Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Roasting of copper pyrites is done |
|
Answer» to remove MOISTURE and VOLATILE impurities |
|
| 3. |
Roasting is used during metallurgical operation using |
|
Answer» Galena |
|
| 4. |
Roasting is used during metallurgical operations for : |
| Answer» Answer :D | |
| 5. |
Roasting is generally done in case of the |
|
Answer» OXIDE ORES ` 2ZnS +3O_2 to2ZnO+ 2SO_2 UARR` |
|
| 6. |
Roasting is generally done in case of : |
|
Answer» OXIDE ores |
|
| 7. |
Roasting is generally done in case of |
|
Answer» OXIDE ORES |
|
| 8. |
Roasting is generally carried out in case of |
|
Answer» OXIDE ores |
|
| 9. |
Roasting is generally carried out in case of.... |
|
Answer» SILICATE ORES |
|
| 10. |
Roasted silver ore +CN-_((aq))+O_(2) to [X]_((aq))+OH_((aq))^(-),[X]_((aq))+Zn to [Y]_((aq))+Agdarr The [X] and [Y] are respectively : |
|
Answer» `[Ag(CN)_(2)]^(-),[Zn(CN)_(6)]^(-4)` |
|
| 11. |
Roasting is generally carried out in case of : |
|
Answer» OXIDE ores |
|
| 12. |
Roasting involves _________. |
|
Answer» only volatilization of volatile IMPURITIES |
|
| 13. |
Roasting is carried out in case of |
| Answer» Answer :D | |
| 14. |
Roasting. |
| Answer» SOLUTION :It is a process of STRONGLY heating a CONCENTRATION ore in the EXCESS of air below melting point of metal, to convert it INOT oxide form. For example, PbS, ZnS ores on roasting form PbO, ZnO respectively. | |
| 15. |
Roasted on one is mixed with coke and time stone and smelted in blast fumice. Whichamong the following does not represent the correct answer about the reactions taking place in the blast fumace. |
|
Answer» In the lower part, called ZONE of fusion temperature (1773 K) COKE burns to `CO_(2)` producing 97000 calories of beat |
|
| 16. |
Roasted gold ore +CN^(-)water overset(O_(2))to[X]+OH^(-),[X]+Znto[Y]+Au then the coordination number of metal in [X] is 'a' the coordination number of metal in [Y] is 'b' then 'a+b'is |
|
Answer» |
|
| 17. |
Roasted gold ore +CN^(-)+H_(2)O overset(O_(2)) to [x]+OH, [x]+Zn to [y]+Au[x] and |y| are |
|
Answer» `x=[Au (CN)_(2)]^(-) "" y=[Zn(CN)_(4)]^(-2)` `2Na[Au(CN)_(2)]^(-)+Zn to Na_(2)[Zn(CN)_(4)]+2Au` `X=[Au(CN)_(2)]^(-) and Y=[Zn(CN)_(4)]^(-2)` |
|
| 18. |
Roasted gold ore +CN^(-)+H_(2)O overset(O_(2)) to [X]+OH^(-) |
|
Answer» `X=[Au(CN)_(2)]^(-) , Y=[Zn(CN)_(4)]^(2-)` |
|
| 19. |
Roasted gold ore +CN^(-)+H_(2)O+ overset(O_(2)) to x+OH^(-),What is the oxidation at 'Au' is complex 'x |
|
Answer» Oxidation state of Au in the compole is +1 |
|
| 20. |
Roasted copper pyrites on smelting with sand produces_________. |
|
Answer» `FeSiO_(3)` as FUSIBLE slag and `Cu_(2)O` matte |
|
| 21. |
Roasted copper pyrite on smelting with sand produces |
|
Answer» `FeSiO_3` as fusible SLAG and `Cu_2 S` as matte `FeO + UNDERSET("Silica")(SiO_2) to underset("(slag)")underset("Ferrous silicate")(FeSiO_3)` |
|
| 22. |
Roastedcopperpyrite on smeltingwith sandproduces |
|
Answer» `FeSiO_3 `as fusibleslagand` Cu_2 S `as matte |
|
| 23. |
RNH_(2) can be converted into R, NH and no other amine by |
|
Answer» its REACTION with RX |
|
| 24. |
Roasting of HgS in air produces: |
|
Answer» HGO |
|
| 25. |
RNA is different from DNA because RNA contains…….. |
|
Answer» RIBOSE SUGAR and tymine |
|
| 26. |
RNA is different from DNA< because RNA contains: |
|
Answer» RIBOSE SUGAR and thymine |
|
| 27. |
RNA is different from DNA because RNA contains |
|
Answer» RIBOSE SUGAR and thiamine |
|
| 28. |
RNA is : |
|
Answer» Single helix STRAND |
|
| 29. |
RNA differs from DNA with respect to the following |
| Answer» Answer :D | |
| 30. |
RNA and DNA are chiral molecules, there chirality is due to |
|
Answer» CHIRAL PHOSPHATE ESTER units |
|
| 31. |
RNA and DNA are chiral molecules, their chirality is due to |
|
Answer» CHIRAL bases |
|
| 32. |
RNA and DNA are chiral molecules, their chirality is due to : |
|
Answer» Chiral bases |
|
| 33. |
RNA and DNA are chiral molecules , their chirality |
|
Answer» D-sugar component |
|
| 34. |
RMgBr on reaction with an excess of oxygen followed by hydrolysis gives |
|
Answer» RH 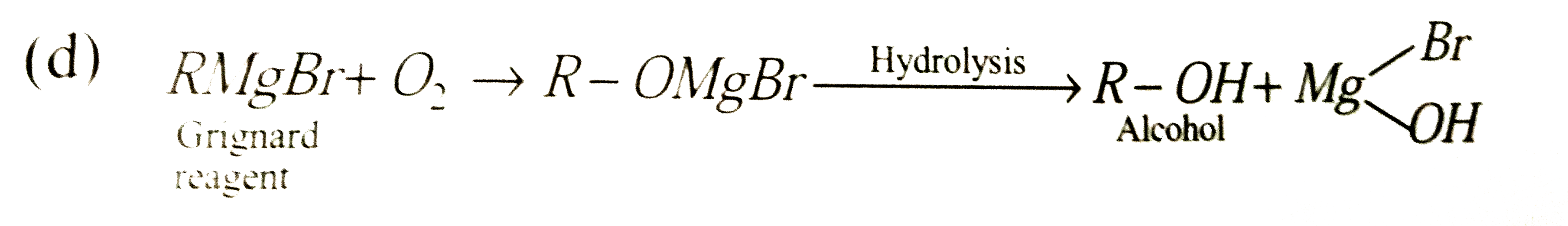
|
|
| 35. |
RMgX on reaction with O_2 followed by hydrolysis gives : |
|
Answer» RH |
|
| 37. |
R=k[O_3]^2 |
|
Answer» `R=k[O_3]^2[O_2]^(-1)` |
|
| 38. |
Rio Tinto process is used for extraction of: |
|
Answer» Cu |
|
| 39. |
Ring test is performed for : |
|
Answer» `NO_3^(-)` ION |
|
| 40. |
Ring test formiratesconformed byacidifyingprepared FeSO_(4) soin a brownring isformedthatto theformation of [Fe(H_(2)O)_(3)NO[SO_(4) Thisrest shouldnot be performed fornitrate ion in presence of |
| Answer» Answer :d | |
| 42. |
Ring structure of glucose is due to formation of hemiacetal and ring formation is in between |
|
Answer» `C_1 and C_5` |
|
| 43. |
Ring-opening reactions of epoxides can proceed by either S_(N)2 or S_(N)1 mechanisms, depending on the nature of the epoxide and on the reaction conditions. If the epoxide is asymmetric, the structure of the product will very according to which mechanisms dominates. When an symmetric epoxide undergoes solvolysis in basic methanol, ring-opening occurs by and S_(N)2 mechanisms, and the less subsitution carbon is the site of nucleophilic attack. Conversely, when solvolysis occurs in acidic methanol, the reaction occurs by a mechanisms with substantial S_(N)1 character, and the more substituted carbon is the site of attack. These are both good examples of regioselective reactions, examine the basic, S_(N)2 case first. The leaving group is an alkoxide anion, because there i s not acid avaialble to protonate the oxygen prior to ring opening. An alkoxide is a poor leaving group, and thus the ring is unlikely to open without a'push' from the nucleophile. Like in other S_(N)2 reactions, nucleophilic attack takes place from the backside, resulting in inversion t the electrophilic carbon. The best way to depict the acid-catalyzed epoxide ring-opening reaction is as a hybrid, or cross, between an S_(N)2 and S_(N)1 mechanism. First, the oxygen is protonated, creatinga good leaving group. Then the carbon-oxygen bond begins to break and positive charge begins to build up on the more substituted carbon. answer the following based on the above : The product formed after nucleophilic attack and mild hydrolysis is |
|
Answer»
|
|
| 44. |
Ring-opening reactions of epoxides can proceed by either S_(N)2 or S_(N)1 mechanisms, depending on the nature of the epoxide and on the reaction conditions. If the epoxide is asymmetric, the structure of the product will very according to which mechanisms dominates. When an symmetric epoxide undergoes solvolysis in basic methanol, ring-opening occurs by and S_(N)2 mechanisms, and the less subsitution carbon is the site of nucleophilic attack. Conversely, when solvolysis occurs in acidic methanol, the reaction occurs by a mechanisms with substantial S_(N)1 character, and the more substituted carbon is the site of attack. These are both good examples of regioselective reactions, examine the basic, S_(N)2 case first. The leaving group is an alkoxide anion, because there i s not acid avaialble to protonate the oxygen prior to ring opening. An alkoxide is a poor leaving group, and thus the ring is unlikely to open without a'push' from the nucleophile. Like in other S_(N)2 reactions, nucleophilic attack takes place from the backside, resulting in inversion t the electrophilic carbon. The best way to depict the acid-catalyzed epoxide ring-opening reaction is as a hybrid, or cross, between an S_(N)2 and S_(N)1 mechanism. First, the oxygen is protonated, creatinga good leaving group. Then the carbon-oxygen bond begins to break and positive charge begins to build up on the more substituted carbon. answer the following based on the above : |
|
Answer»
|
|
| 45. |
Ring structure of glucose is due to formation of hemiacetal and ring formation between : |
|
Answer» `C_1` and `C_5` |
|
| 46. |
rignard reagent is used to prepare carboxylic acids. What is the other reagent in this reaction ? |
| Answer» SOLUTION :CARBON DIOXIDE | |
| 47. |
Riddish brown gas is obtain with the following are treated with cone H_(2)SO_(4) |
|
Answer» `BR^(Θ)` |
|
| 48. |
Rickets and Xerophthalmia are caused due to which two deficiencies ? |
|
Answer» |
|
| 49. |
Rickets may be caused by the deficiency of which vitamin ? |
|
Answer» VITAMIN D |
|
| 50. |
Rickets is caused due to the deficiency of |
|
Answer» Vit-A |
|







