Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
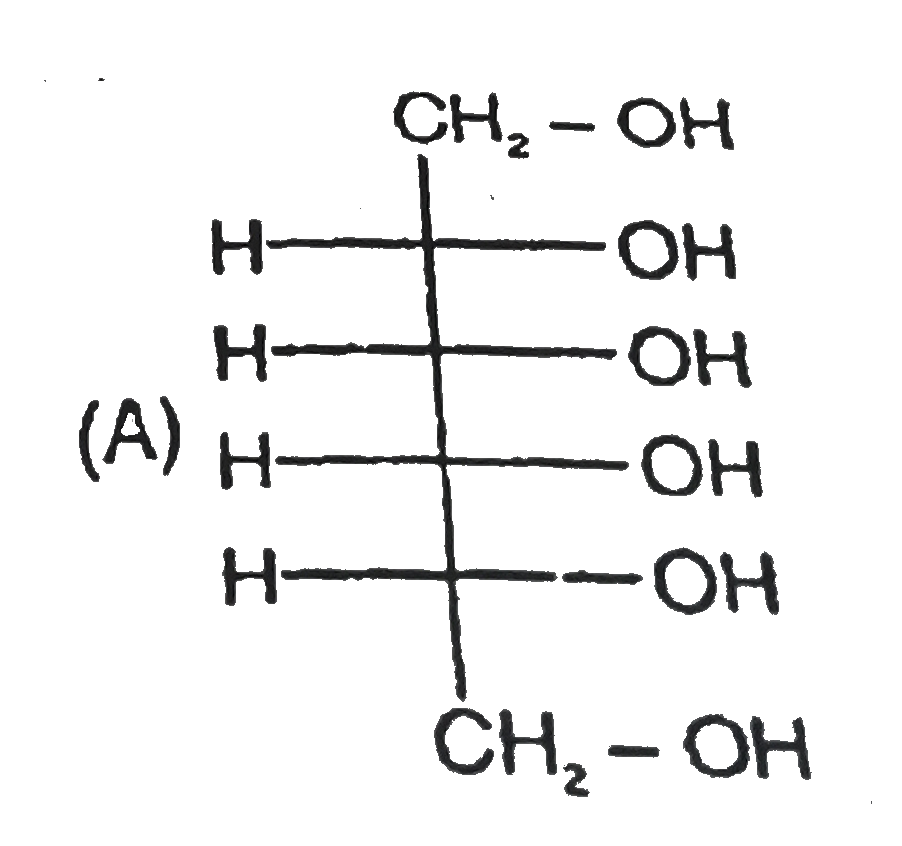
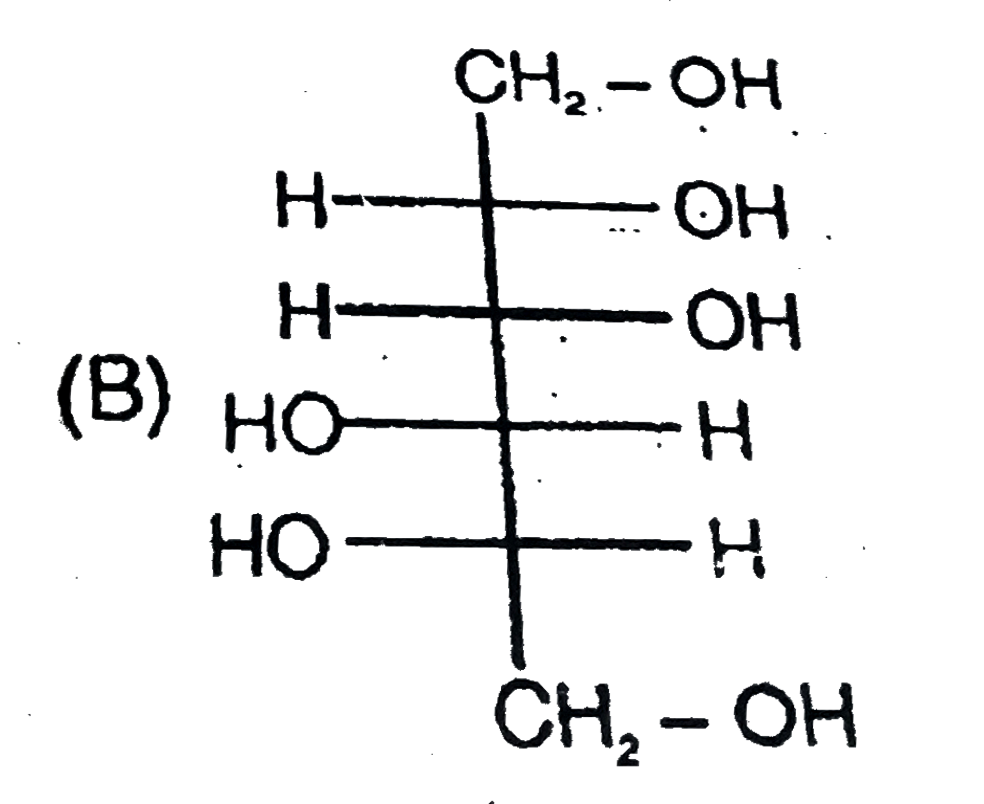
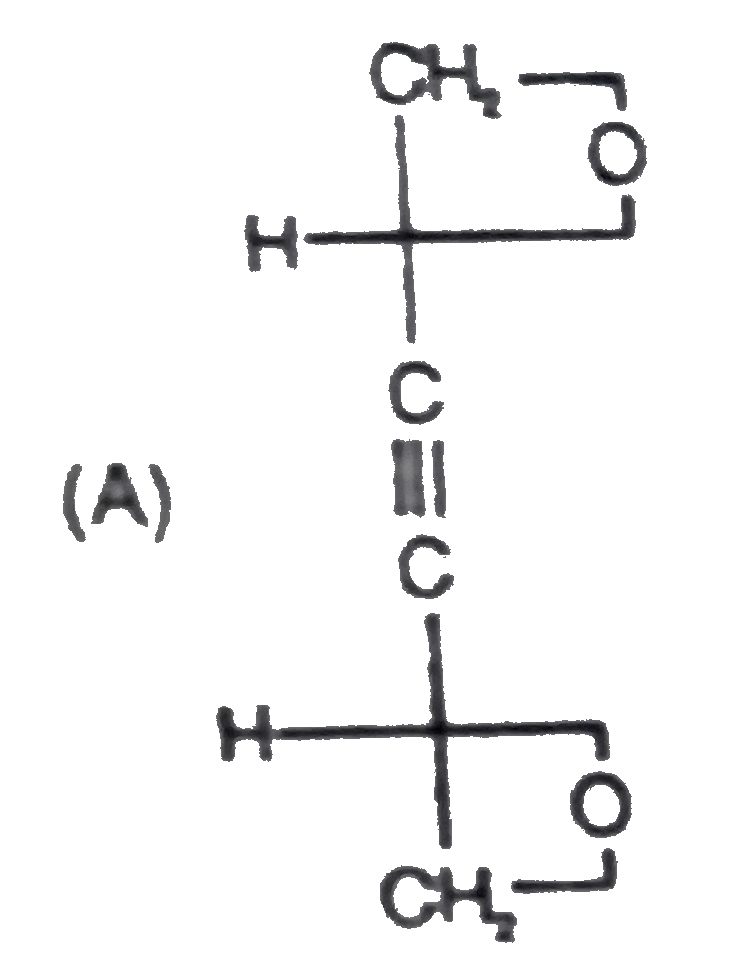
Read the following passage carefully and answer the questions : CH_(2)=CH-underset(H_(1)C)underset(|)(C )=underset(CH_(3))underset(|)(C )-CH=CH_(2) , How many total monochloro structure isomers obtained on chlorination of product (N). |
Answer» SOLUTION :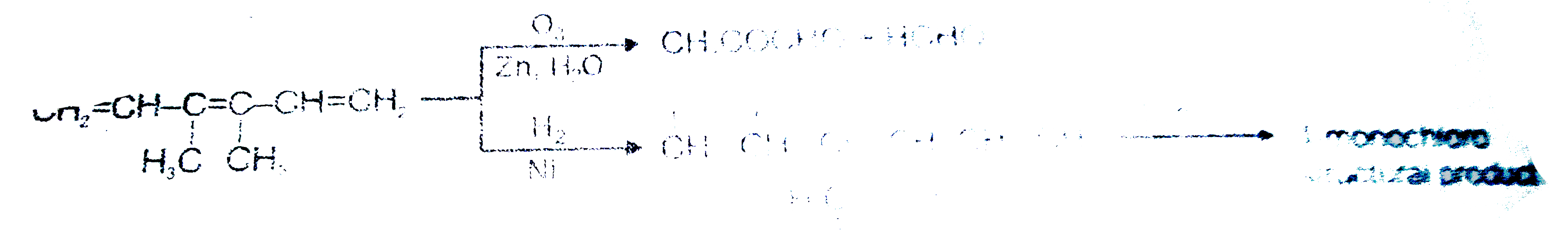
|
|
| 2. |
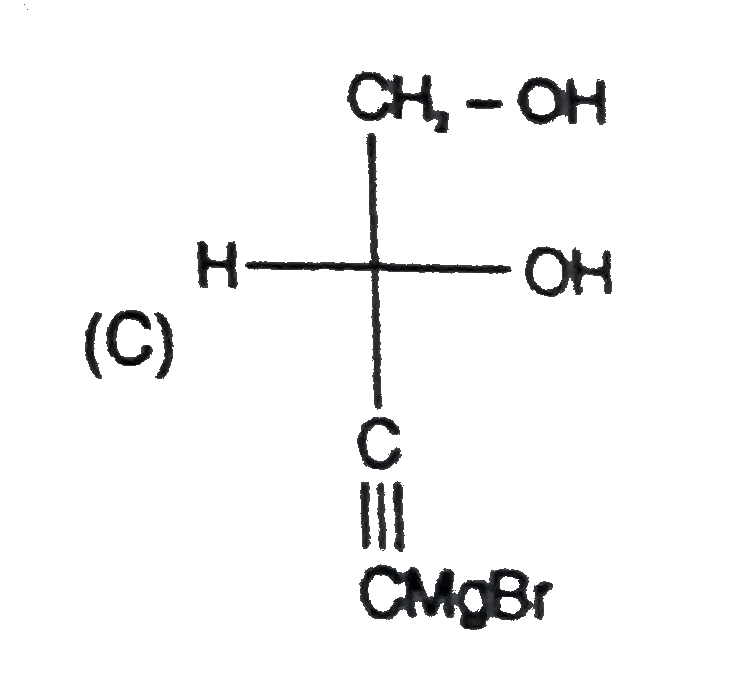
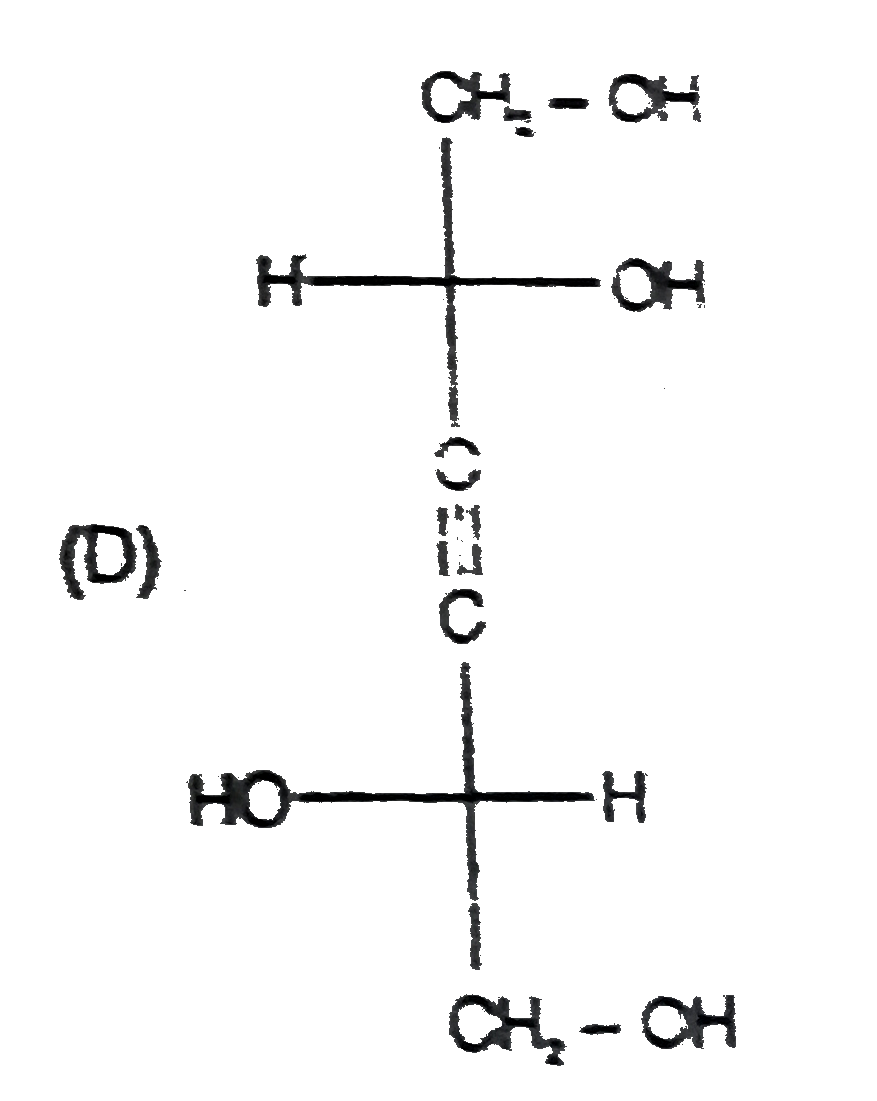
Read the following reaction CH-=CHoverset(NaOCl)toPunderset(Ether)overset(Mg)toQoverset(undersetunderset(Cl)(|)(2CH_2)-CHO)toRoverset(KOH)toSunderset(H_2O)overset(NaOH)to Tunderset((i)DiL KMnO_4)overset((i)H2//Pd.//BaSO_4)toU & U' 'U' is |
|
Answer»
|
|
| 3. |
Read the following reaction CH-=CHoverset(NaOCl)toPunderset(Ether)overset(Mg)toQoverset(undersetunderset(Cl)(|)(2CH_2)-CHO)toRoverset(KOH)toSunderset(H_2O)overset(NaOH)to Tunderset((i)DiL KMnO_4)overset((i)H2//Pd.//BaSO_4)toU & U' 'S' is the above reaction |
|
Answer»
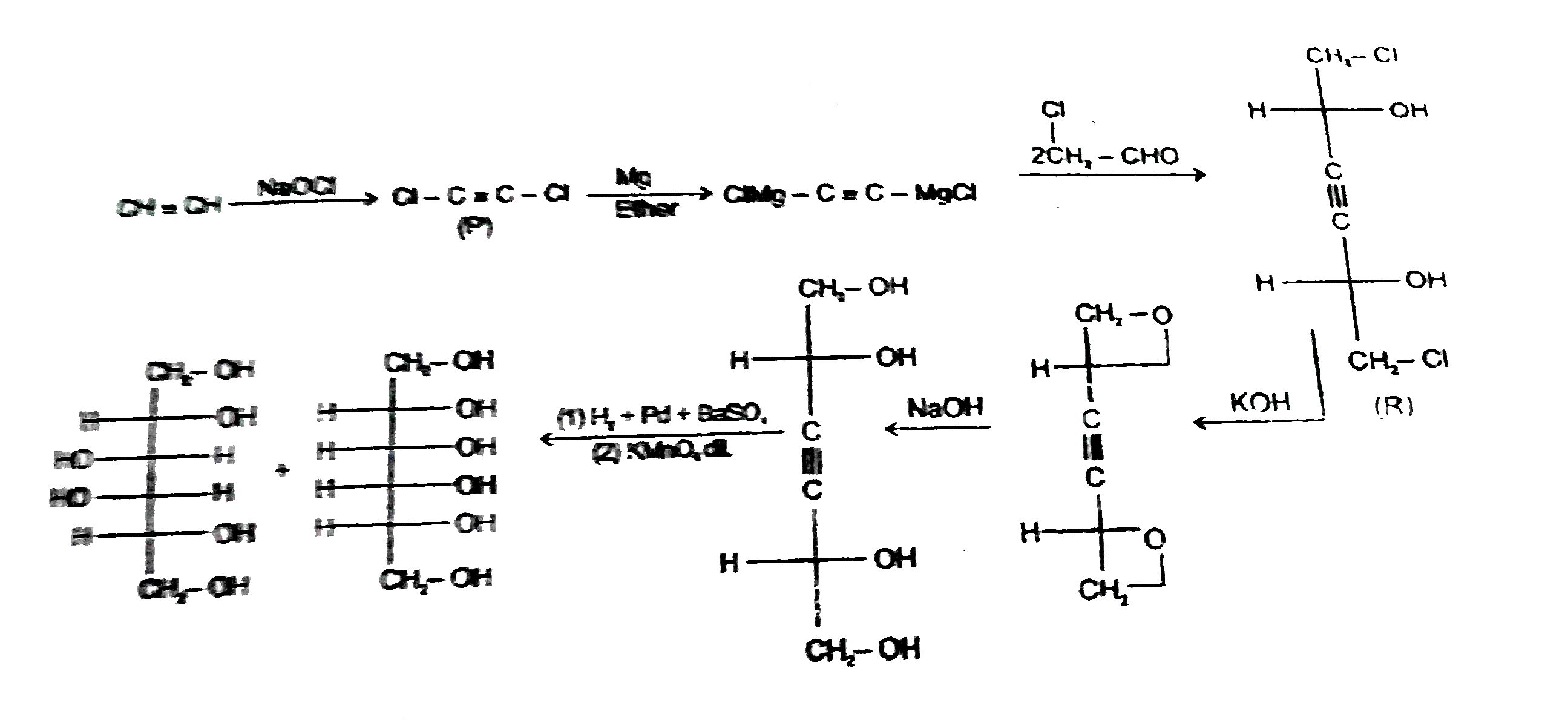
|
|
| 4. |
Read the following passage carefully and answer the questions : CH_(2)=CH-underset(H_(1)C)underset(|)(C )=underset(CH_(3))underset(|)(C )-CH=CH_(2) , Number of moles of ozone used for one mole of the given unsaturated hydrocarbon ? |
Answer» SOLUTION : 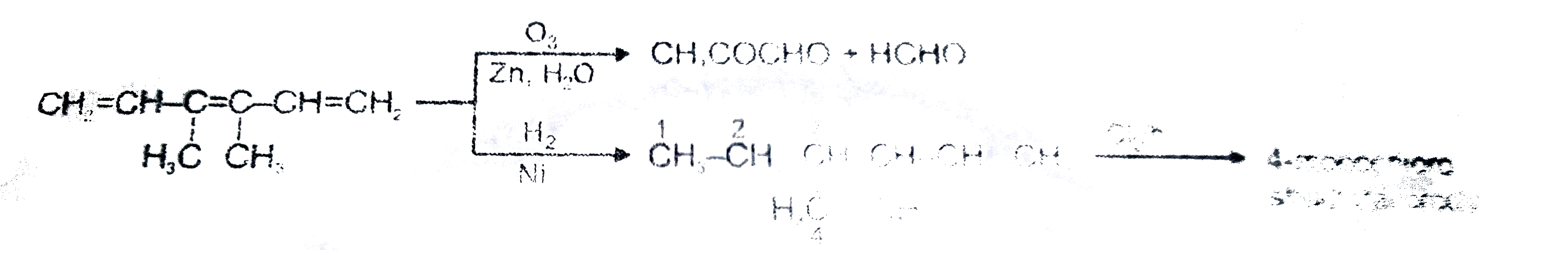
|
|
| 5. |
Read the following passage carefully and answer the questions : CH_(2)=CH-underset(H_(1)C)underset(|)(C )=underset(CH_(3))underset(|)(C )-CH=CH_(2) , Product M cannot respond with : |
|
Answer» `2,4`-DNP 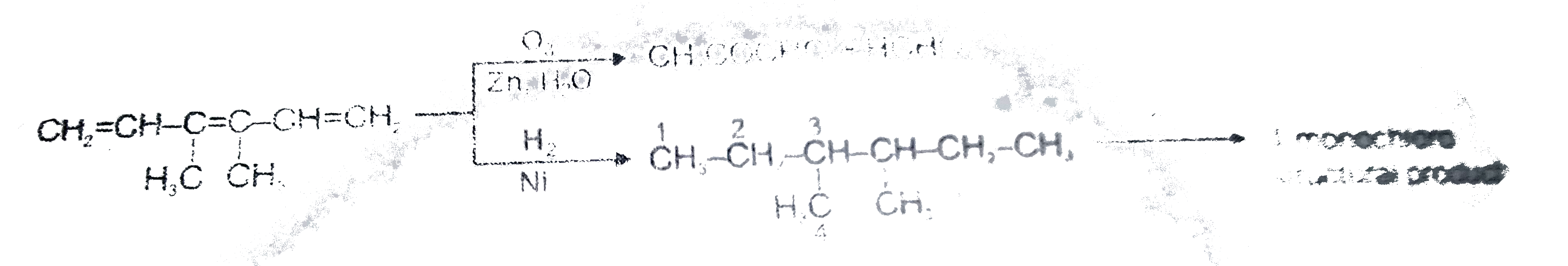
|
|
| 6. |
Read the following passage and answer the questions , Bodenstein carried out the determination of equilibrium constants of phosgene equilibrium by introducing CO and CI_2 at know pressure in a reaction bulb and measuring the equillibrium pressure from the attached monometer. in one experiment, CO at 342 nm and CI-2 at 351.4 nm were introduced . The equilibrium pressure was found to be 439.5nm . at equilibrium if partial pressure of COCI_2 "be" x "mm at " 127^@C. When he gas added in this above equilibrium at constant pressure |
|
Answer» MOLES of `COCI_2` INCREASED |
|
| 7. |
Read the following passage and answer the questions , Bodenstein carried out the determination of equilibrium constants of phosgene equilibrium by introducing CO and CI_2 at know pressure in a reaction bulb and measuring the equillibrium pressure from the attached monometer. in one experiment, CO at 342 nm and CI-2 at 351.4 nm were introduced . The equilibrium pressure was found to be 439.5nm . at equilibrium if partial pressure of COCI_2 "be" x "mm at " 127^@C. DeltaH of above equilibrium is equal to ............when K_p becomes 6.75 atm^(-1) "at" 227^@C |
|
Answer» `-50 kJ//mol` |
|
| 8. |
Read the following passage and answer the questions , Bodenstein carried out the determination of equilibrium constants of phosgene equilibrium by introducing CO and CI_2 at know pressure in a reaction bulb and measuring the equillibrium pressure from the attached monometer. in one experiment, CO at 342 nm and CI-2 at 351.4 nm were introduced . The equilibrium pressure was found to be 439.5nm . at equilibrium if partial pressure of COCI_2 "be" x "mm at " 127^@C. The volume of reacting vessel decreased by 3 times , K_p changed by |
|
Answer» 3 TIMES increased |
|
| 9. |
Read the following paragraph and answer the questions given below:Some of the most important reactions involving the Lewis and Bronsted acidity of inorganic compounds occur at solid surfaces. For example, 'surface acids' which are solid with a high surface area and Lewis acid sites are used as catalysts in the petrochemical industry for the interconversion of hydrocarbons. The surfaces of many materials that are important in the chemistry of soil and natural water also have Bronsted and Lewis acid sites. Silica surfaces do not readily produce Lewis acid sites because -OH group remain tenaciously attached at the surface of SiO_(2) derivatives, as a result Bronsted acidity is dominant. Alumino silicates display strong Bronsted acidity. When surface OH groups are removed by heat treatment, the alumino silicates surfaces possess strong Lewis acid sites. Surface reactions carried out using the Bronsted acid sites of silica gel are used to prepare thin coatings of a wide variety of organic groups using surface modification reactions. Thus, silica gel can be modified to have affinities for specific classes of molecules. Choose the correct option:QWhich of the following statement is correct? |
|
Answer» Compound which acts as Bronsted acid ALWAYS ACT as LEWIS acid, |
|
| 10. |
Read the following passage and answer the questions , Bodenstein carried out the determination of equilibrium constants of phosgene equilibrium by introducing CO and CI_2 at know pressure in a reaction bulb and measuring the equillibrium pressure from the attached monometer. in one experiment, CO at 342 nm and CI-2 at 351.4 nm were introduced . The equilibrium pressure was found to be 439.5nm . at equilibrium if partial pressure of COCI_2 "be" x "mm at " 127^@C. The K_p for the above reaction is |
|
Answer» `0.029 ATM^(-1)` |
|
| 11. |
Read the following passage and answer the questions , Bodenstein carried out the determination of equilibrium constants of phosgene equilibrium by introducing CO and CI_2 at know pressure in a reaction bulb and measuring the equillibrium pressure from the attached monometer. in one experiment, CO at 342 nm and CI-2 at 351.4 nm were introduced . The equilibrium pressure was found to be 439.5nm . at equilibrium if partial pressure of COCI_2 "be" x "mm at " 127^@C. Reaction is CO+CI_2 hArr COCI_2 C Value of x |
|
Answer» 88.1 MM |
|
| 12. |
Read the following paragraph and answer the questions given below:Some of the most important reactions involving the Lewis and Bronsted acidity of inorganic compounds occur at solid surfaces. For example, 'surface acids' which are solid with a high surface area and Lewis acid sites are used as catalysts in the petrochemical industry for the interconversion of hydrocarbons. The surfaces of many materials that are important in the chemistry of soil and natural water also have Bronsted and Lewis acid sites. Silica surfaces do not readily produce Lewis acid sites because -OH group remain tenaciously attached at the surface of SiO_(2) derivatives, as a result Bronsted acidity is dominant. Alumino silicates display strong Bronsted acidity. When surface OH groups are removed by heat treatment, the alumino silicates surfaces possess strong Lewis acid sites. Surface reactions carried out using the Bronsted acid sites of silica gel are used to prepare thin coatings of a wide variety of organic groups using surface modification reactions. Thus, silica gel can be modified to have affinities for specific classes of molecules. Choose the correct option:QDue to availability of Lewis as well as Bronsted sites of natural materials |
|
Answer» DIFFERENT places have different pH of soil |
|
| 13. |
Read the following paragraph and answer the questions given below:Some of the most important reactions involving the Lewis and Bronsted acidity of inorganic compounds occur at solid surfaces. For example, 'surface acids' which are solid with a high surface area and Lewis acid sites are used as catalysts in the petrochemical industry for the interconversion of hydrocarbons. The surfaces of many materials that are important in the chemistry of soil and natural water also have Bronsted and Lewis acid sites. Silica surfaces do not readily produce Lewis acid sites because -OH group remain tenaciously attached at the surface of SiO_(2) derivatives, as a result Bronsted acidity is dominant. Alumino silicates display strong Bronsted acidity. When surface OH groups are removed by heat treatment, the alumino silicates surfaces possess strong Lewis acid sites. Surface reactions carried out using the Bronsted acid sites of silica gel are used to prepare thin coatings of a wide variety of organic groups using surface modification reactions. Thus, silica gel can be modified to have affinities for specific classes of molecules. Choose the correct option:QWhen surface OH groups are removed by heat treatments from alumino silicates they can |
|
Answer» CATALYSE any reaction |
|
| 14. |
Read the following paragraph and answer the questions given below:Some of the most important reactions involving the Lewis and Bronsted acidity of inorganic compounds occur at solid surfaces. For example, 'surface acids' which are solid with a high surface area and Lewis acid sites are used as catalysts in the petrochemical industry for the interconversion of hydrocarbons. The surfaces of many materials that are important in the chemistry of soil and natural water also have Bronsted and Lewis acid sites. Silica surfaces do not readily produce Lewis acid sites because -OH group remain tenaciously attached at the surface of SiO_(2) derivatives, as a result Bronsted acidity is dominant. Alumino silicates display strong Bronsted acidity. When surface OH groups are removed by heat treatment, the alumino silicates surfaces possess strong Lewis acid sites. Surface reactions carried out using the Bronsted acid sites of silica gel are used to prepare thin coatings of a wide variety of organic groups using surface modification reactions. Thus, silica gel can be modified to have affinities for specific classes of molecules. Choose the correct option:QWhen -OH group strongly attached with SiO_(2) derivatives, its |
|
Answer» ACIDIC STRENGTH REMAINS same .. |
|
| 15. |
Read the following paragraph and answer the questions given below:Some of the most important reactions involving the Lewis and Bronsted acidity of inorganic compounds occur at solid surfaces. For example, 'surface acids' which are solid with a high surface area and Lewis acid sites are used as catalysts in the petrochemical industry for the interconversion of hydrocarbons. The surfaces of many materials that are important in the chemistry of soil and natural water also have Bronsted and Lewis acid sites. Silica surfaces do not readily produce Lewis acid sites because -OH group remain tenaciously attached at the surface of SiO_(2) derivatives, as a result Bronsted acidity is dominant. Alumino silicates display strong Bronsted acidity. When surface OH groups are removed by heat treatment, the alumino silicates surfaces possess strong Lewis acid sites. Surface reactions carried out using the Bronsted acid sites of silica gel are used to prepare thin coatings of a wide variety of organic groups using surface modification reactions. Thus, silica gel can be modified to have affinities for specific classes of molecules. Choose the correct option:Q Alumino silicates are also called |
|
Answer» autocatalysts |
|
| 16. |
Read the following paragraph and answer the questions given below,The dissociation of nitrosyl chloride into nitric oxide and chlorine takes place according to the equation, 2NOCI(g) hArr 2NO(g) +CI_2 (g) varying amounts of the three gases placed in a container and allowed to come to equilibrium at two different temperature. The equilibrium concentrations of the three gases obtained are tabulated below, The reaction is carried outin sealed vessel at constant temperature . if the pressures in the vessels were halved by removal of half the contents and equilibrium were then re-established at the same temperature, then the concentration of chlorine would be halved. |
|
Answer» 1 |
|
| 17. |
Read the following paragraph and answer the questions given below,The dissociation of nitrosyl chloride into nitric oxide and chlorine takes place according to the equation, 2NOCI(g) hArr 2NO(g) +CI_2 (g) varying amounts of the three gases placed in a container and allowed to come to equilibrium at two different temperature. The equilibrium concentrations of the three gases obtained are tabulated below, The enthalphy of dissociation of the NOCI is 38 kJ per mole. |
|
Answer» 1 |
|
| 18. |
Read the following paragraph and answer the questions given below,The dissociation of nitrosyl chloride into nitric oxide and chlorine takes place according to the equation, 2NOCI(g) hArr 2NO(g) +CI_2 (g) varying amounts of the three gases placed in a container and allowed to come to equilibrium at two different temperature. The equilibrium concentrations of the three gases obtained are tabulated below, The above equilibrium reaction is endothermic. |
|
Answer» 1 |
|
| 19. |
Read thefollowing paragraph and answer the questions given below , Stable equilibrium is of various type . Mechanical equilibrium is achieved when all particles are at rest and total potential energy of the system is minimum . At any stage where particles are at rest but the system is not at stabel equilibrium as it can reduce its potential energy by reverting to another position, is called metastable equilibrium . Thermal equilibrium is result from the absence of temperature gradients in the system. chemical equilibrium is obtained when no further reaction occurs between reacting substances, i.e forward and reverse rates of reaction are equal. when steam with solid iron at high temperature Fe_3O_4(s) and hydrogen gas are produced . But the reaction never goes to completion. this is because as the products are formed the reaction proceedsin reverse direction and when rate of reverse reaction is equal to rate of forward reaction , the concentration of reactants and products become constant and equilibrium is reached. When two reactants (A) and (B) are mixed to give products (C) and (D) the concentration quotient (Q) at initial stage of the reaction |
|
Answer» is zero |
|
| 20. |
Read the following paragraph and answer the questions given below, for a general reaction, aA+bB hArr cC=dD, equilibriumconstant K_c is given by K_c=([C]^c[D]^d)/([A]^a[B]^b) However, when all reactants and products are gases the equilibrium constant is generally expressed in terms of partial pressure. K_p=(P_C^cxx P _D^d)/(P_A^axxP_B^b) For the reaction, C(s) +CO(g) hArr 2CO(g) the partial pressure of CO_2 and CO are 2 and 4 atm at equilibrium . the K_p for the reaction is |
|
Answer» 0.5 |
|
| 21. |
Read the following paragraph and answer the questions given below: According to Maxwell distribution, the area under the curve is equal to the total number of molecules in collision. On increasing temperature, fraction of molecules having speed equal to ump (most probable speed) decreases. The speed distribution also depends on the mass of the molecules along with the temperature. In general, the distribution depends upon the value of (M)/(T) (where M is molar mass and T is temperature in kelvin). Choose the correct option: On increasing temperature, which of the following statement is incorrect? |
|
Answer» Area under the CURVE at DIFFERENT temperature are different. |
|
| 22. |
Read the following paragraph and answer the questions given below: According to Maxwell distribution, the area under the curve is equal to the total number of molecules in collision. On increasing temperature, fraction of molecules having speed equal to ump (most probable speed) decreases. The speed distribution also depends on the mass of the molecules along with the temperature. In general, the distribution depends upon the value of (M)/(T) (where M is molar mass and T is temperature in kelvin). Choose the correct option: If U_(mp)" at "T_(1) K" is "U_(1), then value of U_(rms)" at "T_(2) is |
|
Answer» `3sqrt((T_(2))/(T_(1)))u_(1)` |
|
| 23. |
Read the following paragraph and answer the questions given below: According to Maxwell distribution, the area under the curve is equal to the total number of molecules in collision. On increasing temperature, fraction of molecules having speed equal to ump (most probable speed) decreases. The speed distribution also depends on the mass of the molecules along with the temperature. In general, the distribution depends upon the value of (M)/(T) (where M is molar mass and T is temperature in kelvin). Choose the correct option: On increasing the temperature, number of molecules having speed between u_(1) and u_(2) |
|
Answer» INCREASES |
|
| 24. |
Read the following paragraph and answer the question given below: Freezing point of benzene is 278.4K and heat of fusion of benzene is 10.042 KJ/mol. Acetic acid exists partly as dimer in benzene solution. The freezing point of 0.02 mol fraction of acetic acid in benzene is 277.4 K. The equilibrium constant for dimerisation of acetic in benzene is |
|
Answer» 2.4 |
|
| 25. |
Read the following paragraph and answer the questions given below: According to Maxwell distribution, the area under the curve is equal to the total number of molecules in collision. On increasing temperature, fraction of molecules having speed equal to ump (most probable speed) decreases. The speed distribution also depends on the mass of the molecules along with the temperature. In general, the distribution depends upon the value of (M)/(T) (where M is molar mass and T is temperature in kelvin). Choose the correct option: Relationship between T_(1) and T_(2) a is given as |
|
Answer» `T_(1)=T_(2)` |
|
| 26. |
Read the following paragraph and answer the question given below: Freezing point of benzene is 278.4K and heat of fusion of benzene is 10.042 KJ/mol. Acetic acid exists partly as dimer in benzene solution. The freezing point of 0.02 mol fraction of acetic acid in benzene is 277.4 K. The degree of dimerisation of acetic acid in benzene is |
|
Answer» `0.24` |
|
| 27. |
Read the following paragraph and answer the question given below: Freezing point of benzene is 278.4K and heat of fusion of benzene is 10.042 KJ/mol. Acetic acid exists partly as dimer in benzene solution. The freezing point of 0.02 mol fraction of acetic acid in benzene is 277.4 K. The molal cryoscopic constant of benzene in K "molality"^(-1) is |
|
Answer» `4.0` |
|
| 28. |
Read the following information about ionic compound- (i) For formation of ionic compound ionisation potential of metal should be high. (ii) Ionic bond has non directional nature. (iii) For completeion of octetionic bond can represent as a coordinate bond. (iv) Ionic compound does not conduct electricity in solid state but conduct electricity in molten state. (v) During the solubility of ionic compound if lattice energy gt Hydration energy then compound is insoluble in water. The statements which are correct is: |
|
Answer» two |
|
| 29. |
Read the following details and decide the correct answer(s) given with each question and then select correct option given below the questions. (i) What is the method to obtain gas in laboratory ? (a) NH_(4)Cl(aq) + NaNO_(2)(g) to Na_(2)(g) + 2H_(2)O(l) + NaCl(aq) (b) 2KClO_(3)(s) overset("Heat")underset([MnO_(2))]to 2KCl(s) + 3O_(2)(g) ( c) 2PbO_(2)(s) overset(Delta)to 2PbO(s) + O_(2)(g) (ii) Which of the following does not have allotropes ? (a) Oxygen (b) Phosphorous (c) Nitrogen (d) Bismuth (iii) XeF_6 reacts with water to produce........... (a) XeO_(3) (b) XeO_(2)F_(2) ( c) XeOF_(4) (d) XeO (iv) Required concentration of 02 to substain of marine and aquatic living beings is............. (a) 3.08 ppm (b) 3.80% w/w (c) 3.80% v/v (d) 3.08% v/v |
|
Answer» (i)a, B (II) rarr d, a (III) rarr b, d, C, a (iv) rarr d |
|
| 30. |
Read the following industrial methods for the preparartion of H_(2)SO_(4) and answer the question at the end. Professor Molina of the Massachusetts Institute of Technology won the 1995 Noble prize in Chemistry for his work on atmospheric chemistry . One reaction that he studied in detail is the acid rain reaction which produces H_(2)SO_(4) in the atmosphere. He proposed two possible stoicchiometric reactions: Proposal A: H_(2)O(g) +SO_(3)(g) rarr H_(2)SO_(4)(g) Proposal B: 2H_(2)O(g) +SO_(3)(g) rarr H_(2)SO_(4)(g) +H_(2)O(g) Using the simple collision theory, what reaction orders would be expected for proposal B? ProposalB is thought to proceed by the following two-step process: SO_(3)+2H_(2)Ounderset(k_(-1))overset(k_(1))hArr SO_(3).2H_(2)O""(Fast) SO_(3).2H_(2)O overset(k_(2))rarr H_(2)SO_(4)+H_(2)O "" (slow) (SO_(3).2H_(2)O is a complex which is stabilized by hydrogen bonds and k_(2) lt lt k_(1) or k_(-1)) |
|
Answer» `k[H_(2)O][SO_(3)]` |
|
| 31. |
Read the following graph and answer the following question To make the following reduction process spontaneous, temperature should be: |
|
Answer» `LT 1000^(@)C` |
|
| 32. |
Read the following graph and answer the following question Which of the following statement is true? |
|
Answer» |
|
| 33. |
Read the following : (i) The half - life period of a radioactive element X is same as the mean-life time of another radioactive element Y. Initially both of them have the same number of atoms. ThemY will decay at a faster rate than X. (ii) The electron emitted in beta radiation originates from decay of a neutron un a nucleus. (iii) The half-life of^(215) At is 100 ms. The time taken for the radioacivity of a sample "of"^(215) At to decay to 1//16th of its initial value is 400 ms. (iv) The volume (V) and mass (m) of a nucleus are related as V prop m. (v) Given a sample of Radium-226 having half-life of4 days. The probability, a nucleus disintegrates within 2 half llives is 3/4. Select the correct code for above. |
|
Answer» T T T T T |
|
| 34. |
Read the following : |
|
Answer» The half -life period of a radioactive ELEMENT X is same as the mean-lifetime of another radioactive element Y. Initially both of them have the same number of atoms. Then Y will decay at a faster rate than X. `:.y` DECOMPOSES at a faster rate than x (B) ln nucleus `._(0)n^(1)rarr._(1)P^(1) ._(+1)e^(0)` so during `beta` - decay radiation electron emitted originates from decay of neutronin a nucleus. (C) ln four half life times the activity left will be `=(A_(0))/(2^(4))=(1)/(16)A_(0)` (`A_(0)rarr` Initial activity) (D) radius of nuclei`=r_(0)(A)^(1//3)"" Ararr` mass number Volume of Nuclei ` alpha r^(3)propA` Mass of nuclei ` alphaA` `:.` Volume of nuclei ` prop` mass of nuclei |
|
| 35. |
Read of the following statements and choose the correct code w.r.t. true(T) and false (F). (I) manganese salts give a violet borax bead test in reducing flame (II) from a mixed precipitate of AgCl and AgI, ammonia solution dissolves only AgCl (III) ferric ions give a deep green precipitate, on adding potassium ferrocyanide solution (IV) on boiling the solution having K^(+), Ca^(2+) and HCO_(3)^(–) we get a precipitate of K_(2)Ca(CO_(3))_(2) |
|
Answer» TTFF |
|
| 36. |
Read name silica covers an entire group of minereal, which have the general formual SiO_(2), the mole common of which is quartz. Quartz is form work silicane with SiO_(4) tetrahedra arranged in spirals. The spirals can turn in a clockwise or anticlockwise direction a feature that results in there being two mirror images, optically active, varieties of quartz. The silicate aion in the mineral kinotite is a Chian of three SiO_(4) tetrahedra that share corners with adjacent tetrahedra. The mineral also contains Ca^(2+) ions, Cu^(2+). ions and water molecule in a 1:1:1 ratio mineral is represented as |
|
Answer» `CaCuSi_(3)O_(10).H_(2)O` |
|
| 37. |
Read colour sodiumsaltis obtainedby acidformreact with . |
|
Answer» `Na_(2)SO_(4)` |
|
| 38. |
reacts with : gamma-Butyroactone (ester) What are the producut in each case ? |
| Answer» Answer :A::C | |
| 39. |
…………….reacts with alkali on boiling in an inert atmosphereliberating phosphine. |
| Answer» SOLUTION :WHITE PHOSPHOROUS | |
| 40. |
Reactivity order with respect to alkyl group of Hunsdicker reaction is |
|
Answer» `1^@ GT2^@ GT 3^@` |
|
| 41. |
Reactivity of transiton elements decreases almost regularly from Sc to Cu . Explain. |
| Answer» Solution :Reactivity of transition elements depends mainly upon their upon their ionization enthalpies. As we move from SC to Cu, ionization enthalpies increase ALMOST REGULARLY . Hence, their reactivity DECREASES almost regularly from Sc to Cu. | |
| 42. |
Reactivity order of the following towards NaOEt, EtOH |
|
Answer» `III GT II gt I` |
|
| 43. |
Reactivity order of halides for dehydrohalogenation is |
|
Answer» `R-FgtR-ClgtR-BrgtR-I` |
|
| 44. |
Reactivity of transition elements decreases almost regularly from Sc to Cu. Explain. |
| Answer» Solution :REACTIVITY of an ELEMENT is dependent on the value of ionisation enthalpy. In moving from Sc, the FIRST element to Cu, the ionisation enthalpy increases regularly. THEREFORE, the reactivity decreases as we move from Sc to Cu. | |
| 45. |
Reactivity of NO is due to: |
|
Answer» Its LOW MOLECULAR weight |
|
| 46. |
Reactivity of order of halides for dehydrohalogenation is |
|
Answer» `R-F gtR- Cl gt R - Br gt R -I` THUS, the reactivity order is `R- I gt R - Br gt R - Cl gt R - F`. |
|
| 47. |
Reactivity of hydrogen atoms attached to different atoms in alkanes has the order: |
|
Answer» `3^@ GT 1^@gt2^@` |
|
| 48. |
Reactivity of different types of hydrogen during halogenation of alkanes follows the order- |
|
Answer» `2^@H gt 1^@ HGT 3^@H` |
|
| 49. |
Reactivity of borazole is greater than that of benzene because. |
|
Answer» BORAZOLE is nor-polar compound |
|