Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select the incorrect statement about metal carbonyl complex compounds : |
|
Answer» Metal carbon bond in metal carbonyls possess both `sigma" and "Pi` characters |
|
| 2. |
Select the incorrect statement about silicate's :- |
|
Answer» Beryl an EXAMPLE of cyclic silicate |
|
| 3. |
Select the INCORRECT statement about inert gases. |
|
Answer» The ionization enthalpy of each INERT gas is the HIGHEST in its period. |
|
| 4. |
Select the incorrect statement about hydrides of group 15 elements. |
|
Answer» The CENTRAL atom in the hydride is `SP^(2)`-hybridised |
|
| 5. |
Select the incorrect statement about cathode rays. |
|
Answer» Deflected by ELECTRIC and MAGNETIC field |
|
| 6. |
Select the incorrect statement about beryllium and aluminium? |
|
Answer» Chlorides of BERYLLIUM and aluminium are soluble in organic solvents |
|
| 7. |
Select the incorrect statement about (A) and ( C) compounds. |
|
Answer» Aqueous solution of (C ) PRODUCES white PRECIPITATE with dilute HCl (2M) |
|
| 8. |
Select the incorrect statement: |
|
Answer» Schottky defect is shown by CSCL |
|
| 9. |
Select the incorrect statement. |
|
Answer» HGO gives oxygen on heating. |
|
| 10. |
Select the incorrect property for Fe_(4)[Fe(CN)_(6)]_(3) complex |
|
Answer» <P>`d^(2)sp^(3)` Hybridisation of coordinated FE atom `10.2Z^(2)=w+KE_("max")2` `lambda_(DB)=(h)/(p)=(h)/(sqrt(2m(K in))),(lambda_(1))/(lambda_(2))=sqrt((Kin_(2))/(K_(in_(1))))=2.3` `KE_(2)=5.25KE_(1)` `10.2z^(2)=3xx 13.6,z=2 " "&" " w=3eV` |
|
| 11. |
Select the incorrect statement ? |
|
Answer» `Ag^(+)` ions with an excess of a solution containing `CN^(-)` ions form a complex having coordination number two. |
|
| 12. |
Select the incorrect reduction process. |
|
Answer» `2[Ag(CN)_(2)]^(-) +Zn to [Zn(CN)_(4)]^(2-)+2Ag ` Thus , hydrogen hasno ability to reduce `Zn^(2+)` to Zn. Cu being less reactive is reduced by hydrogen . Hence reaction 'c' is not possible . |
|
| 13. |
Select the incorrect orders from the following . |
|
Answer» `CIO^(-) LT BrO^(-) lt IO^(-)` - disproportionation (D)HOCI gt HOBr gt HOI. As ELECTRONEGATIVITY of halogens increases the acidic character increases GIVEN (B) and (D) are correct order . |
|
| 14. |
Select the incorrect match: |
|
Answer» `[Co(NO_(2))(H_(20O)(en)_(2)]Cl_(2),[CoCl(NO_(2))(en)_(2)]Cl*H_(2)O`: HYDRATE isomerism |
|
| 15. |
Select the given code of reagents for following canversion and write your answer as abcd. If reagentis used once then you need not to take again. (1) H_(2)So_(4)(2) ZnHg//HCl (3) NHO_(3)//NH_(3)So_(4)(4)NaNO_(2)//HCl0^(@)-5^(@)C (5) NaH_(4)//H_(2)O_(2)(6) NBS (7) Me-overset(O)overset(||)C-Cl//AlCl_(3)(8) Me_(3)CO^(Θ)Na^(o+) |
|
Answer» |
|
| 16. |
Select the given code of reagents for following conversion and write your answer as abcd. If reagent is used once then you need not to take it again. |
|
Answer» |
|
| 17. |
Select the group of oxides that can not be reduced by carbon to give the respective matals. |
|
Answer» `CaO,K_(2)O` |
|
| 18. |
Select the flow diagram which is correct for extraction of particular metal from its ore :- |
|
Answer» `"Tin stone" overset("Magnetic")overset("separation")to SnO_(2)overset("Self")overset("reduction")tounderset(SN("pure"))underset(""darr"distillation")(Sn("impure"))` |
|
| 19. |
Select the example of desproportionatonreaction |
|
Answer» `BaCl_(2)+H_(2)SO_(4)rarrBaSO_(4)+2HCL` |
|
| 20. |
Select the ether among following that yields methanol as one of the products on reaction with cold hydroiodic acid |
|
Answer» 1-Methoxybutane |
|
| 21. |
select the cottect IUPAC name of [Co(H_(2)NCH_(2)CH_(2)NH_(2))_(3)]_(2)(SO_(4))_(3) is : |
|
Answer» TRIS (ethane-1, 2-diamine) cobalt (III) sulphate |
|
| 22. |
Select the diamagnetic complex ion amongst the following complexes (At.no : Fe = 26 , CO = 27) |
|
Answer» `K_(3)[FE(CN)_(6)]` (a) `K_(3) [Fe (CN)_(6)] i.e. , [Fe(CN)_(6)]^(3-) `ion `""_(26)Fe = [Ar] 3d^(6) 4s^(2) implies Fe^(3+) = [Ar] 3d^(5) , 4s^(0)`  (b) `[Co(NH_(3))_(6)]Cl_(3)` i.e, `[Co(NH_(3))_(6)]^(3+)` ion `""_(27)Co = [Ar] 3d^(3) 4s^(2) implies Co^(3+) = [Ar] 3d^(6) 4s^(0)`  (c) `K_(3) [Fe F_(6)] i.e., FeF_(6)^(3-)` ion `""_(26)Fe = [Ar] 3d^(6) 4s^(2) implies Fe^(3+) = [Ar] 3d^(5) 4s^(0)`  (d) `K_(3) [CoF_(6)]i.e., CoF_(6)^(3-) `ion `""_(27)Co = [Ar] 3d^(7) 4s^(2) implies Co^(3+) = [Ar] 3d^(6)`  Therefore `, [Co(NH_(3))_(6)] CF_(3)` diamagnetic in nature . |
|
| 23. |
Select the element which does not form double bond |
|
Answer» Nitrogen |
|
| 24. |
Select the correct statements(s): |
|
Answer» At Boyle's TEMPERATURE a real gas behaves like an IDEAL gas at low pressure |
|
| 25. |
Select the correct synthesis products |
|
Answer»
`AtoHBO` `BTO` Oxymeruration Demercuration `Cto` Epoxidation `DtoSN`- Reaction of AROMATIC compound.  
|
|
| 26. |
Select the correct statements(s) |
|
Answer» when `Tto prop` or `E_(a)to0`, then K=A |
|
| 27. |
Select the correct statement(s) with respect to the p pi-dpi dative bond. |
|
Answer» In `(Ph_3Si)_2O`, the Si-O-Si group is nearly linear (B)Due to stabilization of the conjugate base anion by O `(ppi)to Si(dpi)`bonding MOTION. ( C)In is PYRAMIDAL because `ppi-dpi` bonding is not effective due to the bigger size of phosphorus atom (D)Most effective due to small size of chlorine. |
|
| 28. |
Select the correct statement(s) related to hexagonal close packing of identical spheres in three dimensions : |
|
Answer» In ONE unit cell there are 12 octahedral voids and all are COMPLETELY INSIDE the unit cell. For calculating voids between two layers A and B Octahedral voids=3=represented by (*). So total octahedral voids=6= All are completely inside. 
|
|
| 29. |
Select the correct statement(s) regardingthe compounds (W) and (Z) |
|
Answer» COORDINATION number of Al in the COMPOUND (W) is 6 |
|
| 30. |
Select the correct statement(s) regarding BF_(2)NH_(2) molecule: |
|
Answer» FBF bond angle `lt120^(@)` 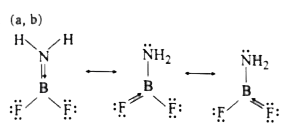 `2p_(pi)-2p_(pi)` back bonding According to Bent.s rule there is more p-character in `B-F` bond than in `B-N` bond, Hence FBF is less than `120^(@)`. As size of `-NH_(2)` group is larger than F-atom hence repulsion between `-NH_(2)` group and F-atom is higher than between two F-atoms. As LONE pair of N-atom in `NH_(2)` group participate in back bonding hence `H-N-H` is greater than `109^(@)28.`. as hybridization of N is changing from `sp^(3)` towards close to `sp^(2)` |
|
| 31. |
Select the correct statements regarding [Pt(en)(pn)Cl_(2)]^(2+) ion. |
|
Answer» It exhibits optical isomerism 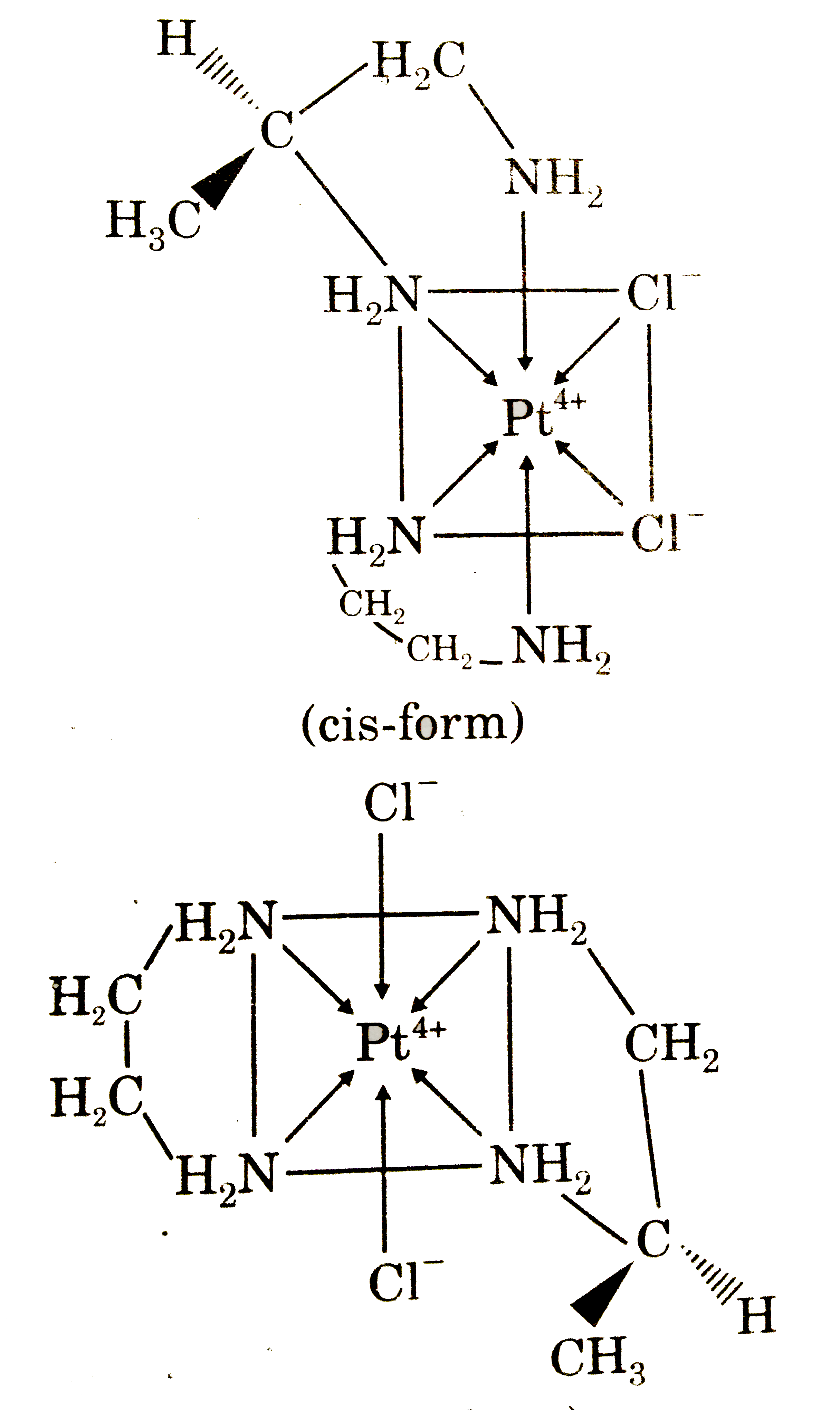 and both geometrical isomers are optically active. |
|
| 32. |
Select the correct statements of the following:- (a) Effective nuclear charge for nitrogen is 3.90 (b) IP of Ne is more than Na^(+) (c ) Order of electronegativity- sp gt sp^(2) gt sp^(3) (d) Order of acidic character- NH_(3) lt PH_(3) lt AsH_(3) |
|
Answer» a, B, d |
|
| 33. |
Select the correct statements : I. Cs^(+) is more highly hydrated that the other alkali metal ions. II. Among the alkali metals. Li, Na, K and Rb, lithium has the highest melting point. III. Among the alkali metals only lithium froms a stable nitride by direct combination with nitrogen. |
|
Answer» I, II and III |
|
| 34. |
Select the correctstatements . (i) Physical adsorption is weak, multilayer, non-directional and non-specific. (ii) Chemical adsorption is strong, unilayer, directional and strong (iii) Chemical adsorption decreases with temperature (iv) Chemical adsorption is more stronger than physical adsorption |
|
Answer» (i) and (III) only |
|
| 35. |
Select the correct statements from the following |
|
Answer» A-methoxy PHENOL is more reactive tha P-nitro phenol towards Reimer-Tiemann reaction. |
|
| 36. |
Select the correct statement(s) for the compound K_(2)[Cr(NO^(+))(NH_(3))(CN)_(4)] |
|
Answer» Its IUPAC name is potassium amminetetracyanidonitrosoniumchromate (I) `mu=sqrt(5(5+2))BM"":.5` unpaired ELEMENTS present. `=sqrt(35)BM` and can GIVE SFL are present `[Ma_(4)bx]^(2+)` inner orbital octahedron complex hybridisation of Cr is `d^(2)sp^(3)`. It is of type `[Ma_(4)bx]^(2+)` type and shows geometrical isomerism. |
|
| 37. |
Select the correct statements for the following decay chain A overset(-alpha)(to) B overset(-beta)(to) C overset(-beta)(to) D |
|
Answer» A and D are isotopes |
|
| 38. |
Select the correct statements : CaCl_(2) doped with NaCl solid solution results in |
|
Answer» substitutional CATION vacancy |
|
| 39. |
Select the correct statement(s) for positron emission by unstable nucleus. |
|
Answer» X-ray EMISSION takes place |
|
| 40. |
Select the correct statement(s) for (NH_(4))_(3)PO_(4) |
|
Answer» Ratio of number of oxygen atoms to number of HYDROGEN atoms is 1:3 |
|
| 41. |
Select the correct statements among the following |
|
Answer» Greater the HUMIDITY, lesser will be the RATE of evaporation |
|
| 42. |
Select the correct statements among the following: |
|
Answer» The DECAY of mass during nuclear fusion and nuclear fission are 0.1% and 0.231% respectively. |
|
| 43. |
Select the correct statements among the following. |
|
Answer» Co (III) is stabilised in presence of weak field ligands, while Co (II) is stabilised in presence of strong field ligabd, |
|
| 44. |
Select the correct statement(s) among following |
|
Answer» Increase in concentration of reactant increases the rate of a zero order reaction |
|
| 45. |
Select the correct statement(s) about the complex, [Pt(S_(5))_(3)]^(2-). |
|
Answer» It contians `S_(5)^(2-)` anion which is a chelating ligand. 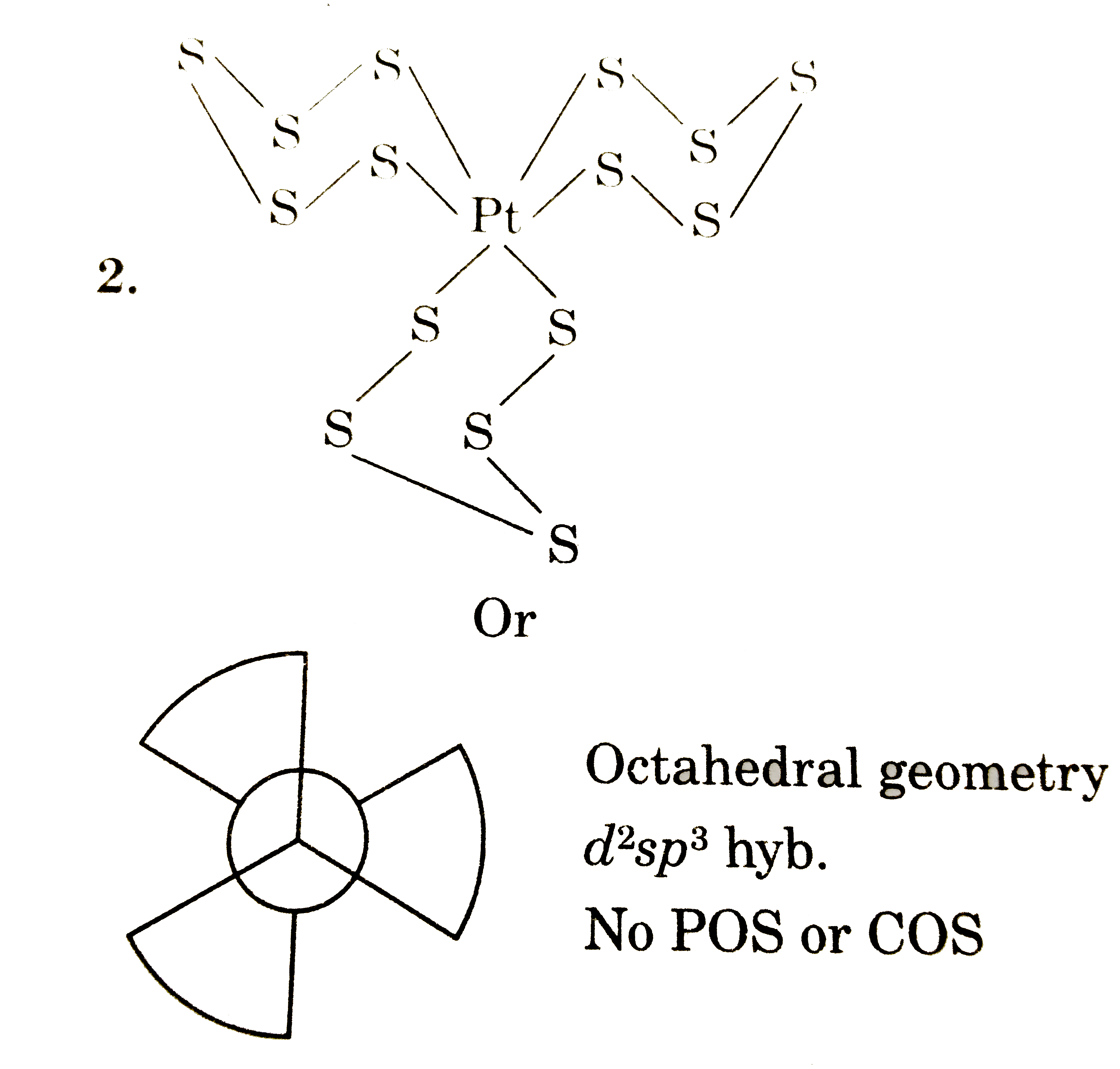
|
|
| 46. |
Select the correct statement(s) about the compound NO[BF_4] |
|
Answer» It has `5sigma and 2pi` bonds 
|
|
| 47. |
Select the correct statements about oxygen molecule. |
|
Answer» It is PARAMAGNETIC. |
|
| 48. |
Select the correct statements about Ma_(2)bcde |
|
Answer» It has 6 cis GEOMETRICAL isomers with RESPECT to two a  (b)  (C ) Stereo`implies 12+3=15` |
|
| 49. |
Select the correct statement(s) about climetidine andrantitidine. |
|
Answer» These are ANTIHISTAMINE drugs |
|
| 50. |
What are the main ingredients of Portland cement :- |
| Answer» | |



