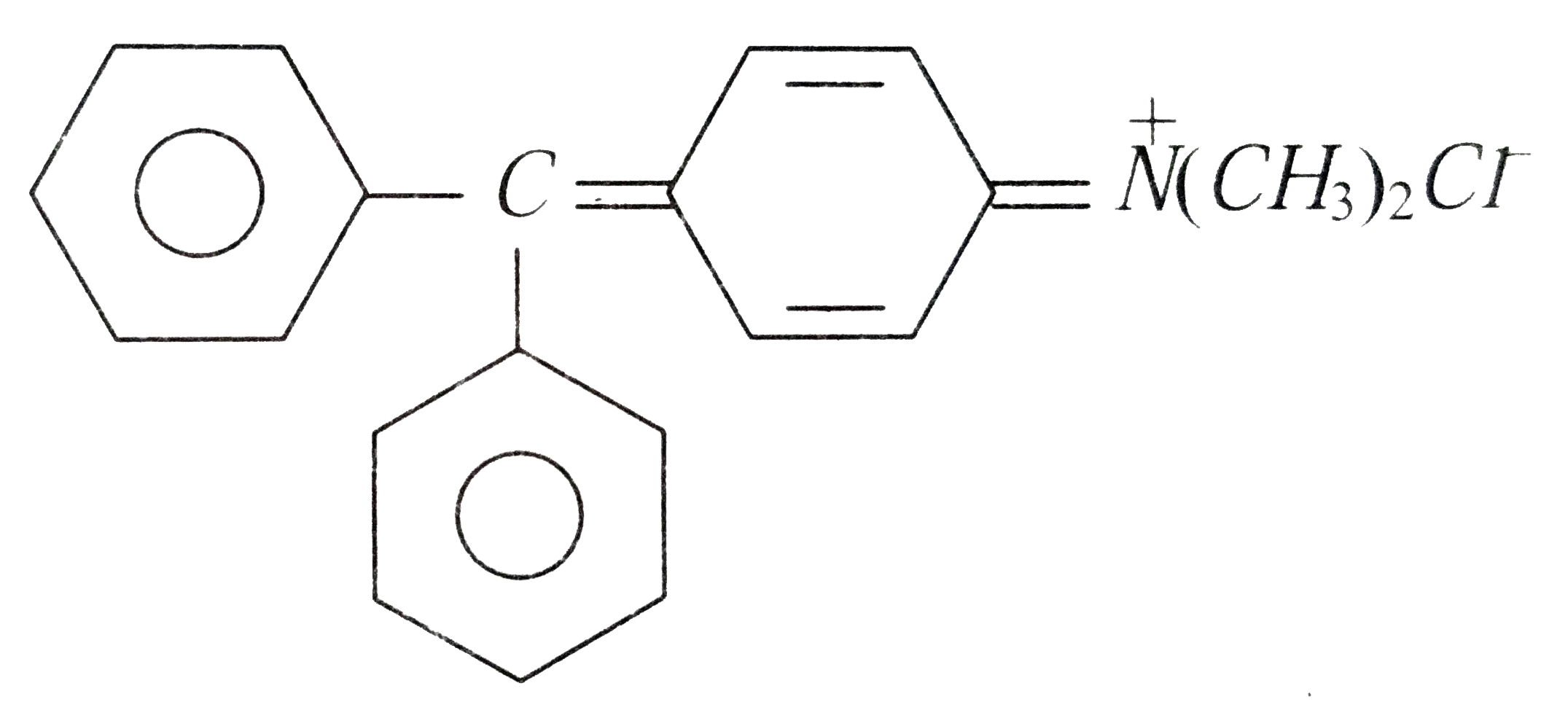
|
|
| 5. |
Select the ambident nucleophile from the following: (i) C bar(N) "" (ii)- CH_3 barO "" (iii) CNbarS (iv) overset(* * )(NH_3) "" (V ) overset(* * )(PH_3) "" (vi )NO_(2)^(-) |
| Answer» SOLUTION :`C BARN, NO_(2)^(-) , CNS^(-)` | |
| 6. |
Select that pairs in which bond angle of first member is higher than second one. |
|
Answer» `BrO_3^(-), CrO_3^(-)` `{:(,CIO_3^(-),107^(@),BrO_3^(-),104^@),((B),AsI_3,100.2^@ ,Sbl_3,90^@),(( C),SbBr_3,98.2^@,Sbl_3,99^@),((D),NH_3 , 106.6^@ ,NF_3,102.2^@):}` |
|
| 7. |
Select th correctstatements among the following |
|
Answer» METHANEIS presentin theatmoshere ofjupiter |
|
| 8. |
Select similarity between : [Co(NH_(3))_(4)(H_(2)O)Cl]Cl_(2)" and "[Co(NH_(3))_(4)Cl_(2)]Cl.H_(2)O |
|
Answer» Oxidation NUMBER and coordination number of metal |
|
| 9. |
Select schemes A, B, C out of : I acid catalysed hydration II HBO III oxymercuration-demercuration |
| Answer» Solution :N//A | |
| 10. |
Select right statement(s) about electrolysis : |
|
Answer» Electric CURRENT is used to DRIVE a non - SPONTANEOUS reaction b) `Delta G = +ve` for chemical process during electrolysis C) at over voltage ` O_2` gas evolved |
|
| 11. |
Select reagent which is/are used in laboratoryto differentiate 1^(@), 2^(@)" and " 3^(@)amines from each other : |
|
Answer» `NeNO_(2)HCl` `2^(@)underset(HCl)overset(NaNO_(2))rarrR-N-N=O`(Oily liquid) `3^(@)underset(HCl)overset(NaNO_(2))rarr`Salt (b) Hinsberg's test can distinguish `1^(@),2^(@)" and "3^(@)` amines. (c ) Carbhylamine test is given by only `1^(@)` amines. (d) Mustard oil REACTION is also given by `1^(@)` amines only. |
|
| 12. |
Select reaction(s) with incorrect major product: |
|
Answer»

|
|
| 13. |
Select reaction(s) incorrectly matched with major product: |
|
Answer»
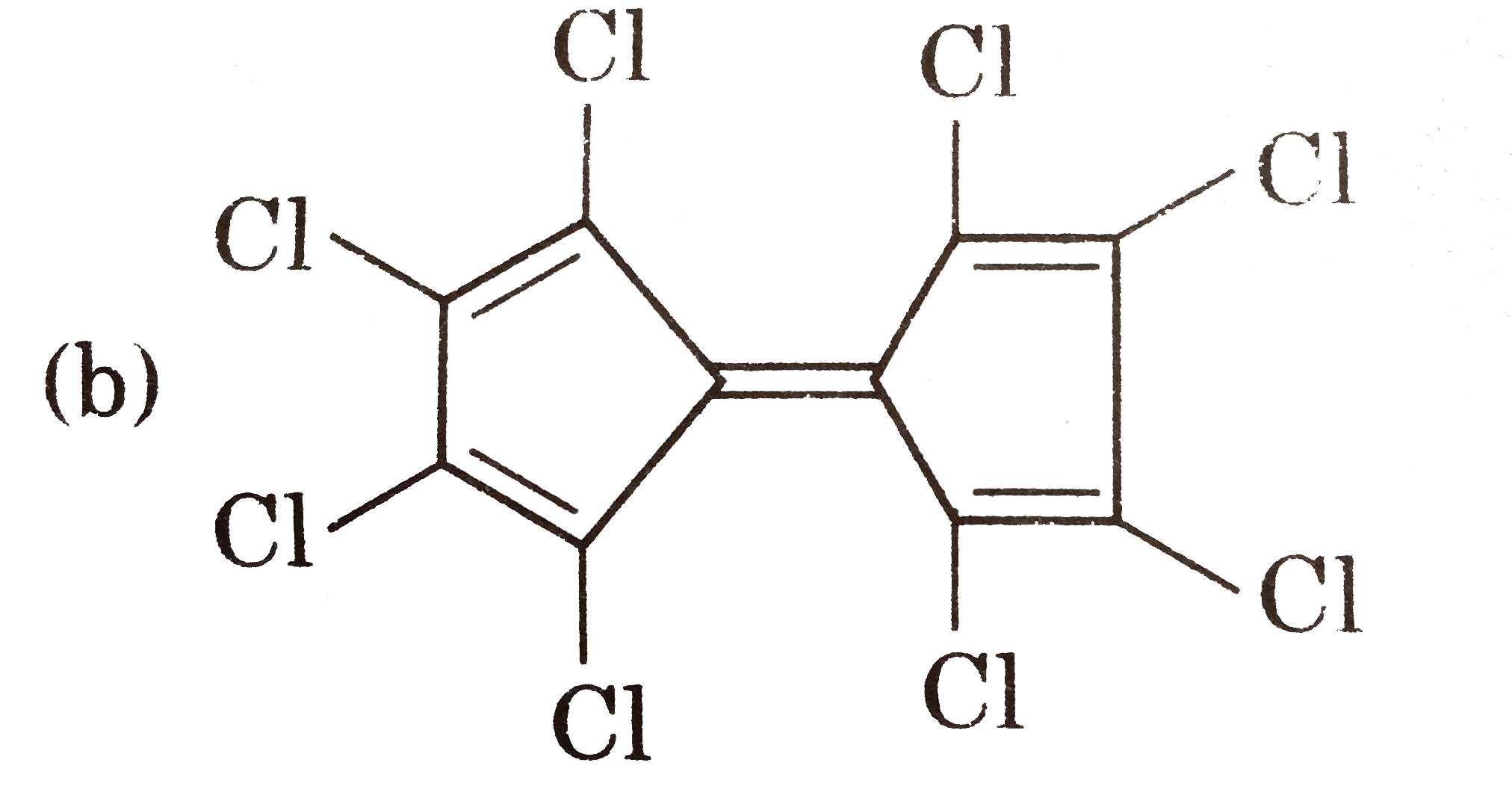 
|
|
| 14. |
Select reaction with incorrect major product : |
|
Answer»
(c ) `SOCl_(2)+Py" gives "S_(N)2` reaction. (d) OH can'tgive the reaction with out TsCl. |
|
| 15. |
Select reaction with correct major product. |
|
Answer»
|
|
| 17. |
Select polybasic Arrhenius acids form the following :H_(3)PO_(2),H_(3)PO_(3),H_(3)BO_(3),HCOOH,(COOH)_(2) b Write conjugate acids of SO_(4)^(2-),RNH_(2),NH_(2)^(-),C_(2)H_(5)OC_(2)H_(5),F^(-) c Write conjugate base of HNO_(2),OH^(-),H_(2)CO_(3),HClO_(4) d Write conjugate acid and conjugate base of following amphoteric species: HS^(-),NH_(3).C_(2)H_(5)OH,H_(2)O e Classify the following into Lewis base :H^(+),FeCl_(3),(CH_(3))_(3)N,F,CH_(2) |
| Answer» Solution :Polybasic Arrhenius acids PRODUCE more than one `H^(+)` ion per acid MOLECULE when dissolved in WATER .On removel of one `H^(+)` from an acid ,we get its CONJUGATE base,On addition of one `H^(+)` to a base we get its conjugate acid . | |
| 18. |
Select reaction with correct major product : |
|
Answer»
|
|
| 19. |
Select reaction with correct majoe product : |
|
Answer»
|
|
| 20. |
Select reaction correctly matched with major product : |
|
Answer» `M-overset(O)overset(||)C-OEt overset(DiBAL-H)toMeCH_(2)OH` 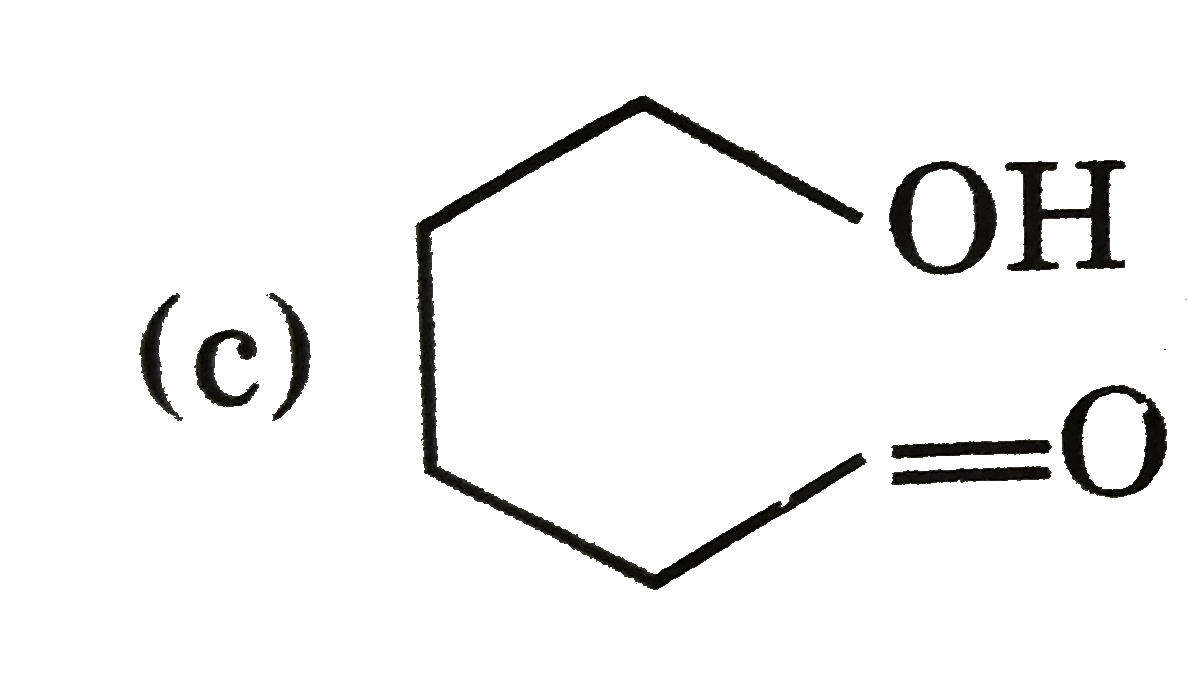 is formed which forms hemi-acetal. is formed which forms hemi-acetal. (d) `Cl-CH_(2)-overset(O)overset(||)C-OH` is formed by HVZ REACTION. |
|
| 21. |
Select option having fiberan polymer . |
|
Answer» Ny-6 |
|
| 22. |
Select option having correctly matched polymer : |
|
Answer» Terelyene`RARR`cloth |
|
| 23. |
Select of the ether among following that yields methanol as one of the products on reaction with cold hydroiodic acid |
|
Answer» 1-methoxy butane 
|
|
| 24. |
Select incorrectly matched (1)One has different nautre of oxide than other two -Zn,Al B (2) One is not extracted by carbon reduction but other two are -Sn, Ag. Zn(from their name ore) (3) One has different magnetic nautre than other two -O_(2), Hg[Co(SCN)_(4)],[NiCl_(4)]^(2-) (4) One has different hydridised central atom than other two -MgO_(4)^(-),CrO_(4)^(-),ClO_(4)^(-) |
|
Answer» One has DIFFERENT natuer of OXIDE than other two -Zn, Al B |
|
| 25. |
Select isoelectronic and isostructural pair: |
|
Answer» `CO_(2),CN_(2)^(2-)` |
|
| 26. |
Select incorrect statments about N_2O_4 |
|
Answer» It self ionises as `NO^(+),NO_(3)^(-)` |
|
| 27. |
Select incorrect statements. |
|
Answer» Methoxy ethane give IODOETHANE with one EQUIVALENT anhydrous HI |
|
| 28. |
Select incorrect statement(s). |
|
Answer» Phenolphthalein is SUITABLE indicator for the titraction of HCl(aq) with `NH_4OH (aq). ` |
|
| 29. |
Select incorrect statement (s) |
|
Answer» `NI^(2+)` compounds TENDS to be thermodynamically more STABLE thean `PT ^(2+)` |
|
| 30. |
Select incorrect statement for [Cr (en)_(3)]^(3+) and [Cr("gly")_(5)] complexes. |
|
Answer» Both show stereo isomerism |
|
| 31. |
Select incorrect statement about the complex : cis-[Rh(H)(CO)(Pme_(3))_(2)] |
|
Answer» It is a chiral complex  `mu NE 0`, complex is ACHIRAL, even in it's trans form. |
|
| 32. |
Select incorrect statement about complex [Cr(NO_(2))(NH_(3))_(5)][Zn(SCN)_(4)]: |
|
Answer» It shows co-ordination isomersm |
|
| 33. |
Select incorrect statement about lanthanides. |
|
Answer» All the LANTHANIDES are non-radioactive (EXCEPT `P_m` ) |
|
| 34. |
Select incorrect statement about followingreaction of ether synthesis : R-X + R' O Na to ROR' + NaX |
|
Answer» It follows `S_N` 2 mechanism |
|
| 35. |
Select incorrect statement about hydroboration-oxidation : |
|
Answer» Addition is against MARKOWNIKOFF rule |
|
| 36. |
Select incorrect statement about alanine. |
|
Answer» it is OPTICALLY active |
|
| 37. |
Which colour emission is radiatedby potassium when excited electrons come back to ground state :- |
|
Answer» CRIMSON red |
|
| 38. |
Half life of long -lived isotopes Francium is :- |
|
Answer» 21 minutes |
|
| 39. |
Select incorrect statement. |
|
Answer» SILICA present in bauxite is removed by addition of NaOH(aq) |
|
| 40. |
Select incorrect statement: |
|
Answer» STABILITY of peroxides and superoxides of alkali metals increase in SIZE of the METAL ion |
|
| 41. |
Select incorrect sequence of solubility product values among the following |
|
Answer» `CoS gt CuS` |
|
| 42. |
Select incorrect statement :- |
|
Answer» `O_3 and O_(2)^(2-)` both are diamagnetic |
|
| 43. |
Select incorrect statement : |
|
Answer» Higher the value of `k_H` (Henry’s LAW constant) at a GIVEN pressure, the lower is the SOLUBILITY of the gas in the liquid |
|
| 44. |
Select incorrect reduction process in respective extraction of an element : |
|
Answer» `8 Al + 3Mn_(3) O_(4) OVERSET(Delta)to 4Al_(2)O_(3) + 9Mn` |
|
| 45. |
Select incorrect reduction process : |
|
Answer» `2[Ag(CN)_(2)]^(-) + Zn rarr [Zn(CN)_(4)]^(2-) + 2Ag` |
|
| 46. |
Select incorrect reduction process |
|
Answer» `2[AG(CN)_(2)]^(-) + Zn rarr [Zn(CN)_(4)]^(2-) + 2AG` |
|
| 47. |
Select incorrect order |
|
Answer» `H_2OgtH_2SgtH_2SegtH_2T_2` (order of bond angle) |
|
| 48. |
Select incorrect opyions about polymerane : |
|
Answer» It is also called as styrofoam. |
|
| 50. |
Select incorrect option: |
|
Answer» `CH_(4) + "AIR" overset(Red hot)underset(Fe tube) (to) CH_(3)OH` |
|