Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Salicylic acid is prepared from phenol by the reaction known as |
|
Answer» WURTZ reaction |
|
| 4. |
Salicylic acid can be prepared using Reimer-Tiemann's reaction by treating phenol with |
|
Answer» methyl CHLORIDE in the presence of anhydrous ALUMINIUM chloride. |
|
| 5. |
Salicyldehyde can be prepared by ....... |
|
Answer» FITTING reaction 
|
|
| 6. |
Salicylaldehyde can be separated from the mixture of salicyladehyde and p - hydroxy benzaladehyde by |
|
Answer» DISTILLATION |
|
| 7. |
Saccharine contains |
|
Answer» C and H 
|
|
| 8. |
Give the structure of the Saccharinand write its one use. |
|
Answer» HEXOSE |
|
| 9. |
Saccharin is a/an________. |
|
Answer» ANALGESIC |
|
| 10. |
Saccharin, butyl hydroxy toluene, aspartane, sucralose, alitame. |
| Answer» Solution :BUTYL hydroxy TOLUENE. It is an ANTIOXIDANT where as others are artificial sweetening AGENTS. | |
| 11. |
Saccharin, an artificial sweetner, is manufacturing from: |
|
Answer» Cellulose |
|
| 12. |
Saccharin, an artificial sweetener is manufactured from |
| Answer» Solution :toluene | |
| 13. |
Saccharin , an artificial sweetener , is manufactured from |
|
Answer» CELLULOSE |
|
| 14. |
Saccharin, an artificial sweetener, is manufactured from |
|
Answer» Cellulose |
|
| 15. |
Saccharification is the process of conversion of : |
|
Answer» SUGAR SOLUTION into alcohol |
|
| 16. |
S_(8) molecule is present in |
|
Answer» Rhombie SULPHUR |
|
| 17. |
S_(2)O_(3)^(2-) + MnO_(4)^(-) + H_(2)O rarr MnO_(2) + S_(2)O_(4)^(2-) + OH^(-). What are the coefficients of S_(2)O_(3)^(2-) and MnO_(4)^(-) in the balance equation ? |
|
Answer» |
|
| 18. |
S_(2) molecule in vapour state is paramagnetic due to the presence of unpaired electrons is |
|
Answer» Bonding `sigma` ORBITALS |
|
| 19. |
S^(2-) Cannot be tested with |
|
Answer» `BaCl_2` solution |
|
| 20. |
S^(2-) " and" SO_(3)^(2-) can be distinguished by : |
|
Answer» `(CH_(3)CO O)_(2) Pb` |
|
| 21. |
S^(2-) and SO_(3)^(2-) canbe distinguished by using |
|
Answer» `(CH_(2)COO)_(2)Pb` |
|
| 22. |
S^(2-) and SO_(3)^(2-) can be distinguihsed by using |
|
Answer» `(CH_(3)COO)_(2)Pb` `S^(2-)+Na_(2)[Fe(CN)_(5)NO]tounderset("violet ppt.")([Fe(CN)_(5)NOS]^(4-))+2Na^(+)` HENCE, they are distinguished by both the REAGENTS as only `S^(2-)` gives precipitate with these reagents. |
|
| 23. |
S^(2–) and SO_(3)^(2–) can be distinguished by using: |
|
Answer» `(CH_(3)COO)_(2)Pb` |
|
| 24. |
S_(1): 2Se_(2)Cl_(2)rarr SeCl_(4)+3Se S_(2) : Dioxides like MnO_(2), PbO_(2) do not form H_(2)O_(2) with dilute acids but they evolve oxygen with concentrated H_(2)SO_(4). S_(3) : Sodium thiosulphate with FeCl_(3) solution develops a pink or violet colour which soon vanishes. S_(4) : White precipitate of PbS_(2)O_(3) get soluble when boiled with water. |
|
Answer» FTTT |
|
| 25. |
S_(1):(HPO_(3))_(n) can be prepared by heating phosphorous acid and bromine in a sealed tube. S_(2) : dry iodine reacts with ozone and formed yellow solid, I_(4)O_(9) S_(3) : B sulphur is stable below 369 K. |
|
Answer» FTF |
|
| 26. |
S_1:The species [CuCl_4]^(2-) exists but [C ul_4]^(2-) does not. S_2:[RhCl(Ph_3P)_3] and [Ni(CO)_4] both are tetrahedral and diamagnetic. S_3:N(Me)_3 and N(SiMe_3)_3 are isostructural |
|
Answer» T T F `S_2:`Both diamagnetic but `[NI(CO)_4]` is tetrahedral and `[RhCl(Ph_3P)_3]` is a square planar. 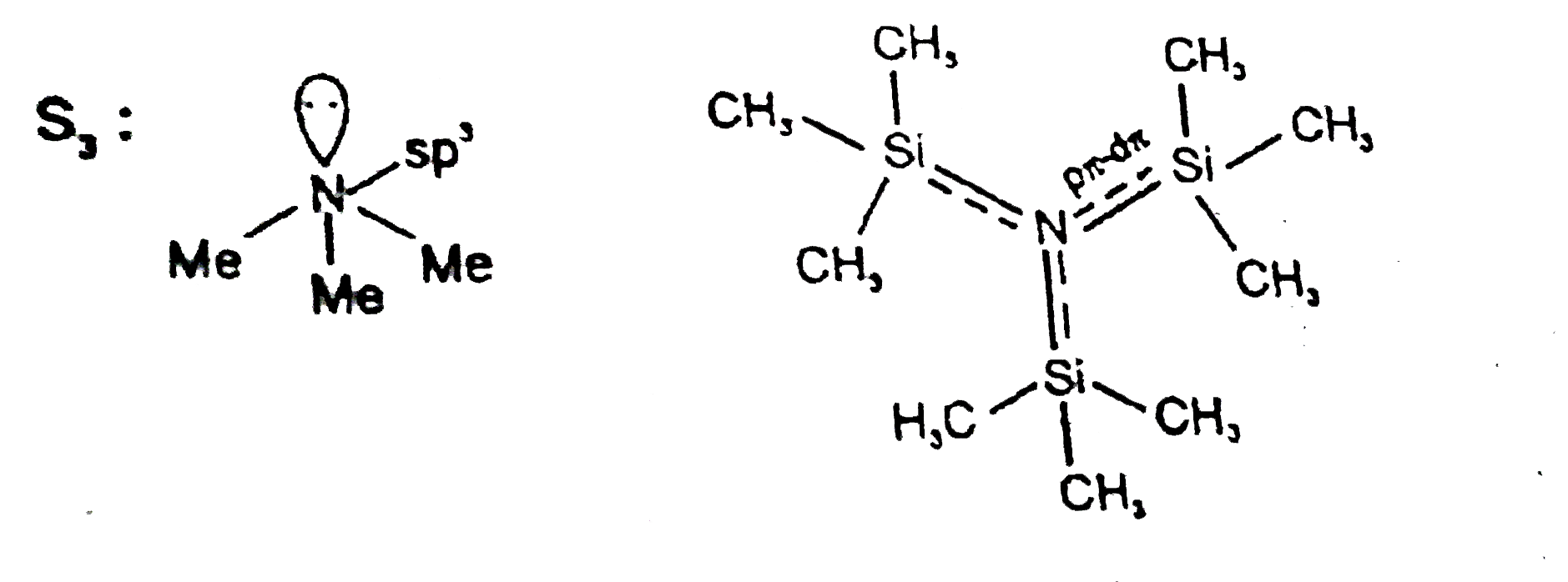
|
|
| 27. |
S, T and U are the aqueous chlorides of the elements X, Y and Z respectively. X, Y and Z are in the same period of the periodic table. U gives a white precipitate with NaOH but this white precipitate dissolves as more NaOH is added. When NaOH is added to T, a white precipitate forms which does not dissolve when more base is added. S does not give precipitate with NaOH. Which of the following statements are correct? |
|
Answer» The three elements are metals |
|
| 28. |
S rarr G + F Time " "t " "prop Rotation of Glucose & Fructose r_(t)" " r_(prop) Find k. |
| Answer» SOLUTION :`K=(1)/(t)" In "(r_(OO))/((r_(oo)-r_(1)))` | |
| 29. |
S-S bond is not present in |
|
Answer» PYRO SULPHUROUS ACID |
|
| 30. |
S-O-O-S bond present in which of the following oxyacid is |
|
Answer» `H_(2)SO_(4)` |
|
| 31. |
S is a well known pain killer which of the following is 'R' ? |
|
Answer» `CH_(3)-overset(O)overset(||)(C)-NH_(2)` |
|
| 32. |
S+(3)/(2)O_(2)rarrSO_(3)+2 x kcalSO_(2)+(1)/(2)O_(2)rarrSO_(3)+y kcalFind out the heat of formation of SO_(2) |
|
Answer» `2X - y` |
|
| 33. |
S+3//2O_2rarrSO_3 + 2x kcal SO_2+ 1/2O_2rarrSO_3 + y kcal: The heat of formation of SO_2 is : |
|
Answer» y-2x |
|
| 34. |
S+2Cl_(2) to X overset(H_(2)O)to Y+HCl, Y to Z+H_(2)O. Oxidation state of S in 'Z' is |
| Answer» ANSWER :D | |
| 35. |
RX + NaOH_(aq) overset(Delta) to ROH + NaX The above reaction proceed by __________ mechanism. |
|
Answer» NUCLEOPHILIC addition |
|
| 36. |
Rydberg constant is |
|
Answer» A UNIVERSAL constant |
|
| 37. |
RX on heating with NH_3 in sealed tube gives : |
|
Answer» `RNH_2` |
|
| 38. |
Rutile is seperated from chlonapatite by |
|
Answer» Froth floation method |
|
| 39. |
Rutile is an ore of: |
|
Answer» Ti |
|
| 40. |
Rutherford's scattering experiment led to the discovery of |
|
Answer» Nucleus |
|
| 41. |
Rutherford's experiment, which established the nuclear model of the atom, used a beam of |
|
Answer» `beta`-particles, which impinged on a METAL FOIL and GOT absorbed |
|
| 44. |
Rusting or iron is catalysed by which of the following |
|
Answer» Fe |
|
| 45. |
Ruthenium carbonyl is : |
|
Answer» RU(CO)_4` |
|
| 46. |
Rusting on iron needs |
|
Answer» DRY air |
|
| 47. |
Rusting of iron is not only on the surface but it eats up iron completely because |
|
Answer» RUST PENETRATES into iron |
|
| 48. |
Rusting of iron is quicker in saline water than in orderinary water. Give reason. |
| Answer» Solution :In saline water, the presence of `Na^(+) and Cl^(-)` ions increases the conductance of the solution in CONTACT with the METAL SURFACE. This accelerates the formation of `Fe^(2+)` ion and HENCE that of rust, `Fe_(2)O_(3).xH_(2)O` | |



