Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Reaction of one molecule of HBr with one molecule of 1,3-butadiene at 40^@ C gives predominantly |
|
Answer» 3-bromobutane under KINETICALLY CONTROLLED conditions |
|
| 2. |
Reaction of one molecule of HBr wit one moleculae 1,3-butadiene at 40^(@)Cgives predominnantly |
|
Answer» 3-bromobutene under kinetically CONTROLLED CONDITIONS |
|
| 3. |
Reaction of nitrous acid on 1^@ aliphatic amines in cold will give : |
|
Answer» A DIAZONIUM SALT |
|
| 4. |
Reaction of methyl bromide with aqueous sodium hydroxide involves …………………. |
|
Answer» RACEMISATION |
|
| 5. |
Reaction of methyl bromide with aqueous sodium hydroxide involves |
|
Answer» RECEMISATION |
|
| 6. |
Reaction of m-dinitro benzene with (NH_(4))_(2)S_(x) gives |
|
Answer»
|
|
| 7. |
Reaction of KMnO_4 in neutral or very weakly acidic solution can be represented as |
|
Answer» `MnO_(4)^(-) + 2H_2O+3e^(-) RARR MnO_(2) + 4OH^(-)` |
|
| 8. |
Reaction of hydrogen bromoide with propene in the absence of peroxide is a/an |
|
Answer» FREE radical addition |
|
| 9. |
Reaction of HNO_(3) withI, S, P and C gives respectively |
|
Answer» `HIO_(3), H_(2)SO_(4), H_(3)PO_(4)` and `CO_(2)` `6HNO_3 + S overset(Delta)(rarr)H_2SO_4 + 6NO_2 + 2H_2O` `20 HNO_3 + P_(4) overset(Delta)(rarr) 4H_3PO_4 + 20 NO_2 + 4H_2O` `4HNO_3 + C overset(Delta)(rarr)CO_2 + 4NO_2 = 2H_2O` |
|
| 10. |
Reaction of HI si easiest with |
|
Answer» `CH_(3)OH` |
|
| 11. |
Reaction of HBr with propene in the presence of peroxide gives |
|
Answer» Allyl bromide Step-1:peroxide undergo FISSION togive free radicals `R-O-O-R to 2 -R-overset(.)O` Step-2:HBr COMBINES with free radical to form BROMINE free radical `R-overset.O+HBr to R-OH+overset.Br` Step-3:`Boverset.r` attacks the double BOND of the alkene to form a more stable free radical 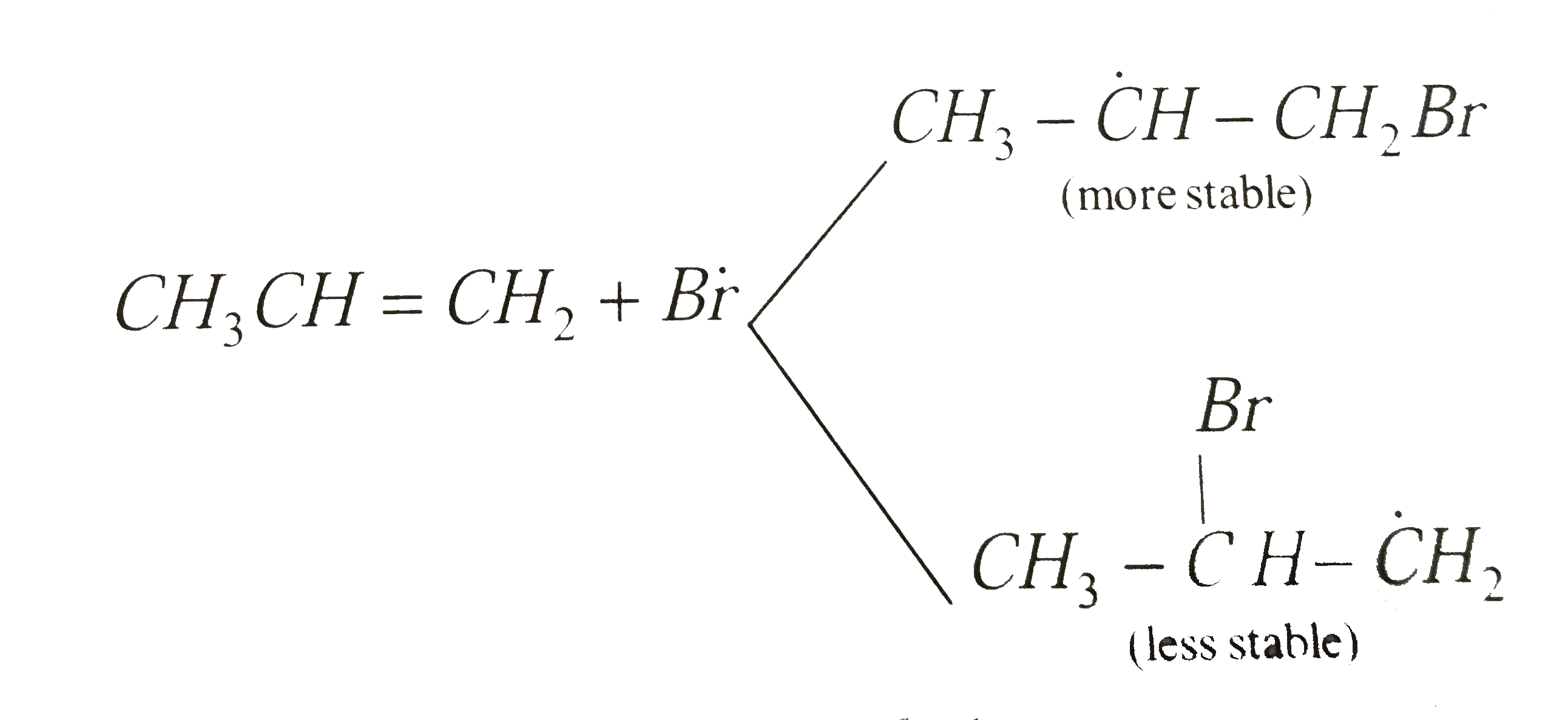 Step-4:More stable free radical attacks the HBr `CH_3-dotCH-CH_2-Br+HBr to underset"n-propyl bromide"(CH_3CH_2CH_2Br+Br)` Step-5: `Bdotr+Bdotr to Br_2` |
|
| 12. |
Reaction of gasous fluorine (F_2) with compound X yields a single product Y, whose mass precent composition is 61.7% F and 38.3% Cl.Calculate Delta_fH^@ (in kJ/mol) for the synthesis of Y using following information 2ClF(g)+O_2(g)toCl_2O(g)+OF_2(g) DeltaH^@=205.6 kJ 2ClF_3(g)+2O_2(g)toCl_2O(g)+3OF_2(g) DeltaH^@=533.0 kJ Delta_f -I^@(OF_2, g)=24.7 kJ/mol |
|
Answer» no. of moles `overset(Cl)(38.7/35.5=1.09)oversetF(61.7/19=3.42)` Simplest RELATIVE ratio 1:3 So, product Yis `CiF_3` & compound X is CIF For the synthesis of Y reaction is `F_2(G)+CIF(g)toCIF_3(g)` `1//2Cl_2O(g)+3//2OF_2(g)toCIF_3(g)+O_2(g) " " DeltaH^@=-533.0//2 KJ implies -266.5 kJ` `CIF(g)+1//2O_2(g)to1//2Cl_2O(g)+1//2OF_2" " DeltaH^@=205.6//2 kJ implies 102.8 kJ` `(F_2(g)+1/2O_2(g)toF_2O(g) " " DeltaH^@=24.7 kJ)/(CIF(g)+F_2(g)toCIF_3(g) " "Delta_fH^@=-139kJ//mol ` |
|
| 13. |
Reaction of glucose with (CH_3CO)_2O suggest that |
|
Answer» pentahydroxy aldehyde |
|
| 14. |
Reactionof ethylformatewith excessof CH_(3)Mglfollowed by hydrolysis gives . |
|
Answer» n-propyl ALCOHOL 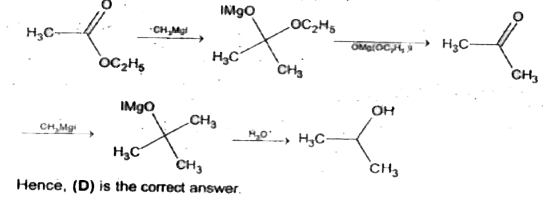
|
|
| 15. |
Reaction of formaldehyde and ammonia gives |
|
Answer» hexamethylene tetramine `6HCHO + 4NH_(3) to UNDERSET("Hexamethylene tetramine")((CH_(2))_(6)N_(4)) + 6H_(2)O` |
|
| 16. |
Reaction of ferrous salts withKMnO_4 is a: |
|
Answer» NEUTRALISATION reaction |
|
| 17. |
Reaction of ethylamine with chloroform in alcoholic KOH gives: |
|
Answer» `C_(2)H_(5)CN` |
|
| 18. |
Reaction of ethyl chloride with sodium leads to : |
|
Answer» Ethane |
|
| 19. |
Reaction of ethyl amine with alkaline chloroform leads to the formation of carbylamine reaction. This reaction involves the attack of an electrophile on ethyl amine, the electrophile is |
|
Answer» `H_(3)O^(+)` |
|
| 20. |
Reaction of ethyl bromide with sodium cyanide gives a mixture of ethyl cyanide and ethyl isocyanide because : |
|
Answer» the products are ISOMERIC |
|
| 21. |
Reaction of ethene with Br_2 in C Cl_4 gives |
|
Answer» bromoethane |
|
| 22. |
Reaction of etherwith HI is preferredthan HBr because _________. |
|
Answer» HI is strongerthan HBr <BR>HI giveshigherconcentrationof oxonium ION |
|
| 23. |
Reaction of dimethyl ether with cold HI is |
|
Answer» `E^2` REACTION |
|
| 24. |
Reaction of cyclohexanone with dimethylamine in the presence of catalytic amount of an acid forms a compound if water during the reaction is continuously removed. The compound formed is generally known as |
|
Answer» an amine 
|
|
| 25. |
Primary amines on reaction with alcoholic KOH and chloroform yields |
|
Answer» Hydrolysis |
|
| 26. |
Reaction of Cl_(2) with slaked lime. (only reaction) |
| Answer» Solution :`2CA(OH)_(2)+2Cl_(2)rarr CA(Ocl_(2))+CaCl_(2)+2H_(2)O` | |
| 27. |
Reaction of CH_3OCH_2CH_3 is maximum with |
|
Answer» HF |
|
| 28. |
Reaction of chloroform with alcoholic KOH in presence of a primary aromatic amine is called: |
|
Answer» Hydrolysis |
|
| 29. |
Reaction of CH_3COOH with CH_3CH_2MgBr yields: |
|
Answer» An ALKENE |
|
| 30. |
Reaction of C_(6)H_(5)CH_(2)Br with aqueous sodium hydroxide follows |
|
Answer» `S_(N)1` mechanism |
|
| 31. |
Reaction of carbonyl compound with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is followed bhy elimination of water. The reagent is |
|
Answer» HYDRAZINE in PRESENCE of FEEBLY ACIDIC solution |
|
| 32. |
Reaction of carboxylic acids with hydrazoic acid in presence of conc. H_(2)SO_(4) gives______with the evolution of CO_(2) and _____ |
| Answer» SOLUTION :PRIMARY amines, `N_(2)` GAS | |
| 33. |
Reaction of C_(6)H_(5)CH_(2)Br with aqueous sodium hydroxide follows_______. |
|
Answer» `S_(N)1` mechanism and hence it undergoes hydrolysis by `S_(N)1` mechanism and form benzyl alcohol as PRODUCT. |
|
| 34. |
Reaction of C_(6)H_(5)CH_(2)Br with aqueous sodium hydroxide follows ……….. |
|
Answer» `S_(N)1` mechanism 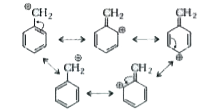
|
|
| 35. |
Reaction of C_(6)H_(5)CH_(2)Br with aqueous sodium hydroxide follows _____ . |
|
Answer» `S_(N)1` mechanism |
|
| 36. |
Reaction of bromine water with glucose suggest that |
|
Answer» `1^(@)` ALCOHOLIC GROUP present in GLUCOSE |
|
| 37. |
Reaction of C_6H_5 CHOwith CH_3NH_2 gives |
|
Answer» `C_6H_5COOH` |
|
| 38. |
Reaction of Br_(2) with Na_(2)CO_(3) in aqueous solution gives NaBr and sodium bormate with evolution of CO_(2) gas. The number of NaBr molecue involved in the balanced chemical equation is …………. |
|
Answer» `{:((i),[Br_(2)+overset(Θ)(O)Hoverset("Hot")toBr^(Θ)+BrO_(3)^(Θ)]),("Note:", "In cold it gives"),((ii), [Br_(2)+Ooverset(Θ)Hoverset("Cold")toBr^(Θ)+BRO^(Θ)]):}]` Both are disproportionation reaction |
|
| 39. |
Reaction of Br_2 on ethylene in presence of NaCl gives |
|
Answer» `BrCH_2-CH_2Br`<BR>`ClCH_2-CH_2Br` |
|
| 40. |
Write the reaction of benzene diazonium chloride with : CuCN |
|
Answer» |
|
| 41. |
Reaction of benzenediazonium chloride with alkaline phenol gives an azo dye. This is an example of |
|
Answer» ELECTROPHILIC substitution |
|
| 42. |
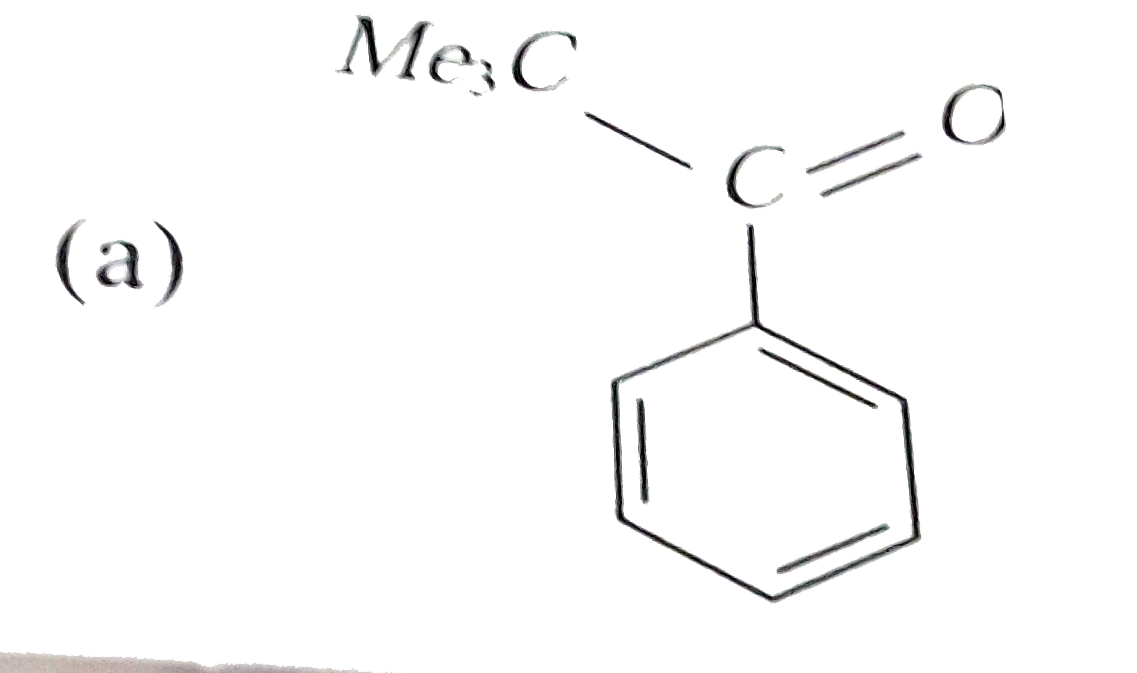
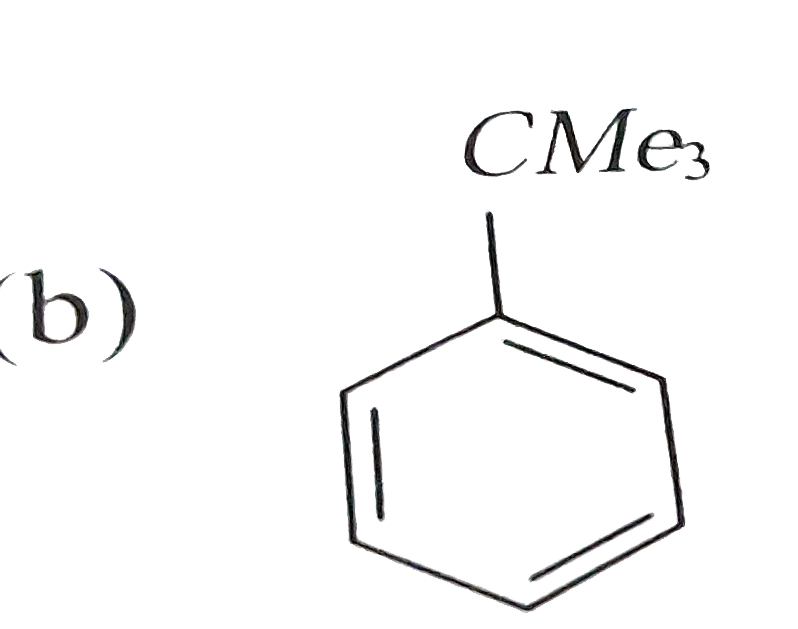
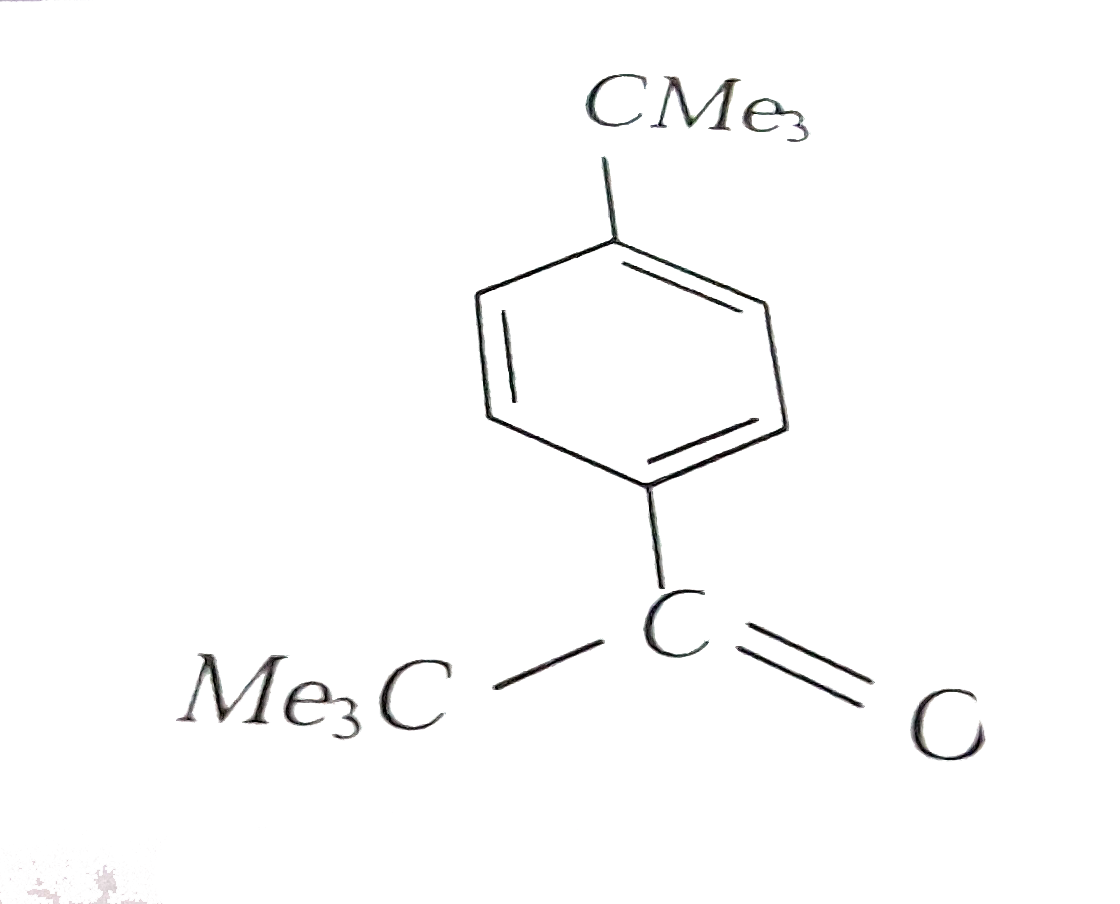
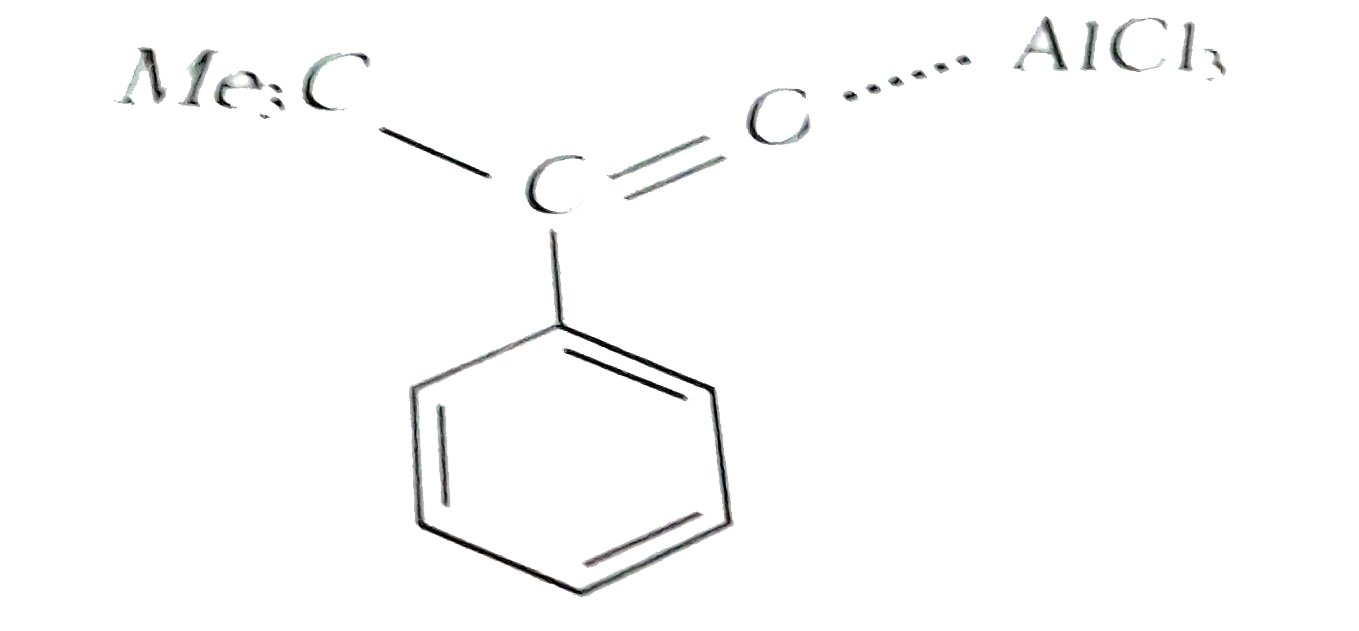
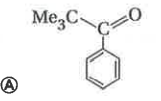
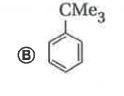
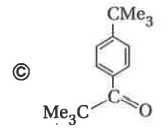
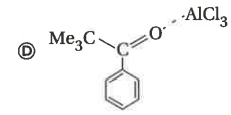
Reaction of benzene with Me_3COCl in the presence of anhydrous AlCl_3 gives |
|
Answer»
|
|
| 43. |
Reaction of benzene with Me_(3)"CCOCl" in the presence of anhydrous AlCl_(3) gives |
|
Answer»
|
|
| 44. |
Reaction of benzene with Me_3C""COCl in the presence of anhydrous AlCl_3 gives- |
|
Answer»
|
|
| 45. |
Reaction of benzene with a mixture of conc.HNO_(3) and ConcH_(2)SO_(4) at about 60^(@)C give nitrobenzene. Nitration of substituted benzenes depends on the nature of the group already present in the ring. Increase of temperature may result in multiple nitration. Which of the following compounds undergo nitration at a rate faster than the other |
|
Answer» Toluene |
|
| 46. |
Reaction of benzene with a mixture of conc.HNO_(3) and ConcH_(2)SO_(4) at about 60^(@)C give nitrobenzene. Nitration of substituted benzenes depends on the nature of the group already present in the ring. Increase of temperature may result in multiple nitration. Nitration of benzene is an |
|
Answer» Elechphilic addition |
|
| 47. |
Reaction of anisole with conc. HNO_(3) and conc. H_(2)SO_(4) will give….. |
|
Answer» PHENOL 
|
|
| 48. |
Reaction of aniline with ......... yields C_6H_5N underset(to )= C and the reaction is known as ........ reaction. |
| Answer» SOLUTION :`CHCl_3 - KOHcarbylamine | |
| 49. |
Reaction of aniline with benzoyl chloride gives : |
|
Answer» Benzoin |
|