Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Reaction of ammonia gas with excess of dichlorine gas produces .......... and ..........products. |
|
Answer» `NCl_(3), H_(2)` |
|
| 2. |
Reaction of aniline with benzaldehyde is |
|
Answer» polymerisation 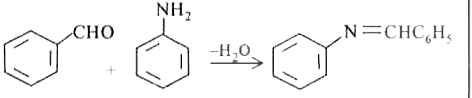
|
|
| 3. |
Reaction of alkyl halides with aromatic compounds in presence of anhydrous AlCl_(3) is known as |
|
Answer» Friedal-Craft reaction `underset("BENZENE")(C_(6)H_(6))+underset("CHLORO methane")(CH_(3)Cl) underset(AlCl_(3))overset(DRY)to underset("Toluene")(C_(6)H_(5)CH_(3))+HCl` |
|
| 4. |
Reaction of aldehyde with NH_(2) - OH will be fastest at which of the following P_(H) |
|
Answer» 1 |
|
| 5. |
Reaction of Alcohol with carboxylic acid in presence of acid catalyst is known as …….. reaction. |
|
Answer» Fermentation |
|
| 6. |
Reaction of acetone with one of the following reagents involves nucleophilic additionfollowed by elimination of water . The reagent is |
|
Answer» grignard reagent |
|
| 7. |
Reaction of acetamide with bromine water and KOH gives: |
|
Answer» `CH_(3)COOH` |
|
| 8. |
The reaction of acetamide with bromine and caustic potash or NaOBr gives : |
|
Answer» ETHANE |
|
| 9. |
Reaction of acetaldehyde with HCN followed by hydrolysis gives a compound which shows |
|
Answer» Optical isomerism |
|
| 10. |
Reaction of acetaldehyde withNH_(2)OH will be fastest at which of the following pH? |
|
Answer» 1 |
|
| 11. |
Reaction of (AAK_MCP_37_NEET_CHE_E37_012_Q01)with HI (one mole) leads to the formation of which one of the following as major product? |
|
Answer»
|
|
| 12. |
Reaction of a compound with conc. KOH gives methanol and potassium formate.Name the compound. |
| Answer» SOLUTION :METHANAL | |
| 13. |
Reaction of a carbonyl compound with one of the following reagents involves nucleophilicaddition followed by elimination of water. The reagent is |
|
Answer» hydrazine in presence of feebly ACIDIC solution |
|
| 14. |
Reaction I^(st) and II^(nd) are |
|
Answer» Both `S_(N)` 1 |
|
| 15. |
Reaction involving carbanion formation is/are: |
|
Answer» CANNIZZARO's reaction |
|
| 16. |
Reaction invoiving anti addition is |
|
Answer» `CH_(2) = CH_(2) overset(H//H_(2)O)to` |
|
| 17. |
Reaction intermediate in hydration of alkene is |
|
Answer» carbanion |
|
| 18. |
Reactioninvolving antiaddition is |
|
Answer» `CH_(2)=CH_(2) overset(H^(+)//H_(2)O)(to)` |
|
| 19. |
Reaction involved during conversion of 3^(@) alkyl amine to 3^(@) nitro alkane |
|
Answer» HYDROLYSIS |
|
| 20. |
Reaction in which yield of prodict will increase with increase in pressure is |
|
Answer» `H_(2(G))+I_(2(g))hArrHI_((g))` Volume is decreasing in forward direction so on increasing pressure the YIELD of product will increase. |
|
| 21. |
Reaction intermediate formed in the formation of salicylaldehyde from phenol (Reimer-Tiemann reaction) is |
|
Answer» Carbocation |
|
| 22. |
Reaction I : CH_(3)CH_(2)-underset(+N(CH_(3))_(3)OH^(-))(CH-CH_(3))overset("heat")to Product Reaction II : CH_(3)CH_(2)-underset(Br)underset(|)CH-CH_(3)overset(CH_(3)ONa)to Product Products are CH_(3)-CH=CH-CH_(3)(X) CH_(3)CH_(2)CH=CH_(2)(Y) |
|
Answer» The major product in reaction 1 is (X) |
|
| 23. |
Reaction,H_(2)(g)+I_(2)(g)rarr2HI(g)DeltaH=-12.40 kcal.According to this, the heat of formation of HI will be |
|
Answer» 12.4 KCAL Hence `DeltaH_(F)^(@)(HI)=-12.40//2=-6.20 kcal`. |
|
| 24. |
Reaction given below is an example of (AAK_MCP_37_NEET_CHE_E37_036_Q01) |
|
Answer» Wolff-Kishner reduction |
|
| 25. |
Reaction given in passage is spontaneous because |
|
Answer» NaOH formed is IONIC |
|
| 26. |
Reaction : CH_(3)OH+O_(2) underset(Ag)overset(600^(@))to productThe product is |
|
Answer» `CH_(2)=C=O` |
|
| 27. |
Reaction CH_(3)COOH+C_(2)H_(5)OH to CH_(3)COOC_(2)H+H_(2)O is an illustration of |
|
Answer» dehydration `CH_(3) COOH + C_(2) H_(5)OH UNDERSET(Delta) OVERSET(CONC. H_(2)SO_(4)) HARR CH_(3)COOC_(2)H_(5)+H_(2)O`. Thus in this reaction , an ester `(CH_(3)COOHC_(2)H_(5))` is formed . This process is called esterfication . |
|
| 28. |
Reaction : Cu_(("solid"))+2Ag_(("aqueous"))^(+) to Cu_(("aqueous"))^(2+)+2Ag_(("Solid")), what is the equilibrium constant for this reaction ? (E^(@)=0.46V" at "298K Temperature). |
|
Answer» `2.0xx10^(10)` `therefore 0.46=(0.0591)/(2)logK_(c)` `therefore logK_(c)=(2xx0.46)/(0.0591)=15.57` or `K_(c)="Antilog "15.57` `=3.7xx10^(15)~~4xx10^(15)`. |
|
| 29. |
Reaction favoured by low pressure is : |
|
Answer» `H_2 + I_2 ⇌ 2HI` |
|
| 30. |
Reaction, CH_3CNO_2 overset rarrgives : |
|
Answer» `CH_3Br` |
|
| 31. |
Reaction by which benzaldehyde cannot be prepared , is |
|
Answer» GATTERMAN - KOCH synthesis  group group
|
|
| 32. |
Reaction by which benzaldehyde cannot be prepared is |
|
Answer»
|
|
| 33. |
Reaction by which benzaldehyde cannot be prepared |
|
Answer»
|
|
| 34. |
Reaction by which benzaldehyde cannot be prepared : |
|
Answer»
|
|
| 35. |
Reaction between solution of phenol in aq.sodium hydroxide and halo alkane will give ether, what is the name of this process of ether preparation ? |
|
Answer» WILLIAMSON's SYNTHESIS |
|
| 36. |
Reaction between NO and O_(2)" to form "NO_(2)" is "2NO + O_(2) rarr2NO_(2) follows the following mechanism No + No k-1 N_(2) O_(2) (in rapid equilibrium) N_(2)O_(2) + O_(2) overset(k_(2))rarr 2NO_(2) (slow) Show that the rate of reaction is given by (1)/(2) ((d[NO_(2)])/(dt))=K[NO]^(2)[O_(2)] |
| Answer» SOLUTION :To PROVE | |
| 37. |
Reaction between sodium ethoxide and bromoethane forms |
|
Answer» ETHYL METHYL ether |
|
| 38. |
Reaction between Grignard's reagent and cyanogen chloride gives : |
|
Answer» ALKANE nitrile |
|
| 39. |
Reaction between ethyl bromide and aqueous KOH follows which order kinetics |
|
Answer» ZERO ORDER `C_2H_5Br + KOHaq to C_2H_5OH + KBR` proceeds by `SN^(2)` mechanism, i.e., it is a second order reaction. |
|
| 40. |
Reaction between barium chloride and sodium sulphate goes to completion bacause |
|
Answer» barium SULPHATE is ALMOST INSOLUBLE |
|
| 41. |
Calculate the heat of hydrogenation , C_(2)H_(4)(g)+H_(2)(g)toC_(2)H_(6)(g) Given that, the heats of combustion of ethylene, hydrogen and ethane are -337.0, -68.4 and -373.0 kcal respectively. |
|
Answer» `-32.4` KCAL |
|
| 42. |
Reaction : A to B follows zero order kinetics and initial concentration of A is 0.01M. If concentration of A is 0.008 M after 10 min, calculate half-life (in minute). |
|
Answer» |
|
| 43. |
Reaction between an acid and alcohol will give |
|
Answer» HIGHER C CONTAINING acid |
|
| 44. |
Reaction A to B carried out inside a container.Concentration varies as C_t=a+bt.Rate of reaction can be expressed in term of pressure at temp T=300 K, Take b=4, R=1/12 atm mol^(-1) k^(-1) Give your Answer divide by 10. |
|
Answer» `C_1=a+bt` p=(a+bt)RT `(DP)/(dt)=dRT " " =4xx1/12xx300=100 "atm" SEC ^(-1)` |
|
| 45. |
Reaction A+BrarrC+D 38 kcal has activation energy 20 kcal. Activation energy for reaction, C+DrarrA+B is |
|
Answer» 20 kcal `(E_a)_f`=20 kcal as `DeltaH=(E_a)_f-(E_a)_b` `-38=20-(E_a)_b` `THEREFORE (E_a)_b`=20+38=58 kcal |
|
| 46. |
Reaction A+B rarr C +D+38kcal has activation energy 20 kcal . Activation energy for reaction C+D rarr A+B is : |
|
Answer» 20 kcal ` =DeltaE +E_(a)` (FORWARD ) = 38 +20 = 58 kcal |
|
| 48. |
Reaction, |
|
Answer» ELECTROPHILIC substitution |
|
| 49. |
Reaction 3ClO^(-)toClO_(3)^(-)+2Cl^(-) occurs in following two steps. (i)ClO^(-)+ClOoverset(K_(1))toClO_(2)^(-)+Cl^(-) (Slow step) (ii)ClO_(2)^(-)+ClO^(-)overset(K_(2))toClO_(3)^(-)+Cl^(-)(Fast step) Then the rate of given reaction =........ |
|
Answer» `K_(1)[CL^(-)]^(2)` |
|
| 50. |
Reaction : - 3ClO^(-)toClO_3^(-) +2Cl^(-)occursinfollowingtwosteps (i)2ClO^(-)rightarrow ClO_(2)^(-) +Cl^(-)( slow step ) (ii)ClO_(2) ^(-)+ClO^(-)overset(K_(2)) toClO_(3)^(-)+Cl^(-)( fast step) thentherateofgivenreaction- ______. |
|
Answer» `K_(1)[CLO^(-) ]^(2)` |
|




 `+ Zn//Hg` and
`+ Zn//Hg` and  `+CrO_(2)Cl_(2)` in `CS_(2)` and `H_(3)O^(+)`
`+CrO_(2)Cl_(2)` in `CS_(2)` and `H_(3)O^(+)` `+ H_(2)` and `Pd-BaSO_(4)`
`+ H_(2)` and `Pd-BaSO_(4)`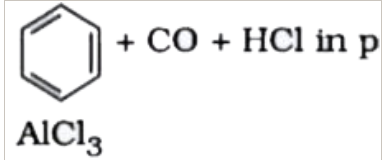 in
in 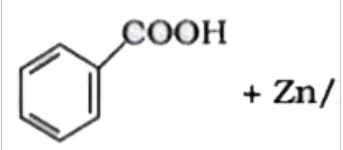 + Zn/Hg and conc.
+ Zn/Hg and conc. 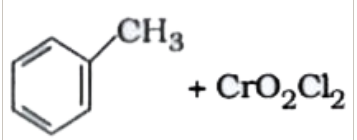 + `
+ `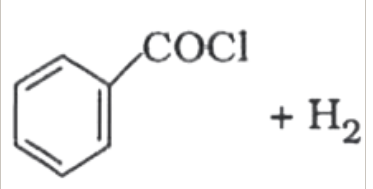 + `H_(2)` in presence of `Pd* BaSO_(4)`.
+ `H_(2)` in presence of `Pd* BaSO_(4)`.