Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Rice is deficient in : |
|
Answer» Lysine |
|
| 2. |
Rice has deficiency of the essential amino acid: |
|
Answer» Alanine |
|
| 3. |
Ribulose is an exampleof ………….. |
| Answer» SOLUTION :KETOPENTOSE | |
| 4. |
Ribulose is ……………. type of sugar |
|
Answer» ALDOPENTOSE |
|
| 5. |
Ribose is an example of ……… . |
|
Answer» keto hexose |
|
| 7. |
Ribose is an example of an |
|
Answer» aldopentose |
|
| 8. |
Riboseand 2 - deoxyribosecanbe differentiated by . |
|
Answer» Fehling's REAGENT  In deoxyribose , one - OH GROUP is MISSING, WHICHWILL pervent the FORMATIONOF osazone. |
|
| 9. |
Ribose is _______ |
|
Answer» KETOPENTOSE |
|
| 11. |
Rhombic sulphur has the following structure |
|
Answer» OPEN chain |
|
| 12. |
Rhombic and monoclinic sulphur are: |
|
Answer» Isobars |
|
| 13. |
[RhF_(6)]^(3-) complex ion is : |
|
Answer» Outer orbital complex |
|
| 14. |
Review the equilibrium and choose the correct statement HClO_(4) + H_(2)O harr H_(3)O^(+) + ClO_(4)^(-) |
|
Answer» `HClO_(4)` is the conjugate ACID of `H_(2)O` 
|
|
| 15. |
Review the equilibrium and choose the correct statement. HClO_(4)+H_(2)OhArr H_(3)O^(+)+ClO_(4)^(-) |
|
Answer» `HClO_(4)` is the conjugate ACID of `H_(2)O` |
|
| 16. |
Reversible and Irreversible Process. |
| Answer» Solution :`{:("REVERSIBLE process","Irreversible process"),("1. The process whose direction can be reversed at any stage by an infinitesimal increase in the opposing force is called a reversible process.","1. the process whose direction cannot be reversed by an infinitesimal increase in the opposing force is called an irreversibel process."),("2. Such a process is not spontaneous and takes place infinitesimally slowly and takes INFINITE time for completion.","2. Such a process is spontaneous and takes finite time for completion."),("3. In this process, the thermodynamic equilibrium is ALWAYS maintained between the system and the surroundings at EVERY step.","3. The thermodynamic equilibrium is attained only at the end of the process."),("4. The opposing force is infinitesimally less than the driving force.","4. The opposing force is significantly less than the driving force."),("5. It is an ideal or hypothetical process.","5. It is a practical or real and spontaneous process."):}` | |
| 18. |
Reverberatory furnace is epmloyedin the metallurgincalprocess mainly for |
|
Answer» REDUCTION of oxide ores |
|
| 19. |
Reverberatory furnace. |
Answer» SOLUTION :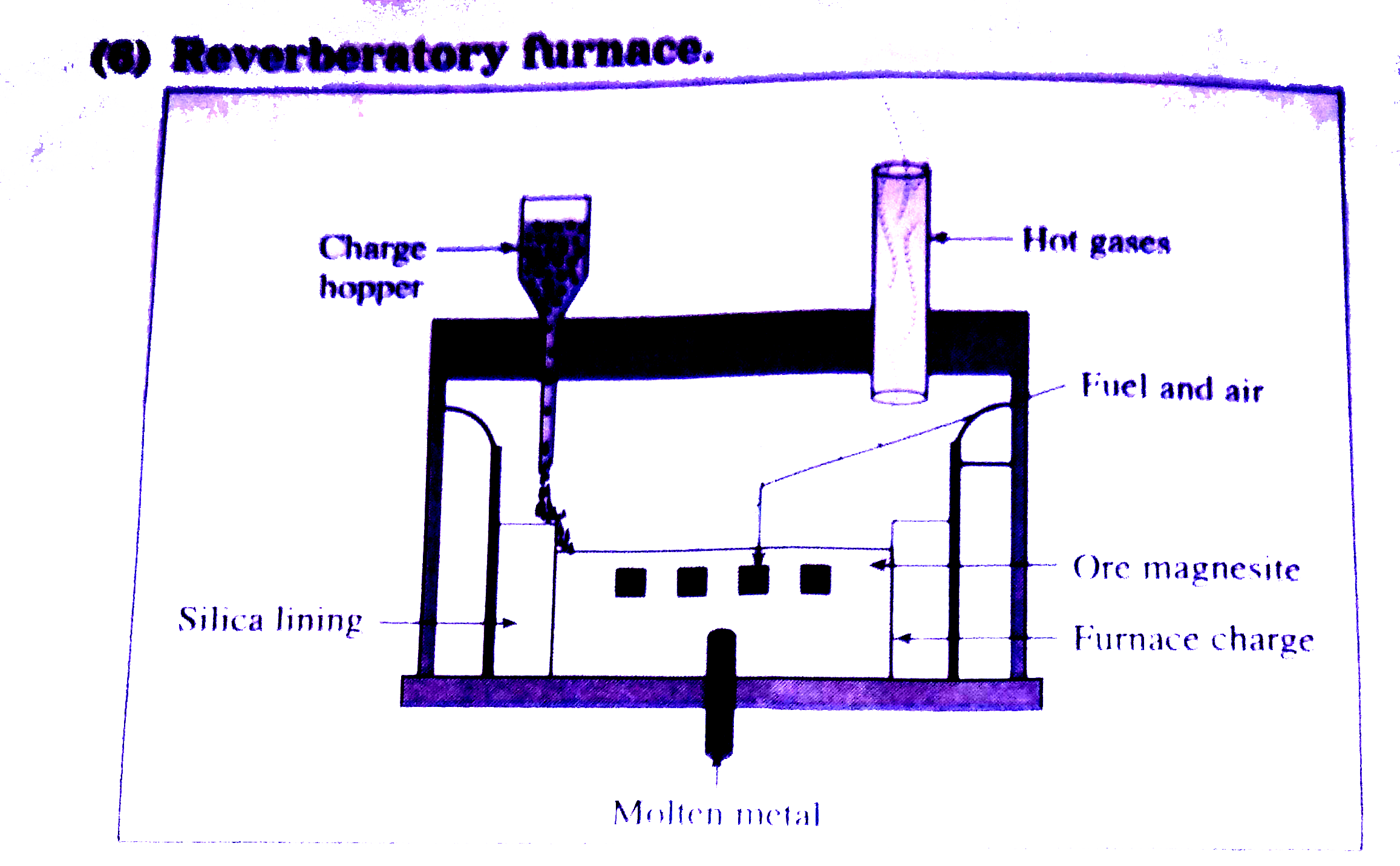
|
|
| 20. |
Resultant solution of electrolysis of concentrated NaCl solution is. . . |
|
Answer» Do not change colour of RED or BLUE LITMUS paper. |
|
| 22. |
Rest mass of 1 mole neutrons (m_(n)=1.675xx10^(-27)kg) is: |
|
Answer» `1.8xx10^(-3)KG` `=1.675xx10^(-27)xx6.023xx10^(23)` `=1.008xx10^(-3)kg` |
|
| 23. |
Respiration ultimately results in : |
|
Answer» `CO_2 + O_2` |
|
| 26. |
Resonance structure of molecule does not have |
|
Answer» Identical arrangement of ATOMS |
|
| 28. |
Resonance is due to : |
|
Answer» DELOCALIZATION of SIGMA electrons |
|
| 29. |
Resonance occurs due to |
|
Answer» IDENTICAL ARRANGEMENT of atoms |
|
| 30. |
Resonance in benzene is accompained by delocalisation of pi- electron is attached with: |
|
Answer» 4 carbon |
|
| 32. |
Resonance energy of benzene is 36 kcal mol^(-1). This means |
|
Answer» The heat of hydrogenation of BENZENE is 36 KCAL LOOK for DEFINATION of resonance energy |
|
| 33. |
Resonance effect is not observed in |
|
Answer» `CH_(2)=CH-C=N` |
|
| 34. |
Resonance arises due to the: |
|
Answer» MIGRATION of atoms |
|
| 35. |
Resistance of cell having 0.02 M KCl solution is 164 Omega. If 0.05 M AgNO_(3) is filled then resistance of cell become 75.8 Omega, then calculate following : [Conductivity of 0.02 M KCl=2.768xx10^(-3)Omega^(-1)cm^(-1)] (i) Conductivity of 0.05 M AgNO_(3). (ii) Molar conductivity of AgNO_(3) solution. |
|
Answer» (II) `Lamda_(m)(AgNO_(3))=115.6Omega" "cm^(-1)mol^(-1)` |
|
| 36. |
Resistnace of a conductivity cell at 298K for 0.0100 M KCl is 161.8Omega. Resistance become 190Omega when 0.005 M NaOH solution is filled in this cell. Then calculate (i) cell constant (ii) specific conductivity for NaOH solution and (iii) molar conductivity. |
|
Answer» (ii) `1.2xx10^(-3)Omega^(-1)CM^(-1)` and (iii) `240" S "cm^(2)mol^(-1)` |
|
| 37. |
Resistivity of 0.5 M electrolytic solution is 10Omegacm, then find out the molar conductivity of solution. |
|
Answer» SOLUTION :`Lamda_(m)=`molar CONDUCTIVITY of solution `KAPPA`=specific conductivity of solution p=resistivity of solution=`10Omegacm=0.1Omegacm` `kappa=(1)/(p)=(1)/(10)=0.1Omega^(-1)cm^(-1)=0.1" S "cm^(-1)` CONCENTRATION of solution : `c=0.5" MOL "L^(-1)=0.5" mol "cm^(-3)` `=1000xx0.5" mol "m^(-3)` `=500.0" mol "m^(-3)` `Lamda_(m)=(1000kappa)/(c)=(1000xx0.1" S "cm^(-1))/(0.05" mol "cm^(-3))` `=200" S "cm^(3)mol^(-1)` |
|
| 38. |
Resistance of conductor having unit meter length and 1 square meter cross sectional area is known as. . . |
|
Answer» SPECIFIC RESISTANCE |
|
| 39. |
Resistance of any equal conductor is . . . . |
|
Answer» Inversely proportional to its LENGTH So, `R prop (l)/(A)`. |
|
| 40. |
Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1M is 100Omega. The conductivity of this solution is 1.29 S m^(-1). Resistance of the same cell when filled with 0.2M of the samesolution is 520Omega. the molar conductivity of 0.02M solution of the electrolyte will be |
|
Answer» `1.24xx10^(-4)Sm^(2)"mol"^(-1)` `=1.29S m^(-1)xx100Omega=129m^(-1)` In the 2nd CASE `x=("cell constant")/("Resistance")` `=(129m^(-1))/(520Omega)= 0.248Omega^(-1)m^(-1)` `^^_(m)=(x)/("MOLARITY")=(0.248Omega^(-1)m^(-1))/(0.02xx10^(3)"mol"m^(-1))` `=124xx10^(-4)Sm^(2)"mol"^(-1)` |
|
| 41. |
Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1M is 100Omega . The conductivity of this solution is 1.29 S m^(-1) . Resistance of the same cell when filled with 0.02M of the same solution is 520 Omega . The molar conductivity of 0.02M solution of the electrolyte will be |
|
Answer» `12.4 XX 10^(-4) Sm^2. "mol"^(-1)` |
|
| 42. |
Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1 M is (100Omega). The conductivity of this solution is 1.29sm^(-1).Resistance of the same cell when filled with 0.2 M of the same solution is 520Omega . The molar conductivity of (0.02)M solution of the electrolyte will be : |
|
Answer» ` 124xx10^(-4) sm^(2) MOL^(-1)` For 0.1M solution `G^ast= (1.29)/(G) = 129m^(-1)`For 0.2M solution `K= Gxx(1)/(R ) = (129)/(520) sm^(-1)` If we DILUTE 0.2 M solution by 10 times, its concentration becomes 0.02 M. Thus number of ions PER unit volume is `(1)/(10)`th of INITIAL one. So K for this solution is `(129)/(520xx10) SM^(-1)` |
|
| 43. |
Resistance of a conductivity cell filled with 0.1 mol L^(-1) KCl solution is 100Omega. If the resistance of the same cell when filled with 0.02 mol L^(-1) KCl solution is 520Omega, calculate the conductivity and molar conductivity of 0.02 M KCl solution. The conductivity of 0.1 M KCl solution is 1.29S/m. |
|
Answer» SOLUTION :(i) For 0.1 M KCL solution, `R=100Omega,kappa=1.29" S "m^(-1)` ,brgt Cell constant=Conductivity`xx`Resistance`=1.29" S "m^(-1)xx100Omega=129m^(-1)xx100Omega=129m^(-1)(S=Omega^(-1))=1.29cm^(-1)` (ii) Conductivity of 0.02 M KCl solution `(kappa)=("Cell constant")/("Resistance")=(129m^(-1))/(520Omega)=0.248Omega^(-1)m^(-1)` or `0.248" S "m^(-1)` Concentration of the solution `=0.02M=0.02" mol "L^(-1)=0.02xx10^(3)" mol "m^(-3)=20" mol "m^(-3)` Molar conductivity`=(kappa)/(c_(m))=(0.248Sm^(-1))/("20 mol "m^(-3))=0.0124" S "m^(2)mol^(-1)=1.24xx10^(-2)SM^(2)mol^(-1)` |
|
| 44. |
Resistance of a conductivity cell filled with 0.1 mol L^(-1) KCl solution is 100Omega. If the resistance of the same cell when filled with 0.02 mol L^(-1) KCl solution is 520 Omega, calculate the conductivity and molar conductivity of 0.02 mol L^(-1) KCl solution. the conductivity of 0.1 mol L^(-1) KCl solution is 1.29 S/m. |
|
Answer» Solution :Calculation for cell constant `G^(**)`: Cell constant `G^(**)`=conductivity `xx`resistance Where, resistivity of resistance cell `R=100Omega` Resistivity of solution `K=1.29"S "m^(-1)` `therefore G^(**)=(1.29"S "m^(-1))xx100Omega` `=129" S "OMEGA" "m^(-1)""["but "1S=1Omega^(-1)]` `=129m^(-1)""["but "1m^(-1)=0.01cm^(-1)]` `therefore G^(**)=1.29cm^(-1)` Calculation for conductivity (k) of KCl solution 0.02 mol `L^(-1)`: conductivity of 0.02 mol `L^(-1)` KCl solution=cell constant/resistance Resistivity of cell `(k)=("Cell constant "(G^(**)))/("Resistance of solution (R)")` Where, `G^(**)=129m^(-1),R=520Omega` `therefore k=(129m^(-1))/(520Omega)` `=0.248Omega^(-1)m^(-1)` `=0.248S" "m^(-1)` `=0.248xx10^(-2)" S "CM^(-1)""(because 1m^(-1)=10^(-2)cm^(-1))` Calculation for molar conductivity `Lamda_(m)` of solution: `Lamda_(m)=(k)/(c)=("Resistivity of solution")/("Molarity of solution")` Molarity of solution `c=0.02" mol "L^(-1)` [But `L^(-1)=1000m^(-1)`] `=1000xx0.02" mol "m^(-3)` =20 mol `m^(-3)` Resistivity of solution `k=0.248"S "m^(-1)` `=0.248xx1000xx10^(-3)" S "m^(-1)` `=248xx10^(-3)S " "m^(-1)` `therefore Lamda_(m)=(248xx10^(-3)S" "m^(-1))/(20" mol "m^(-3))` `=1.24xx10^(-2)" S "m^(2)" "mol^(-1)` `=124xx10^(4)" S "m^(2)" "mol^(-1)`. . . (i) OR Resistivity of 0.02 M KCl `k=("Cell constant "(G^(**)))/("Resistance of solution (R)")` `=(1.29cm^(-1))/(520Omega)` `=0.248xx10^(-2)" S "cm^(-1)` Where, `k=0.248xx10^(-2)" S "cm^(-1)` `Lamda_(m)=(k)/(c)` `c=0.02" mol "L^(-1)` `Lamda_(m)=(0.248xx10^(-2)" S "cm^(-1)xx1000cm^(3)L^(-1))/(0.02" mol "L^(-1))` `Lamda_(m)=124" S "cm^(2)mol^(-1)`. . . (ii) According to And `Lamda_(m)=124xx10^(4)S" "m^(2)mol^(-1)`. . (i) According to Hence, `1.0cm^(2)=1xx10^(-4)m^(2)` |
|
| 45. |
Resistance of 0.2 M solution of an electrolyte is 50 Omega. The specific conductance of the solution is 1.4 S m^(-1). The resistance of 0.5 M solution of the same electrolyte is 280Omega. The molar conductivity of 0.5 M solution of the electrolyte in S m^(2)mol^(-1) is |
|
Answer» `5xx10^(2)` `kappa`=Conductance`xx`Cell const.`=(1)/(R)xx`cell const. ltBrgt `therefore`Cell constant`=kappaxxR=(1.4" S "m^(-1))(50Omega)=70M^(-1)` Case II: `C=0.5" M ",R=280Omega," cell CONT."=70m^(-1)` `kappa=(1)/(R)xx"cell constant"=(1)/(280Omega)xx70m^(-1)` Molar conductivity (in S `m^(2)mol^(-1)`) `=(kappa(S" "m^(-1)))/("MOLARITY"(mol " "L^(-1))xx1000" L "m^(-3))` `=(0.25" S "m^(-1))/(0.5 " mol "L^(-1)xx1000" L "m^(-3))` `=5xx10^(-4)" S "m^(2)mol^(-1)` |
|
| 46. |
Resistance of a conductivity cell filled with 0.1 mol L ^(-1) KCl solution is 100 Omega. If the resistance of the same cell when filled with 0.02 mol L^(-1) KCl solution is 520 Omega, calculate the conductivity and molar conductivity of 0.02 mol L^(-1)KCl solution. The conductivity of 0.1 mol L^(-1) KCl solutin is 1.29 S/m. |
|
Answer» SOLUTION :The cell constant is given by the equation : Cell constant `=G^(**)=` CONDUCTIVITY `xx` RESISTANCE `=1.29 S//m m 100Omega =129m^(-1)=1.29 cm^(-1)` Conductivity of `0.02 mol L^(-1)` Kcl solution = cell constant /resistance `=(G^(**))/(R)=(129m^(-1))/(520Omega)=0.248Sm^(-1)` Concentration `=0.02 mol L^(-1)` `=1000xx0.02 mol m^(-3) =20 mol m^(-3)` Molar conductivity `=^^_(m)=k/c ` `(248xx10^(-3)Sm^(-1))/(20 mol m ^(-3))=124xx10^(-4) Sm^(2) mol ^(-1)` ALTERNATIVELY, `k=(1.29 cm^(-1))/(520Omega)=0.248xx10^(-2) S cm^(-1)` `and ^^_(m) =k xx1000 cm^(3) L-1 "molarity"^(-1)` `=(0.248 xx10^(-2) S cm^(-1)xx1000 cm ^(23) L^(-1))/(0.02 mol L^(-1))` `=124S cm^(2) mol ^(-1)` |
|
| 47. |
Resistance of a conductivity cell filled with 0.1 mol L^(-1)KCl solution is 100 ohms. If the resistance of the same cell filled with 0.02 mol L^(-1)KCl solution is 520 ohms, calculate the conductivity and molar conductivity of 0.02 mol L^(-1) KCl solution. The conductivity of 0.1 mol L^(-1)KCl solution is 1.29 xx 10^(-2) S cm^(-1) |
|
Answer» SOLUTION :Cell constant, `G^(@)` = Conductivity `XX` Resonance `=1.29 xx 10^(-2) S cm^(-1) xx 100` ohm `=1.29 cm^(-1)` Conductivity of 0.02 mol `L^(-1)` KCl `=G^(@)/("Resistance") = (1.29 cm^(-1))/(520 ohm) = 0.00248 S cm^(-1)` `Lambda_(m) = (K xx 1000)/("concentration") = (2.48 xx 10^(-3) xx 1000)/0.02 = 124 S cm^(2) mol^(-1)` |
|
| 48. |
Resistance of a conductivity cell filled with 0.02 M KCl solution is 520 Omega. Calculate the conductivity and molarconductivity of that solution. [Cell constant of the cell = 1.29 cm^(-1)]. |
|
Answer» SOLUTION :Cell CONSTANT =`1.29 cm^(-1)," Resistance" =520Ohm` Molar concentration of KCl=0.02 M `"Conductivity "=("Cell constant")/("Resistance")=(1.29)/(520)=0.248xx 10^(-2)Scm^(-1)` `wedge_(m) (KCl)=(k xx 1000)/(C)=(0.248 xx 1000)/(0.02) =124 Scm^(2)mol^(-1)`. |
|
| 49. |
Resistance of 1 N CH_(3)COOH is 250 ohm. This conductive cell has constant value of 1.15 cm^(-1), then what is equivalent conductivity of 1N CH_(3)COOH ? (Ohm^(-1)" "cm^(2)" "equ^(-1)). |
|
Answer» 18.4 =0.004 `Ohm^(-1)` (ii) Specific conductivity : (K)=conductivity`xx`cell constant `=0.004xx1.15(ohmxxcm^(-1))` `=0.00460ohmxxcm^(-1)` (III) equivalent conductivity : `(LAMDA)=(1000K)/(C)=(1000xx0.0046)/(1)` `=4.6ohm^(-1)CM^(2)equ^(-1)`. |
|
| 50. |
Resistance of a conductivity cell filed with a solution of an cleetrolyto of concentration 0.1M is 100 Omega. The conductivity of this solution is 1.29 S m ^(-1). Resistance of the same cell when filled with 0.2 M of the same solution is 520 Omega. The molar coonductivity of 0.2 M solution os electrolyte will be |
|
Answer» `1.24xx10^(-4) SM^(2) MOL ^(-1)` `l/A`(cell constnat `= 1.29xx10m^(1)` Given, `R =520Omega , C=0.2M,` `mu` (molar conductivity)= ? `mu =k xxV ` (k can be calculated as `k=1/R ((l)/(A))` now cell constnat is known. ) Here, `mu =(1)/(520) xx129 XX(1000)/(0.2)xx10^(-6)m ^(3)` `=12.4xx10^(-4) Sm^(2) mol ^(-1)` |
|



