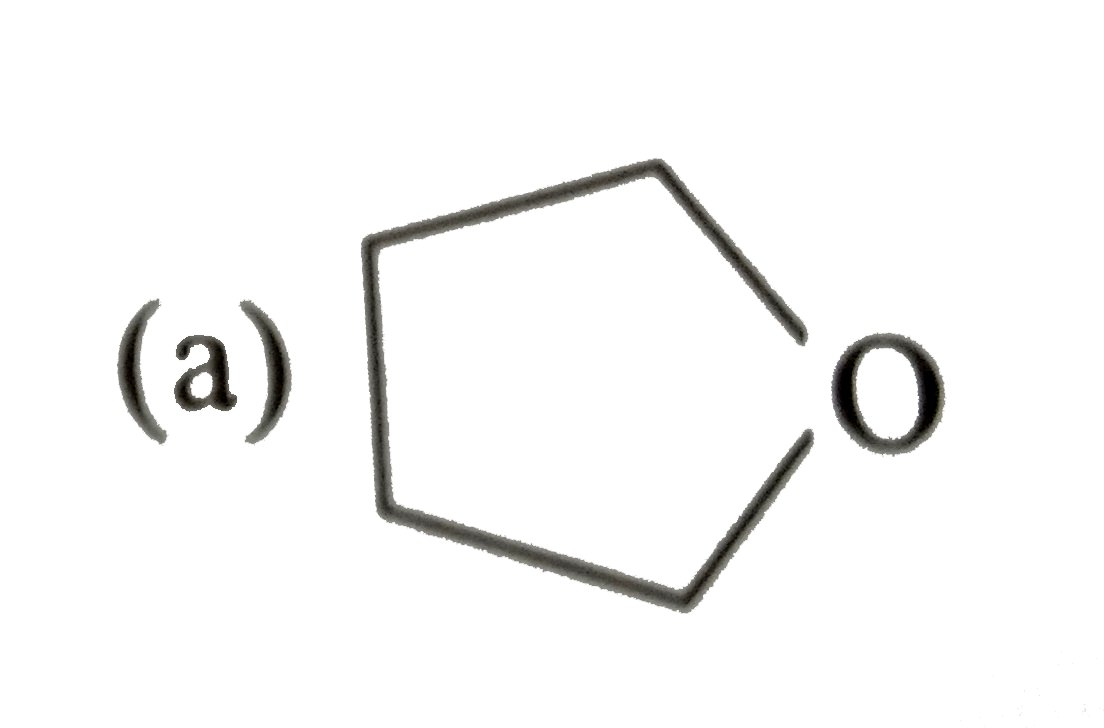
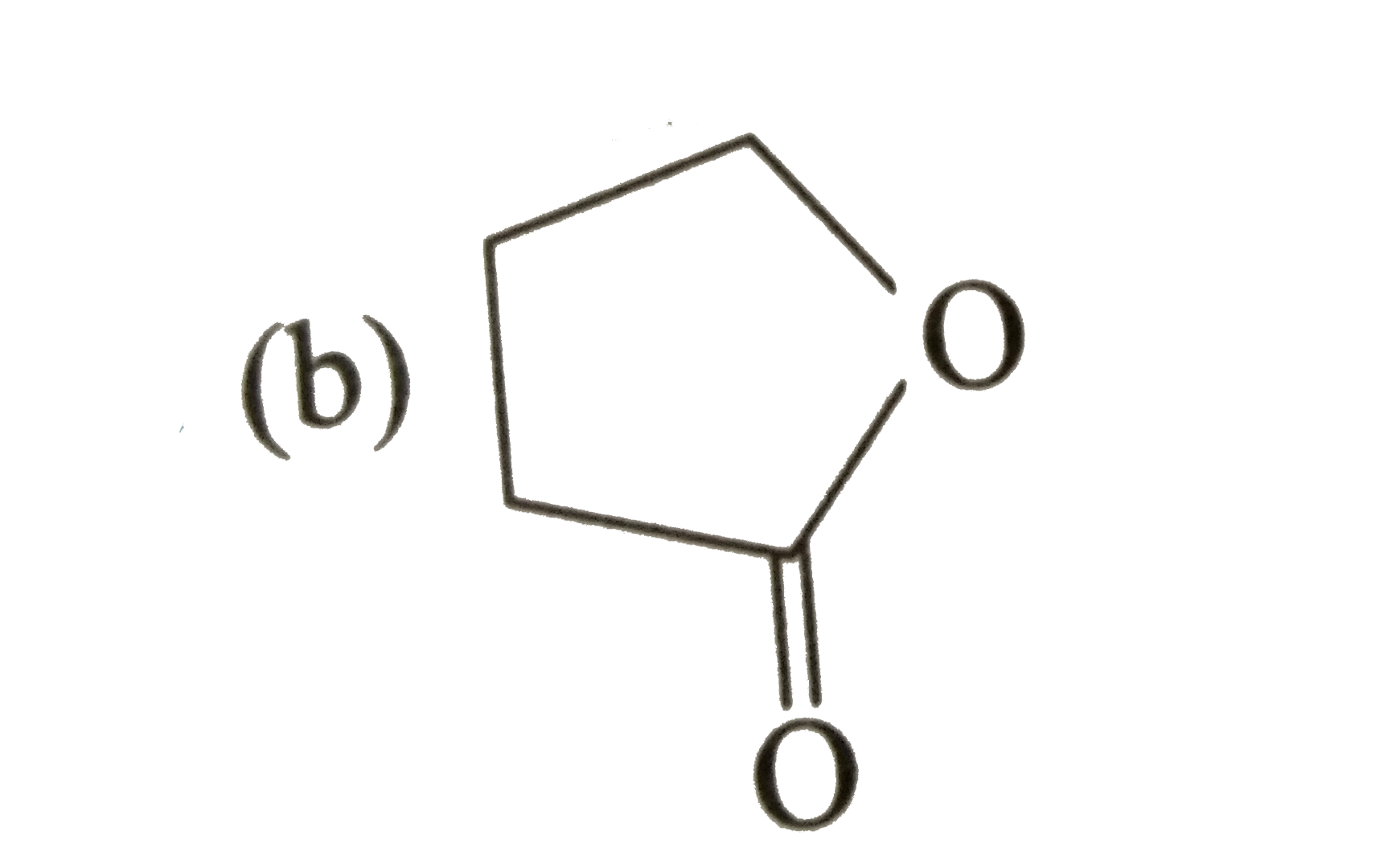
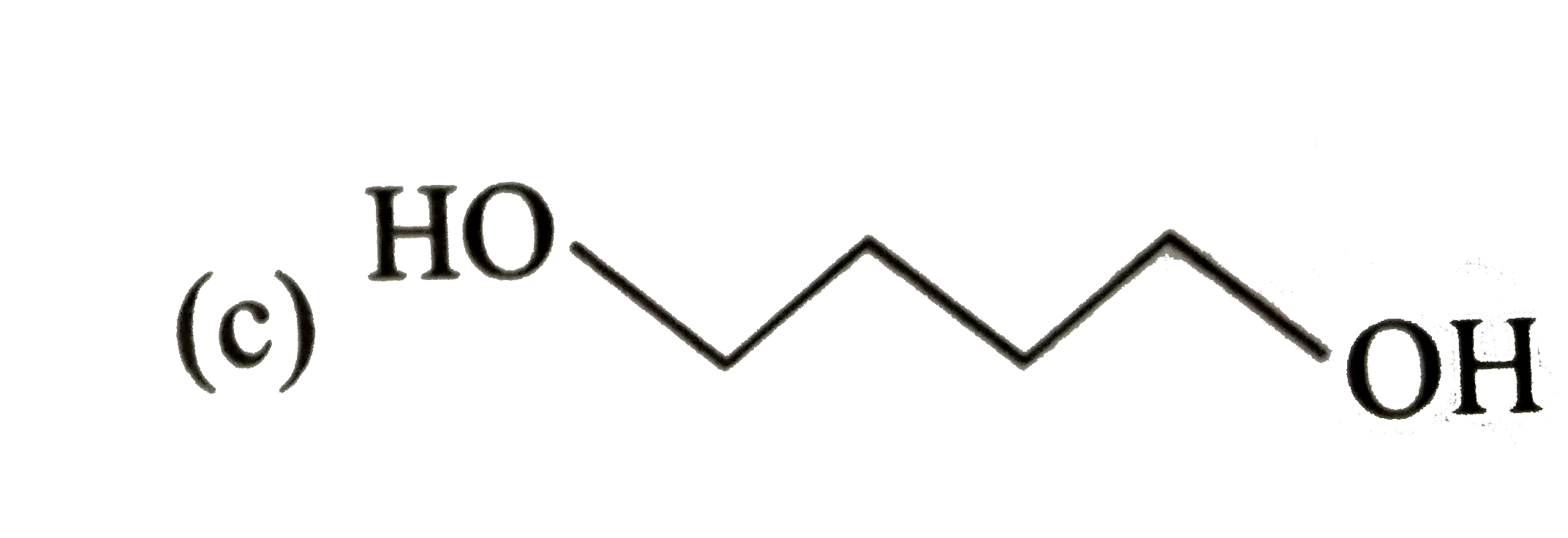
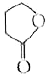Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Refractive index of a solid is observed to have the same value along all directions. Comment on the nature of this solid. Would it show cleavage property? |
| Answer» Solution :As the SOLID has same VALUE of refractive index along all directions, this means that it is isotropic and hence AMORPHOUS. Being an amorphous solid, it would not show a CLEAN CLEAVAGE when cut with a knife. Instead it would break into pieces with irregular surfaces. | |
| 2. |
Refractive index of a solid is observed to have the same value along all directions. Comment on the nature of this solid. Would it show cleavage property? |
| Answer» Solution :As the solid has same value of REFRACTIVE index along all directions, this means that it is isotropic and hence amorphous. Being an amorphous solid, it WOULD not show a clean cleavage when cut with a knife. Instead it would break into PIECES with irregular SURFACES. | |
| 3. |
Refining process of impure metalsdepends upon the differences in properties of the metal. Extraction of Al is usually carried out from its bauxite ore. Extraction of Iron is done by reduction of its oxide ore in blast furnance. Copper is extracted by smelting and heating in reverberatory furnance. Extraction of zinc from zinc oxide is done usingcoke. Several methods are used and employed in refining the metals Match list-I with List-II and select the correct answer using codes given below in the list {:("List _I ","List-I"),("I. Cyanide process","(A) Ultra pure Ge"),("II. Froth Floatation process","(B) Pine oil"),("III. Electrolytic refining","(C) Extraction of Al"),("IV. Zone refining","(D) Extraction of Au"):} |
|
Answer» I-C,II-A,III-D,IV-B |
|
| 4. |
Refining process of impure metalsdepends upon the differences in properties of the metal. Extraction of Al is usually carried out from its bauxite ore. Extraction of Iron is done by reduction of its oxide ore in blast furnance. Copper is extracted by smelting and heating in reverberatory furnance. Extraction of zinc from zinc oxide is done usingcoke. Several methods are used and employed in refining the metals The purification method used for mineral Al_(2)O_(3).2H_(2)O |
|
Answer» FROTH FLOATATION |
|
| 5. |
Refiring of Zn is done using the proces called |
|
Answer» ELECTROLYTIC refining |
|
| 6. |
Refining process for a metal depends upon the difference in properties of the metal and the impurities. |
|
Answer» |
|
| 7. |
Refining of Tin can be done by |
|
Answer» Cupellation |
|
| 9. |
Refer to the graph for trends in M.P. of transition elements of 3d,4d and 5d series. Q. Choose the correct statement(s). |
|
Answer» III REFERS to 3RD transition series of PERIODIC table. |
|
| 10. |
Refer to the graph for trends in M.P. of transition elements of 3d,4d and 5d series. Q. What does maxima at point 'X' indicate? |
|
Answer» One unpaired electron PER d-orbital is particularly FAVOURABLE for strong inteeratomic interaction. |
|
| 11. |
Refer to the graph for trends in M.P. of transition elements of 3d,4d and 5d series. Q. A metal having high boiling point: |
|
Answer» has HIGH ENTHALPY of atomization |
|
| 12. |
Refer to Q. No. 10. Which of the following statements are correct? |
|
Answer» FORMATIONOF `(I)` and `(II)` proceeds VIA `SN^(1)` mechanism. |
|
| 13. |
Refer to Q. No. (8) above. Which statements is/are correct ? |
|
Answer» Both REACTIONS (i) and (ii) PROCEED via `SN^(2)` mechanism. |
|
| 14. |
Refer to Q. 39. The pair of isochoric processes among the transformation of states is |
|
Answer» K to L and L to M cooling at constant volume cooling at constant pressure heating at constant volume |
|
| 15. |
Reetu is purely vegetarian but her husband Rohit likes non-vegetarian food. He regularly buys nonvegetarian food (fish, meet, eggs etc.) and store it in fridge. The intermixing of odours creates problem for Reetu. Their daughter Rihana is a student of science. One day Rihana suggested her mother to put some small pieces of charcoal in the fridge as she thinks that this will solve the problem to some extent. (i) Do you think by putting some small pieces of charcoal in the fridge will solve the problem of intermixing of odours. Give reason for your answer. (ii) What are the values associated with Rihana's suggestion ? |
|
Answer» Solution :(i) Yes, putting some SMALL pieces of CHARCOAL in the fridge can solve the problem of intermixing of odours to some extent. This is because charcoal is a GOOD adsorbent of odours or vapours. Further breaking a big LUMP of charcoal into small pieces will increase the surface area of adsorbent, more is the extent of adsorption. (ii) Values associated with Rihana.s suggestion are (a) Love for her parents (B) Keen observation (c) Use of chemistry in daily life (d) Will to do experiment and explore (e) Presence of mind |
|
| 16. |
Redusing the pressure from 1.0 to 0.5 atm would change the number of molecules in one mole of ammonia to |
|
Answer» 75% of initial value HENCE, (D) is the correct answer. |
|
| 17. |
Reducutionon nitroalkanein neutralmedium(Zn + NH_(4)Cl)froms mainly |
|
Answer» `R -NH_(2)` |
|
| 18. |
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidsing power. {:("Ion",ClO_(4)^(-),IO_(4)^(-),BrO_(4)^(-)),("Reduction",E^(@)=-1.19V,E^(@)=1.65V,E^(@)=1.74V),("potentail"E^(@)//V,,,):} |
|
Answer» `ClO_(4)^(-) GT IO_(4)^(-) gt BrO_(4)^(-)` |
|
| 19. |
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power. |
|
Answer» `ClO_(4)^(-) gt IO_(4)^(-) gt BrO_(4)^(-)` |
|
| 20. |
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power |
|
Answer» `ClO_(4)^(-) gt IO_(4)^(-) gt BrO_(4)^(-)` |
|
| 21. |
Reduction potentials of same ions are given below. Arrange them in decreasing order of oxidising power. {:("Ion",ClO_(4)^(-),IO_(4)^(-),BrO_(4)^(-)),("Reduction potential"E^(@)//V,E^(-)=1.19V,E^(-)=1.65V,E^(-)=1.74V):} |
|
Answer» `ClO_(4)^(–) GT IO_(4)^(–) gt BrO_(4)^(–)` |
|
| 22. |
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power : |
|
Answer» `ClO_(4)^(-) gt IO_(4)^(-) gt BrO_(4)^(-)` |
|
| 23. |
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power. |
|
Answer» `ClO_(4)^(-)gtIO_(4)^(-)gtBrO_(4)^(-)` |
|
| 24. |
Reduction potential oftwo metals M_(1) " and " M_(2) are E_(M_(1)^(2+)|M_(1))^(@)=2.3V " and " E_(M_(1)^(2+)|M_(2))^(@)=0.2V Predict which one is better for coating the surface of iron. Given : E_(Fe^(3+)|Fe)^(@)=-0.44V |
| Answer» SOLUTION :Oxidation potential of `M_(1)` is more +ve than the oxidation potential of FE which indicates that it will prevent iron from rusting. | |
| 25. |
Reduction potential of four elements P,Q,R,S is -2.90,+0.34,+1.20 and -0.76. reactivity decreases in the order |
|
Answer» PgtQgtRgtS |
|
| 26. |
Reduction potential of A,B,C and D are 0.8V, 0.79V, 0.34V and -2.37V respectively. Which element displaces all the other three elements |
|
Answer» B |
|
| 27. |
Reduction of ZnO with carbon is done at 1100^(@)C. Reason : Delta G^(@) is negative at this temperature thus, process is spontaneous. |
|
Answer» If both ASSETION and reason are CORRECT, and reason is the CORRECT explanation of the assertion |
|
| 28. |
Reductionof the followingcompound wouldyieldmixtureof N-ethyl hydroxyl amineand acetaldoxime |
|
Answer» `C_(2)H_(5) - NH_(2)` |
|
| 30. |
Reduction of the metal centre in aqueous permanganate ion involves |
|
Answer» 3 electrons in neutral medium `MnO_(4)^(-) + 2H_(2)O + 3e^(-) rarrMnO_(2) + 4OH^(-)` In alkaline medium `MnO_(4)^(-) + e^(-) rarr MnO_(4)^(2-)` But `MnO_(4)^(2-)` is further reduced to `MnO_(2)` as shown below `MnO_(4)^(2-)+ 2H_(2)O + 2E^(-) rarr MnO_(2) + 4OH^(-)` Complete reaction is `MnO_(4)^(2-) + 2H_(2)O+ 3e^(-) rarr MnO_(2) + 4OH^(-)` which is the same as that for neutral medium. In acidic medium `MnO_(4)^(-) + 8 H^(+) + 5e^(-) rarr Mn^(2+) + 4H_(2)O` HENCE, the number of electrons involved in the reductionof metalcentre in aqueous `MnO_(4)^(-)` ION in neutral, alkaline and acidicmedia are 3,3 and 5 respectively. |
|
| 31. |
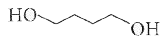
Reduction of the lactol Swith sodium borohydride gives |
|
Answer»
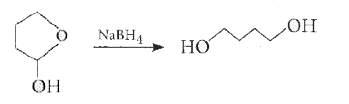
|
|
| 32. |
Reduction of ______ takes place in fuel cell. |
|
Answer» `O_(2)` |
|
| 33. |
Reduction of RCHO to RCH_(3)" by "N_(2)H_(4)" in "C_(2)H_(5)ONa is ________ reaction. |
|
Answer» STEPHEN REDUCTION |
|
| 34. |
Reductionof nitroparaffinegives |
| Answer» Answer :A | |
| 35. |
Reduction of nitrobenzene with LiAlH_4 is ether yields |
|
Answer»
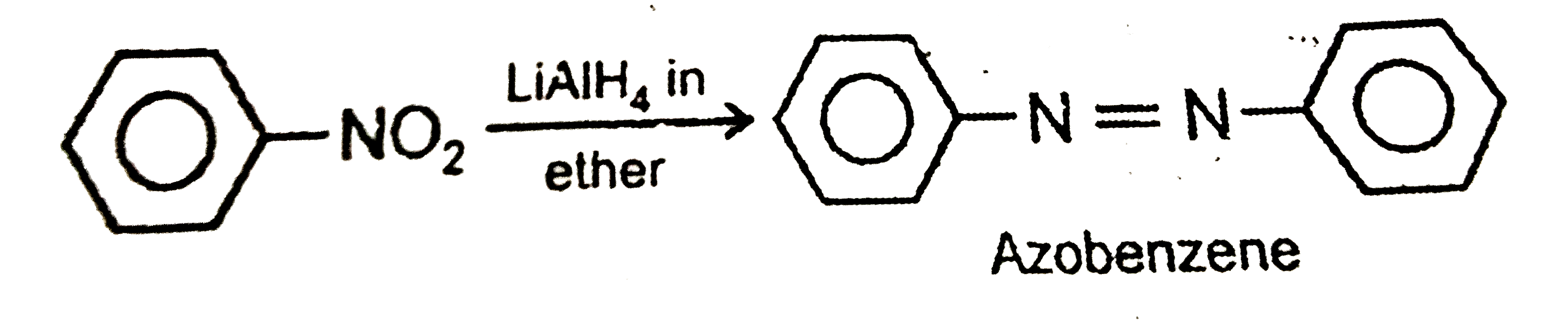
|
|
| 36. |
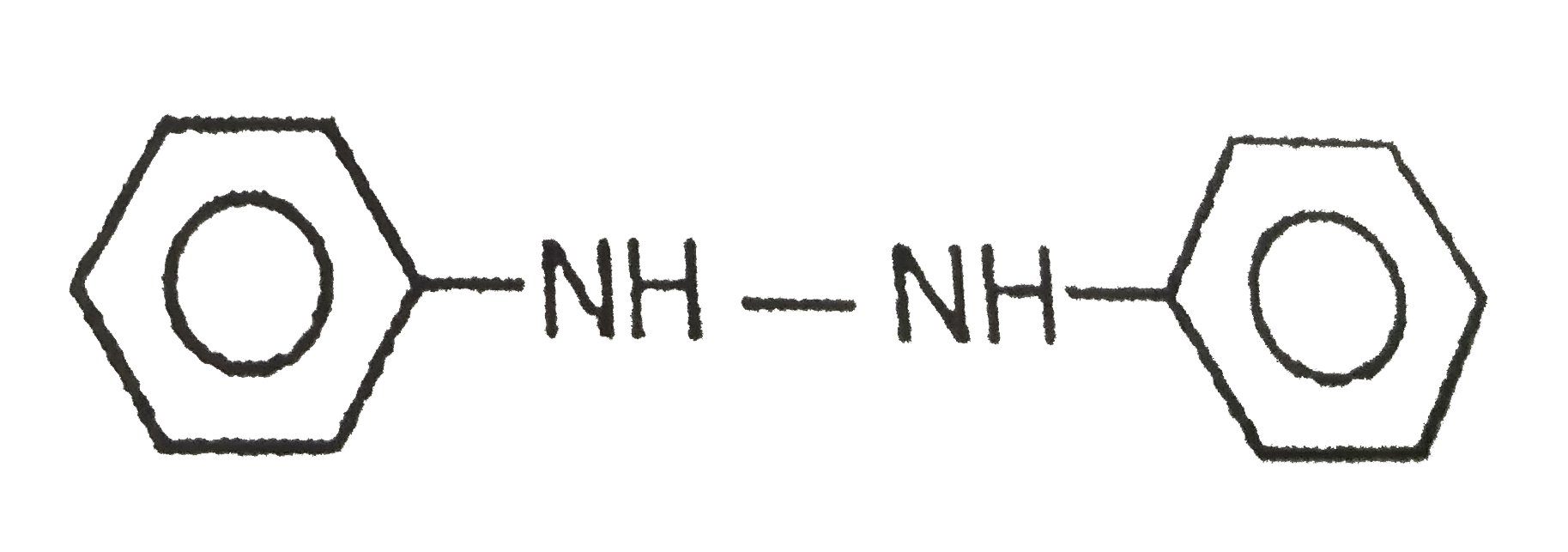
Reduction of nitrobenzene with LiAlH_(4) gives azobenzene. The number of hydrogen atoms involved in the formation of the product is |
|
Answer» |
|
| 37. |
Reduction of nitrobenzene with H_2in presence of platinum gives underline ("nitrosobenzene"). |
| Answer» SOLUTION :REDUCTION of NITROBENZENE with H, in presence of platinum GIVES aniline. | |
| 38. |
Reduction of nitroalkanes in neutral medium (e.g. Zn/NH_(4)Cl) forms mainly |
|
Answer» `R-NH_(2)` |
|
| 39. |
Reduction of nitroalkane produces (1) 1^(@) -amines (2) N-alkyl hydroxyl amine (3) oxime |
|
Answer» 1,3 |
|
| 40. |
Reduction of nitrobenzene using LiAlH_(4) gives |
|
Answer» Azobenzene 
|
|
| 41. |
Reduction of nitrobenzene by which of the following reagent gives aniline? |
|
Answer» Sn/HCl |
|
| 42. |
Reduction of MnO_(4)^(-) in strongly basic medium gives : |
| Answer» Answer :C | |
| 43. |
Reduction of N-methylacetamide with LiAlH_(4) in ether gives_______ |
| Answer» SOLUTION :ETHYLMETHYLAMINE (N-methylethanamine). | |
| 44. |
Reduction of ketones canot be carried out with which of the following reagents? |
|
Answer» HYDROGENIN the presence of palladiun deposited over barium sulphate in the presence of quinoline |
|
| 45. |
Reduction of metal sulphides directly with carbon is not possible. Why? |
| Answer» Solution :In the REDUCTION of metal SULPHIDES with CARBON, carbon disulphide `(CS_(2))`is supposed to be formed . `CS_(2)` is thermodynamically less stable than metal sulphides. Thus reduction of metal sulphides directlywith carbon is notpossible | |
| 46. |
Reduction of metal oxidesby thermitprocess becomes faster justafterignition . Why ? |
|
Answer» Solution :Aluminium is used as reductantin thermitprocess and the process is HIGHLY exothermic `Fe_(2) O_(3) + 2A1 overset(1000 - 1500^(0) C) to A1_(2) O_(3) + 2Fe` The ignition helps to overcomethe energy of activitation and hence the REACTION is fast. |
|
| 47. |
Reduction of lactic acid with excess of HI gives : |
|
Answer» PYRUVIC ACID |
|
| 48. |
Reduction of ketones cannot be carried out with which of the following reagents? |
|
Answer» Hydrogen in presence of palladium in barium sulphate and quinoline |
|
| 49. |
Reduction of ketone gives _______ . |
|
Answer» PRIMARY alcohol |
|