Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Schiff's reagent gives pink colour with |
|
Answer» Aldehydes |
|
| 2. |
Schiff's bases or anils are formed when aniline reacts with : |
|
Answer» ALCOHOLS |
|
| 3. |
Schiff's reagent gives pink colour with : |
|
Answer» ALCOHOLS |
|
| 4. |
Schiff's bases are formed when aniline reacts with |
|
Answer» Aromatic aldehydes |
|
| 5. |
Schiff's base is substituted |
|
Answer» imine |
|
| 6. |
Schiff's base and Schiff's reagent are: |
|
Answer» Same compounds |
|
| 7. |
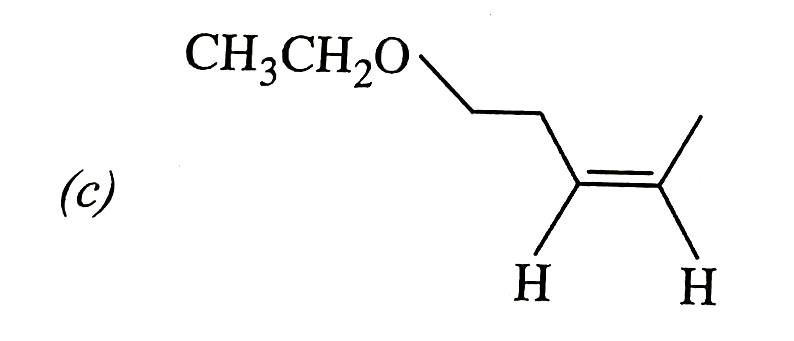
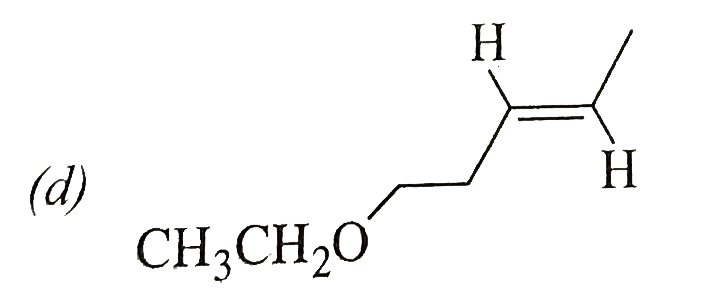
Schemes 1 and 2 decribe sequential transformation of alkynes M and N. Consider only the major products formed in each step for both the schemes. The product X is : |
|
Answer»
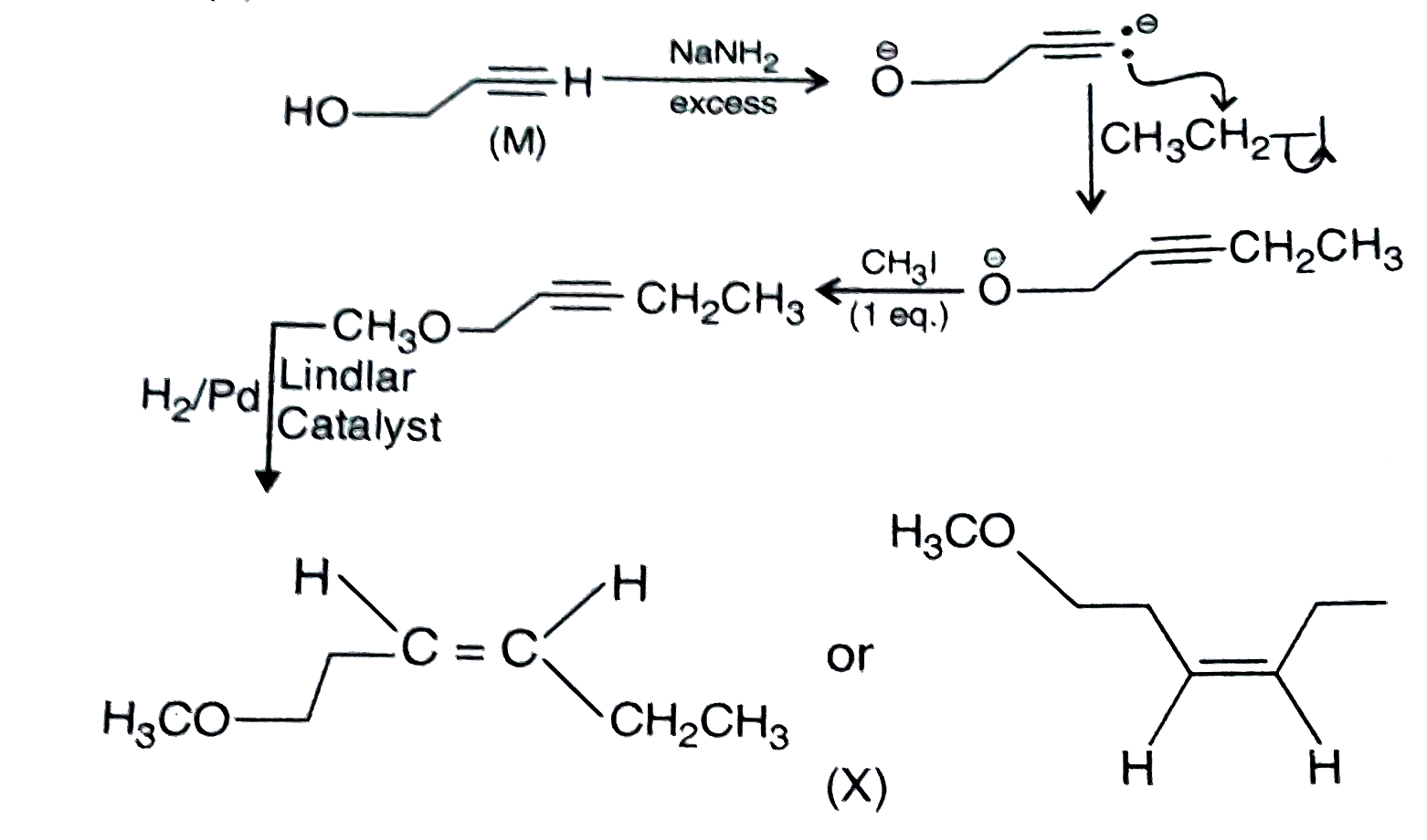
|
|
| 8. |
Schiff bases or anils are formed, when aniline reacts with |
|
Answer» ALIPHATIC ALDEHYDES `{:(ArCHO+RNH_(2)RARR ArCH(OH)NHR),(""darr-H_(2)O),(""ArCH=NR),("""Schiff-base"):}` |
|
| 9. |
Schemes 1 and 2 decribe sequential transformation of alkynes M and N. Consider only the major products formed in each step for both the schemes. The correct statement with respect to product Y is : |
|
Answer» It gives a positive TOLLEN's test and is a functional isomer of X. 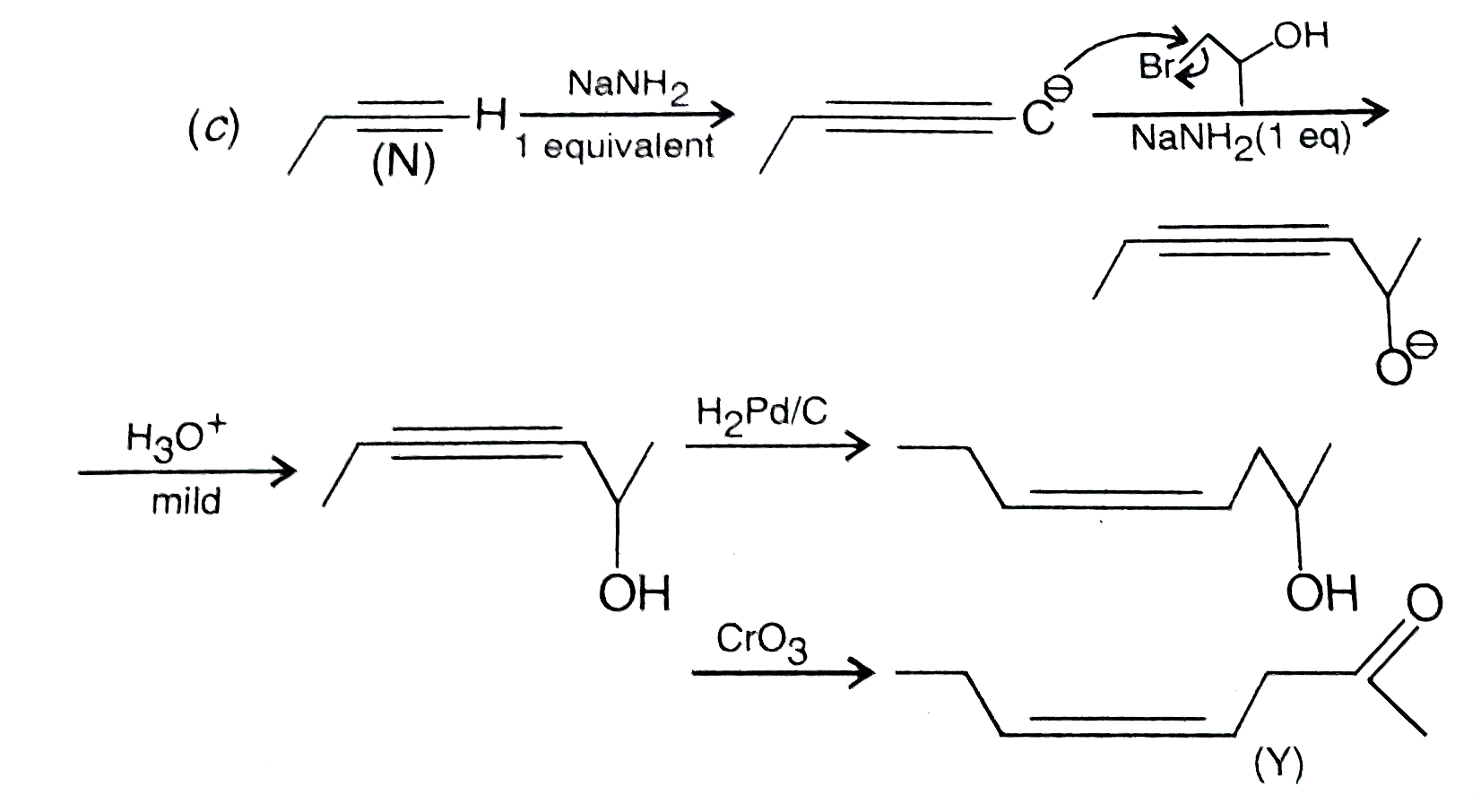 Both (X) and (Y) are functional isomers. COMPOUND (Y) gives positive iodoform test (yellow ppt.) |
|
| 10. |
Schematic alignment of magnetic moments of ferromagnetic, antiferromagnetic and ferrimagnetic substances are given below. Identify each of them. |
| Answer» SOLUTION :FERROMAGNETIC | |
| 11. |
Scheel's green, formerly used as a green pigment for colouring wall paper is. |
|
Answer» SODIUM ARSENITE `(Na_3AsO_3)` |
|
| 12. |
Schematic alignment of magnetic moments of ferromagnetic, antiferromagnetic and ferrimagnetic substances are given below. Identify each of them. |
| Answer» SOLUTION :FERRIMAGNETIC | |
| 13. |
Schematic alignment of magnetic moments of ferromagnetic, antiferromagnetic and ferrimagnetic substances are given below. Identify each ofthem. |
| Answer» Solution :i)antifero magnetism ii)Ferrimagnetism III)FERROMAGNETISM | |
| 15. |
Scarlet flame colour of bunsen flame is characteristic of : |
|
Answer» Sn |
|
| 16. |
Scandium resembles with aluminium in the following properties except : |
|
Answer» the NATURE of bonding in both `Al^(3+)` and `Sc^(3+)` compounds in MAINLY ionic |
|
| 17. |
Scandium forms no coloured ionis, yet it is regarded as a transition elements. Explain why ? |
| Answer» Solution :Scandium in the ground state has ONE ELECTRON int eh 3d-subshell . HENCE,it is regarded as a transitio elements. However, in its common oxidation state `+3`, it has no electron in 3dsusshell `( 3d^(0))` .Hence, it does not FORM coloured ion. | |
| 18. |
Sc^(3+), Ti^(4+), V^(5+) are diamagnetic. Give reason. |
|
Answer» SOLUTION :(i) `Sc^(3+), Ti^(+4), V^(5+)`have `d^(0)` ELECTRONIC configuration. n = 0 (ii) `mu = sqrt(0(0+2)) = 0 mu_(B)`.So they are diamagnetic. |
|
| 19. |
Sc (Z=21) is a transition elements but Zn(Z=30) is not because…. |
|
Answer» Both `Sc^(3+) and Zn^(2+)` ions are colourless and form with compounds |
|
| 20. |
Sc^(3+) ions are colourless where as V^(3+) ions are coloured. Give reason. |
|
Answer» Solution :`SC^(3+)` ions does not contain any UNPAIRED electrons HENCE d-d transition does not TAKE place. `V^(3+)` ION contain two unpaired electrons, hence d-d transition takes place. Therefore `V^(3+)` ions are coloured. |
|
| 21. |
Sc (Z = 21) is a transition element but Zinc (z = 30) is not because |
|
Answer» both `Sc^(3+)` and `Zn^(2+)` ions are colourless and form white compounds, |
|
| 22. |
Sc (Z = 21) is a transition element but Zn(Z = 30) is not because |
|
Answer» Both `SC^(3+)` and `ZN^(2+)` ions are colourless and form white compounds |
|
| 23. |
SbF_(5) reacts with XeF_(4) and XeF_(6) to form ionic compounds [XeF_(3)^(+)][SbF_(6)^(-)] and [XeF_(5)^(+)][SbF_(6)^(-)\ then molecular shape of [XeF_(3)^(+)] ion and [XeF_(5)^(+)] ion respectively : |
|
Answer» SQUARE PYRAMIDAL, T-shpaed 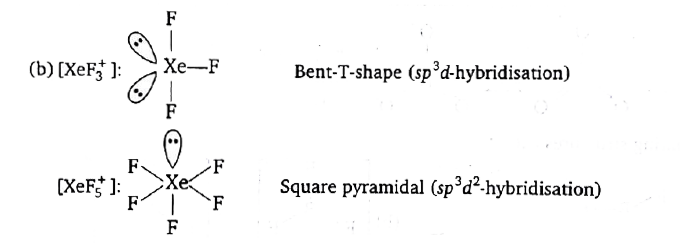
|
|
| 24. |
SBR(GRS,Buna-S,Cold Rubber)is obtained by free radical intiator.The most commenly used free radical initiator is: |
|
Answer» Buta`-1,3-` dinene `(70%)` and `30%` phenyl ethene (STYRENE) |
|
| 25. |
SbF_(5) reacts with XeF_(4) to form an adduct. The shapes of cation and anion in the adduct are respectively : |
|
Answer» square PLANAR, TRIGONAL bipyramidal |
|
| 26. |
[Sc (H_(2)O)_(6)]^(3+)ion is :- |
|
Answer» COLOURED and paramagnetic |
|
| 28. |
Saturated solution of KNO_(3) is used to make 'salt-bridge' because |
|
Answer» Velocity of `K^(+)` is greater than that of `NO_(3)^(-)` |
|
| 29. |
Saturated solution of NaCl on heating becomes: |
|
Answer» SUPER SATURATED |
|
| 30. |
To form a super saturated solution of salt one must: |
|
Answer» SUPER SATURATED |
|
| 31. |
Saturated solution of AgCl at 25^(@)C has specific conductance of 1.12xx10^(-6)ohm^(-1)cm^(-1). The lamda_(oo)Ag^(+) and lambda_(oo)Cl^- and 54.3 and 65.5 ohm^(-1)cm^2// equi. Respectively. Calculate the solubility product of AgCl at 25^(@)C |
|
Answer» |
|
| 32. |
Saturated solution of KNO_(3) is used to make salt bridge because: |
|
Answer» velocity of `K^(+)` is greater than that of `NO_(3)^(-)` |
|
| 33. |
Saturated solution of KCl (or) Na_2SO_4 is used to make salt bridge because |
|
Answer» VELOCITY of `K^+` is greater than that of `CL^-` |
|
| 34. |
Saturated solution of KNO_3 is used to inake salt bridge because |
|
Answer» VELOCITY of `K^(+)` is GREATER than that of `NO_(3)^(-)` |
|
| 35. |
Saturated monocarboxylic acid is second oxidative product of |
| Answer» Answer :C | |
| 36. |
Saturated mono carboxylic acid are also called |
|
Answer» Paraffin |
|
| 37. |
Saturated fatty acids are represented by which of the forumula |
|
Answer» `C_(n)H_(n)O_(2)` |
|
| 38. |
Saturatd solution of KNO_3 is used to mke salt bridge because . |
|
Answer» Velocity of `K^(+)` is GREATER than that of `NO_3^(2)` |
|
| 39. |
Sapphire is a valuable precious stone containing___________. |
|
Answer» Cu |
|
| 40. |
Saponiflication of ethyl acetate (CH_(3)COOC_(2)H_(5)) by NaOH is studied by titration of the reaction mixture, have 1 : 1 molar ratio of the reactants. If 10ml of 1 N HCl is required by 5ml of the solution at the start and 8ml of 1N HCl is required by another 5ml after 10 minutes, then rate constant is |
|
Answer» `k=(2.303)/(10)log.(10)/(8)` |
|
| 42. |
Saponification of ethyl benzoate with caustic soda as alkali gives |
|
Answer» Benzyl alcohol and ethanoic ACID |
|
| 43. |
Saponification of ethyl acetate (CH_(3)COOC_(2)H_(5)) by NaOH (Saponification of ethyl acetate by NaOH is second order reaction) is studied by titration of the reaction mixture initially having 1:! molar ratio of the reactants. If 10 mL of 1 N HCl is required by 2 mL of the solution at the start and 8 mL of 1 N HCl is required by another 5 mL after 10 minutes, then rate constant is : |
|
Answer» `K=(2.303)/(10)"LOG"(10)/(8)` |
|
| 44. |
Saponificationof an ester(A) followed by neutralizationgivesa compound (B), Which givesvioletcolourationwithFeCl_(3). The ester (A) is . |
| Answer» Answer :D | |
| 45. |
Sanger's reagent reacts with functional group in a peptide ? |
|
Answer» FREE amino groups 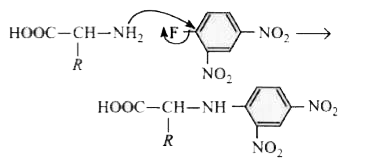
|
|
| 46. |
Saponification means hydrolysis of an ester with : |
|
Answer» Enzymc |
|
| 47. |
Sanger's reagent is used for the identification of |
|
Answer» N -terminal of a peptide chain |
|
| 48. |
Sandmeyer'sreactionof benzene diazoniumcholdrideis usedin thepreparationof |
| Answer» Answer :A | |
| 49. |
Sandmeyer's reaction occurs in the presence of………and…………. |
|
Answer» CUPROUS HALIDE and haloacid |
|