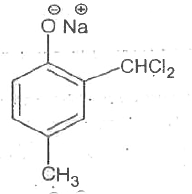Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Relation between partial pressure and mole fraction is stated by |
|
Answer» Graham's law `p = P_(x)^(@)` where, p= Partial pressure of componem `p ^(@)=` Vapour pressure of component in pure form x = mole fraction of component in solution. |
|
| 2. |
Relation between hydrolysis constant and dissociation constant are given. Which is the correct formula for MgCl_(2). |
|
Answer» `K_(h) = (K_(w))/(K_(a))` `K_(h) = (K_(w))/(K_(b))`. |
|
| 3. |
Relation between magnetic moment (spin-only formula) and number of unpaired electrons is given by |
|
Answer» `mu=sqrt(N(n+2))` |
|
| 4. |
Relation between(A) and (B) is |
|
Answer» Anomers |
|
| 5. |
Relation between degree of dissociation (alpha) and vant Hoff's factor for BaCI_(2) is |
|
Answer» `I = 1 + alpha` van't-Hoff FACTOR `i=1-alpha +alpha + 2alpha = 1 + 2alpha` |
|
| 6. |
Relation between amino acid and protein is similar to the one between |
|
Answer» Glucose and fructose |
|
| 7. |
Reimer-Tiemann reaction involves an aryl carbanion/enolate anion and also -"CCl"_(3) derived from the action of strong bases on CHCl_(3), though the latter has only a transient existence decomposing to "CCl"_(2), a highly electron deficient electrophile that attacks the aromatic nucleus, the product from phenoxideion is after acidification, very largely the o-aldehyde plus just a small amount of p-isomer. Reimer-Tiemann reaction involves |
|
Answer» nucleophilic substitution |
|
| 8. |
Reimer-Tiemann reaction involves an aryl carbanion/enolate anion and also -"CCl"_(3) derived from the action of strong bases on CHCl_(3), though the latter has only a transient existence decomposing to "CCl"_(2), a highly electron deficient electrophile that attacks the aromatic nucleus, the product from phenoxideion is after acidification, very largely the o-aldehyde plus just a small amount of p-isomer. CO_(2) can attack in a similar mannerto : "CCl"_(2) on phenoxide ion. Following-a similar mechanism, major product will be |
|
Answer» o-hydroxybenzoate |
|
| 9. |
Reimer Tiemann reaction is an electrophillic substitution reaction in which the electrophile is |
|
Answer» `^+C CI_(3)` |
|
| 10. |
Reimer-Tiemanns reaction involves a |
|
Answer» Carbonium ION INTERMEDIATE 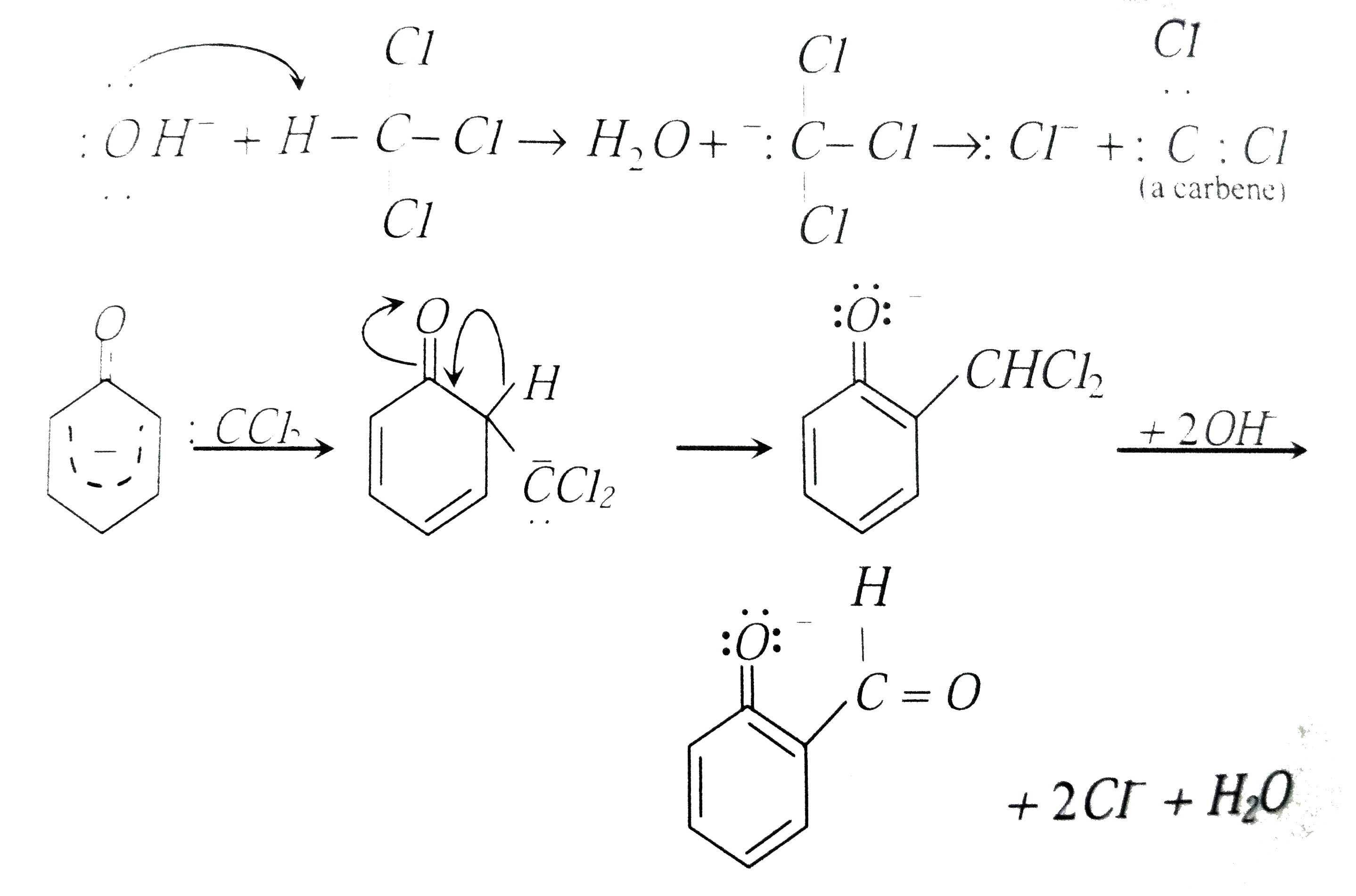
|
|
| 11. |
Reimer-Tiemann reaction involves an aryl carbanion/enolate anion and also -"CCl"_(3) derived from the action of strong bases on CHCl_(3), though the latter has only a transient existence decomposing to "CCl"_(2), a highly electron deficient electrophile that attacks the aromatic nucleus, the product from phenoxideion is after acidification, very largely the o-aldehyde plus just a small amount of p-isomer. The product of the reaction in the reaction in the above paragraph is largely |
|
Answer» m-salicylaldehyde |
|
| 12. |
Reimer-Tiemann reaction involves an aryl carbanion/enolate anion and also -"CCl"_(3) derived from the action of strong bases on CHCl_(3), though the latter has only a transient existence decomposing to "CCl"_(2), a highly electron deficient electrophile that attacks the aromatic nucleus, the product from phenoxideion is after acidification, very largely the o-aldehyde plus just a small amount of p-isomer. Name the electrophile attacking carbanion in the above reaction. |
|
Answer» Chloroform |
|
| 13. |
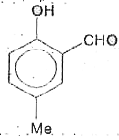
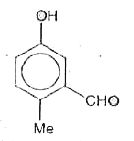
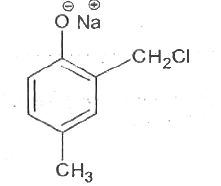
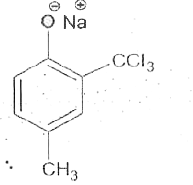
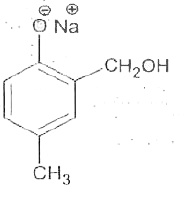
Reimer-Tiemann reaction involves an aryl carbanion/enolate anion and also -"CCl"_(3) derived from the action of strong bases on CHCl_(3), though the latter has only a transient existence decomposing to "CCl"_(2), a highly electron deficient electrophile that attacks the aromatic nucleus, the product from phenoxideion is after acidification, very largely the o-aldehyde plus just a small amount of p-isomer. If the above reaction is carried out on the anion of p-hydroxytoluene, what will be theproduct/products? |
|
Answer»
|
|
| 14. |
Reimer-Tiemann reaction introduces an aldehyde group, on to the aromatic ring of phenol, ortho to the hydroxyl group. This reaction involves electrophilic aromatic substitution. This is a general method for the synthesis of substituted salicyladehydes as depicted below. The structure of the intermediate I is |
|
Answer»
|
|
| 15. |
Reimer-Tiemann reaction introduces an aldehyde group, on to the aromatic ring of phenol, ortho to the hydroxyl group. This reaction involves electrophilic aromatic substitution. This is a general method for the synthesis of substituted salicyladehydes as depicted below. The electrophile in this reaction is |
|
Answer» : CHCL |
|
| 16. |
Reimer-Tiemann reaction introduces an aldehyde group, on to the aromatic ring of phenol, ortho to the hydroxyl group. This reaction involves electrophilic aromatic substitution. This is a general method for the synthesis of substituted salicyladehydes as depicted below. Which one of the following reagents is used in the above reaction ? |
|
Answer» `"AQ. NAOH"+CH_(3)Cl` |
|
| 17. |
Reimer-Tiemann reaction introduces an aldehyde group on to the aromatic ring of phenol, ortho to the hydroxyl group. Thi reaction involves electrophilic aromatic substitution. It is a general method for the synthesis of substituted salicylaldehydes as depicted below: Q. The structure of intermediate (I) is: |
|
Answer»
|
|
| 18. |
Reimer-Tiemann reaction introduces an aldehyde group on to the aromatic ring of phenol, ortho to the hydroxyl group. Thi reaction involves electrophilic aromatic substitution. It is a general method for the synthesis of substituted salicylaldehydes as depicted below: Q. Which one of the following reagents is used ini the above reaction? |
|
Answer» AQ. `NaOH+CH_(3)CL` |
|
| 19. |
Reimer-Tiemann reaction introduces an aldehyde group on to the aromatic ring of phenol, ortho to the hydroxyl group. Thi reaction involves electrophilic aromatic substitution. It is a general method for the synthesis of substituted salicylaldehydes as depicted below: Q. The electrophilic in this reaction is- |
| Answer» Answer :C | |
| 20. |
Reimer Tieman reaction involves intermediate |
|
Answer» Carbonium ion intermediate |
|
| 21. |
Regular use of which of the following fertilizer increases the acidity of soil |
|
Answer» POTASSIUM nitrate `(NH_(4))_(2) SO_(4) to 2NH_(4)^(+) + SO_(4)^(2-)` `NH_(4)^(+) +H_(2) hArr NH_(4)OH +H^(+)` |
|
| 22. |
Reimer and Tiemann reaction is used to prepare |
|
Answer» SALICYLIC acid |
|
| 23. |
Regular use of which of the following fertilizers increases the acidity of soil? |
|
Answer» Urea |
|
| 24. |
Regent 'A' in the followingreaction is H - CHO + CH_(3) - NO_(2) overset(A) to H - overset(OH)overset(|)underset(H)underset(|)(C)- CH_(2) - NO_(2) |
|
Answer» 20%KOH |
|
| 25. |
Regardint the isolation of noble gases fromatomosphric air by Ramsay - Rayleigh's second method four equations are givenbelow I) N_2 + O_2 + 2NOII) 2NO+ O_2 to 2NO_2 III) 2NO_2+ 2NaOH to NaNO_2+ NaNO_3 + H_2OIV) CaC_2 + N_2 overset(Delta)to CaCN_2+ C In this method, the reactions truely involved are |
|
Answer» Both I and II |
|
| 26. |
Regarding XeF_2the correct combination is |
|
Answer» `sp^3d - 4L.P` |
|
| 27. |
Regarding the structure of cyanamide ion, pick out the wrong statement |
|
Answer» It has one carbon with a NEGATIVE charge |
|
| 28. |
Regarding XeF_2, the correct combination is (L.P = lone pairs mu = Dipolemoment) |
|
Answer» `sp^3d-4L.P` |
|
| 29. |
Regarding the electrolysis of Alumina, the correct statements A) Cryolite is added to reduce the fusion temp B) Impure aluminium is mixed with copper and silicon to increase the density C) Fluorspar is added to reduce the fusion temp D) Electrolyte is mixture of fluorides of sodium, Barium and Aluminium saturated with Alumina |
|
Answer» Only B, C and D are CORRECT |
|
| 30. |
Regarding the electrolysis of Alunina, the correct statements A) Cryolite is added to increase the fusion temp B) Impure aluminium is mixed with copper and silicon to decrease the density C)Fluorspar is added to reduce the fusion temp D) Electrolyte is mixture of oxides saturated with alumina |
|
Answer» Only B, C and D are CORRECT |
|
| 31. |
Regarding tetraamminedithiocyanato-S cobalt tris (oxalato) Cobaltate (III) the correct statements are |
|
Answer» FORMULA of complex in `[Co(SCN)_(2)(NH_(3))_(3)(CN)_(3)]` |
|
| 32. |
Regarding starch the wrong statement is |
|
Answer» It is PRESENT in SORGHUM |
|
| 33. |
Regarding SF _(6), the wrong statement is |
|
Answer» It is inert and does not UNDERGO hydrolysis |
|
| 34. |
Regarding secondary structure of a protein, correct statement(s) is/are (A) peptide bonds possess regional planarity(B) C = O and -NH- of different peptide chains are held by Van der Waal attarctions (C ) closely packed arrangement so as to minimise repulsion between ''R'' groups. |
|
Answer» only C |
|
| 35. |
Regarding phenol the correct statements areA) It reacts with B_2//CS_2to give a mixture of o-bromophenol and p-bromophenolB) In Riemer-Tiemann reaction with CHCl_3 a substituted benzalchloride intermediate is formedC) In Kolbe's reaction gives salicylic acidD) On oxidation with it gives a conjugated diketone |
| Answer» Answer :C | |
| 36. |
Regarding ozone the wrong statement is |
|
Answer» The BOND angle is `116^(0)49^(1)` |
|
| 37. |
Regarding oxygen the correct statements are A) It is an important constituent of rocket fuelsB) It is used for artificial respriration C) It is used in oxyacetylene welding. |
|
Answer» A,B |
|
| 38. |
Regarding lactose some statements are given below (A) On hydrolysis lactose gives beta- D-galactose and beta-D-glucose In lactose C_(1) " of " beta - D- galactose has acetal structure and C_(1) " of " beta - D-glucose hashemiacetal structure (C ) In lactose molecule beta- D-galactose is a nonreducing unit and beta-D- glucose is a reducing unit The correct statements are |
|
Answer» A, C |
|
| 39. |
Regarding molarity, which of the following statements are correct (a) Units of molarity mol kg^(-1) (b) Molarity of dibasic acid is half of its normality (c) Normality x GEW / GMW (d) Molarity always equals to its molality |
|
Answer» a,b |
|
| 40. |
Regarding H_(3)PO_(5) the wrong statement is |
|
Answer» It's basicity is three |
|
| 41. |
Regarding enzymatic reactions, the 4 steps are shown below A) E + S rarr E -S B) E-P rarr E + P (C) E - I rarr E - P D) E - S rarr E-I The correct sequence of the steps is |
| Answer» Answer :A | |
| 42. |
Regarding enzymes, incorrect statement is |
|
Answer» an enzyme is GENERALLY a protein |
|
| 43. |
Regarding diethyl ether, the wrong statement is |
|
Answer» It is slightly soluble in water |
|
| 44. |
Regarding cross-linked or network polymers, which of the following statements is incorrect ? |
|
Answer» They contain strong covalent bonds in their polymer chains. In this TYPE of polymers, bifunctional or trifunctional monomer combines with covalent BOND INBETWEEN the long polymer chain. Long polymer chain is joined by strong covalent bond and forms a net like structure. 
|
|
| 45. |
Regarding cross-linked or network polymers, which of the following statement is incorrect? |
|
Answer» EXAMPLES are Bakelite and melamine |
|
| 46. |
Regarding amylopectin, the wrong statement is |
|
Answer» it is a branched CHAIN polysaccharide |
|
| 47. |
Refractory metals are used in construction of furnaces because |
|
Answer» they can withstand HIGH temperature |
|
| 48. |
Refractory materials are used for the construction of furnaces because they : |
|
Answer» are light in weight |
|
| 49. |
Refractive index of a solid is observed to have the same value along all directions. Comment on the nature of this solid. Would it show cleavage property ? |
| Answer» Solution :As the SOLID has the same value of refractive index along all DIRECTIONS, it is isotropic. It is therefore amorphous solid. Amorphous SOLIDS when CUT with a knife or on breaking do not show a clean cleavage. | |