Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Resistance of 0.2M solution of an electrolyte is 50Omega. The specific conductance of the solution of 0.5M solution of the same electrolyte is 280Omega. The molar conductivity of 0.5M solution of the electrolyte is S m^(2)mol^(-1) is |
|
Answer» `5xx10^(-4)` `R=50Omega` `M=0.2 ` `K=(1)/(R)xx(l)/(A)` `implies(l)/(A)=1.4xx50m^(-1)`. Now, NEW solution has M=0.5, `R=280Omega` `impliesK=(1)/(R)xx(l)/(A)=(1)/(280)xx1.4xx50=(1)/(4)` `implies lamda_(M)=(K)/(1000xxM)=((1)/(4))/(1000xx0.5)=(1)/(2000)=5xx10^(-4)` |
|
| 2. |
Resistance of 0.2 M solution of an electrolyte is 50 Omega. The specific conductance of the solution of 0.5 M solution of same electrolyte is 1.4 S m^(-1) and resistance of same solution of the same electrolyte is 280 Omega. The molar conductivity of 0.5 M solutions of the electrolyte is 5 m^(2) mol^(-1) is |
|
Answer» `5 XX 10^(-4)` |
|
| 3. |
Resistance of 0.2 m solution of an electrolyte is 50 Ohm^(-1). The specific conductance of the solution is 1.3 Sm^(-1). If resistance of 0.4 m solution of the same electrolyte is 260 Ohm^(-1), its molar conductivity is ………… . |
|
Answer» `62.5 Sm^2 mol^(-1)` |
|
| 4. |
Resistance of 0.2 M electrolytic solution is 50Omega. And its specific conductivity is 1.4 S m^(-1). If specific resistance of 0.5 M same electrolytic solution is 280Omega. Then what is the molar conductivity of 0.5 M electrolytic solution in S m^(2) ? |
|
Answer» `5XX10^(3)` |
|
| 5. |
Resistance of 0.05 M electrolytic solution at 298K temperature is 30.0 Omega. The cross sectional area of conductivity cell having Pt electrode is 3.8cm^(2) and distance between two electrode is 1.5 cm, then what is the molar conductivity of electrolytic solution ? |
|
Answer» SOLUTION :Conductivity constant for cell : `G^(**)=(l)/(A)=(1.5cm)/(3.8cm^(2))=0.3947cm^(-1)` Where, R=resistance of solution=`30.0Omega` c=concentration of solution=0.05 mol `L^(-1)` Conductivity of solution : `kappa=(G^(**))/(R)=(1.5cm)/(3.8cm^(2))xx(1)/(30Omega)` `=0.013158cm^(-1)Omega^(-1)~~0.0132" S "CM^(-1)` Molar conductivity : `Lamda_(m)=(1000kappa)/(c)=(1000xx0.0132" S "cm^(-1))/(0.05" mol "cm^(-3))` `=264" S cm"^(2)mol^(-1)` |
|
| 6. |
Resemblance between Nb and Ta is because they |
|
Answer» belong to same group of perioidc table |
|
| 7. |
Required amount of crystalline oxalic acid (eq. eq.=63) to prepare N/10 250 ml oxalic acid solution is |
|
Answer» 0.158 g |
|
| 8. |
Representative elements are elements of |
|
Answer» s - BLOCK |
|
| 9. |
Representative elements belong to |
|
Answer» s and p-block |
|
| 10. |
Represent the galvanic cell in which the reaction Zn(s)+Cu^(2+) (aq) to Zn^(2+) (aq)+Cu(s) takes place. |
|
Answer» SOLUTION :MAY be represented as: `ZN(s)//Zn^(2+) (aq)||CU^(2+) (aq) |Cu(s)` |
|
| 11. |
Represent the galvanic cell in which the following reaction takes place : Zn(s)+2Ag^(+)(aq)toZn^(2+)(aq)+2Ag(s) (i) The reaction taking place at each of its electrodes. (ii) The carriers of current within this cell. |
|
Answer» SOLUTION :(i) At ANDOE, `ZntoZn^(2+)+2E` At cathode, `Ag^(+)+etoAg` (ii) `Zn^(2+)andAg^(+)ions`. |
|
| 12. |
Represent the galvanic cell based on the cell reaction given below:Cu+2Ag^+to Cu^(2+)+2Ag |
| Answer» SOLUTION :`Cu|Cu^(+2)||Ag^+|Ag` | |
| 13. |
Represent the chemical forula of iron rust. |
|
Answer» |
|
| 14. |
Represent the cell in which the following reaction takes place: Mg(s)+2Ag^(+)(0.0001M)toMg^(2+)(0.130M)+2Ag(s) Calculate its E_(cell). Given that E_(Mg^(2+)//Mg)^(@)=-2.37V and E_(Ag^(+)//Ag)^(@)=+0.80V |
|
Answer» Solution :Here, we are given reduction potential as `E_(Mg^(2+),Mg)=-2.37,E_(AG^(+),Ag)=+0.80V` As the emf of the cel must be positive, this can be so only if oxidation takes PLACE at the MAGNESIUM electrode, hence, the electrode reaction will be `MgtoMg^(2+)+2e^(-)` (At anode) `2Ag^(+)+2e^(-)to2Ag` (At cathode) Thus, the cell may be REPRESENTED as `Mg|Mg^(2+)(0.130M)||Ag^(+)(0.0001M)|Ag` Standard emf of the cell will be: `E_(cell)^(@)=`Std. Red. Pot. Of R.H.S. electrode-Std. Red. Pot. Of L.H.S. electrode=0.80-(-2.37)=3.17V The overall reaction is: `Mg+2Ag^(+)hArrMg^(2+)+2Ag""(n=2)` Applying nernst eqn., we get `E_(cell)=E_(Cell)^(@)-(0.0591)/(n)"log"([Mg^(2+)])/([Ag^(+)]^(2))` `E_(cell)=E_(cell)^(@)-(0.0591)/(2)"log"(0.130)/((10^(-4))^(2))=E_(Cell)^(@)-(0.0591)/(2)"log"(0.130)/(10^(-8))` `=3.17-0.02955log(1.30xx10^(7))=3.17-0.02955xx(7.1139)=3.17-0.21=2.96`volt. ALTERNATIVELY, this problem may be solved by first calculating the electrode potentials of the two electrodes separately and then calculating the emf from the electrode potentials. |
|
| 15. |
Represent the cell in which the followig reaction takes place, Mg_((s))+2Ag_((0.0001M))^(+)to Mg_((0.130M))^(2+)+2Ag_((s)) Calculate its E_(cell) if E_(cell)^(Theta)=3.17V |
|
Answer» Solution :In this cell `Mg_((s))|Mg^(2+)` is anode because Mg get oxidized and also `Ag^(+)` is reduced in Ag so on cathode `Ag^(+)|Ag` will be on RIGHT side. Between these two ELECTRODES salt bridge is DENOTED by two verticle line || and hence the symbolic representation of cell will be `Mg_((s))|Mg_((0.130M))^(2+)||Ag_((0.0001M))^(+)|Ag_((s))` Calculation for `E_(cell)` potential is as follows: `E_(cell)=E_(cell)^(Theta)-(RT)/(nF)LN([Mg^(2+)])/([Ag^(+)]^(2))` `therefore E_(cell)=3.17V-(0.0591)/(2)"log"([Mg^(2+)])/([Ag^(+)]^(2))` `=3.17V-(0.0591)/(2)"log"(0.130)/((0.0001)^(2))` `=3.17-0.2955log(1.33xx10^(+7))` `=3.17-0.2955(+7.1139)` `=3.17-0.2099=3.17-0.21=2.96V` |
|
| 16. |
Represent the cell in which the following reaction takes place Mg(s)+2Ag^(+)(0.001M) to Mg^(2+)(0.130)+2Ag(s) Calculate E_("cell")" if "E_("cell")^(0)=3.17V. |
|
Answer» Solution :Represent of the cell `Mg//Mg^(2+)(0.130M)||Ag^(+)(0.01M)//Ag` Cell reaction `Mg+2Ag^(+) to Mg^(2+)+2Ag` FORMULA: `E_("cell")=E_("cell")^(0)-(0.059)/(N)LOG""([Mg^(2+)])/([Ag^(+)]^(2)]` Substitution `E_("cell")=3.17-(0.059)/(2)log"" ([0.130])/([0.0001)]^(2))` =2.96V |
|
| 17. |
Replacement of -N_2^+Cl^- from benzene diazonium chloride by iodine can be done by using |
|
Answer» HI |
|
| 19. |
Replacement of Cl of chlorobenzene to give phenol requires drastic conditions but chlorine of 2,4-dinitrochlorobenzene is readily replaced because |
|
Answer» `NO_(2)` make ring electron rich at ORTHO and para 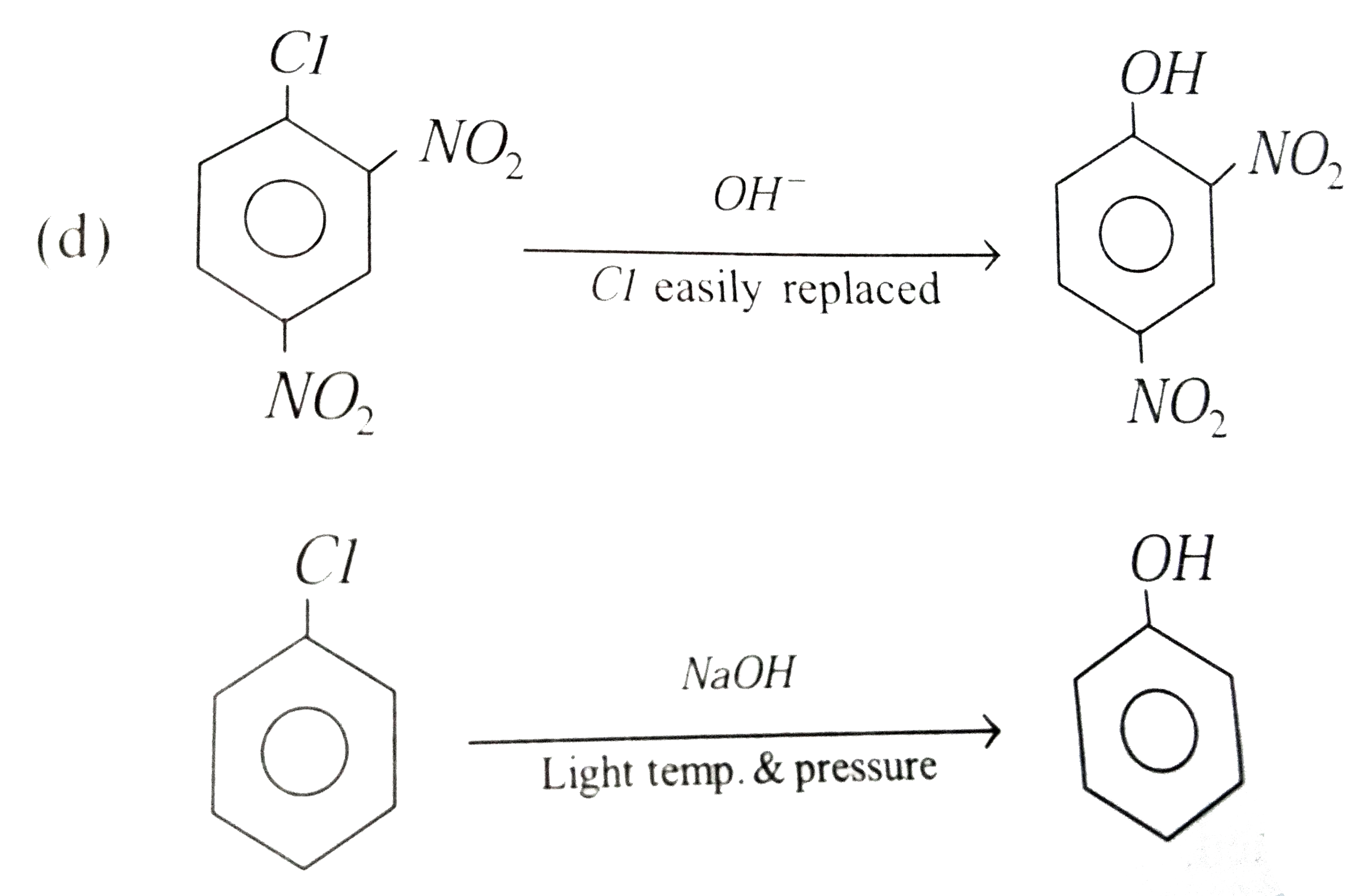
|
|
| 20. |
Replacement of diazonium group by halogen atom can be done by the reaction |
|
Answer» Ullmann reaction |
|
| 21. |
Replacement of diazonium group by chlorine atom can be carried but by use of |
|
Answer» AgCl |
|
| 22. |
Replacement of Cl of chlorobenzene to give phenol requires drastic conditions, but Cl of 2,4-dinitrochlorobenzene is readily replaced. This is because |
|
Answer» `-NO_(2)` group makes the RING electron rich at ORTHO and para position |
|
| 23. |
Replacement of CI of chloro benzene to give phenol requires drastic condition but CI of 2. 4-6-trinitro chloro benzene is really replaced because |
|
Answer» `NO_2`makes ring electrons RICH at ortho and para positions |
|
| 24. |
Replacement- N_(2)^(+)Cl^(-) frombenzene diazonium chloride byhydrogenatomcanbedone by using |
| Answer» Answer :D | |
| 25. |
Repeateduse of which one of the following fertilizers would increase the activity of the soil ________ |
|
Answer» AMMONIUM sulphate |
|
| 26. |
Repeating units teflon is |
|
Answer» `-CF_3-CF_3-` |
|
| 27. |
Replacement by -N_(2)^(+)Cl^(-) frombenzenediazonium choridebycan bedone byusing |
|
Answer» HI |
|
| 28. |
Reonance is due to |
|
Answer» Delocalization of SIGMA ELECTRONS |
|
| 29. |
Remsay was awarded Noble Prize for the discovery of rare gases in |
| Answer» SOLUTION :N//A | |
| 30. |
Removal of unwanted meterial from the ore is called……………. |
| Answer» SOLUTION :CONCENTRATION | |
| 31. |
Removal of unreacted oxide ore, other metals, non metals associated with isolated crude metal is called ________ |
|
Answer» leaching |
|
| 32. |
Removal of hydrogen atom is easier when it is attached to: |
|
Answer» `1^@` CARBON |
|
| 33. |
Removal of a hydride ion from a methane molecule will give a: |
|
Answer» METHYL radical |
|
| 34. |
..............relieve pain and produces steeps and they are additive. |
| Answer» SOLUTION :NARCOTIC Analgegics (or) OPIOIDS | |
| 35. |
Relative rate of SN^(1) reaction for t-butyl bromide? |
|
Answer» 37 |
|
| 36. |
Relative rate of methyl bromide in SN^(2) reaction is |
|
Answer» 1 |
|
| 37. |
Relative lowering of vapour pressure of a dilutesolutionof glucose dissolved in 1 kgof wateris 0.002. The molality of the solution is . |
|
Answer» 0.004 Molalityof solution MEANS the numberof MOLES of glucose PRESENTIN 100 g of water . `w_(1)= 1000 g, n_(1) = (1000)/(18) = 55.55` `(n_(2))/(n_(2) + 55.55) = 0.002` For dilutesolution , 55.55 + `n_(2) = 55.55` `THEREFORE n_(2) = 0.002 xx 55.55 = 0.1111` |
|
| 38. |
Relative lowering of vapour pressure is ____ the mole fraction of the solute. |
| Answer» Solution :equal to | |
| 39. |
Relative lowering of vapour pressure of al dilute solution of gluscose dissolved in 1kg of water is 0.002.The molality of the solution is |
|
Answer» `0.004` Given: `(p^(@)-p_(s))/(p^(@)) = 0.002, n_(1(H_(2)O)) = (1000)/(18)` moles For dilute solution, `n_(2)` is molality `= 0.002 XX = (1000)/(18)` m = 0.111 m |
|
| 40. |
Relative lowering of vapour pressure is a colligative property because …………….. . |
|
Answer» It DEPENDS on the CONCENTRATION of a non-electrolyte solute in solution and does not depend on the nature of the solute MOLECULES |
|
| 41. |
Relative lowering of vapour pressure is a colligative property because |
|
Answer» It depends on the xoncentration of a non-electrolytesolute in solution and does not depend on the NATURE of the SOLUTE MOLECULES |
|
| 42. |
Relative lowering of vapour pressure, elevation of boiling point, depression of freezing point and osmotic pressure are important colligative properties of dilute solutions. Relative lowering of vapour pressure of an aqueous dilute solution of glucose is 0.018 What is the mole fraction of glucose in the solution ? |
| Answer» Solution :Mole fraction of glucose-Relative LOWERING of VAPOUR PRESSURE= 0.018 | |
| 43. |
Relative lowering of vapour pressure is a colligative property because …….. |
|
Answer» It depends on the concentration of a non - ELECTROLYTE solute in SOLUTION and does not DEPEND on the nature of the solute molecules. |
|
| 44. |
Relative decrease in vapour pressure of an aqueous solution containing 2 moles [Cu(NH_(3))_(3)Cl]Cl in 3 moles H_(2)O is 0.50. on reaction with AgNO_(3), this solution will from |
|
Answer» `1 MOL AGCL` |
|
| 45. |
Relationship between products (Q) and (S) is |
|
Answer» POSITIONAL isomer |
|
| 47. |
Relationship between DeltaG^(@) and K_(P) is……. |
|
Answer» <P>`DeltaG^(@)`=RT In `K_(P)` |
|
| 48. |
Relation between standard cell potential and equilibrium constant is |
|
Answer» `E^(@) = (N RT)/(F)` In K |
|
| 49. |
Relation between standard e.m.f. of a cell and equilibrium constant is ________. |
| Answer» SOLUTION :`(E_(CELL)=(2.303RT)/(NF)IogK_c)` | |