Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Rate of subsitution reaction in phenol is |
|
Answer» <P>Slower than the rateof benzene 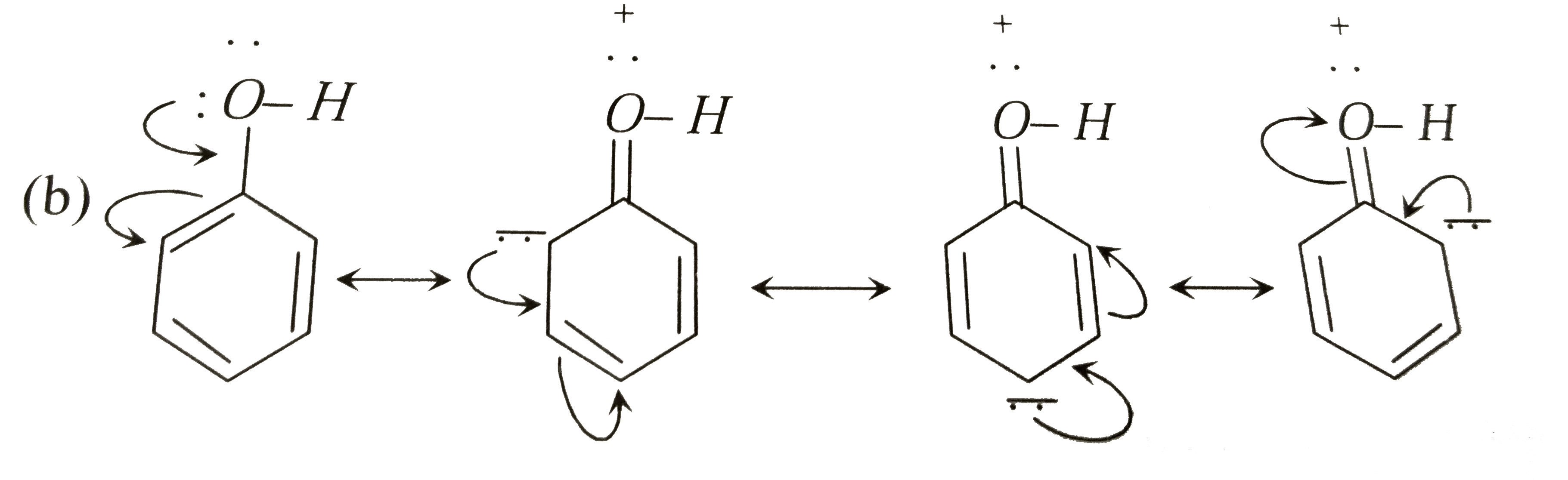 Rate of electrophillic substitution reaction in phenol is faster than in benzene presence of `-OH` group increases electron density at o- and p- positions. |
|
| 2. |
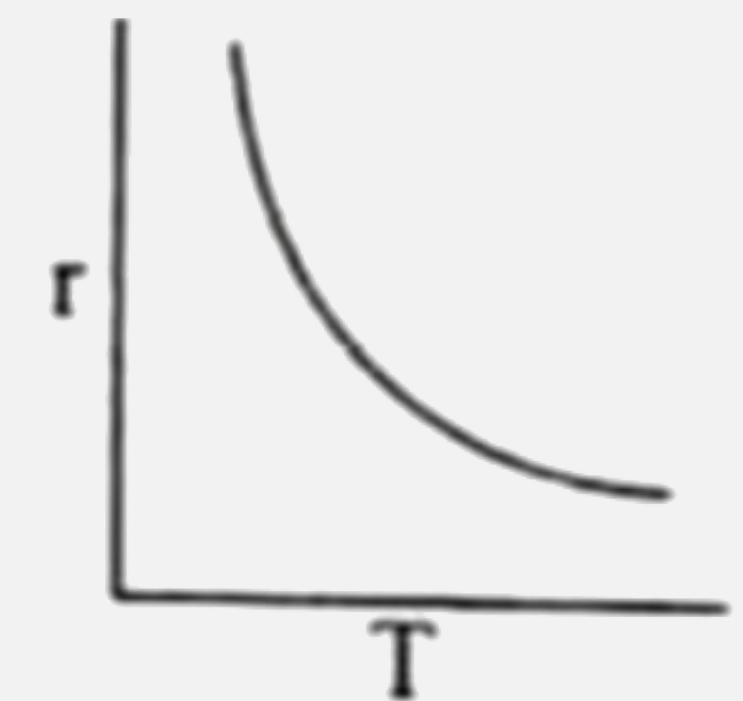
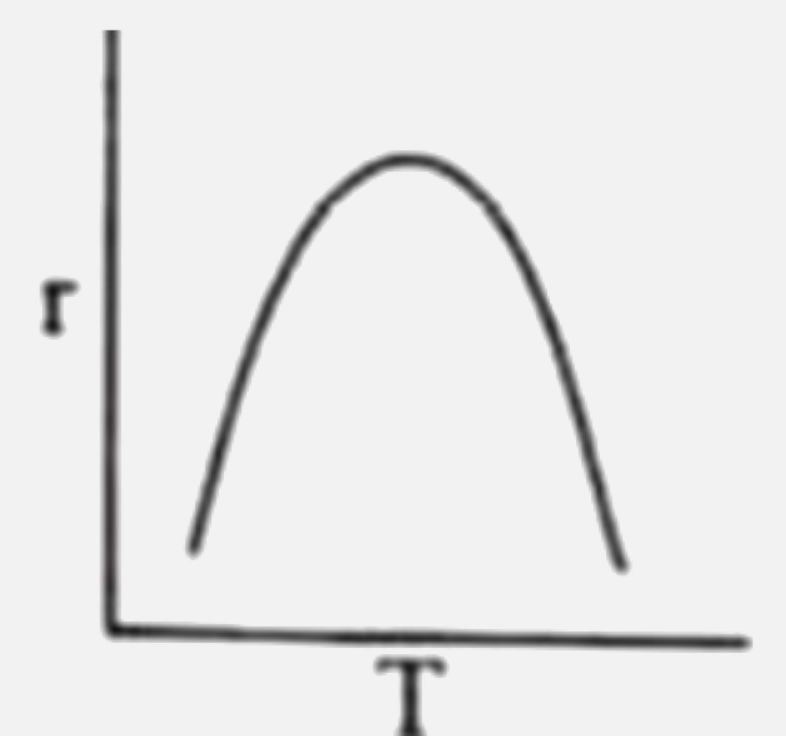
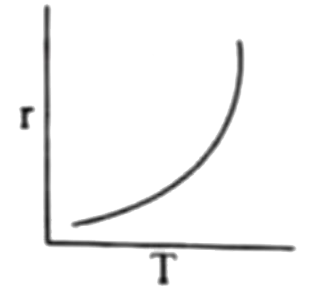
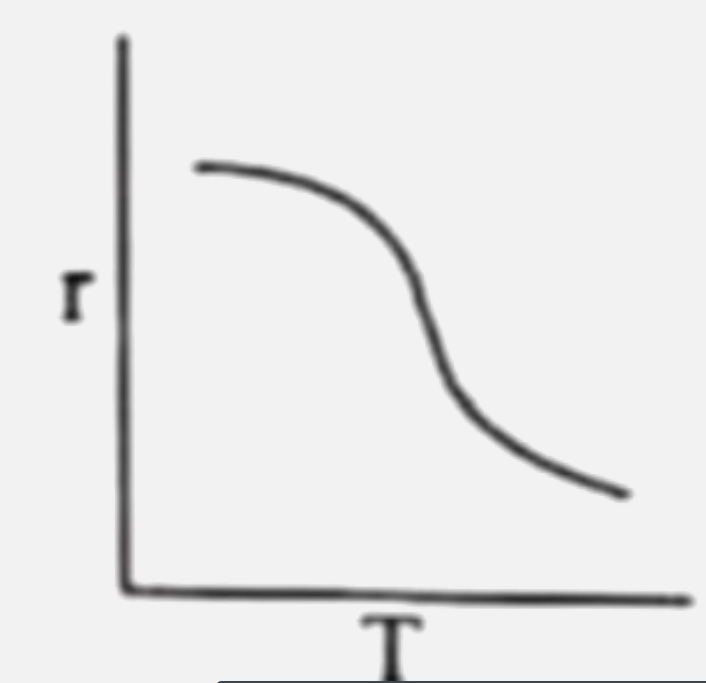
Rate of reaction (r ) is plotted against temperature (T) for an enzyme catalysed reaction. Which of the following is correct representation ? |
|
Answer»
|
|
| 4. |
What is Lucas reagent? |
| Answer» Solution :RATE of REACTION of 3^@ ALCOHOL is fastest with LUCAS reagent. | |
| 5. |
Rate of reaction ofalcohols with Lucas reagent: |
|
Answer» p > s > t |
|
| 6. |
Rate of reaction for reaction .B. is double at constant concentration of .A. then what will change in rate constant value? |
|
Answer» REMAIN CONSTANT |
|
| 7. |
Rate of reaction is influenced by_____. |
| Answer» SOLUTION :CONCENTRATION of REACTANTS and TEMPERATURE | |
| 8. |
Rate of reaction decreases as temperature increases.is it true or false |
| Answer» SOLUTION :INCREASES | |
| 9. |
Rate of reaction can be expressed by Arrhenius equation as k = Ae^(–E//RT) , In this equation, E represents |
|
Answer» the energy below which colliding molecules will not REACT |
|
| 10. |
Rate of reaction can be expressed by Arrhenius equation as k= Ae^(-E//RT) . In this reaction E represents |
|
Answer» The ENERGY below which colliding molecules will not react |
|
| 11. |
Rate of radiocative decay for a radioactive element depends on : |
|
Answer» amount of RADIOACTIVE ELEMENT |
|
| 12. |
Rate of reaction….. |
|
Answer» DECREASES with INCREASE in temperature. |
|
| 13. |
Rate of physisorption increases with: |
|
Answer» DECREASE in TEMPERATURE |
|
| 14. |
Rate of physisorption increases with |
|
Answer» Incresaes in T |
|
| 15. |
Rate of physisorption increases with : |
|
Answer» DECREASE in TEMPERATURE |
|
| 16. |
Rate of physisorption always increases with decrease in temperature. Explain. |
| Answer» | |
| 17. |
Rate of physical adsorption increase with .................. |
|
Answer» INCREASE in TEMPERATURE |
|
| 18. |
Rate of most of reactions increases when temperature of reaction mixture is increased. Why? In what units is the rate of reaction expressed? |
|
Answer» Solution :Because KINETIC energy of molecules increases therefore number of moleculus POSSESSING ACTIVATION energy increases, which increases number of collisions,HENCE rate of REACTION increases. `mole lit^-1S^1` |
|
| 19. |
Rate of which reactions increases with temperature: |
|
Answer» Solution :Because kinetic ENERGY of molecules INCREASES therefore number of moleculus possessing ACTIVATION energy increases, which increases number of collisions,hence rate of reaction increases. `MOLE lit^-1S^1` |
|
| 20. |
Rate of osmosis increases with increase in temparature. |
| Answer» | |
| 21. |
Rate of hydration of with be in order, |
|
Answer» `I LT II lt III` |
|
| 22. |
Rateof hydration of : |
| Answer» Solution :N//A | |
| 23. |
Apparent expansion of a liquid depends upon a) nature of liquid b) nature of vessel c) temperature rise d) scale of temperature |
|
Answer» a, B only |
|
| 24. |
Rateof disappearance of the reactant A at two different temperature is given by A rarr B (-d[A])/(dt)=2 xx 10^(-2) "sec"^(-1)[A]-4xx10^(-3)"sec"^(-1)[B] at 300 K (-d[A])/(dt)=4 xx 10^(-2) "sec"^(-1)[A]-16xx10^(-4)"sec"^(-1)[B] at 400 Kheat of reaction in the given temperature range, when equilibrium is set up is : |
|
Answer» `(2.303 XX 2 xx 300 xx 400)/(100) "log"50` CAL |
|
| 25. |
Rate of electrophilic substitution reaction in phenol is |
|
Answer» EQUAL to that to benzene |
|
| 26. |
Rate of effusion of a gas is : |
|
Answer» 1. directly proportional to its DENSITY |
|
| 27. |
Rate of diffusion of two gases are r_(A)and r_(B), molecular weights are M_(A) andM_(B), then partial pressure P_(A) is ( if number of moles are n_(A) and n_(B). |
|
Answer» `( r_(A))/( r_(B)) xx sqrt((M_(A))/( M_(B)) ) xx( n_(B))/( n_(A) + n_(B)) xx P_("TOTAL")` |
|
| 28. |
Rate of diffusion of NH_(3) is twice that of X. What is the molecular mass of X |
|
Answer» 68 Molecular MASS of `NH_(3),M_(NH_(3))=(N)+3(H)=14+3(1)=17` Molecular mass of X,`M_(X)= ?` By Graham's law of diffusion `:. (r_(NH_(3)))/(r_(X))=sqrt((M_(X))/(M_(NH_(3))))` or ` 2= sqrt((M_(X))/(M_(NH_(3)))) ` `:. 4 xx M_(NH_(3))IMPLIES M_(X)=4 xx 17 = 68` |
|
| 29. |
Rate of chemical reaction nA rarr Product, is doubled when the concentration of A is increased four times. If the half-life time of the reaction at any temperature is 16 minutes, then the time required for 75% of the reaction to complete is |
|
Answer» 24.0 minutes |
|
| 30. |
Rate of chemical reaction is not uniform throughout. Justify you answer. |
| Answer» Solution :RATE of a REACTION at any time depends on the concentration of the reactants which keepts on decreasing with time. | |
| 32. |
"Rate of adsorption is directly proportional to the fraction of area uncovered and rate of desorption is directly proportion to the fraction of area covered". This statement is true for |
|
Answer» FREUNDLICH ADSORPTION ISOTHERM |
|
| 33. |
Rate of a zero order reaction slowly decreases with the progress of a the reaction. |
| Answer» | |
| 34. |
Rate of a reaction is the change in concentration of any one of the reactants or any one of the products in unit time.N_2O_(5(g))to2NO_(2(g))+1/2O_(2(g))is a first order reaction. Find the unit of k |
| Answer» SOLUTION :`s^(-1)or TIME^(-1)` | |
| 35. |
Rate of a reaction is the change in concentration of any one of the reactants or any one of the products in unit time. Calculate the time required for the completion of 90% of a first order reaction(k=0.2303s^(-1)) |
| Answer» SOLUTION :`thereforet=2.303/0.2303xx log100/10`=10 SECONDS | |
| 36. |
Rate of a reaction is the change in concentration of any one of the reactants or any one of the products in unit tim.Express the rate of the following reaction in terms of reactants and products2NO(g)+O_(2(G))to2NO_(2(G)) |
| Answer» SOLUTION :RATE=`(-1)/2(d[NO])/DT=(-d[O_2])/dt=(-1)/2(d[NO_2])/dt` | |
| 37. |
Rate of a reaction is the change in concentration of any one of the reactants or any one of the products in unit time. 2HI to H_(2)+I_(2) Whether the molecularity and the order of the above reaction are the same? Give reason. |
| Answer» SOLUTION :The ORDER and MOLECULARITY of the given REACTION are the same. Order and molecularity are the same because it is an elementary reaction. The reaction involves the collision of two molecules of the reactant HI. | |
| 38. |
Rate of a reaction is the change in concentration of any one of the reactants or any one of the products in unit time. Define order of a reaction. |
| Answer» Solution :The ORDER of a REACTION is DEFINED as the sum of the POWERS of the concentration terms in the rate EQUATION. | |
| 39. |
Rate of a reaction is the change in concentration of any one of the reactants or any one of the products in unit time. If rate expression for the above reaction is rate=k(HI)^2,what is the order of the reaction? |
| Answer» SOLUTION :ORDER of the reaction=2 | |
| 40. |
Rate of a reaction is given by the equation : Rate =k[A]^(2)[B] What are the units for the rate and the rate constant for this reaction ? |
|
Answer» SOLUTION :UNITS of RATE `="mol L"^(-1)s^(-1)` Units of `K=("Rate")/([A][B]^(2))=("mol L"^(-1)s^(-1))/(("mol L"^(-1))("mol L"^(-1))^(2))=L^(2)"mol"^(-2)s^(-1).` |
|
| 41. |
Rate constant of a reaction can be expressed by Arrhenius equation as K=Ae(-E//R). In this equation, E represents |
|
Answer» The energy above which not all the COLLIDING molecules will react |
|
| 42. |
Rate of a reaction is the change in concentration of any one of the reactants or any one of the products in unit time. Express the rate of the following reaction in terms of reactants and products:2HItoH_2+I_2 |
| Answer» SOLUTION :`Rate=-1/2 (d[H])/DT=(d[H_2])/dt=(d[I_2])/dt` | |
| 43. |
Rate of a reaction can be expressed by Arrhenlus equation, k=Ae^(-E_(a)//RT).Here, E is |
|
Answer» the total energy of the reacting MOLECULES at a temperature T |
|
| 44. |
Rate of a reaction can be expressed by Arrhenius equation as k=Ae^(-E_a/(RT)) . In this equation , E_a represents: |
|
Answer» The TOTAL energy of reactingmolecules at a TEMPARATURE T |
|
| 45. |
Rate of a reaction |
|
Answer» DECREASES with INCREASE in temperature. |
|
| 46. |
Rate of a reaction can be expressed by Arrhenius equation as: k = Ae^(-E//RT). In this equation, E represents |
|
Answer» the energy below which colliding molecules will not react |
|
| 47. |
Rate of a reaction, A+Bto Product is given as a fundtion of different initial concentrations of A and B {:("[A]|mol.L"^(-1),"[B]|mol.L"^(-1),"Rate "["mol.L"^(-1)."min"^(-1)]),(0.01,0.01,0.005),(0.02,0.01,0.010),(0.01,0.02,0.005):} The half - life of in the reaction is |
|
Answer» `0.693` MIN |
|
| 48. |
Rate of reaction, Ararrr B increases two times by increasing the concentration 'A' by four times, what is the order of a reaction? |
| Answer» SOLUTION :RATE = `K[A]^x`WhenA = 4, rate = 2k giventherefore2k =`k[4]^x,2^1 = 2^2x,x = 1/2` | |
| 49. |
Rate of a reaction |
|
Answer» increases with INCREASE in temperature As the temperature increase KINETIC energy of molecules increases and thus EFFECTIVE COLLISION increase and thus rate of reaction increases. |
|
| 50. |
Rateof areaction |
|
Answer» INCREASES withincrease intemperature |
|