Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Reactivity of halogens towards alkanes is in order |
|
Answer» 'Butene GT PROPENE gt ETHENE' |
|
| 2. |
Reactivity of alcohol in breaking of C-O bond is |
|
Answer» `1^@ GT 2^@ gt 3^@ ` |
|
| 3. |
Reactions that have standard free energy changes lessthan zero always have equlibrium constnat equal to |
|
Answer» Unity When `DeltaG^(@)lt0i.e.-ve, then K_(c)GT1.` In this case forward REACTON is FEASIBLE showing THEREBY a large concentration of products. |
|
| 4. |
Reactivity of acids in esterification follows the order: |
|
Answer» HCOOH `GT CH_(3)`COOH `gt RCH_(2)` COOH`gt R_(2)`CHCOOH `gt R_(3)`CCOOH |
|
| 5. |
Reactivity of alcohol in breaking O-H bond is |
|
Answer» `1^@ GT 2^@ gt 3^@ ` |
|
| 6. |
Reactivity of acids in esterification follows the order : |
|
Answer» `HCOOHgtCH_3COOHgtRCH_2COOHgtR_2CHCOOHgtR_3C COOH` |
|
| 7. |
Reactivating in halogen substituted benzene rings is controlled by |
|
Answer» RESONANCE |
|
| 8. |
Reactions taking place in a fuel cell are: |
|
Answer» `O_(2)(g)+2H_(2)O(L)+4E^(-)to4OH^(-)(aq)` at the CATHODE |
|
| 9. |
Reactions of zeolite catalysts depend on............... |
|
Answer» pores |
|
| 10. |
Reactions involving stereoisomers To study the involvement of stereoisomers in chemical reactions we shall take up following points: (i)The conversion of an achiral molecule into a chiral molecule, with the generations of a chiral centre. Secondary butyl chloride exist as two enantiomers but mixture is optically inactive due to formation of a racemic mixture.Both enantiomers are obtained in equal amounts.Racemic mixture is an eqnimolar mixture of two enantiomers. (ii)Conversion of chiral molecule into chiral and achiral molecules. Monochlorination of which of the following will give only an achiral products ? |
|
Answer» n-Pentane |
|
| 11. |
Reactions involving stereoisomers To study the involvement of stereoisomers in chemical reactions we shall take up following points: (i)The conversion of an achiral molecule into a chiral molecule, with the generations of a chiral centre. Secondary butyl chloride exist as two enantiomers but mixture is optically inactive due to formation of a racemic mixture.Both enantiomers are obtained in equal amounts.Racemic mixture is an eqnimolar mixture of two enantiomers. (ii)Conversion of chiral molecule into chiral and achiral molecules. Which of the following reactant will give chiral products on monochlorination ? |
|
Answer» `CH_3-CH_2-CH_3` |
|
| 12. |
Reactions involving gold have been of particular interest to a chemist. Consider the following reactions, {:(Au (OH)_(3) + 4 HCl rarr HAuCl_(4) + 3 H_(2) O",",Delta H = - 28 k Cal,),(Au (OH)_(3) + HBr rarr HAuBr_(4) + 3 H_(2)O",",Delta H = -36.8 k Cal,):} In an experiment there was an absorption of 0.44 k Cal when one mole of HAuBr_(4) was mixed with 4 moles of HCl. what is the percentage conversion of HAuBr_(4) into HAuCl_(4) ? |
|
Answer» 0.005 `HAuBr_(4)+4HCL rarr HAuCl_(4)+4HBr` `DeltaH=8.8` `%` CONVERSION `=100/8.8 xx0.44=5%` |
|
| 13. |
Reactions involving heterolytic fission are said to proceed via: |
|
Answer» IONIC mechanism |
|
| 14. |
reactions I and II are limited mainly to naphthalene compounds. These reactions are called, respectively: |
|
Answer» Buchere REACTION and its reversal. |
|
| 15. |
Reactions in Zeolite catalyst depend on, |
|
Answer» PORES |
|
| 16. |
'Reaction with zero order is possible but zero molecularity is not''.Justify the statement. |
|
Answer» SOLUTION :MOLECULARITY of a reaction is the NUMBER of molecules, atoms or ions which collide simultaneously to bring about reaction. So it is always a natural number. Zero molecularity is no possible. Order is the sum of power of concentration term in rate LAW. It can be zero. |
|
| 17. |
Reaction which will produce chiral product : |
| Answer» Answer :A::B::C::D | |
| 18. |
Reaction used to convert acid to 1^@ alcohol is |
|
Answer» OXIDATION |
|
| 19. |
Reaction used for the preparation of ethers is |
|
Answer» Reimer-Tiemann reaction |
|
| 20. |
Reaction that takes place at graphite anode in dry cell is |
|
Answer» `Zn^(2+)+2d^(-)rarrZn(s)` `Zn(s)rarrZn^(2+)(AQ)+2e^(-)` |
|
| 21. |
Reaction taking place at anode in dry cell is : |
|
Answer» `ZN^(2+) +2E rarr Zn(s)` |
|
| 22. |
Reaction taking place at anode in Daneil cell |
|
Answer» `ZN^(2+) + 2 e to Zn(s)` `E_(Cu^(2+) | Cu) gt E_(Zn^(2+) | Zn)` |
|
| 23. |
Reaction rate between two substance A and B is expressed as following, rate=k[A]^(n)[B]^(m) If the concentration of A is doubled and concentration of B is made half of initial concentration, the ratio of the new rate to the earlier rate will be, |
|
Answer» `m+n` `R_(2)=k[2A]^(n)[(1)/(2)B]^(m)` `THEREFORE (R_(2))/(R_(1))=(k[2A]^(n)[(1)/(2)B]^(m))/(k[A]^(n)B^(m))=2^(n)((1)/(2))^(m)=2^(n)*2^(-m)=2^(n-m)` |
|
| 24. |
Reaction takes place at anode is |
| Answer» Solution :Oxidation occurs at anode. | |
| 25. |
Reaction of oxirane with RMgX followed with hydrolysis produces : |
|
Answer» `RCHOHCR_3` |
|
| 26. |
Reaction of which among the following ethers with Hl in cold leads to formation of methyl alcohol? |
|
Answer» ETHYL METHYL ETHER |
|
| 27. |
Reaction of which among the followingetherswith HI in cold leads to the formation of methyl alcohol? |
|
Answer» etyhy METHYL ETHER |
|
| 28. |
Reaction of which among the following ethers with HI in cold leads to formation of alcohol? |
|
Answer» ethylmethyl ether |
|
| 29. |
Reaction of t-butyl bromide with sodium methoxide produces |
|
Answer» SODIUM TERTIARY butoxide |
|
| 30. |
Reaction of t-butyl bromide with sodium methoxide produces : |
|
Answer» Isobutane |
|
| 31. |
Reaction of solid KMnO_(4) with conc. H_(2)SO_(4) produces manganeses heptoxide (Mn_(2)O_(7)) in |
|
Answer» Solution state `2KMnO_(4)+3H_(2)SO_(4)rarr2KHSO_(4)+(MnO_(3))_(2)SO_(4)+2H_(2)O` `(MnO_(3))_(2)SO_(4)+H_(2)Orarrunderset("liquied")underset("red-brown")(Mn_(2)O_(7))+H_(2)SO_(4)` `Mn_(2)O_(7)rarr2MnO_(2)+(3)/(2)O_(2)` `therefore Mn_(2)O_(7)` so formed is a red-brown LIQUID. |
|
| 32. |
Reaction of solid KMnO_4 with conc H_2SO_4 produces an explosive substance |
|
Answer» `MnO_2` |
|
| 33. |
Reaction of solid KMnO_4 with conc H_2SO_4 produces maganese heptoxide (Mn_2O_7) in: |
|
Answer» SOLUTION state |
|
| 34. |
Reaction of ROH with R'MgX produces |
|
Answer» RH 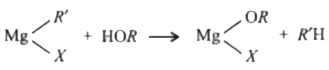
|
|
| 35. |
Reaction of SO_(2) with water and alkali is similar to that of |
| Answer» ANSWER :A | |
| 36. |
Reaction of SO_(2) with water na alkali is similar to that of |
| Answer» ANSWER :A | |
| 37. |
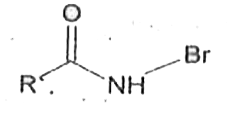
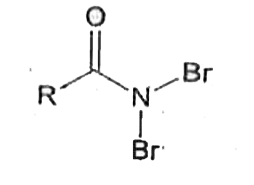
Reaction of RCONH_(2) with a mixture of Br_(2) and KOH gives RNH_(2) as the main product. The intermediates involved in the reaction are |
|
Answer» `R-OVERSET(O)overset(||)C-NHBr` |
|
| 38. |
Reaction of R - overset(O)overset(||)(C) - NH_(2)with a mixtureof Br_(2)andKOHgives R-NH_(2)as the mainproduct . The intemediates (s) involved in thisreaction is/are. |
|
Answer»
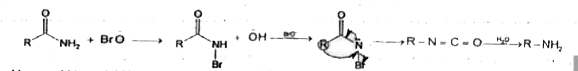
|
|
| 39. |
Reaction of RCNH_(2) with a mixture of Br_(2)and KOHgives R-NH_(2) as the main product the intermediate involved in the reaction is |
|
Answer» `R-OVERSET(O)overset(||)C-Br` |
|
| 40. |
Reaction of R-COOH with N_(3)H gives RNH_(2) as the main product The intermediates involved in this reaction are: |
|
Answer» `RNHNH_(2)` |
|
| 41. |
Reactionof R'- OH withR' - Mg Xproduces : |
|
Answer» R-H |
|
| 42. |
Reaction of phenol with dil. HNO_(3) gives |
|
Answer» <P>p and m-nitrophenols 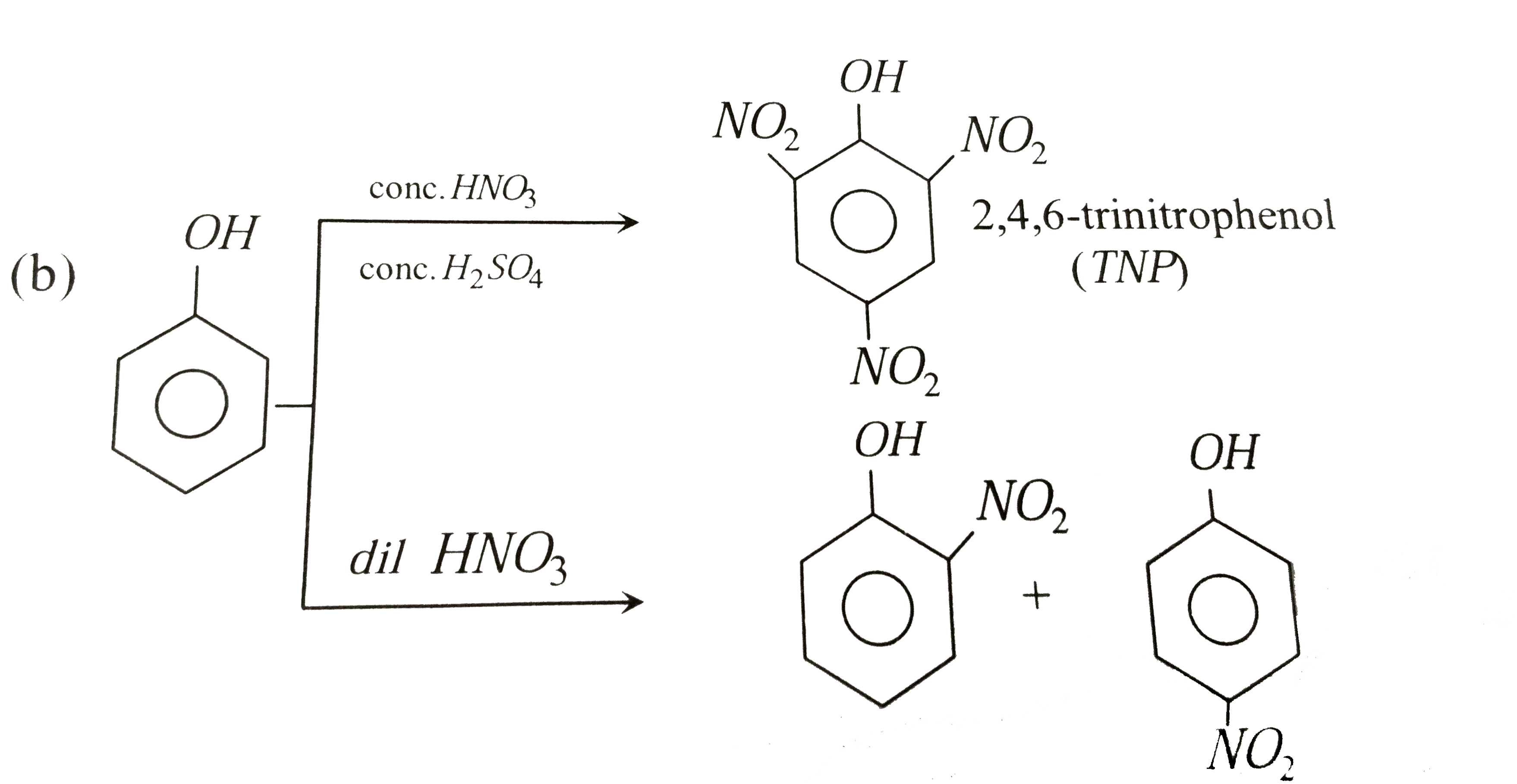
|
|
| 43. |
Reaction of primary amine with aldehydes gives : |
|
Answer» Amide |
|
| 44. |
Reaction of phenol with chloroform/sodium hydroxide to give o-hydroxy benzaldehyde involves the formation of |
|
Answer» DICHLORO CARBINE |
|
| 45. |
Reaction of phenol with chloroform in presence of dilute sodium hydroxide finally introduces which one of the following functional group |
|
Answer» `-CH_(2)Cl` 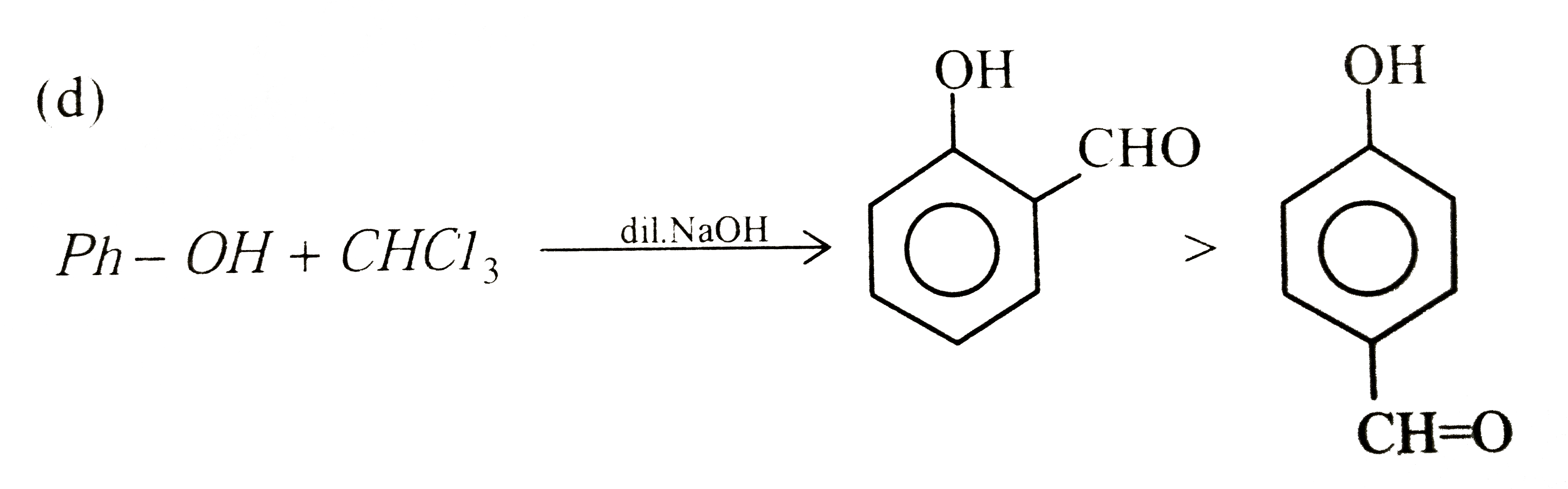
|
|
| 46. |
Reaction of phenol with chloroform/sodium hydroxide to give o- hydroxy benzaldehyde involves the formation of |
|
Answer» Dichloro carbene `OH^(-)+CHCl_(3) hArr HOH+:"CC"l_(2)^(-) to CL^(-)+:"CC"l_(2)` |
|
| 47. |
Reaction of phenol with chloroform in presence of dilute sodium hydroxide finally introduces which one of the following functional group ? |
|
Answer» `-CHCl_2` |
|
| 48. |
Reaction of phenol with chloroform in presence of dilute sodium hydroxide finally introduces which one of the following functional group? |
|
Answer» `-COOH` |
|
| 49. |
Reaction of phenol with chloroform in presence of dilute sodium hydroxide finally introduce which one of the following fuctional group? |
|
Answer» `-CHCl_2` |
|