Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Sample containing ""_(88)^(234)Ra, which decays by alpha-particle emission, is observed to disintegrate at the following rate expressed as counts per minute (cpm). Calculate half-life of this nuclide. t=0,1000 cpm, t=1h, 992 cpm, t= 10h, 924 cpm, t=100h, 452cpm, t= 250h, 138 cpm |
|
Answer» |
|
| 2. |
Sample of 2, 3-dibromo-3-methylpentane is heated with dust. The resulting product is isolated and heated with HI in presence of phosphorus. Indicate which is the structure represent the final organic product formed in the reaction |
|
Answer» `CH_3-CH_2-underset(CH_3)underset|CH-CH_2-CH_3` 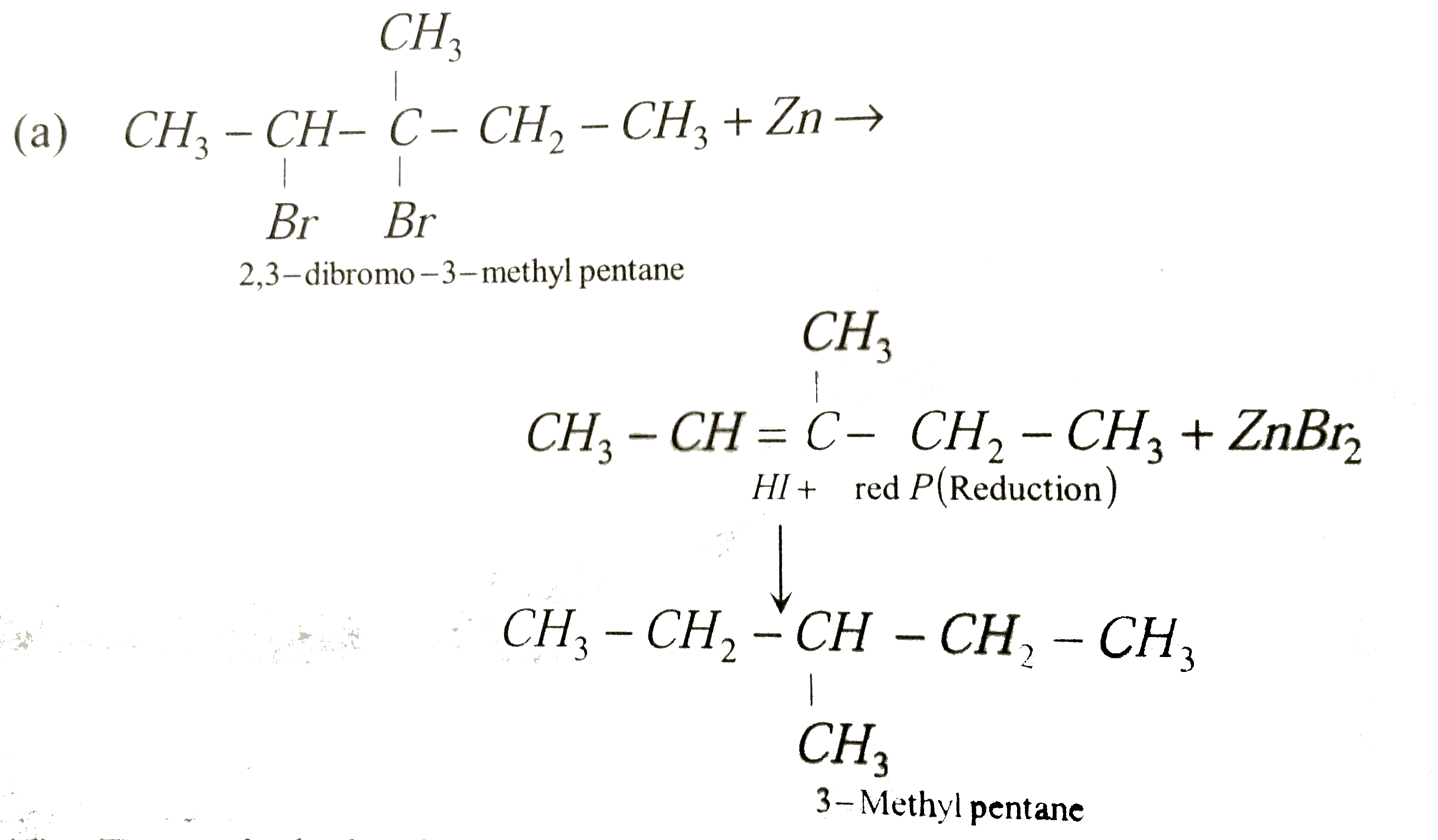
|
|
| 3. |
Same quantity of electricity was passed through solutions of salts of elements A,B and C with atomic weights 7, 27 and 48 respectively. The masses of A,B and C deposited were 2.1 g,27.7g and 7.2 respectively. The valencies of A,B and C respectively are |
|
Answer» 3,2 and 1 `("Mass of A")/("Eq. wt. of A")=("Mass of B")/("Eq. wt of B")=("Mass of C")/("Eq. wt of C")` `therefore(2.1)/(7//a)=(2.7)/(27//b)=(7.2)/(48//c)` or 0.3 a=0.1, b=0.15c, or 3a=b=1.5c. thus, if b=1, then `a=(1)/(3)`, `c=(1)/(1.5)=(2)/(3)` `therefore`Ratio `a:b:c=(1)/(3):1:(2)/(3)=1:3:2` |
|
| 4. |
Same mass of CH_(4) and H_(2)is taken in container. The partial pressure caused by H_(2) is |
|
Answer» `8 //9` `N_(H_(2))= ` number of moles of `H_(2) = (m)/(2)` fractionpartial pressure of `H_(2)` is `H_(2)=(n_(H_(2)))/(n_(H_(2))+n_(CH_(4)))=((m)/(2))/((m)/(2)+(m)/(16))=((m)/(2))/((9M)/(16))=(8)/(9)` |
|
| 5. |
Same quantity of electrical charge that deposited0.583 g of silver was passed through a solution of gold salt. If 0.335 g of gold is deposited, calculate the oxidation state of gold in the given salt. At wt of Au = 197. |
|
Answer» |
|
| 6. |
Same mass of CH_(4) and H_(2) is taken in container. The partial pressure caused by H_(2) is |
| Answer» Answer :A | |
| 7. |
Same amount of electricity is passed through solution of ferrous chloride and ferric chloride . Ratio of weight of iron deposited in the two solution is |
|
Answer» `1:1` `2E^(-) xx 3 : 3e^(-) xx 2 therefore 3:2` |
|
| 8. |
Same amount of electric current is passed through solutions of AgNO_(3) and HCl. If 1.08g of silver is obtained in the first case, the amount of hydrogen liberated at S.T.P. in the second case is |
|
Answer» `224cm^(3)` `("Mass of Ag")/("Mass of H")=("EQUIVALENT mass of SILVER")/("Equivalent mass of hydrogen")` or `(1.08)/("Mass of H")=(108)/(1)` or mass, `H=0.01g` `because2.00` gm of `H_(2)` occupies 22400 ml at STP `therefore 0.01`gm of `H_(2)` will OCCUPY`=(22400xx0.01)/(2)` `=112cm^(3)`. |
|
| 10. |
Same amount of electric current is passed through solutions of AgNO_3and HCl. If 1.08 g of silver is obtained in the first case, the volume of hydrogen liberated at S.T.P. in the second case is |
|
Answer» `112 cm^3` |
|
| 11. |
Salvarsan is arsenic containing drug which was first used for the treatment of.________. |
|
Answer» SYPHILIS |
|
| 12. |
Salvarsan is arsentic containing drug which was first usedfor the treatmentof "……." |
|
Answer» syphilis Paul EHRLICH investigated arsenic based structure in order to produce less toxic substances for the treatement of syphillis as he got Nobel PRIZE for medicine in `1908` for this discovery. Salvarsan was the firsteffective medicine discoveredfor syphilis. ALTHOUGH salversan is toxic to human beings its effect on bacteria is much greater than on human being. Strucutre of salvarsanis - 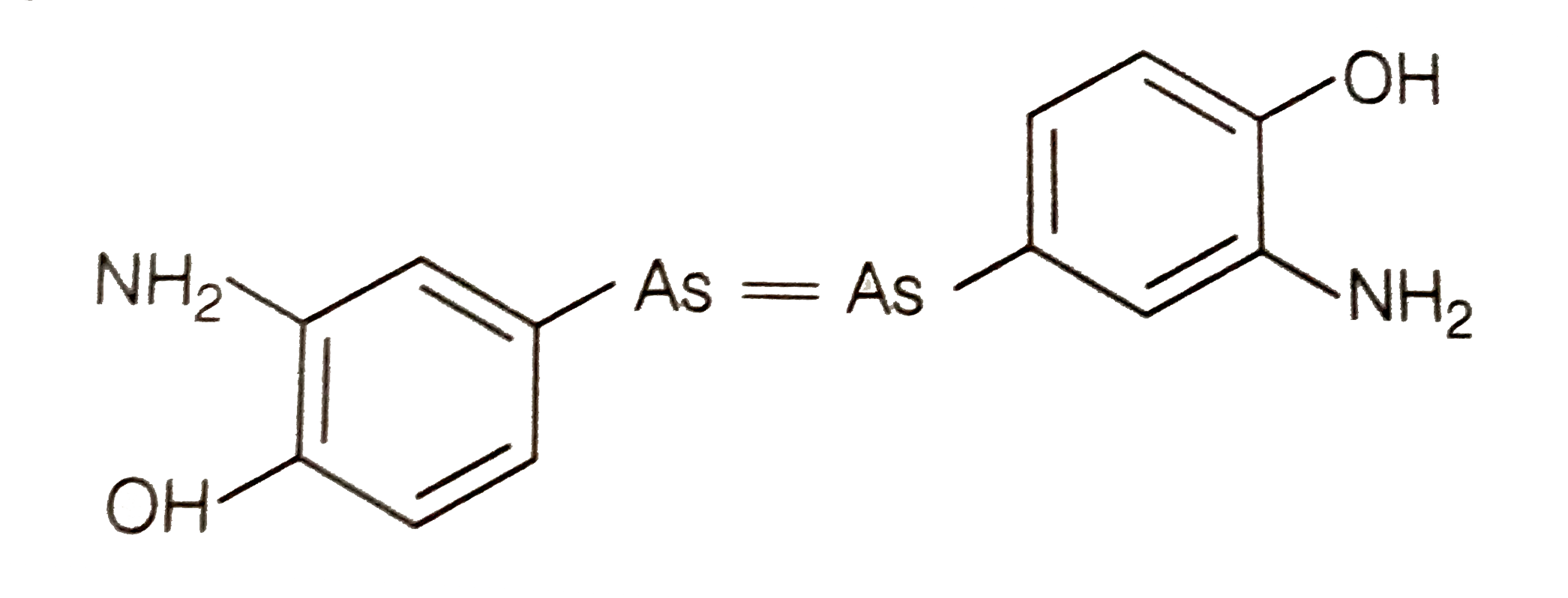
|
|
| 13. |
Salvarsan is having similarity in structure as that of |
|
Answer» azodye |
|
| 14. |
Salvarsanis arsenic containing drug which was firstused for the treatment of ___________ |
|
Answer» SYPHILIS |
|
| 15. |
Salvarsan is arsenic containing drug which was first used the treatment of |
|
Answer» syphilis |
|
| 16. |
Salvarsan is arsenic containing drug which was first used for the treatment of .. |
|
Answer» SYPHILIS |
|
| 17. |
Salvarsan is arsenic containing drug which was first used for the treatment of |
|
Answer» syphilis |
|
| 18. |
Salvarsan is arsenic containing drug which was first used for the treatment of _____ |
|
Answer» syphilis |
|
| 19. |
Salts of which of the following elements are mostly blue in colour? |
|
Answer» Cu |
|
| 20. |
Salvarsan is arsenic containing drug which was first used for the treatment of ________ |
|
Answer» syphilis |
|
| 21. |
Salts of sorbic acid and propionic acid are used as: |
|
Answer» ANTIOXIDANTS |
|
| 22. |
Salts of metals X, Y and Z are electrolysed under identical condition using same quantity of electricity . Ifwas observed that 4.2 g of X, 5.4 g of Y and 19.2 g of Z were deposited at respetive cathode . Ifthe atomic weights of X , Y, Z are 7, 27 and 64 respectively , then their ratio of valencies is |
|
Answer» `1:2:3` |
|
| 23. |
Salts of sorbic acid and propionic acid are used as |
|
Answer» Antioxidants |
|
| 24. |
Salts can be obtained from a concentrated sea water by : |
|
Answer» Catalyst |
|
| 25. |
Salt 'X' + excess NaOH solution overset("warm")rarr'Y' ("gas")+'Z' ("solution") 'Z'("solution")+BaCl_(2)rarr"white ppt" Which of the following salt follow above reaction. |
|
Answer» `NA(NH_(4))HPO_(4).4H_(2)O` |
|
| 26. |
Salts of A (atomic weight 7), B (atomic weight 27) and C (atomic weight 48) were electrolysed under identical condition using the same quantity of electricity. It was found that when 2.1 g of A was deposited, the eight of B and C deposited were 2.7 and 7.2 g. The valencies of A,B and C are respectively : |
|
Answer» `3,1 and 2` |
|
| 27. |
Salt with which one of the following anion produce deep blue colour with starch and Kl is |
|
Answer» CHLORIDE |
|
| 29. |
Salt of the following is used as a water softner |
|
Answer» `H_(4)P_(2)O_(6)` |
|
| 30. |
Saltmixture overset(dil. HCI) to Saltis consistingof cation |
|
Answer» `Pb^(2+)` and `HG^(2+)` |
|
| 31. |
Salt cake is |
|
Answer» SODIUM SULPHATE |
|
| 32. |
Salt bridge is used to |
|
Answer» CONNECT two half cells |
|
| 33. |
Salt and water is formed by acid-base neutralisation reaction. If ratio of moles of acid & base taken is not similar to the ratio of their stoichiometric coefficient, then one of the component is limiting reagent.Assume no dissociation of water in following reactions. (Base is 80% pure only, take impurity present as inert & non electrolytic ) Select the correct combination for the resulting acidic solution. |
|
Answer» <P>(I)(ii)(S) `{:(NaOH+,HCl to ,NaCl,+H_2O),(8/40=0.2 "mole",(8xx500)/1000,-,-):}` =0.4 mole `=0.2 "mole" " " 0.2"mole"` BASE in L.R.,`[H^+]=0.2 M " " [Na^+]=0.2 M [Cl^(-)]=0.4 M` (ii)KOH pure `=(28xx80)/100=22.4 gm` `{:(KOH+,HNO_3to,KNO_3,+H_2O),(22.4/56=0.4 "mole",(2.5xx500)/1000,-,-):}` =0.1 mole `=0.3 "mole" " " 0.1"mole"` Acid in L.R.,`[OH^(-)]=0.3 M " " [K]=0.4 M [NO_3^(-)]=0.1 M` (iii) `{:(Ca(OH)_2+,H_2SO_4to,CaSO_4,+2H_2O["Pure" Ca(OH)_2=37xx0.8=29.6 gm]),(29.6/74=0.4 ,(0.8xx500)/1000=0.4,-,-):}` `[Ca^(2+)]=[SO_4^(2-)]=0.4M` (iv) `{:(Ba(OH)_2+,2HBrto,BaBr_2,+2H_2O["Pure" Ba(OH)_2=342xx0.8=,273.6 gm]),(273.6/171=1.6 ,(6.4xx500)/1000=3.2,-,-),(-,-, ,1.6 "mole"):}` `[Ba^(2+)]=1.6 M " " [Br^(-)]=3.2M` |
|
| 34. |
Salt and water is formed by acid-base neutralisation reaction. If ratio of moles of acid & base taken is not similar to the ratio of their stoichiometric coefficient, then one of the component is limiting reagent.Assume no dissociation of water in following reactions. (Base is 80% pure only, take impurity present as inert & non electrolytic ) Select incorrect combination |
|
Answer» (I)(II)(P)<BR>(II)(i)(R) `{:(NaOH+,HCl to ,NaCl,+H_2O),(8/40=0.2 "mole",(8xx500)/1000,-,-):}` =0.4 mole `=0.2 "mole" " " 0.2"mole"` Base in L.R.,`[H^+]=0.2 M " " [Na^+]=0.2 M [Cl^(-)]=0.4 M` (ii)KOH pure `=(28xx80)/100=22.4 gm` `{:(KOH+,HNO_3to,KNO_3,+H_2O),(22.4/56=0.4 "mole",(2.5xx500)/1000,-,-):}` =0.1 mole `=0.3 "mole" " " 0.1"mole"` Acid in L.R.,`[OH^(-)]=0.3 M " " [K]=0.4 M [NO_3^(-)]=0.1 M` (iii) `{:(Ca(OH)_2+,H_2SO_4to,CaSO_4,+2H_2O["Pure" Ca(OH)_2=37xx0.8=29.6 gm]),(29.6/74=0.4 ,(0.8xx500)/1000=0.4,-,-):}` `[Ca^(2+)]=[SO_4^(2-)]=0.4M` (iv) `{:(Ba(OH)_2+,2HBrto,BaBr_2,+2H_2O["Pure" Ba(OH)_2=342xx0.8=,273.6 gm]),(273.6/171=1.6 ,(6.4xx500)/1000=3.2,-,-),(-,-, ,1.6 "mole"):}` `[Ba^(2+)]=1.6 M " " [Br^(-)]=3.2M` |
|
| 35. |
Salt and water is formed by acid-base neutralisation reaction. If ratio of moles of acid & base taken is not similar to the ratio of their stoichiometric coefficient, then one of the component is limiting reagent.Assume no dissociation of water in following reactions. (Base is 80% pure only, take impurity present as inert & non electrolytic ) Select the correct combination for the resulting basic solution. |
|
Answer» (I)(iii)(S) `{:(NaOH+,HCL to ,NaCl,+H_2O),(8/40=0.2 "mole",(8xx500)/1000,-,-):}` =0.4 mole `=0.2 "mole" " " 0.2"mole"` Base in L.R.,`[H^+]=0.2 M " " [Na^+]=0.2 M [CL^(-)]=0.4 M` (ii)KOH pure `=(28xx80)/100=22.4 gm` `{:(KOH+,HNO_3to,KNO_3,+H_2O),(22.4/56=0.4 "mole",(2.5xx500)/1000,-,-):}` =0.1 mole `=0.3 "mole" " " 0.1"mole"` Acid in L.R.,`[OH^(-)]=0.3 M " " [K]=0.4 M [NO_3^(-)]=0.1 M` (iii) `{:(Ca(OH)_2+,H_2SO_4to,CaSO_4,+2H_2O["Pure" Ca(OH)_2=37xx0.8=29.6 gm]),(29.6/74=0.4 ,(0.8xx500)/1000=0.4,-,-):}` `[Ca^(2+)]=[SO_4^(2-)]=0.4M` (iv) `{:(BA(OH)_2+,2HBrto,BaBr_2,+2H_2O["Pure" Ba(OH)_2=342xx0.8=,273.6 gm]),(273.6/171=1.6 ,(6.4xx500)/1000=3.2,-,-),(-,-, ,1.6 "mole"):}` `[Ba^(2+)]=1.6 M " " [Br^(-)]=3.2M` |
|
| 36. |
Salt bridge is used because it |
|
Answer» maintains the electrical neutrality of the TWO half cells |
|
| 37. |
Salt A overset("Layer test") to If reddish brownlayer come first , then |
|
Answer» `BR^(Θ)` PRESENT `I^(Theta)`ion is oxidised preferably COMPOUND to `Br^(Theta)`,Hence `I^(Theta)` ID absent 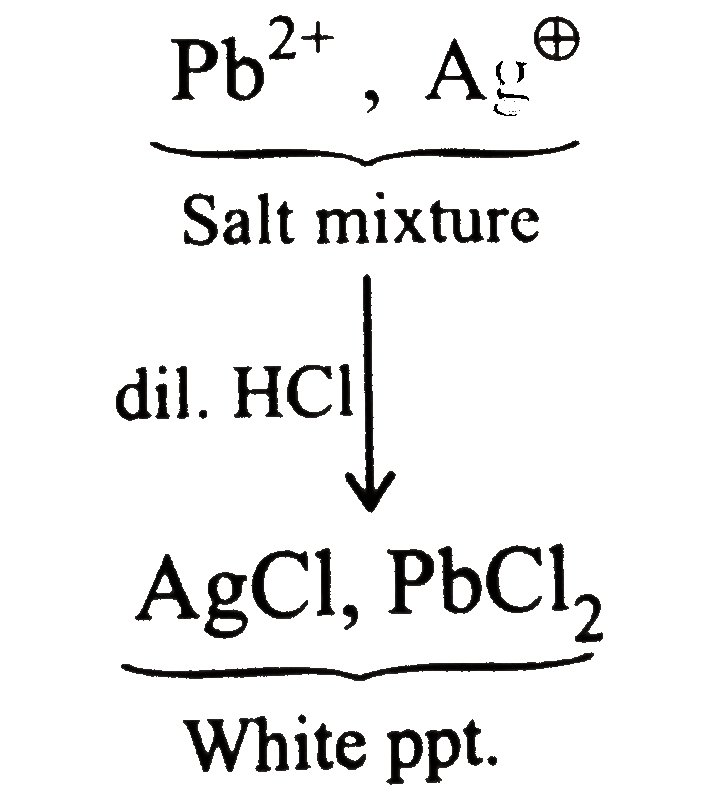 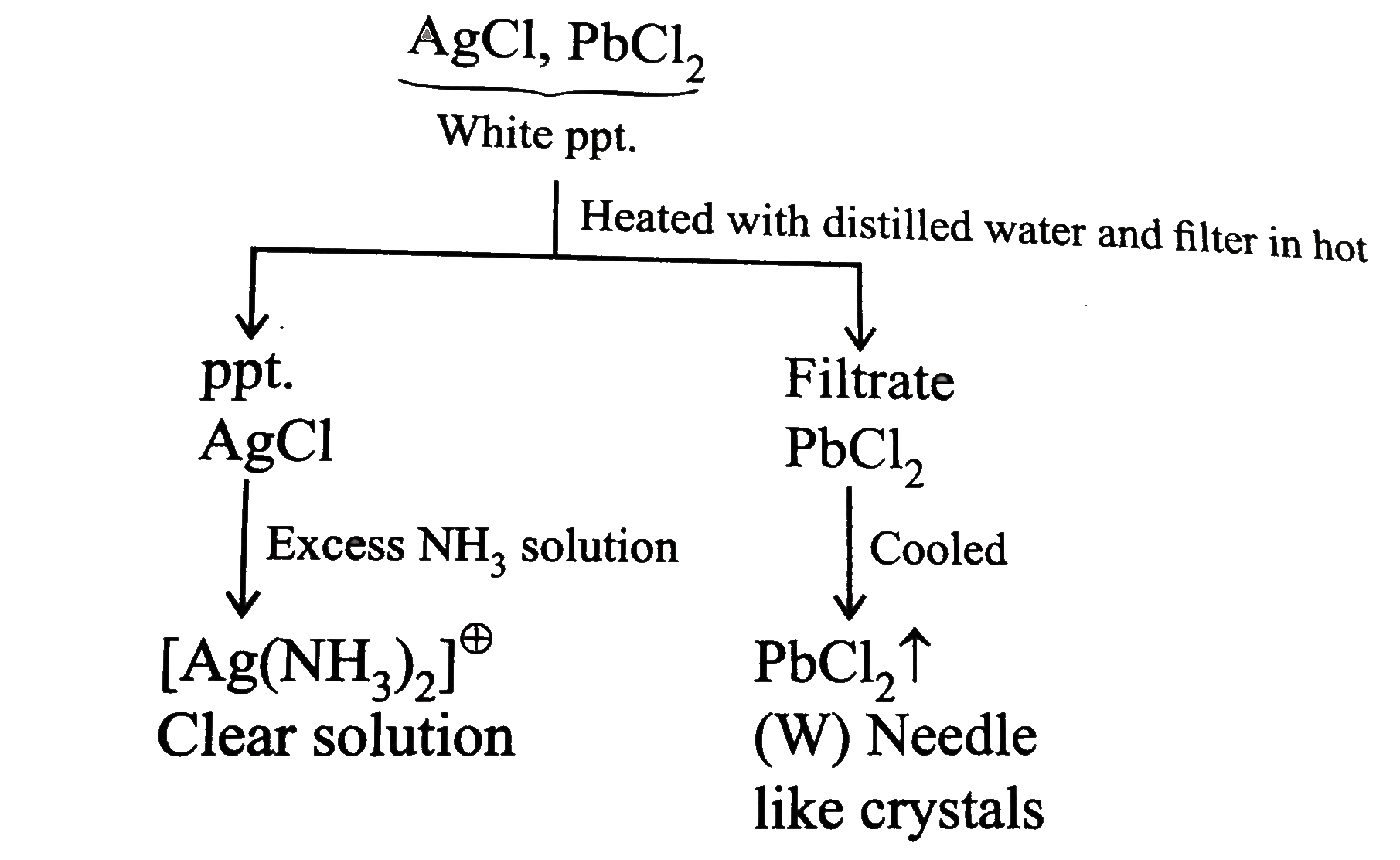
|
|
| 38. |
Salt (A) makes part of electrode and is insoluble in water (A) is blackened by NH_(3) forming (B),(B) is soluble is equa regia forming (C ),(C ) gives orange ppt with KI but ppt dissolves in extcess of KIforming (D ) , identify (A),(B),(C ) and (D) |
|
Answer» Solution :(A) `Hg_(2)CI_(2)` (CALOMEL) (B ) `(HgNH_(2)CI+ Hg)` (C ) `HgCI_(2)`(D) `H_(2)HgI_(4)` |
|
| 39. |
Salt (A) is a white deliquescent solid. On heating the solid, brown fumes were evolved. On adding NaOH to the salt solution of (A), a white gelatinous ppt. was obtained. On adding, a drop of blue litmus solution to the ppt., a blue lake was obtained. On adding DPA to the salt solution, a deep blue colouration was obtained. Identify the salt and give the equations involved. |
|
Answer» Solution :(i) `2AL(NO_(3))_(3)OVERSET(Delta)toAl_(2)O_(3)+6NO_(2)uarr+(3)/(2)O_(2)uarr` `(ii) `Al(NO_(3))_(3)+3NaOHtoAl(OH)_(3)darr+3NaNO_(3)` `Al(OH)_(3)+"Blue litmus solution" to"Blue lake"` [Note: A lake is a ppt. floating in a clear liquid.] (iii) `NO_(3)^(-)+(C_(6)H_(5))_(2)NHtoNO_(2)^(-)+(C_(6)H_(3))_(2)N-N(C_(6)H_(5))_(2)+H_(2)O` |
|
| 40. |
Salol (phenyl salicylate) is used as an : |
|
Answer» Insecticide |
|
| 41. |
Salol can be used as: |
|
Answer» ANTISEPTIC |
|
| 42. |
Salol can be used as : |
|
Answer» ANTISEPTIC |
|
| 43. |
Saliva contains: |
|
Answer» Amylases |
|
| 44. |
Saline soil contains : |
|
Answer» HIGH CONCENTRATION of salt |
|
| 45. |
Salicylic acid on brornination gives: |
|
Answer» 2,4,6-tribromosalicylic ACID |
|
| 46. |
Saline hydrides are : |
|
Answer» STRONG oxidants |
|
| 47. |
Salicylic acid on heating with sodalime forms: |
|
Answer» PHENOL |
|
| 48. |
Salicylic acid on heating with soda lime (CaO+NaOH) gives : |
|
Answer» Benzene 
|
|
| 49. |
Salicylic acid is produced when phenol in alcoholic KOH is treated with |
| Answer» Answer :D | |