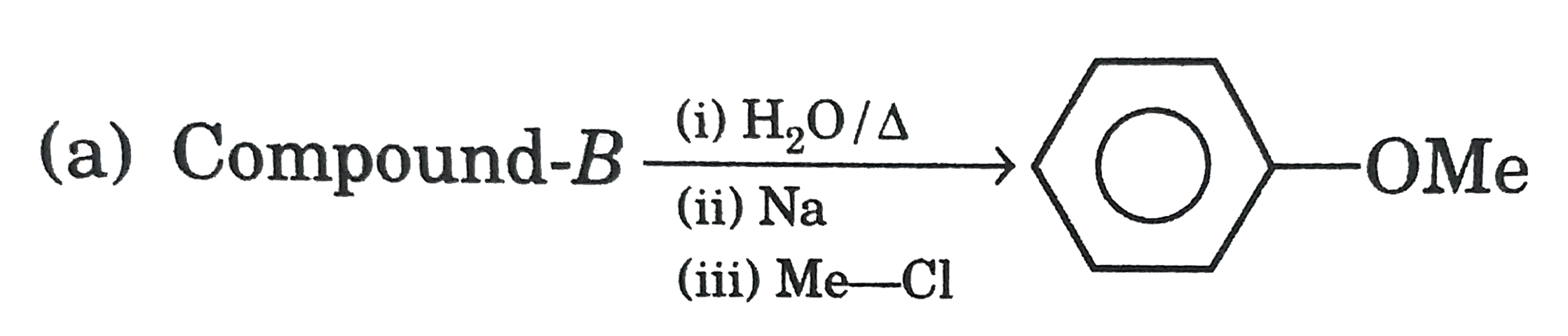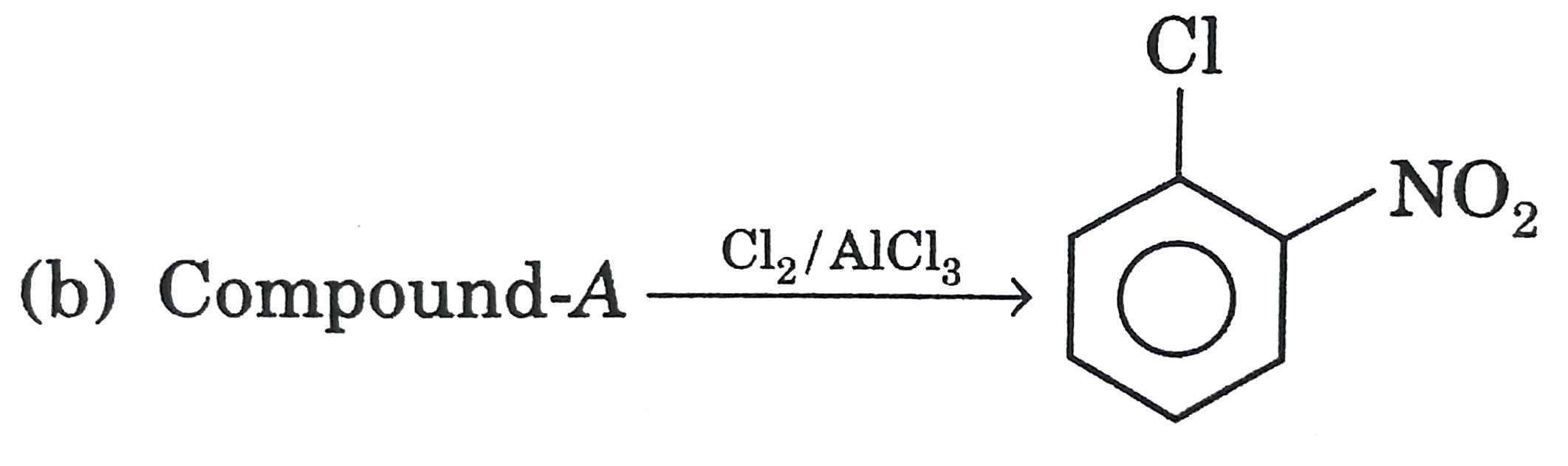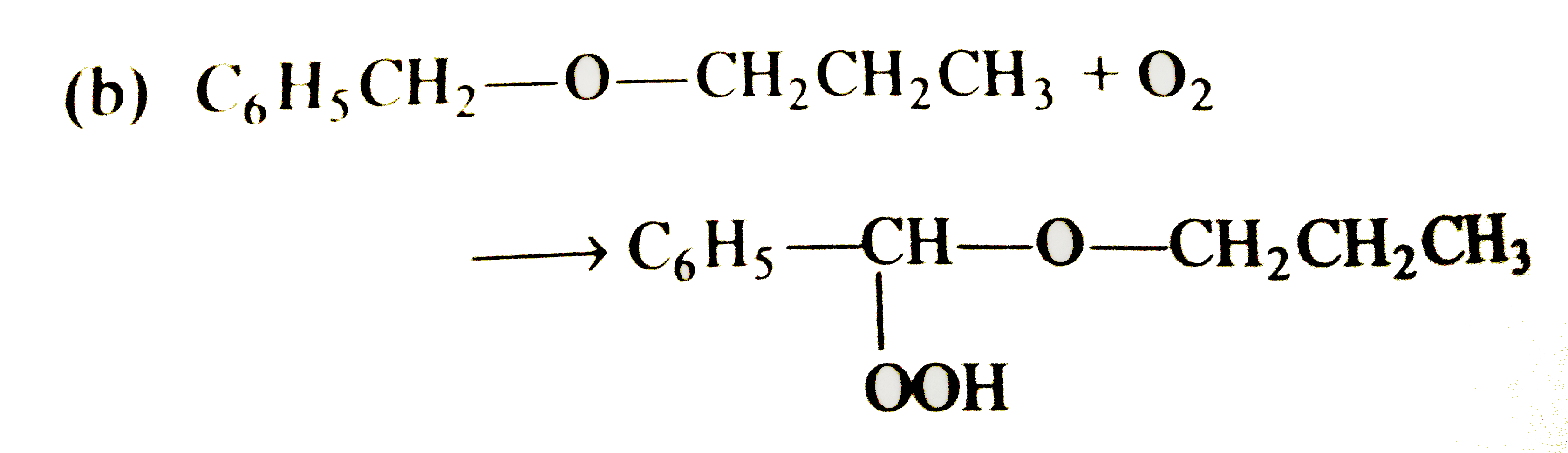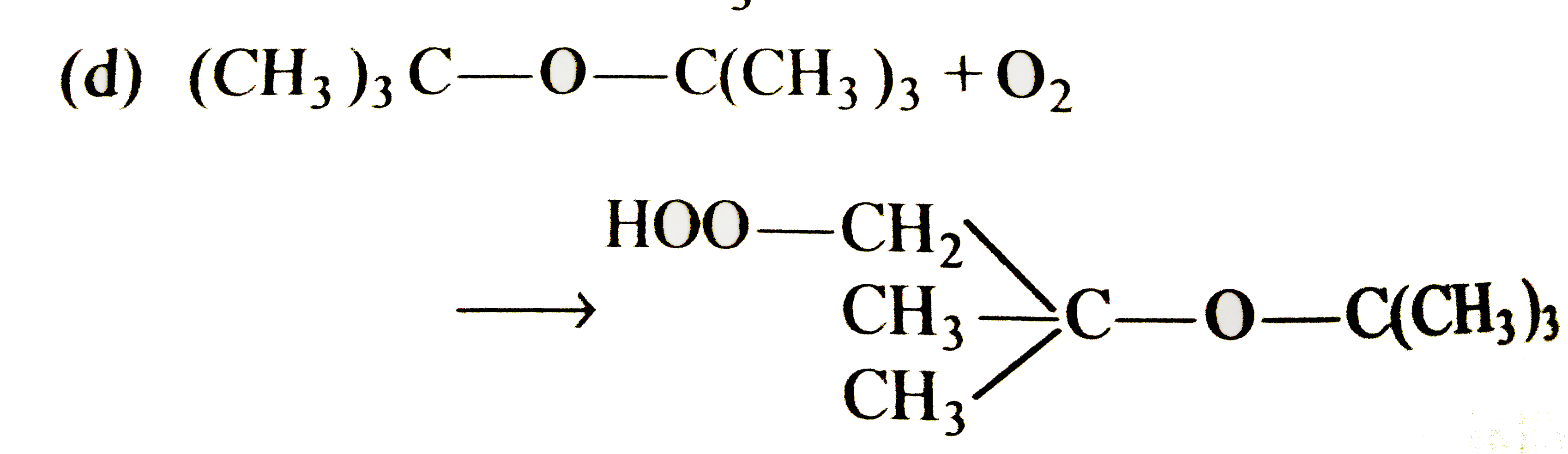Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select the correct statements: |
|
Answer» |
|
| 2. |
Select the correctstatement(s). |
|
Answer» `dG = V dp - SdT` is applicaleforall process involvingboth 'PV'and non `dG= dU +PdV +VdP -TdS-SdT` `=q + W + PdV + VdP -TdS - SdT` For a reversiblepath q= TdS If there isno non-PVwork w = - PdV `dG = VdP - SdT `(D) `G = H -TS + U+ PV -TS` at constant 'T' & 'P' `dG_("SYS") =dU + PdV -TdS_("sys")= q +w_(pv) +w_("non-PV") + PdV -TdS_("sys")` SINCE`W_(PV) = - PdV` andin theabsence of `w_(non-PV)` `dG_("sys") = q -TdS_("sys")` ` dG_("sys") = - T(dS_("sys") + dS_("surr"))` |
|
| 3. |
Select the correct statements? |
|
Answer» `Cl_(2)O` and `ClO_(2)` are used as bleaching agents and germicides |
|
| 4. |
Select the correct statement(s). |
|
Answer» `[Co(EDTA)]^(-)` has two optical ISOMERS. |
|
| 5. |
Select the correct statements. |
|
Answer» The molecularity of an elementary reaction indicates how many reactant species TAKE part in the step. |
|
| 6. |
Select the correct statements. (1) Ferric bromide is obtained when chlorine gas is bubble through a solution of ferrous bromide (2) Sodium sulphate is obtained when SO_(2)(g) , H_(2)O(g) and air are passed over heated sodium chlorine (3) H_(2)S gas is liberated when aluminiumsuphide becomes damp. |
|
Answer» statements 1 and 2 are CORRECT |
|
| 8. |
Select the correct statement whenlower the position of a metal line in the Ellingham diagram |
|
Answer» The greater is the stability of it's oxide for example the line for AL (oxidation of Al) is FOUND to be below that for Fe (formation of `Fe_(2)O_(3))` |
|
| 9. |
Select the correct statement (s) - (a) The C.N. of cation occupying a tetrahedral hole is 4. (b) The C.N. of cation occupying a octahedral hole is 6. (c) In schottky defects, density of the lattice decreases. (A) a, b(B) b, c (C) a, b, c (D) a, c |
|
Answer» Solution : (C) Since tetrahedral holes are surrounded by 4 NEAREST neighbours. So, the C.N. of cation occupying tetrahedral hole is 4. Since OCTAHEDRAL hole is surrounded by six nearest neighbours. So, C.N. of cation occupying octahedral is 6. In SCHOTTKY a pair of anionand cation leaves the lattice. So, density of lattice DECREASES. |
|
| 10. |
Select the correct statement (s): |
|
Answer» Lower value of quantum number l indicates that there is a higher probability of finding the 3selectron close to the nucleus than those of 3p and 3d electrons Energy order `rarr3s lt 3p lt 3d lt 3f,"3s is CLOSER to nucleus"therefore"is most stable."` Whereas 3f is farthest from nucleus and hence has maximum energy and is least stable. |
|
| 11. |
Select the correct statement (s). |
|
Answer» The value of rate constatnt cannot exced the value of Arrhenius factor. |
|
| 12. |
Select the correct statement (s) : |
|
Answer» `alpha`- PARTICLES are simply dipositive helium nucleus |
|
| 13. |
Select the correct statement regarding [Pt(CH_(3)-CH(NH_(2))COO)Cl_(2)]^(-) |
|
Answer» It EXHIBITS both GEOMETRICAL and OPTICAL isomerism 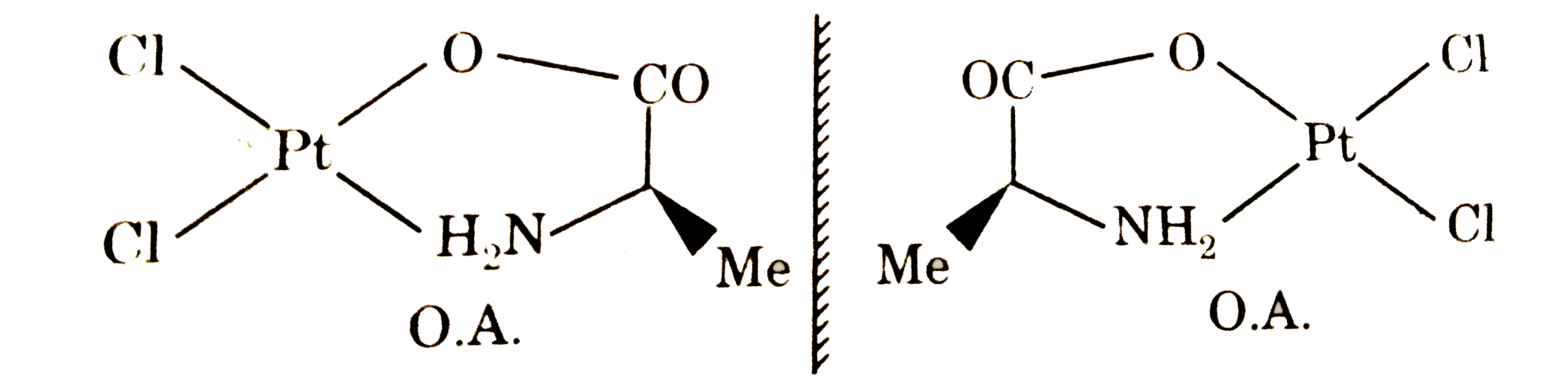
|
|
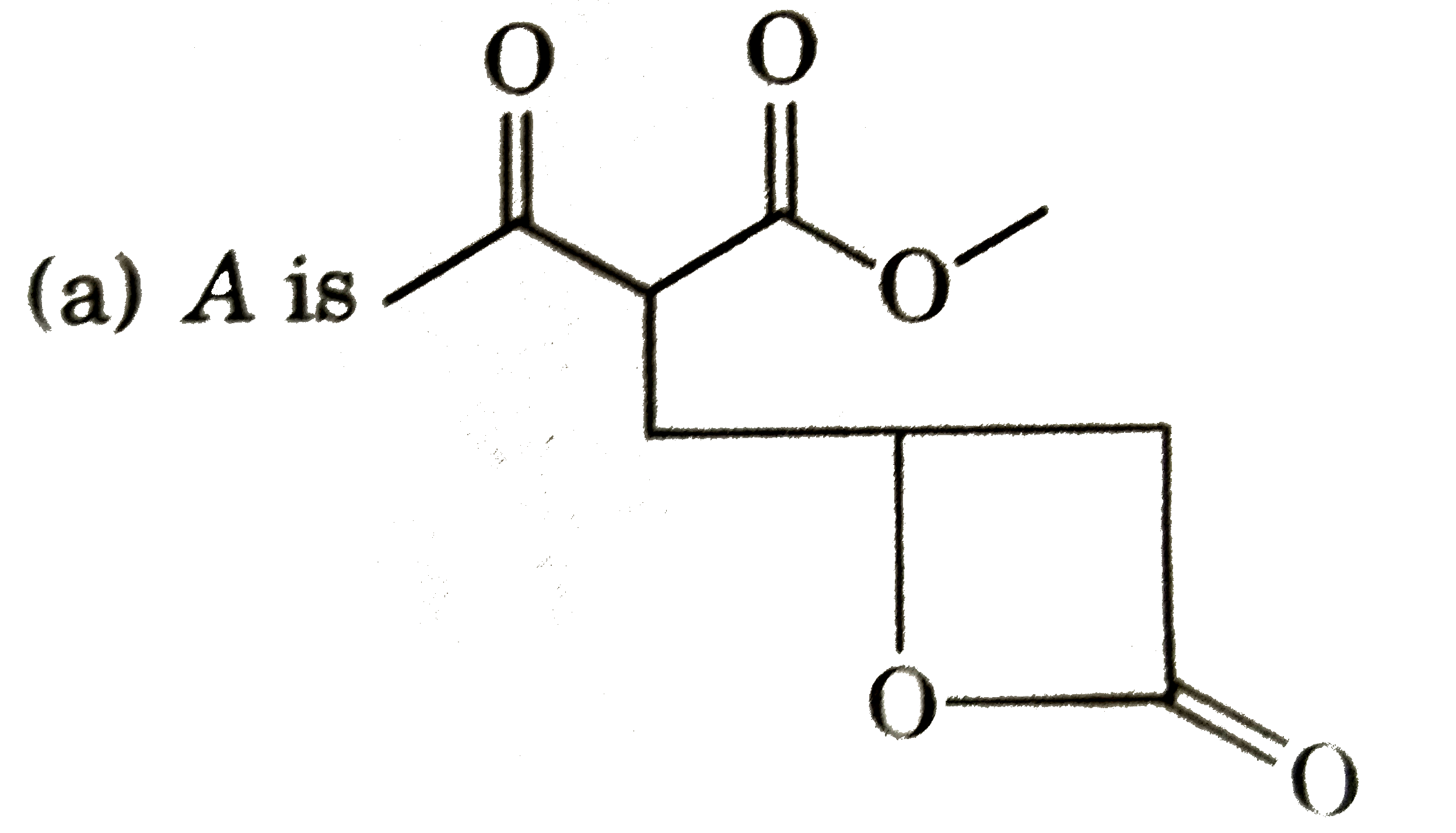
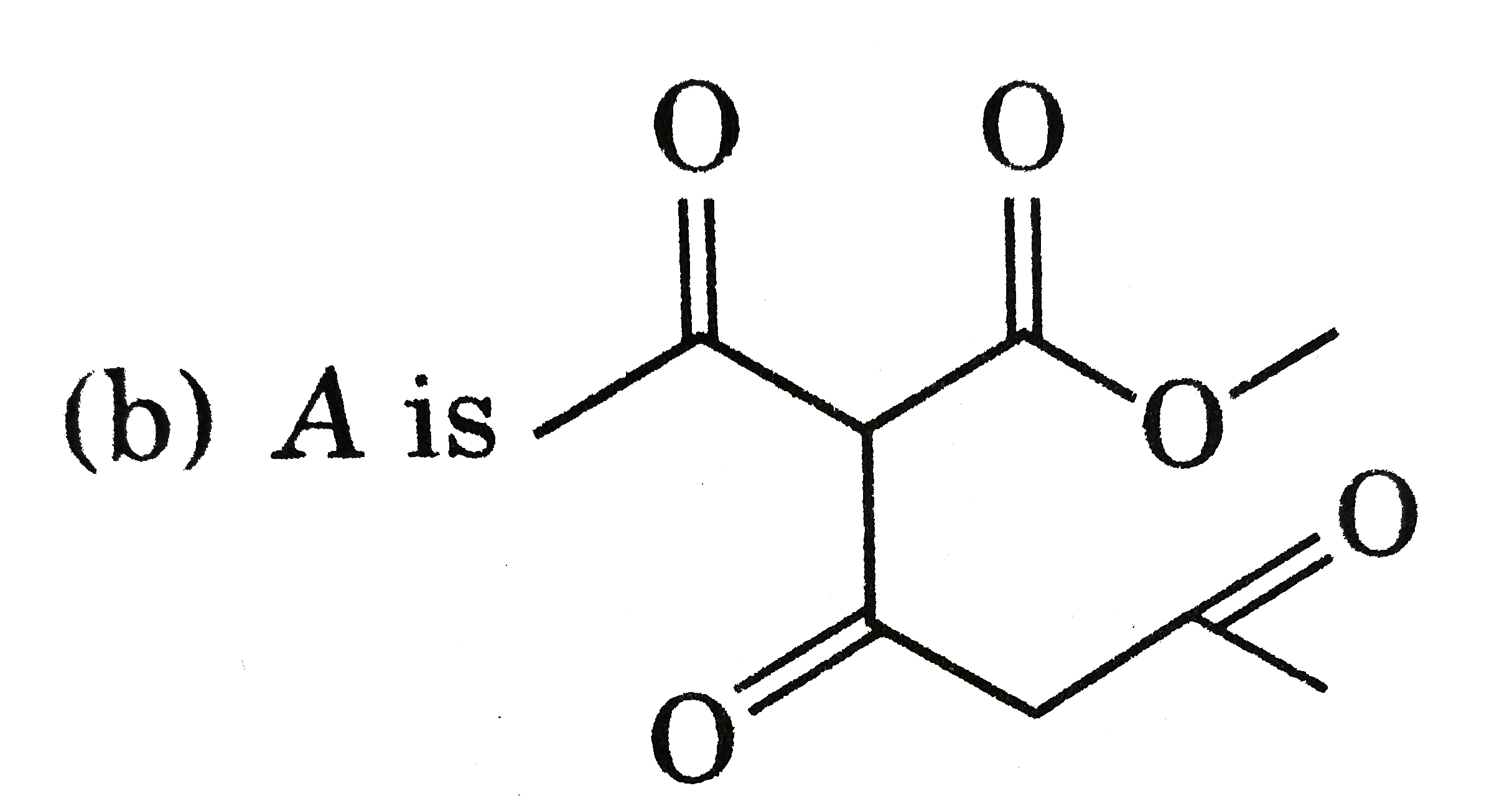
| 14. |
Select the correct statement regarding below figure if in this figure C_(1) has sp^(2) (S + P_(y) + P_(z)) hybridisation and C_(4) has sp (SP_(x) hybridisation. H_(2)underset(1)(C )= underset(2)(C ) = underset(3)(C ) = underset(4)(C ) = underset(5) F_(2) |
|
Answer» Over all molecule is planar 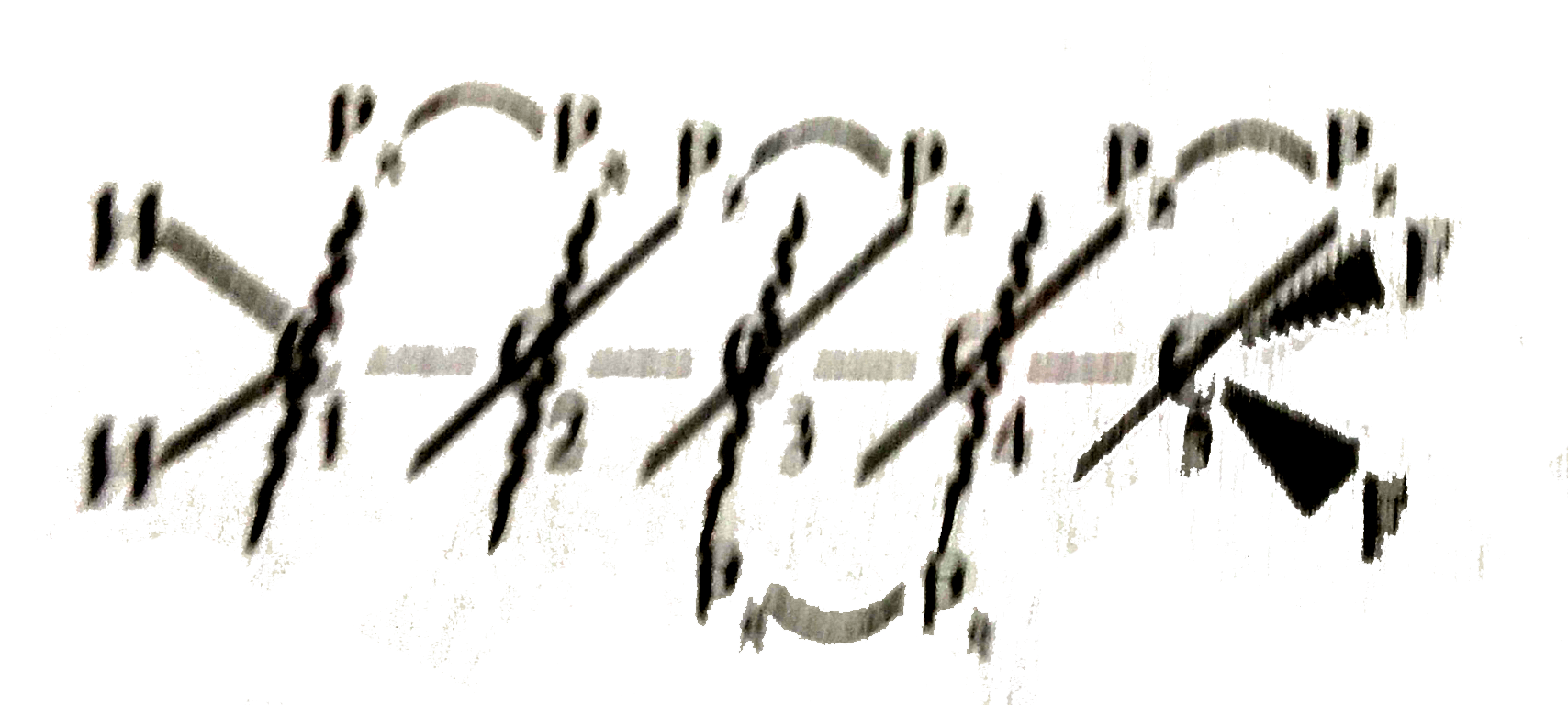
|
|
| 15. |
Select the correct statement regarding salt solution of weak acid and strong base. |
|
Answer» Solution will be neutral |
|
| 16. |
Select the correct statement regarding formation of micelle. |
|
Answer» Association formed during micelle formation, the polar head REMAINS outside TOWARDS surface. |
|
| 17. |
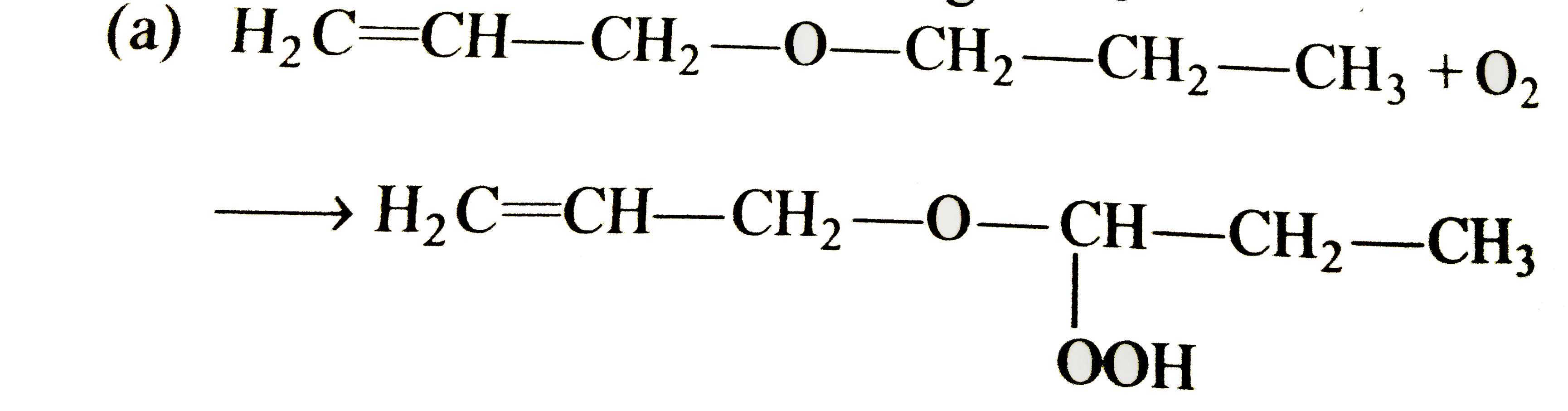
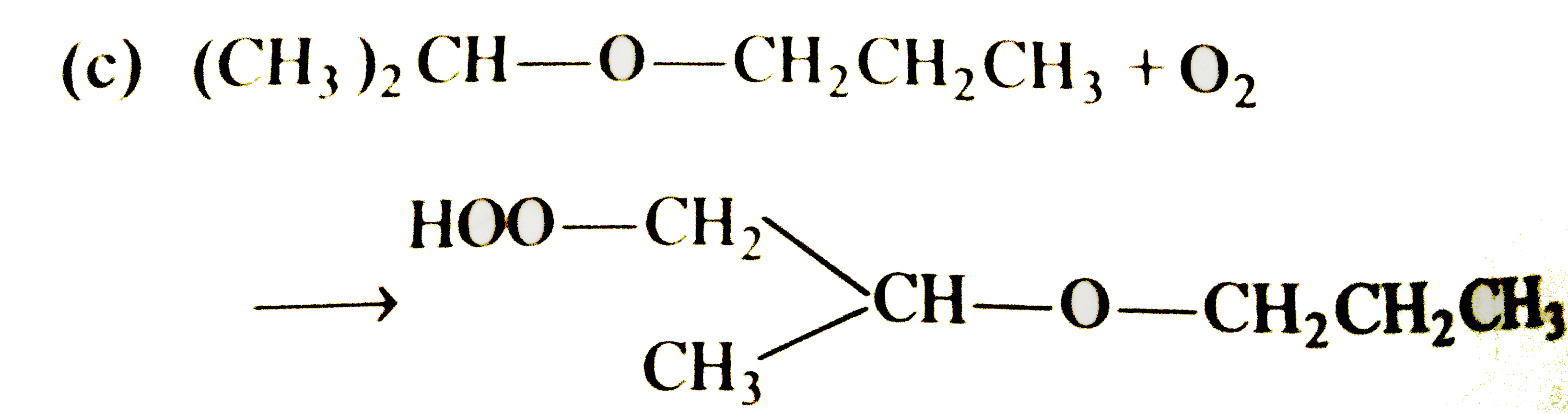
Select the correct statement/reaction among following |
|
Answer»
|
|
| 18. |
Select the correct statement regarding above solution. |
|
Answer» Anion will UNDERGO HYDROLYSIS producing `OH^(-)` & solution is expected to be basic. |
|
| 19. |
Select the correct statement out of the following : |
|
Answer» `BETA`-D(+) glucopyranose is more STABLE than `alpha`-D(+) glucopyranose |
|
| 20. |
Select the correct statement in the case of reduction of FeO at a temperature indicated by the point D- |
|
Answer» `DeltaG` of the overall reduction reaction INVOLVING CO is 0. |
|
| 21. |
Select the correct statement from the following? |
|
Answer» `[Sc(H_(2)O)_(6)]^(3+) and [Ti(H_(2)O)_(6)]^(3+)` both are colourless<BR>`[Co(NH_(3))_(4)Br_(2)]CL`show IONIZATION isomers and geometrical isomers |
|
| 22. |
Select the correct statement from the following : |
|
Answer» `[Sc(H_(2)O)_(6)]^(3+)" and "[Ti(H_(2)O)_(6)]^(3+)` both are colourless. |
|
| 23. |
Select the correct statement from the fllowing |
|
Answer» EQULIBRIUM constant changes with ADDITION of catalyst |
|
| 24. |
Select the correct statement for Zeise's salt : |
|
Answer» The C=C bond is present in same plane to the `PtCl_(3)` group |
|
| 25. |
Select the correct statement for the complexes [Cr(H_(2)O)_(6)]Cl_(3) and [Cr(H_(2)O)_(5)Cl]Cl_(2). H_(2)O |
|
Answer» There can be DIFFERENTIATED by amount. |
|
| 26. |
Select the correct statement for clous seeding |
|
Answer» It promotes rainfall |
|
| 27. |
Select the correctstatement about thefollowingreaction. [(CH_(3))_(3)NCH_(2) - CH_(3)]^(+)OH^(-) to (CH_(3))_(3)N + CH_(2) = CH_(2) + H_(2)O . |
|
Answer» THISIS calledHofmannelimination REACTION |
|
| 28. |
Select the correct statementabout the following reaction CH_3-undersetunderset(CH_3)(|)CH-CH_2-CH=CH_2overset(H_2//Ni)to(Y)overset(Cl_2//hv)toMonochloro products (X) |
|
Answer» The reactant (X) has four positional ISOMERS ( includeing 'X' itself) Positional Isomers of X : (4) `CH_3-undersetunderset(CH_3)(|)CH-CH_2-CH=CH_2,CH_3-undersetunderset(CH_3)(|)CH-CH=CH=CH_3` `CH_3-undersetunderset(CH_3)(|)C=CH-CH_2-CH_3,CH_2-undersetunderset(CH_3)(|)C-CH_2-CH_2-CH_3` Positional isomers of Y : (2) `CH_3-undersetunderset(CH_3)(|)CH-CH_2-CH_2-CH_3,CH_3-CH_2-undersetunderset(CH_3)(|)CH-CH_2-CH_3` Monochloro products : (5) `CH_3-undersetunderset(CH_3)(|)CH-CH_2-CH_2-oversetoverset(Cl)(|)CH_2,CH_3-undersetunderset(CH_3)(|)CH-CH_2-oversetoverset(Cl)(|)CH-CH_3` `CH_3-undersetunderset(CH_3)(|)CH-oversetoverset(Cl)(|)(CH)-CH_2-CH_3,CH_3-undersetunderset(CH_3)(|)oversetoverset(Cl)(|)C-CH_2-CH_2-CH_3,oversetoverset(Cl)(|)CH_2-undersetunderset(CH_3)(|)CH-CH_2-CH_2-CH_3` Alkyne : (2) `CH_3-undersetunderset(CH_3)(|)CH-CH_2-C=CH,CH_3-undersetunderset(CH_3)(|)CH-C-=C-CH_3` |
|
| 29. |
Selectthe correct statement about the complex [Co(NH_(3))SO_(4)]Br. |
|
Answer» Its ionisation isomer is `[CO(NH_(3))_(5)BR]SO_(4)` |
|
| 30. |
Select the correct statement about Na_(2)S_(2)O_(3).5H_(2)O? |
|
Answer» It is also called as hypo `2Na_(2)S_(2)O_(3)+I_(2) to Na_(2)S_(4)O_(6)+2NaI` |
|
| 31. |
Select the correct statement about given square planar complex. |
|
Answer» It has no GEOMETRICAL isomer |
|
| 32. |
Select the correct statement about elements of group 15^(th) |
|
Answer» The order of stability of oxidation state for +3 is `Bi^(3+) gt Sb^(3+) gt As^(3+)` and for `+5` is `Bi^(5+)lt Sb^(5+) lt As^(5+)` (B)`3HNO_2 to HNO_3+H_2O+2NO` (C )This is due to the PRESENCE of completely FILLED d and / or f orbitals in heavier elements. Covalent radius/ pm , N=70, P=110, As=120, Sb=140, Bi=150 |
|
| 33. |
Select the correct statement about A and B . |
|
Answer» B is OPTICALLY active but `A` does not 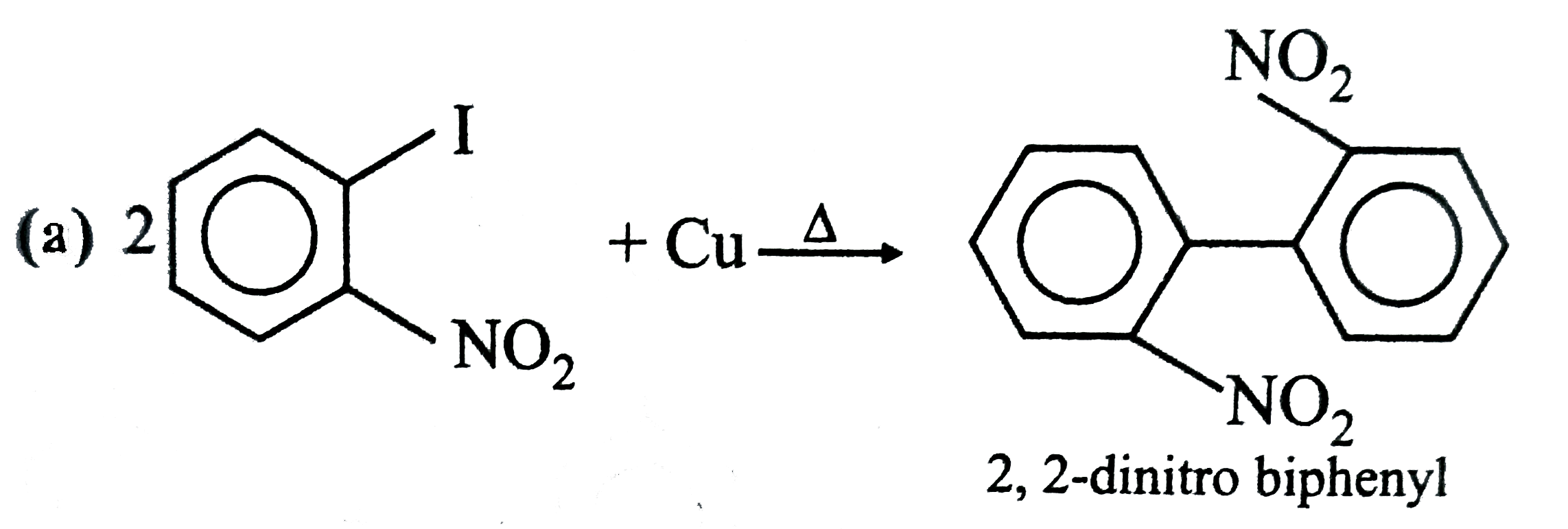 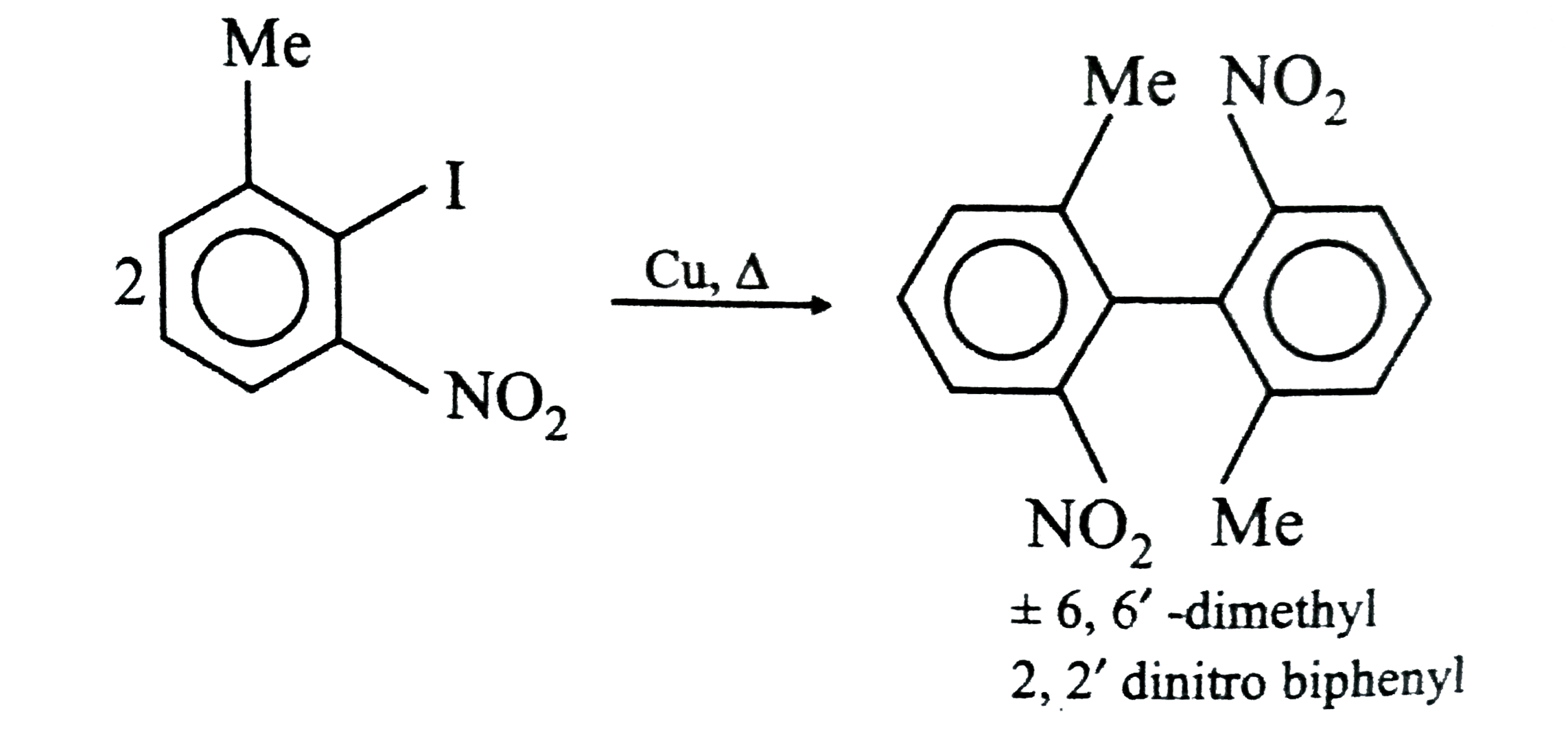 . .
|
|
| 34. |
Select the correct statement about [CoF_(6)]^(3-) : |
|
Answer» It is labile COMPLEX |
|
| 35. |
Write equations for complete process of prepration of Na_2CO_3 :- |
| Answer» | |
| 36. |
Select the correct statement: |
|
Answer» Be and Al SHOW DIAGONAL relationship |
|
| 37. |
Select the correct statement - |
|
Answer» GEOMETRICAL isomer may differ in DIPOLE moment and visible/UV spectra. |
|
| 38. |
Select the correct statement : |
|
Answer» POTASSIUM ferrocyanide and potassium ferricyanide can be differentiated by measuring the solid STATE magnetic moment. |
|
| 39. |
Select the correct statement . |
|
Answer» Complex ion `[MoCl_(6)]^(3-)` is paramagnetic `(b) d^(6), t_(2g)^(2,2,2)eg^(0,0)` |
|
| 40. |
Select the correct set of answer: {:("ColumnI","Column - II"),("(A) Tyrosine "," (P) Essential amino acid"),("(B) Serin "," (Q) Ceric Ammonium Nitrate"),("(C) Tryptophane"," (R) Neutral "FeCl_(3)),("(D) Proline "," (S) Carbaylamine Test - Negative "):} Select the correct set of answer : |
|
Answer» <P> A - R, B - Q, C - P, D - S |
|
| 42. |
Select the correct relation of interaction energy of dipole-dipole force in stationary polar molecule with internuclear distance ® |
|
Answer» INTERACTION ENERGY `PROP 1/r` |
|
| 43. |
Select the correct reduction processes. |
|
Answer» `ZnO + H_(2) to Zn + H_(2)O` |
|
| 44. |
Select thecorrect plot of radial probability function (4pir^(2)R^(2)) for 2s - orbital. |
|
Answer»
|
|
| 45. |
Select the correct priority for citation as principal group: |
|
Answer» `-OVERSET(O)overset(||)(C)-Ohgt-overset(O)overset(||)(C)-gt-overset(O)overset(||)(C)-HGT-Ohgtgt-SH` |
|
| 46. |
Select thecorrect order sequence(s). |
|
Answer» `C-C` bond length : (c) B.P. : `BiH_(3)gtSbH_(3)gtNH_(3)gtAsH_(3)gtPH_(3)` |
|
| 47. |
Select the correct order(s). |
|
Answer» `HOCl gt HOBR gt HOI`-Acid strength |
|
| 48. |
Select the correct order of solubility of AgCN(s) at 25^(@)C. Given that at this temperature its solubility is S_(1)(in presence of 1 M AgNO_(3)) , S_(2)(in presence of 1 M NH_(3) (aq)), S_(3)(in presence of pure water) and S_(4)(in presence of 1.5 M KCN). |
|
Answer» `S_(1) lt S_(2) lt S_(3) lt S_(4)` |
|
| 49. |
Select the correct order of sweetness for the given compounds. (i) Aspartem (ii) Saccarine (iii) Sucrolose (iv) Alitame |
|
Answer» (i) LT (i) lt (III) lt (IV) |
|