Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 3. |
Rusting of iron is a chemical reaction . The reaction is : |
|
Answer» Analysis |
|
| 4. |
Rusting of iron becomes quicker in saline medium. Explain. |
| Answer» Solution :Saline MEDIUM has extra salts such as sodium chloride dissolved in water. This means that it has a GREATER concentration of ELECTROLYTE than ordinary medium. The ions present will favour the formation of more electrochemical CELLS and will thus promote rusting or CORROSION. | |
| 5. |
Rusting of iron in moist air involves: |
|
Answer» LOSS of ELECTRONS by Fe |
|
| 6. |
Rust is a mixture of |
|
Answer» `FEO and FE(OH)_(2)` |
|
| 7. |
Rust is a mixture of ________. |
|
Answer» `Fe_(2)O_(3)+FE(OH)_(3)` |
|
| 8. |
Rust is |
|
Answer» `FeO+FE(OH)_(2)` |
|
| 9. |
Rust is mixture of: |
|
Answer» `FEO` and `Fe(OH)_3` |
|
| 10. |
Rust is a mixture of : |
|
Answer» `Fe_2O_3cdotxH_2O` |
|
| 11. |
Ruby is aluminium oxide (Al_(2)O_(3)) containing about 0.5-1% Cr^(3+) ions (d^(3)), which are randomly distributed in positions normally occupied by Al^(3+) choose the only incorrect statement : |
|
Answer» `Cr^(3+)` is incorporated into the alumina lattice as octahedral chromium (III) complexes |
|
| 12. |
Ruby is alumina containing 1% chromic salt. Discuss the type of complex involved in the colour. |
| Answer» Solution :OCTAHEDRAL COMPLEX with `d^(3)` configuration of `CR^(3+)` | |
| 13. |
Rubber plating and chrome tanning of leather are based upon: |
|
Answer» Electro-osmosis |
|
| 15. |
Rubberlatex is which type of emulsion |
|
Answer» OIL in oil |
|
| 16. |
Rubber latex is which type of emulsion |
|
Answer» OIL in oil TYPE |
|
| 17. |
Rubber latex is |
|
Answer» Emulsion of polyhydrocarbon droplets in an aqueous solution |
|
| 18. |
Rubber is heated with sulphur and the process is known: |
|
Answer» Galvanization |
|
| 19. |
Rubber is a natural polymer obtained from the bark of rubber trees vuclanisation improves elasticity of rubber. What is vulcanisation? |
| Answer» SOLUTION :HEATING RUBBER with SULPHER is KNOWN as vuclanisation | |
| 20. |
Rubber is a solidsol |
| Answer» SOLUTION :Solidfoam | |
| 21. |
Rubber is a natural polymer obtained from the bark of rubber trees Name the monomer of natural rubber |
| Answer» SOLUTION :ISOPRENE (2-methyl I-I, 3 BUTADIENE) | |
| 23. |
Rubber is a ......... colloid. |
|
Answer» LYOPHOBIC |
|
| 24. |
Ru and Os have highest oxidation state in which compounds? Explain with example. |
|
Answer» Solution :(i) Ru and Os have +8 as the highest oxidation STATE. (ii) The highest oxidation state of 4d and 5d elements are FOUND in their compounds with the higher electronegative elements like O, F and Cl. For example: `RuO_(4), OsO_(4)` |
|
| 25. |
Replacement of Cl of Chlorobenzene to give phenol requires drastie conditions, but Cl of 2, 4 -dinitro chlorobenzene is readily replaced. This is because, |
|
Answer» `-NO_(2)` group makes the ring electron RICH at ORTHO and para positions |
|
| 26. |
R.R.D. sir had a great interest in performing the experiment when he used to study at IT BHU. Once He saw the dissociation of PCl_5and concluded that it was 40% dissociated at 4 atm. Calculate the pressure at which he will see 80% dissociation of PCl_5, temperature remaining the same ? |
| Answer» SOLUTION :(0.43 ATM) | |
| 27. |
Round up upto three significant figures : (i) 34.216 (ii) 10.4107 (iii) 0.04597 (iv) 2808 |
| Answer» SOLUTION :`"(i) 34.2(II) 10.4(III) 0.0460(IV) 2810"` | |
| 28. |
Rotational energy levels of diatomic molecules are well described by the formula E_(J)=BJ(J+1), where J is the rotational quantum number of the molecule and B its rotational constant. B is related to the reduced mass mu and the bond length R of the molecule through the equation B=h^(2)/(8pi^(2)muR^(2)). In general, spectroscopic transitions appear at photon energies which are equal to the energy difference between appropriate states of a molecule (hv=DeltaE). The observed rotational transitions occur between adjacent rotational levels, hence DeltaE=E_(J+1)-E_(J)=2B(J+1). Consequently, successive rotational transitions that appear on the spectrum (such as the one shown here) follow the equation h(Deltaupsilon)=2B. By inspecting the spectrum provided, determine the following quantities for ^(2) C ^(p)C with appropriate units (a) Deltaupsilon (b) B (c) R |
|
Answer» {:b) `B=(hDeltav)/(2)=(6.63xx10^(-34)xx115'xx10^(9))/(2)=3.81xx10^(-23) J` {:c) `mu=(m(C)xxm(O))/(m(CO))=(12xx16)/(28)=6.86 a.u.=1,14 xx10^(-26) kg` For interatomic distance R: `R=(H)/(2pisqrt(2muB))=(3.63xx10^(-34))/(2xx3.14sqrt(2xx1.14xx10^(-26)xx3.81xx10^(-23)))=1.13xx10^(-10) m =1.13 Å` |
|
| 29. |
Rotation of plane polarized light can be measured by: |
|
Answer» Manometer |
|
| 30. |
Rotation of plane polarised light is measured by |
|
Answer» Manometer 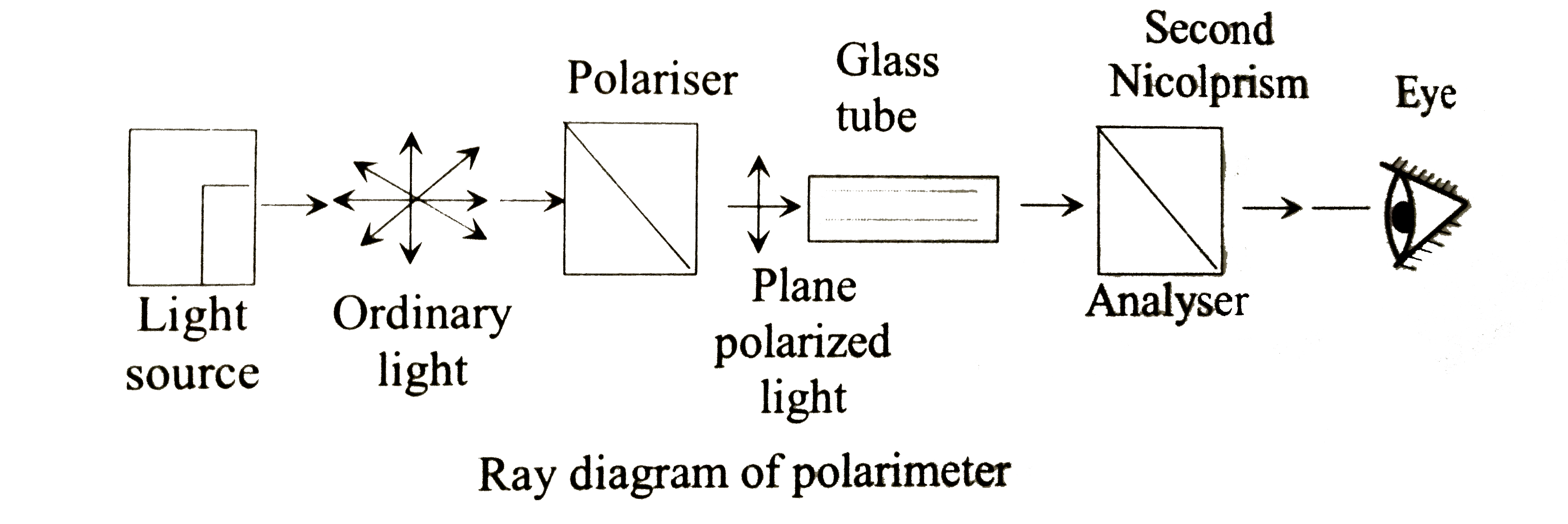
|
|
| 31. |
Rotation around the bond (between the underlined atoms) is restricted in : |
|
Answer» `ulC_2H_4` |
|
| 32. |
Rosstingofsulphidesgivesthe gas X as a byproduct .The is a colourless gaswithchoking small of burnt sulphurand cause great damage to respectively ornge as a result of acid rain its aquneous solutionis acidic , acts as a reducing agent and its acid has neverisolated .The gas X is |
|
Answer» `CO_(2)` i. Sulphideson reastingnecverform `SO_(3)` since in is PRODUCED byreacting `SO_(2)` with `O_(2)` in prenceof a catalyat`(PT , or V_(2)O_(3))` and atoptimumtemperature `(720K)` andhighpresence `(2 bar)`  ii. `SO_(2)` and `SO_(2)` bothare COLOURLESS gas but `SO_(2)` has achoking smell of `SO_(2),NO_(2)` and `SO_(2)`from atmosphareafteroxidation and reactionwithwater aremajorcontributor to acid rain `2SO_(2(G)) + O_(2(g)) + 2H_(2)O_((1)) rarr2H_(2)SO_(4(Aq))` `2NO_(2(g)) +_O_(2(g)) + 2H_(2)O_((1))RARR 2HNO_(3(aq))` So `X` bo `SO_(2)` not `SO_(3)` iii. Aqueoussolution of both `SO_(2)` and `SO_(3)` are acidic `SO_(2)` acts as reducingagent (undergo) oxidationfrom `+4 to +6`oxidation sate) but `SO_(4)` does acts as readicingagent (i.e. it has maximum oxidation sate of `+6)` So `X` can be `SO_(2)` bot `SO_(2)` iv `SO_(2)` reacts with `H_(2)O` as given below and `H_(2)SO_(3)` isneverisolated  Hence the anwer is (d), i.e. `SO_(2)` |
|
| 33. |
Rosenmund's reduction is used to prepare |
|
Answer» ALCOHOL |
|
| 34. |
Rosenmund's reductionconvert |
|
Answer» carboxylic ACID to aldehyde |
|
| 35. |
Rosenmund's reductioncarried out by using |
|
Answer» `H_(2)//Ni` |
|
| 36. |
Rosenmund reduction of acyl chloride gives : |
|
Answer» an ALCOHOL |
|
| 37. |
Rosenmund reaction can be used to obtain: |
|
Answer» Alkanes |
|
| 38. |
Rose metal is an alloy of : |
| Answer» Answer :A | |
| 39. |
Rosasting of copper pyrites is done: |
|
Answer» to remove MOISTURE and volatile impurities |
|
| 40. |
Root of valerian plant contains |
|
Answer» `C_(3) H_(7) COOH` |
|
| 41. |
Roosting is carried out to |
|
Answer» CONVERT sulphide to oxide and sulphate |
|
| 42. |
Role of limestone used in re extraction |
|
Answer» OXIDATION of FE ore |
|
| 43. |
Role of ATP in bioenergetics is |
|
Answer» releasing energy |
|
| 44. |
ROH cannot be converted into RCl on treatment with KCl, however reaction takens place on treatment with HCl. Explain |
| Answer» SOLUTION :`Cl^(-)` is a WEAK nuleophile than `OH^(-)` and hence cannot replace. HOWEVER when Hcl is added, `H^(+)` of HCl protonates on oxygen atom and `H_(2)O` becomes good LEAVING group and gives `R-Cl` | |
| 45. |
Rochelle salt is |
|
Answer» AMMONIUM tartarate |
|
| 46. |
Roasting of sulphides gives the gas X as a byproduct. This is a colourless gas with choking smell of burnt sulphur and causes great damage to respiratiory organs as a result of acid rain. Its aqueous solution is acidic, acts as a reducing agent and its acid has never been isolated. The gas X is |
|
Answer» `CO_(2)` `{:(""2ZnS + 3O_(2)rarr 2ZnO + underset((X))(2SO_(2))),(2SO_(2)+2H_(2)O+O_(2)rarr 2H_(2)SO_(4)("acid rain")),(""SO_(2)+H_(2)O rarr underset("SULPHUROUS acid")(H_(2)SO_(3))):}` Sulphurous acid acts as a reducing agent. Being unstable it has NEVER been isolated. |
|
| 47. |
Roasting results in the production of metal in the case of : |
|
Answer» IRON PYRITES |
|
| 48. |
Roasting process is applied to which of the following ores |
|
Answer» Galena |
|
| 49. |
Roasting of sulphide ore is done before smelting |
|
Answer» to remove moisture and volatile impurities |
|