Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Presently which reagent is used for separation of 1^(@),2^(@) and 3^(@) amines |
|
Answer» p-toluene SULPHONYL CHLORIDE |
|
| 2. |
Prespex or plexiglass is a polymer of: |
|
Answer» METHYL methacrylate |
|
| 3. |
…………………….present in the animals act as an oxidising agent and ……………………catalyses the oxidation of toxic alcohol, the liver produces an enzyme called …………………… . |
| Answer» SOLUTION :`NAD^(+),ADH` | |
| 4. |
Presence of water can be tested with |
|
Answer» adding a pinch of ANHYDROUS COPPER sulphate which changes its colour from WHITE to blue |
|
| 5. |
Presence of three unpaired electrons in phosphorus (Z=15) can be explained by |
|
Answer» AUFBAU principle |
|
| 6. |
presence of small impurity usually makes a metal quite hard because the impurities |
|
Answer» CHANGE the lattice structure of metals |
|
| 7. |
Presence of nitrogen in which among the following compounds can not be detected by Lassaigne method ? |
|
Answer» Hydrazine (a) `underset("Hydrazine")(NH_(2)-NH_(2))`  Since , hydrazine is not an organic compound , thus it cannot detected by Lassaigne method . |
|
| 8. |
Presence of peroxides affects the addition of |
| Answer» Solution :Peroxide RULE is APPLICABLE only to HBr and not for HCl , HF and HI | |
| 9. |
Presence of peroxide effects the addition of: |
|
Answer» HBr |
|
| 10. |
Presence of peroxide in ethers of old stock can be tested by first treating them with FeSO_4 solution and then adding an aqueous solution of ………&from red colour solution |
| Answer» ANSWER :D | |
| 11. |
Presence of ___________ group at o- and p-positions withdraws the electron density from the benzene ring. |
|
Answer» |
|
| 12. |
Presence of Fe^(3+) ions in FeSO_4 can be checked with |
|
Answer» BLUE litmus paper `Fe^(3+) +3CNS^(-) to UNDERSET("Blood red colouration")(Fe(CNS)_(3))` |
|
| 13. |
Presence of halogen in organic compounds can be detected using |
|
Answer» Leibig's test<BR>Duma's tes |
|
| 14. |
Presenceof electron - withdrawing substituents |
|
Answer» STABILIZES carbocations , carbanions as WELL as free radicals. |
|
| 15. |
Presence of electron withdrawing group on the phenyl of aromatic carboxylic acid ___________their acidity. |
| Answer» SOLUTION :INCREASES | |
| 16. |
Prepare the following from 1-pentene: i) CH_(3)CH_(2)CH_(2)DCH_(3) (A) ii) CH_(3)CH_(2)CH_(2)CH_(2)D (B) iii) CH_(3)CH_(2)CH_(2)CHDCH_(2)D (C ) |
Answer» SOLUTION :
|
|
| 17. |
Prepare the ethers by Williamson.s ether synthesis CH_(3)-O-underset(CH_(3))underset(|)overset(CH_(3))overset(|)C-CH_(3) |
Answer» Solution :There are twoapproaches for PREPARING each for preparing each of the above mentioned ETHERS by williamson.s ether synthesis. These are PROVIDED below :  Out of the two approaches (i) is more FAVORABLE. |
|
| 18. |
Prepare the ethers by Williamson.s ether synthesis |
Answer» Solution :There are twoapproaches for preparing each for preparing each of the above mentioned ethers by williamson.s ETHER SYNTHESIS. These are provided below :  Only APPROACH (i) works because aryl halide will not undergo `S_(N)2` reaction since it is resonance stablized. |
|
| 19. |
Prepare pure sample of 1^(@)amine from 1^(@) alkyl halide. |
Answer» SOLUTION :By Grbriel PHTHALIMIDE REACTION 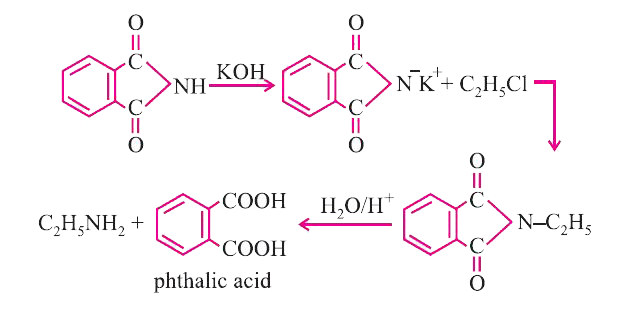
|
|
| 20. |
Prepare the ethers by Williamson.s ether synthesis |
Answer» Solution :There are TWOAPPROACHES for preparing each for preparing each of the above mentioned ETHERS by williamson.s ether synthesis. These are provided below :  The approach (i) is more favorable because `CH_(3)-I` is more reactive TOWARD `S_(N)2` reaction (due to LESS steric hindrance) |
|
| 21. |
Prepare butane fromchoroethane usingthecorey - house synthesis . |
|
Answer»
` (ii)2CH_(3)CH_(2)Li+Cu L tounderset("Lithiumdiethy CUPRATE ")(Li(CH_(3)CH_(2))_(2)Cu+Lil)` ` (iii)Li(CH_(3)CH_(2))_(2)Cu+CH_(3)CH+_(2)Cl to underset("Bulane")(CH_(3)CH_(2)CH_(2)CH_(3))+CH_(3)CH_(2)CU+LiCl` |
|
| 22. |
Prepare / convert nitrobenzene into aniline. |
Answer» SOLUTION :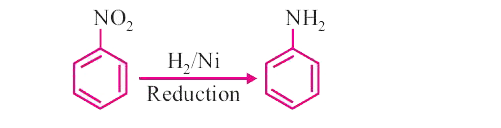
|
|
| 23. |
Prepare a reaction-coordinate diagram for the reaction 2H_2O_2 to 2H_2O_2+ O_2 , Delta H = -191.78 kJin which energy of activation for uncatalysed and catalysed reactions are 75.3 kJ "mole"^(-1) and 56.6 kJ "mole"^(-1)respectively at 298 K.(a) What is the ratio of the rate constant for the forward catalysed reaction to thatfor the forward uncatalysed reaction?(b) By what factor will the rate constant for the reverse catalysed reaction increase compared to that for the reverse uncatalysed reaction?(c) Hence prove that the catalyst increases both the forward and reverse reactions by the same factor. |
| Answer» SOLUTION :(a) 1970 (B) 1970 | |
| 24. |
Preparation of salysilic acid from phenol is known as …… process. |
|
Answer» Dow |
|
| 25. |
Preparation of Na_(2)CO_(3) extract is made for acid radical analysis because |
|
Answer» all anion REACT with Na to GIVE water SOLUBLE compounds |
|
| 26. |
Preparation of looking mirrors involves the use of : |
|
Answer» RED lead |
|
| 27. |
Preparation of looking mirrors involves the use of: |
|
Answer» RED LEAD |
|
| 28. |
Preparation of lakyl halide in laboratory is least preferred by, |
|
Answer» direct halogenation of ALKANE |
|
| 29. |
Preparation of cabalt Metaborate involves the following steps of reactions: (i) Ca_(2)B_(6)O_(11) + Na_(2)CO_(3)(aq) overset("Boiled") to CaCO_(3) "(insoluble)" + Na_(2)B_(4)O_(7) + NaBO_(2) (ii) Na_(2)B_(4)O_(7) overset(Delta)to NaBO_(2) + B_(2)O_(3) (iii) CoO + B_(2) O_(3) overset(Delta) to Co(BO_(2))_(2). (Atomic weight: B=11, Co=59) Mass of Ca_(2)B_(6)O_(11) in kg requried to produce 14.5 kg of Co(BO_(2))_(2), assuming 100% yield of each reaction is |
|
Answer» 32.2 |
|
| 30. |
Preparation of cabalt Metaborate involves the following steps of reactions: (i) Ca_(2)B_(6)O_(11) + Na_(2)CO_(3)(aq) overset("Boiled") to CaCO_(3) "(insoluble)" + Na_(2)B_(4)O_(7) + NaBO_(2) (ii) Na_(2)B_(4)O_(7) overset(Delta)to NaBO_(2) + B_(2)O_(3) (iii) CoO + B_(2) O_(3) overset(Delta) to Co(BO_(2))_(2). (Atomic weight: B=11, Co=59) If the yield of reaction (i),(ii) & (iii) 60% (200)/(3)% & 32.2% respectively , then mass of Ca_(2)B_(6)O_(11) in kg required to produce 14.5 kg of Co(BO_(2))_(2) is |
|
Answer» 250 |
|
| 31. |
Preparation of ethers by acid dehydration of secondary and tertiary alcohols is not a suitable method. Give reason. |
Answer» Solution :Acid-catalysed dehydration of `1^(@)` alcohols to ethers occurs by `S_(N)2` reaction involving nucleophilic attack by the alcohol molecule on the protonated alcohol molecule. 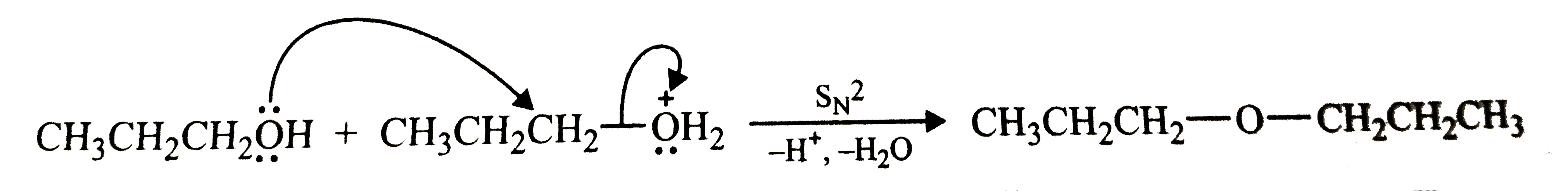 Under these conditions, `2^(@) and 3^(@)` alcohols, however, give alkenes rather than ethers. the reason being that due to sterric hindrance, nucleophilic attack by the alcohol molecule on the protonated alcohol molecule does not occur, INSTEAD protonated `2^(@) and 3^(@)` alcohols lose a molecule of water to FORM stable `2^(@) and 3^(@)` carbocations. these carbocations prefer to loss a proton to form alkenes rather than UNDERGOING nucleophilic attack by alcohol molecule to form ethers. 
|
|
| 32. |
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason. |
Answer» Solution :ACID - catalysed dehydration of `1^(@)` alcohols to ethers occurs by `S_(N)2` reaction involving nucleophilic attack by the alcohol molecule on the protonated alcohol molecule.  However `2^(@)` and `3^(@)` alcohols, give alkenesrather than ether under these conditions. Due to steric hindrance, nucleophilic attack by the alcohol molecule on the protonated alcohol molecule does not occur. Instead protonated `2^(@) and 3^(@)` carbocations. These carbocations prefer to lose a proton to from alkenes rather than undergoing nucleophilic attack by alcohol molecule to FORM ethers, as shown below taking the example of propan -2- ol, a `2^(@)` alcohol.  SIMILARLY, `3^(@)` alcohols give alkenes rather than ethers, again due to steric hindrance.  `underset("(Not formed)")underset("prooxy) propane")underset("2-Methyl-2-(2-methyl")(CH_(3)-overset(CH_(3))overset("|")underset(CH_(3))underset("|")"C "-O-overset(CH_(3))overset("|")underset(CH_(3))underset("|")"C "-CH_(3))overset((CH_(3))(3)COH)underset(-H^(+))rarrunderset("carbocation")underset("tert-Butyl")(CH(3)-underset(CH_(3))underset("|")overset(CH_(3))overset("|")(C^(+))underset(-H^(+))rarr)underset("2-Methylprop-1-ene")(CH_(3)-overset(CH_(3))overset("|")"C "=CH_(2))` |
|
| 33. |
Preparation of ether from ethanol by continuous etherification process is |
|
Answer» `SN^1` REACTION |
|
| 34. |
Preparation of beta-hydroxy ester is favoured by: |
|
Answer» CANNIZZARO's REACTION |
|
| 35. |
Preparation of alkyl halides in laboratory is least preferred by: |
|
Answer» Halide exchange |
|
| 36. |
Preparation of alkyl halides in laboratory is least preferred by |
|
Answer» TREATMENT of ALCOHOLS |
|
| 37. |
Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon. Explain its mechanism. |
|
Answer» Solution :`to`Step-1: Electrophilic attack by hydronium ion (`H_(2)O^(+)`) on alkene gives an intermediate. CARBOCATION. `(H_(2)SO_(4) to H^(+)+ ""^(-)OSO_(2)OH)` `H- underset(* *)overset(* *) O -H+ H^(+) to underset("Hydrominum ion")( H-underset(* *)overset(H)overset(|)overset(+)O- H(H_(3)O^(+)))`  Step-2 : Nucleophilic attack by water on carbocation to yield protonated ALCOHOL.  Step-3: Deprotonation (loss of proton) to form an alcohol. 
|
|
| 38. |
Prefix are correctly named for which of the following side-chain attached to the present chain/ring? |
|
Answer»
|
|
| 40. |
Preence of N is hydroxylamine hydrochloride cannot be detected by Lassaigne's test- why? |
| Answer» Solution :To hydroxylamine hydrochloride `(NH_(2)OH*HCI)` is fused with metalic SODIUM, NaCN is not OBTAINED as the compound CONTAINS no carbon. Thus, the presence of NITROGEN in this compound cannot to detectedby Lassaigne's TEST | |
| 41. |
Predit the rate law of the following reactions based on the data given below: 2A + B to C+D |
|
Answer» rate = `K[A][B]^(2)` `therefore` ORDER w.r.t. [A]=1 By comparing (i) and (iii) reaction rate becomes four timew when [B] is made twice while [A] is made twice while [A] is kept constant `therefore` order w.r.t. [B]=2 `therefore` rate `=k[A][B]^(2)` |
|
| 42. |
Predominantly ionic halide of N-family is - |
|
Answer» `BiF_(3)` |
|
| 43. |
Predict which pairs of ions is not coloured in aqueous solutions ? |
|
Answer» `Ti^(3+), V^(3+)` |
|
| 44. |
Predict which of the following will be coloured in aqueous solution? Ti^(3+), V^(3+), Cu^(+), Sc^(3+), Mn^(2+), Fe^(3+) and Co^(2+). Give reason for each |
Answer» Solution : `CU^(+) and Sc^(3+)` are COLOURLESS while all other ions are coloured. |
|
| 45. |
Predict which of the following will be coloured in aqueous solution ? Ti^(3+), V^(3+), Cu^(+), Sc^(3+), Mn^(2+), Fe^(3+) and Co^(2+). Give reasons for each. |
|
Answer» Solution :Only those IONS will be coloured which have incomplete d-orbitals, Ions which has complete or complete d-orbitals are COLOURLESS. `{:(Ti^(3+) = [AR]3D^(1),"Purple"),(V^(3+) = [Ar]3d^(2),"Green"),(Cu^(+) = [Ar]3d^(10),"Colourless"),(Sc^(3+) = [Ar],"Colourless"),(Mn^(3+) = [Ar]3d^5,"Pink"),(Fe^(3+) = [Ar] 3d^6,"Brown"),(Co^(2+) = (Ar) 3d^7,"Pink"):}` As `Sc^(3+)` and `Cu^(+)` have `3d^(0) and 3d^(10)` configuration in their valence shell so their aqueous solution are colourless. All other i.e., `Ti^(3+), V^(3+), Mn^(2+), Fe^(3+) and Co^(2+)` are coloured in aqueous medium. |
|
| 46. |
Predict which of the following will be coloured in aqueous solution ? Ti^(3+),V^(3+),Cu^(+),Sc^(3+),Mn^(2+),Fe^(3+) and Co^(2+). Give reasons for each. |
| Answer» SOLUTION :Only those iions which have incompletely filled d-orbitals are coloured. Those with fully-filled or empty d-orbitals are colourless. Thus, `Ti^(3+),V^(3+),Mn^(2+),Fe^(3+)` and `Co^(2+)` are all coloured DUE to d-d TRANSITION. `MnO_(4)^(-)` is also colouredbut due to charge transfer. Only `Sc^(3+)(3d^(0))` is colourless. | |
| 47. |
Predict which of the following will be coloured in aqueous solution? Ti^(3+) , V^(3+) , Sc^(3+) , Mn^(2+) , Fe^(3+) , Co^(2+)and MnO_(4)^(-) |
| Answer» Solution :Only those ions will be coloured which have INCOMPLETELY filled d-orbitals. Those withfully - filled or empty d-orbitals are colourless. Thus, `Ti^(3+) , V^(3+), Mn^(2+), FE^(3+)` and `Co^(2+)`are all coloured due to d-d transition. `MnO_(4)^(-)` is also colouredbut due to CHARGE transfer ( see pages) . Only `SC^(3+) ( 3d^(0))` is colourless. | |
| 48. |
Predict which of the following will be coloured in aqueous solution Ti^(2+), V^(3+), Se^(4+), Cu^(+), Sc^(3+), Fe^(3+), Ni^(2+), and Co^(3+) |
|
Answer» Solution :Among`Ti^(2+), V^(3+), Se^(4+), Cu^(+), Sc^(3+), Fe^(3+), Ni^(2+)`, and `Co^(3+)` in aqueous solution state, Ti (Z = 22) `Ti^(2+)` -Electronic configuration is `[AR] 3D^(2)` V (Z = 23) `V^(3+)` -Electronic configuration is `[Ar]3d^(2)` Sc (Z = 21)`Se^(4+)`-Electronic configuration is `[Ar] 1s^(2) 2s^(2) 2p^(6) 3s^(2) 3p^(5)` Cu (Z = 29) `Cu^(+)`-Electronic configuration is `[Ar] 3d^(10)` Sc (Z = 21) `Sc^(3+)`-Electronic configuration is `[Ar] 3d^(0)4s^(0)` Fe (Z = 26)`Fe^(3+)` Electronic configuration is `[Ar] 3d^(5)` Ni (Z = 28) `Ni^(2+)`-Electronic configuration is `[Ar] 3d^(6)` Co (Z = 27)`Co^(3+)`-Electronic configuration is `[Ar] 3d^(5)` A TRANSITION metal ion is coloured if it has one or more unpaired electrons in (n-1) d orbital. i.e. 3d orbitals in the case of first transition series, when such species are exposed to visible radiation, d-d transition take place and the species are coloured. 
|
|
| 49. |
Predict whether zinc nd silver react with 1 M sulphuric acid to give out hydrogen gas or not. Given that the standard reduction potential of zinc and silver are -0.76 volt and 0.80 volt respectively. |
|
Answer» Solution :(a) To predict reaction of zinc with SULPHURIC acid: if it reacts, the following reaction should take place: `Zn+H_(2)SO_(4) to ZnSO_(4)+H_(2),"i.e.,"Zn+2H^(+)TOZN^(2+)+H_(2)` By convention, the cell will be represented as: `Zn|Zn^(2+)||H^(+)|H_(2)`. Std. EMF of the cell `(E_(cell)^(@))=["Std. redn. pot. of R.H.S. electrode"]-["Std. redn. pot. of L.H.S. electrode"]=0-(-0.76)=+0.76` volt. THUS, the EMF of the cell comes out to be positive. HENCE, the reaction takes place (b) To predict the reaction of silver with sulphuric acid: if it reacts, the following reaction should take place: `2Ag+H_(2)SO_(4)toAg_(2)SO_(4)+H_(2),"i.e.,"2Ag+2H^(+)to2Ag^(+)+H_(2)` By convention, the cell may be represented as : `AG|Ag^(+)||H^(+)|H_(2)` `E_(cell)^(@)=E_(H^(+),H_(2))^(@)-E_(Ag^(+),Ag)^(@)=0-0.80=0.80` volt. |
|
| 50. |
Predict whether Tit will disproportionate in aqueous solution, given the following standard reduction potential E^(0)TI // TI ==0.34 V, E^(0)TI^(3)//ETI-1.25 V |
|
Answer» Solution :`2TI^(+)+2eto2TI``E^(@)` = -0.34 V `TItoTI^(3+)+2e``E^(@)` = -1.25 V `3TI^(-)to2TI+TI^(3+)``E^(@)`= -1.59 V Negative VALUE of Eo indicates that disproportion is not possible. THUS, Ti is more STABLE than `TI^(3+)`, hence `TI^(+)` will not oxidize. |
|
 ,
,  , Prefix : Methoxycarbonyl
, Prefix : Methoxycarbonyl , Prefix :
, Prefix :  , Prefix : Carbamoyl
, Prefix : Carbamoyl