Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Reaction :2NO_(2)Fto2NO_(2)+F_(2) Rate : K[NO_(2)F] .What is step of deciding rate? |
|
Answer» `NO_(2)+F_(2)toNO_(2)F+F` |
|
| 2. |
Reaction : 2Fe^(3+)+Zn to Zn^(2+)+2Fe^(2+), is carried out in electrochemical cell, then on increasing concentration of Fe^(2+) what will be observed ? |
|
Answer» CELL POTENTIAL increases From above equation, on increasing `Fe^(2+)` concentration, `E_(cell)` VALUE decreases by subtracting from `E_(cell)^(@)` value. |
|
| 3. |
Reactio of trans 2-phynyl-1- bromocyclopentane on reaction with alcoholic KOH produces |
|
Answer» 4-phenylchlopentene 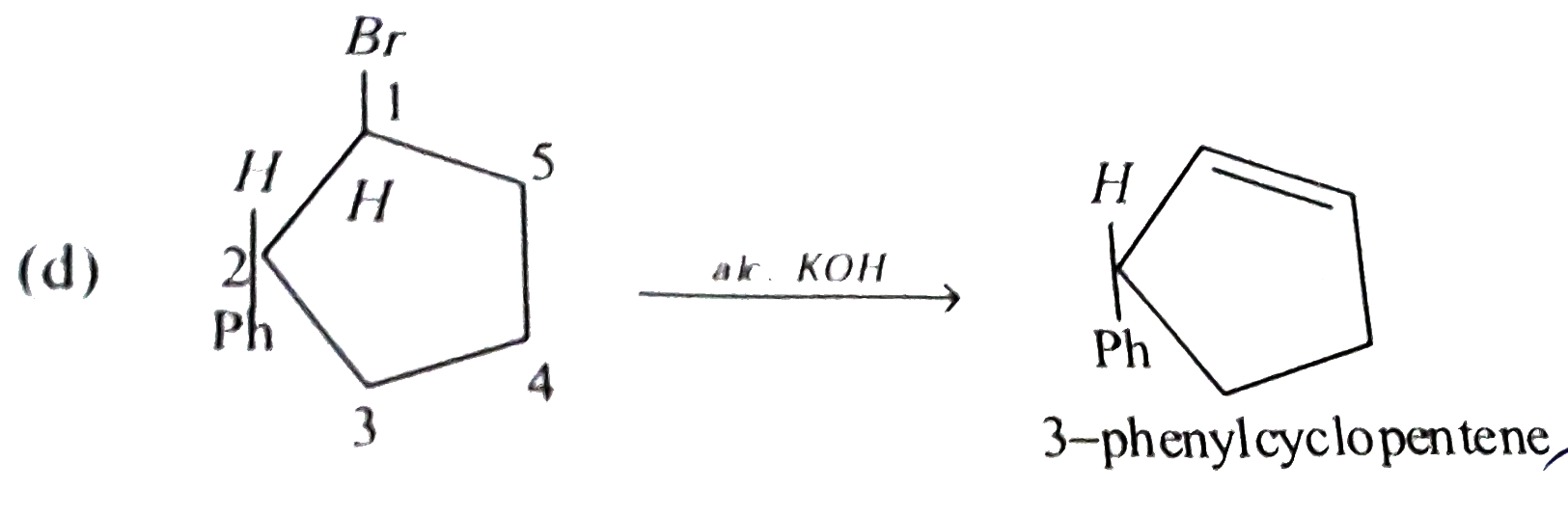 It's antielimination reaction so hydrogen atom from SECOND CARBON can not ELIMINATE as it is in syn-position rather hydrogen atom from `5^(th)` carbon will eliminate. |
|
| 4. |
Reaction 1 : trans-2-butene overset(HCO_3H)underset(H_2O)(to) Reaction 2 : cis-2-pentene overset(KMnO_4 //OH^(-))(to) Which of the following statements are incorrect about the above reactions ? |
|
Answer» REACTION 1 leads to racemic MIXTURE 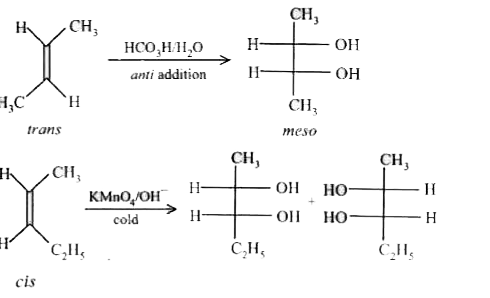
|
|
| 5. |
Reactants of reaction -I are : CH_(3)CONH_(2),KOH,Br_(2) Reactants of reaction-II are : CH_(3)NH_(2), CHCl_(3), KOH The intermediate species of reaction-I and reaction-II are respectively. |
|
Answer» Carbonium ION, carbene |
|
| 6. |
Reactants rarr products DeltaG=+63KJ" mole"^(-1). This reaction is made to take place by coupling with |
|
Answer» `ATPoverset(H_(2)O)rarrADP` |
|
| 7. |
Reactant. A which form B on ozonolys is (AAK_MCP_37_NEET_CHE_E37_030_Q01) |
|
Answer» `(AAK_MCP_37_NEET_CHE_E37_030_A01)` |
|
| 8. |
Reactant 'A' is |
|
Answer»
`n_(H^(+))=1.5xx2` `n_(OH^(-))- =4` `[OH^(-)]=(4-3)/(5)=0.2` `"pOH = - log 0.2=0.7pH = 14 - 0.7 =13.3"` pH=13.3 |
|
| 9. |
RDX is otherwise named as .............. |
|
Answer» Cyclonite |
|
| 10. |
RDX is also called ................ or ........... |
| Answer» SOLUTION :Cyclonite(or)cyclotrimethylene trinitramine | |
| 11. |
RDX has many limitary and civilian applications but these days, it is being misused for terrorist attacks. What does it stand for and how is it prepared? |
Answer» Solution :RDX stands for ROYAL Demolition EXPLOSIVE or reaserch development explosive. It is also called cyclonite or cycloterimethylenetrinitramine. It is prepared by NITRATION of hexamethylenetetramine.  . .
|
|
| 12. |
RCOOR+H_(2)Ooverset(HCl)(to)RCOOH+ROH follows……….reaction kinetics |
|
Answer» `2^(" ND")` order |
|
| 13. |
RCOOHrarrRCH_(2)OH. This mode of reduction of an acid to alcohol can be affected by |
|
Answer» Zn/HCl |
|
| 14. |
RCOOH to RCH_(2)COOH. This conversion is known as reaction: |
|
Answer» Arndt-Eistert reaction |
|
| 15. |
RCOOH after treatment with PCl_(5) and KCN is subjected to hydrolysis followed by Clemmension's reduction, product obtained as : |
|
Answer» `RCH_(2)-COCL` |
|
| 16. |
RCONH_(2) is converted into RNH_(2) by means of Hofmann bromamide dehydration In this reaction, RCONHBr is formed from which this reaction has derived its name. Electron donating group at phenyl activities the reaction. Hofmann degradation reaction is an intramolecular reaction. What are the constituent amines formed when the mixture of (i) and (ii) undergoes Hofmann degradation ? |
|
Answer»
 SINCE the over all reaction is intermolecular there will be no EFFECT on PRODUCT formation. |
|
| 17. |
RCOOH + N_3H overset(H_2SO_4) underset(conc.)rarr RNH_2 + CO_2 + N_2 The above reaction is called : |
|
Answer» HVZ REACTION |
|
| 18. |
RCONH_(2) is converted into RNH_(2) by means of Hofmann bromamide dehydration In this reaction, RCONHBr is formed from which this reaction has derived its name. Electron donating group at phenyl activities the reaction. Hofmann degradation reaction is an intramolecular reaction. Which is the rate determining step in Hofmann bromamide degradation ? |
|
Answer» Formation of (i)<BR>Formation of (ii) |
|
| 19. |
RCONH_(2) is converted into RNH_(2) by means of Hofmann bromamide dehydration In this reaction, RCONHBr is formed from which this reaction has derived its name. Electron donating group at phenyl activities the reaction. Hofmann degradation reaction is an intramolecular reaction. How can the conversion of (i) to (ii) be brought about ? |
|
Answer» `KBR` 
|
|
| 20. |
RCOCl+2Me_(2)NHrarrA+Me_(2)NH_(2)Cl^(-) . Here A is |
|
Answer»

|
|
| 21. |
RCl + NaIoverset(CH_3COCH_3)rarr R-I + NaCl this reaction is |
|
Answer» WURTZ REACTION |
|
| 22. |
RCI is hydrolysed to ROH slowly but the reaction is rapid if catalytic amount of KI is added to reaction mixture. Explain. |
|
Answer» Solution :`I^(-)` is a powerful NUCLEOPHILE which REACTS rapidly with RCI to form RI. `KI to K^(+) +I^(-)` `RCI + I^(-) to RI + Cl^(-)` Iodide is a better leaving group than CHLORIDE and therefore, RI is hydrolysed rapidly to form ROH. `RI + OH^(-) to ROH + I^(-)` The `I^(-)` formed in the above reaction recycles and this explains the catalytic effect. |
|
| 23. |
RCH_(2)OHoverset([O])(to)RCHO Which of the following oxidising agent is most suitable for above reaction ? |
| Answer» Answer :A | |
| 25. |
RCH_2C Cl_2R overset(X)to R - C-=C - R The reagent 'X' is |
|
Answer» Na |
|
| 26. |
RCH_2CH_2OH can be converted to RCH_2CH_2COOH by the following sequence of steps: |
|
Answer» `PBr_3,KCN,H_3O+` |
|
| 27. |
RCH = O + 2H rarr RCH_2OH : For this reaction catalyst is : |
|
Answer» NI only |
|
| 28. |
Strutures of products , Starting materials or reagents RCH = CHCHO +H_(2)NCONHNH_(2)overset(H^(+))rarr |
| Answer» Solution :`underset("2-alkenal SEMICARBAZONE")(R-CH=CH-CH=N-NH-OVERSET(O)overset(|)(C)-NH_(2))` | |
| 29. |
RC-=N can be converted to RCH_(2)NH_(2) by _________. |
|
Answer» REDUCTION |
|
| 30. |
RbO_(2) is |
|
Answer» Peroxide and PARAMAGNETIC |
|
| 31. |
Rbl crystallizes in bcc structure in which each Rb^(+) is surrounded by eight iodide ions each of radius 2.17 Å. Find the length of one side of RbI unit cell. |
|
Answer» SOLUTION :`(r_(rb^(+)))/(r_(R))`=0.732"" ? "" `r_(Rb^(+))=0.732xx2.17=1.59 Å` `asqrt(3)=2[r_(Rb^(+))+r_(I^(-))]a=(2)/(SQRT(2))[1.59+2.17]` 4.34 Å |
|
| 32. |
Rayon yarns are obtained from |
|
Answer» Polymethylene |
|
| 33. |
Rayon is |
|
Answer» natural SILK |
|
| 34. |
Rayon is a |
|
Answer» NATURAL polymer |
|
| 35. |
Raw material used for preparation nylon 6 is |
|
Answer» `EPSILON`-caprolactum |
|
| 36. |
Raw linseed oil is present in a paint as: |
|
Answer» Drier |
|
| 37. |
Ratio of velocity of electron in 5^(th) excited state and 3^(rd) energy level for He^(+) atom is |
|
Answer» `(1)/(4)` |
|
| 38. |
Ratio of the rate of effusion of oxygen gas at 1.5 atm to that of helium gas at 4.5 atm will be |
|
Answer» `1 : 6sqrt2` |
|
| 39. |
Ratio of the everagespeed of He and SO_2 at 27^o C will be |
| Answer» Answer :A | |
| 40. |
Ratio of sigma & pi bond in P_(4)O_(10) is :- |
|
Answer» 4 |
|
| 41. |
Ratio of masses of Na_(3)PO_(4) and Ca_(3)(PO_(4))_(2) each containing 31gm of P is ________ |
|
Answer» <P>0.8 `n_(p)=n_(Ca_(3))(PO_(4))_(2)xx2 (W_(Na_(3)PO_(4)))/(164)xx2xx(W_(Ca_(3))(PO_(4))_(2))/(310)` `(W_(Na_(3)PO_(4)))/(W_(Ca_(3))(PO_(4))_(2))=(2xx164)/(310)=(328)/(310)=1.058` |
|
| 42. |
Ratio of DeltaT_(b)//K_(b both are non-electrolytes) is 1 molal in both cases. Hence atomic masses of A and B are respectively: |
|
Answer» `60,90` |
|
| 43. |
Ratio of loss in solvent to gain in CaCl_(2) tube is |
|
Answer» <P>`(p^(o))/(p^(s))` |
|
| 44. |
Ratio of everage kinetic energy of 14 g of N_2 at 27^oC to 24 g of O_2 at 227^oC is |
| Answer» ANSWER :D | |
| 45. |
Ratio of C_(p) and C_(v) of a gas 'X' is 1.4. The number ofatoms of the gas 'X' present in 11.2litres of it at N.T.P. is |
|
Answer» `6.02 xx 10^(23)` If VOLUME is 11.2It then , no. of moles `=(1)/(2)` ` :. ` no. of MOLECULES `=(1)/(2) xx` Avogadro's No. no. of atoms `= 2 xx `no. of molecules `2 xx (1)/(2) xx `AVAGADRO's No. `= 6.023 x 10^(23)` |
|
| 46. |
Ratio at DeltaT_(b)//K_(b) of 6% AB_(2) and 9%A_(2)B(AB_(2) and A_(2)B both are non-electolytes) is 1 "mol"//kg in both cases. Hence, atomic masses of A and B are respectivly : |
|
Answer» `60,90` `(DeltaT)/(K_(b))(AB_(2)=(1000xx6)/(m_(1)XX100)=1` `therefore m_(1)(AB_(2))=60=A+2B` `(DeltaT)/(K_(b)_(A_(2)B)=(1000xx9)/(m_(1)xx100)=1` `therefore m_(1)(A_(2)B)=90=2A+B` `therefore A=40,B=10` |
|
| 47. |
Ratio between potential energy, kinetic energy and total energy of electron in hydrogen atom are |
|
Answer» `(KE)/( PE ) = - ( 1)/( 2)` |
|
| 48. |
Rates of reaction double with every 10^@ rise in temperature. If this generalization holds for a reaction in the temperature ranges 298 K to308 K, what would be the value of activation energy for their reaction ?R = 8.314 J K^(-1) "mol"^(-1). |
|
Answer» Solution :`logK_2/K_1=E_a/(2.303R)[1/T_1-1/T_2]` Here , `T_1`=298 K , `T_2`=308 K, `R=K^(-1) "mol"^(-1)` `K_2/K_1=2` `log2=E_a/(2.303xx8.314)[1/298-1/308]` `0.3010=E_a/(2.303xx8.314)[10/(298xx308)]` `E_a=(0.3010xx2.303xx8.314xx298xx308)/10` `=52898 "J mol"^(-1)` `=52.898 "KJ mol"^(-1)` |
|
| 49. |
Rate of the given reaction (i)A + B overset (r_(1) = 0.05)(to) X(ii)X + B overset(r_(2) = 0.89)(to) Y (iii) Y+ A overset(r_(2) = 0.001)(to) (AY) (iv) AY + B overset(r_(4) = 0 .10)(to) AYB will be determined by |
|
Answer» Step(i) Because the reaction starts with the formation of X |
|