Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Primary and secondary alcohols are dehydrogenated by copper at 573 K to aldehydes and ketones respectively. In contrast tertiary alcohols are dehydrated to alkenes by heating with copper at 573 K. Similarly, primary alcohols are easily oxidised to form first an aldehyde and then a carboxylic acid while secondary alcohols are oxidised to ketones which are further oxidised to form a mixture of acids. Tertiary alcohols are oxidised with difficulty and with strong oxidising agents in acidic medium. They form first ketones and then acids. In the case of alcohols containing carbon-carbon double bond, some oxidising agents oxidise both double bond and OH group while other reagents donot affect C-Chond. The reagent which oxidises 1^(@) alcohol to aldehyde without affecting C=C double bond is |
|
Answer» `CrO_(3)`aqueous acetone solution |
|
| 2. |
Primary amines react with benzaldehyde to from : |
|
Answer» AZO dyes |
|
| 3. |
Primary amineson treatmentwith HNO_(2)gibes |
|
Answer» PRIMARY ALCOHOLS |
|
| 4. |
Primary amines on oxidation with acidified KMnO_(4), followed by hydrolysis gives: |
|
Answer» ALDEHYDES only |
|
| 5. |
Primary amino group is absent in |
|
Answer» p-amino phenol |
|
| 6. |
Primary amines in heating with CS_(2) followed by excess of mercuric chloride yields isothiocyanates. The reation is called |
|
Answer» HOFMANN MUSTARD OIL reaction |
|
| 7. |
Primary amines on heating with chloroform and alcoholic potash give____and the reaction is known as______. |
| Answer» SOLUTION :ISOCYANIDES or CARBYLAMINES, CARBYLAMINE REACTION. | |
| 8. |
Primary amines have lower boiling points than |
|
Answer» corresponding ALKANES |
|
| 9. |
Primary amines have higher boiling points than tertiary amines why? |
| Answer» Solution :DUE to the presence of two H-atom on N-atoms of primary AMINES they UNDERGO extensive INTERMOLECULAR H-bonding while TERTIARY amines due to the absence of a H-atom on the N-atom do notundergo H-bonding. Asa result primary amines have higher boiling points than tertiary amines. | |
| 10. |
Primary amines can be distinguished from secondary and tertiary a mines by reacting with |
|
Answer» Chloroform and alcoholic KOH |
|
| 11. |
Primary amines can be distinguished from secondary amines by |
|
Answer» Gabriel-pbthalimide REACTION |
|
| 12. |
Primary amines give ___________ test. |
|
Answer» Iodoform |
|
| 13. |
Primary amine reacts with CHCl_3"/"KOH the product formed is………….and the reactions is called………….reaction. |
|
Answer» CYANIDE, CARBYL amine |
|
| 14. |
Primary amine reacts with an aldehyde to give: |
|
Answer» amide |
|
| 15. |
What happens when ethyl amine is treated with NaNO_2 and HCl ? |
|
Answer» NITRO compound |
|
| 16. |
Primary amineforms |
|
Answer» DIACETYL DERIVATIVE |
|
| 17. |
Primary amine on treatment with NaNO_2 and HCI yields: |
|
Answer» NITRO compound |
|
| 18. |
Primary amine acts as |
|
Answer» Electrophile |
|
| 19. |
Primary alkyl halides undergoes substitution by …………………. . |
|
Answer» `SN^(1)` REACTION |
|
| 20. |
Primary alkyl halide C_4H_9Br (X) reacts with alc. KOH to give compound (Y). (Y) reacts with HBr to give compound (Z) which is an isomer of (X). When (X) reacts with Na metal it gives compound (P). (X), (Y), (Z) and (P) are |
|
Answer» `{:(""X,""Y,""Z," "P),(CH_3-UNDERSET(CH_3)underset(|)(CH)-CH_2Br,CH_3-underset(CH_3)underset(|)C=CH_2,CH_3-underset(CH_3)underset(|)overset(BR)overset(|)C-CH_3,C_8P_18):}` 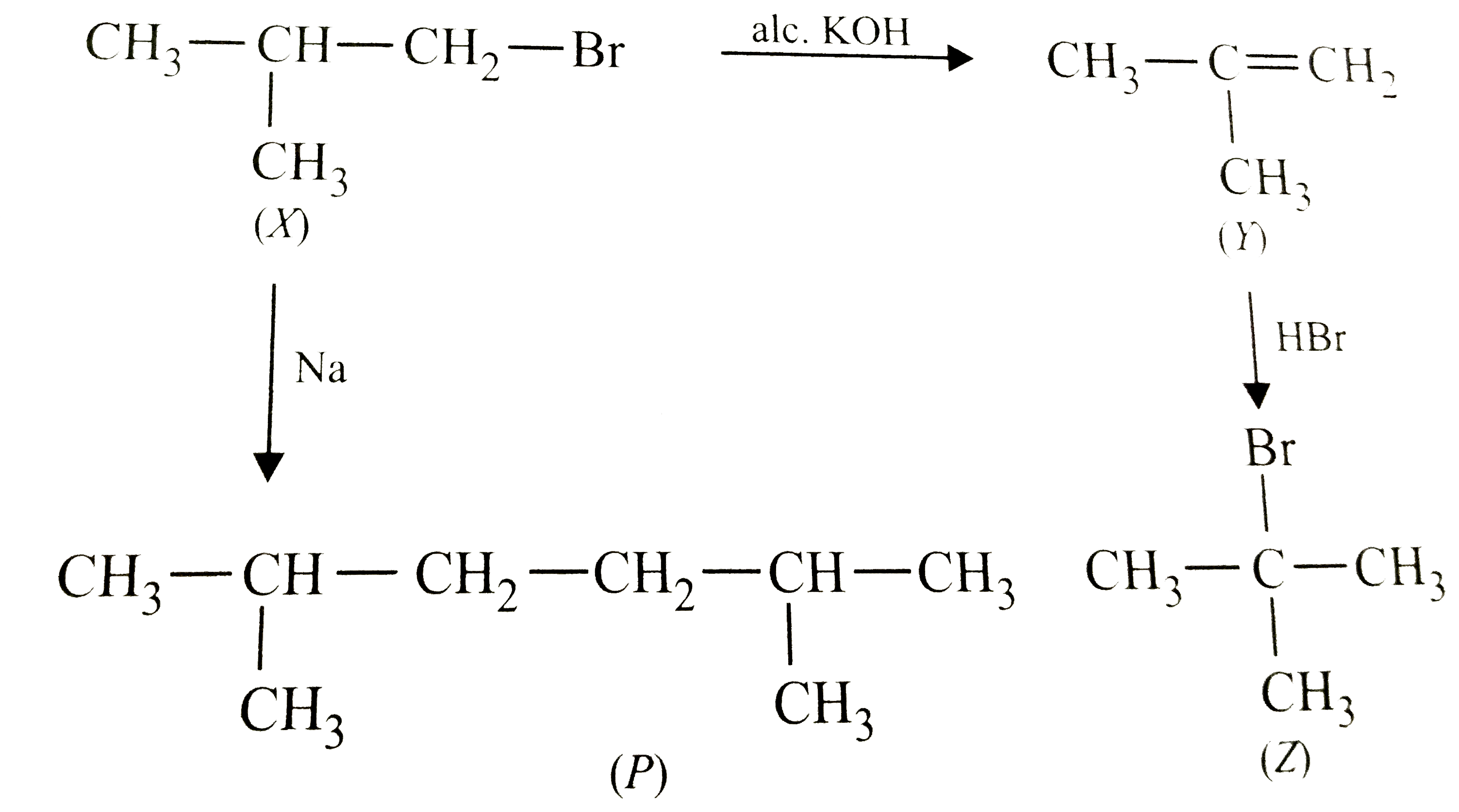
|
|
| 21. |
Primary alkyl halide C_(4)H_(9)Br (a) reacted with alcoholic KOH to give compound (b). Compound (b) is reacted with HBr to give (c ) which is an isomer of (a). When (a) is reacted with sodium metal it gives compound (d), C_(8)H_(18) which is different from the compound formed when n-butyl bromide is reacted with sodium. Give the structural formula of (a) and write the equations for all the reactions. |
|
Answer» SOLUTION :The possible structure of (A) are : `UNDERSET((i))(CH_(3)-underset(CH_(3))underset("|")("C "H-CH_(2)-Br` and `underset((ii))(CH_(3)CH_(2)CH_(2)Br)` When (A) is reacted with Na, the wurtz reaction takes place. However it is given that (A) on reaction with Na metal, gives alkane `C_(8)H_(18)` which is not the same as the one obtained from the wurtz reaction of n-butyl bromide. Thus, (A) is not (ii) 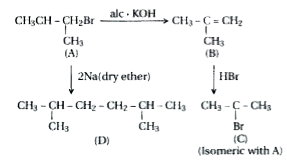
|
|
| 22. |
Primary alkyl halide C_(4)H_(9)Br(a) reacted with alcoholic KOH to give compound (b). Compound (b) is reacted with HBr to give (c) which is an isomer of (a). When (a) is reacted with sodium metal, it gives a compound (d), C_(8)H_(18)which is different from the compound formed when n-butyl bromide is reacted with sodium. Give the structural formula of (a) and write the equations for all the reactions. |
|
Answer» SOLUTION :Two primary ALKYL halideshaving the molecular formula, `C_(4)H_(9)Br `are `underset("n -Butyl bromide") (CH_(3)CH_(2)CH_(2)CH_(2)Br) "and " underset("Isobutyl bromide") (CH - overset(CH_(3)) overset(|) CH - CHBr) ` Compound (a) when reacted with Na metal gives a compound (d) with molecular formula `C_(8)H_(18) `which is different from the compound obtained when n-butyl bromide is reacted with Na metal. THEREFORE, (a) must be isobutyl bromide and compound (d) must be 2, 5- dimethylhexane. `underset("n-Butylbromide "(1^(@))) (2CH_(3)CH_(2)CH_(2)Br ) + Na underset(-2NaBr) overset("Wurtz reaction ") to underset("n -octane") (CH_(3)CH_(2)CH_(2)CH_(2)CH_(2)CH_(2) CH_(2)CH_(3))underset("Isobutyl bromide (a) "(1^(@))) (CH_(3) - overset(CH_(3)) overset(|) CH - CH_(2)Br ) + 2Na underset(-2 NaBr) overset("Wurtz reaction ") to underset("2, 5- Dimethylhexane") (CH_(3) - overset(CH_(3))overset(|) CH - CH_(2) - CH_(2) - overset(CH_(3)) overset(|) CH - CH_(3)) ` With the identification of (a) and (d) , other compounds i.e., (B) and (c ) can be identified as under : `underset((a)) underset ("Isobutyl chloride ") (CH_(3) - overset(CH_(3)) overset(|) C= CH_(2)Br )underset("Dehydrohalogenation ") overset(KOH (alc), Delta ) tounderset((b)) underset("2 - METHYL -1-propane") (CH_(3) - overset(CH_(3)) overset(|) C = CH_(2)) ` Thus, (b) is 2 - methyl -1- propene. The compound (b) on treatment with HBr gives compound (c ) in accordance with Markovnikov.s rule . Therefore , compound (c ) is tert - butyl bromide which is an isomer of compound (a) i.e., isobutyl bromide . `underset("2 - Methyl -1-propene ") (CH_(3) - overset(CH_(3)) overset(|) C = CH_(2)) underset("Markovnikov addition ") overset("HBr") to underset(["an isomer of compound (a) "]) underset("tert -Butyl bromide (c )") (CH_(3) - underset(Br) underset(|) overset(CH_(3)) overset(|) C = CH_(3)) ` Thus , (a) is isobutyl bromide, (b) is 2- methyl -1- propene, (c ) is tert - butylbromide and (d) is 2 , 5 - dimethylhexane. |
|
| 23. |
Primary alkyl halide C_(4)H_(9)Br (a) reacted with alcoholic KOH to give compound (b). Compound (b) is reacted with HBr to give (c) which is an isomer of (a). When (a) is reacted with Na metal, it gives a compound (d), C_(8)H_(18) which is different from the compound formed when n-butyl bromide is reacted with sodium. gove the structural formula of (a) and write the equations for all the reactions. |
|
Answer» Solution :(i) Two primary alkyl halides having the molecular FORMULA `C_(4)H_(9)Br` are `underset("n-Butyl bromide")(CH_(3)CH_(2)CH_(2)CH_(2)Br)"and"underset("Isobutyl bromide")((CH_(3)-overset(CH_(3))overset(|)(C)H-CH_(2)Br)` Thus, compound (a) is either n-butyl bromide or isobutyl bromide. (ii) Since compound (a) when reacted with Na metal gives a compound (d) with M.F. `C_(8)H_(18)` which is different from the compound obtained when n-butyl bromide is reacted with Na metal, therefore, (a) must be isobutyl bromide and compound (d) must be 2,5-dimethylhexane. `underset("n-Butyl bromide "(1^(@)))(2CH_(3)CH_(2)CH_(2)CH_(2)Br)+2Na underset(-2NaBr)overset("Wurtz reaction")to underset("n-Octane")(CH_(3)CH_(2)CH_(2)CH_(2)CH_(2)CH_(2)CH_(2)CH_(3))` `underset("Isobutyl bromide "(a)" "(1^(@)))(CH_(3)-overset(CH_(3))overset(|)(C)H-CH_(2)Br)+2Na underset(-2" "NaBr)overset("Wurtz reaction")to underset("2,5-Dimethylhexane (d), M.F. "C_(8)H_(18))(CH_(3)-overset(CH_(3))overset(|)(C)H-CH_(2)-CH_(2)-overset(CH_(3))overset(|)(C)H-CH_(3))` (iii) If compound (a) is isobutyl bromide then compound (b) which it gives on treatment with alcoholic KOH must be 2-methyl-1-propene `underset("Isobutyl chloride (a)")(CH_(3)-overset(CH_(3))overset(|)(C)H-CH_(2)Br) underset(("Dehydrohalogenation"))overset(KOH(alc),Delta)to underset("2-Methyl-1-propene (b)")(CH_(3)-overset(CH_(3))overset(|)(C)=CH_(2))` (iv) The compound (b) on treatment with HBr gives compound (c) in accordance with Markovnikov's RULE. therefore, compound (c) is tert-butyl bromide which is an isomer of compound (a), i.e., isobutyl bromide. `underset("2-Methyl-1-propene")(CH_(3)-overset(CH_(3))overset(|)(C)=CH_(2)) underset(("Mark. ADDN."))overset(HBr)to underset("tert-Butyl bromide (c) (an isomer of compound (a)")(CH_(3)-underset(Br)underset(|)overset(CH_(3))overset(|)(C)-CH_(3))` thus, (a) is isobutyl bromide, (b) is 2-methyl-1-propene, (c) is tert-butyl bromide and (d) is 2,5-dimethylhexane. |
|
| 24. |
Primary alkyl halide C_(4)H_(9)Br (A) is reacted with alcoholic KOH to give compound (B). The ompound (B) is reacted with HBr to give (C) which is an isomer of compound (A). Write the structures of the compounds (A), (B) and (C). |
Answer» SOLUTION : . .
|
|
| 25. |
Primary alkyl halide (a) C_(4)H_(9)Br was reacted with alcoholic KOH to ive compound (b). Compound (b) was reacted with HBr to give (c) which was an isomer of (a). When (a) was reacted with sodium metal, it have a compound (d) C_(8)H_(18), that was different than the compound when n-butyl bromide was reacted with sodium. give the structural formula of (a) and write the equations for all the reactions. |
Answer» SOLUTION :The TWO primary ALKYL bromides are possible from the molecular formula (a) `C_(4)H_(9)BR` these are: 
|
|
| 26. |
Primary aldehyde on oxidation gives |
|
Answer» Esters |
|
| 27. |
Primary aliphatic or aromatic amines can be distinguished from secondry and tertiary amines by reacting with : |
|
Answer» CHLOROFORM and ALCOHOLIC KOH |
|
| 28. |
Primary aliphatic amines can be distinguished from secondary and tertiary arnmes by heating with |
|
Answer» chloroform and alcoholic KOH |
|
| 29. |
Primary aliphatic amines can be distinguished from secondary and tertiary amines by reaching with |
| Answer» Answer :D | |
| 30. |
Primary alcohols on dehydration give |
|
Answer» alkenes |
|
| 31. |
Primary alcohols can be obtained from the reaction of RMgX with ……………… . |
|
Answer» `CO_2` |
|
| 32. |
Primary alcohol to carboxylic acid |
| Answer» SOLUTION :`CH_3-CH_2-OH UNDERSET(H^+)OVERSET(KMnO_4)(RARR)CH_3COOH` | |
| 33. |
Primary alcohol undergo dehydration by …………………… . |
|
Answer» `E_1` MACHANISM |
|
| 34. |
Primary alcohols can be obtained form the reaction of the RMgX with: |
|
Answer» `HCHO` |
|
| 35. |
Primary alcohol is : |
|
Answer» Propan-3-ol |
|
| 36. |
Primary (1^@) amine group is formed in : |
|
Answer»
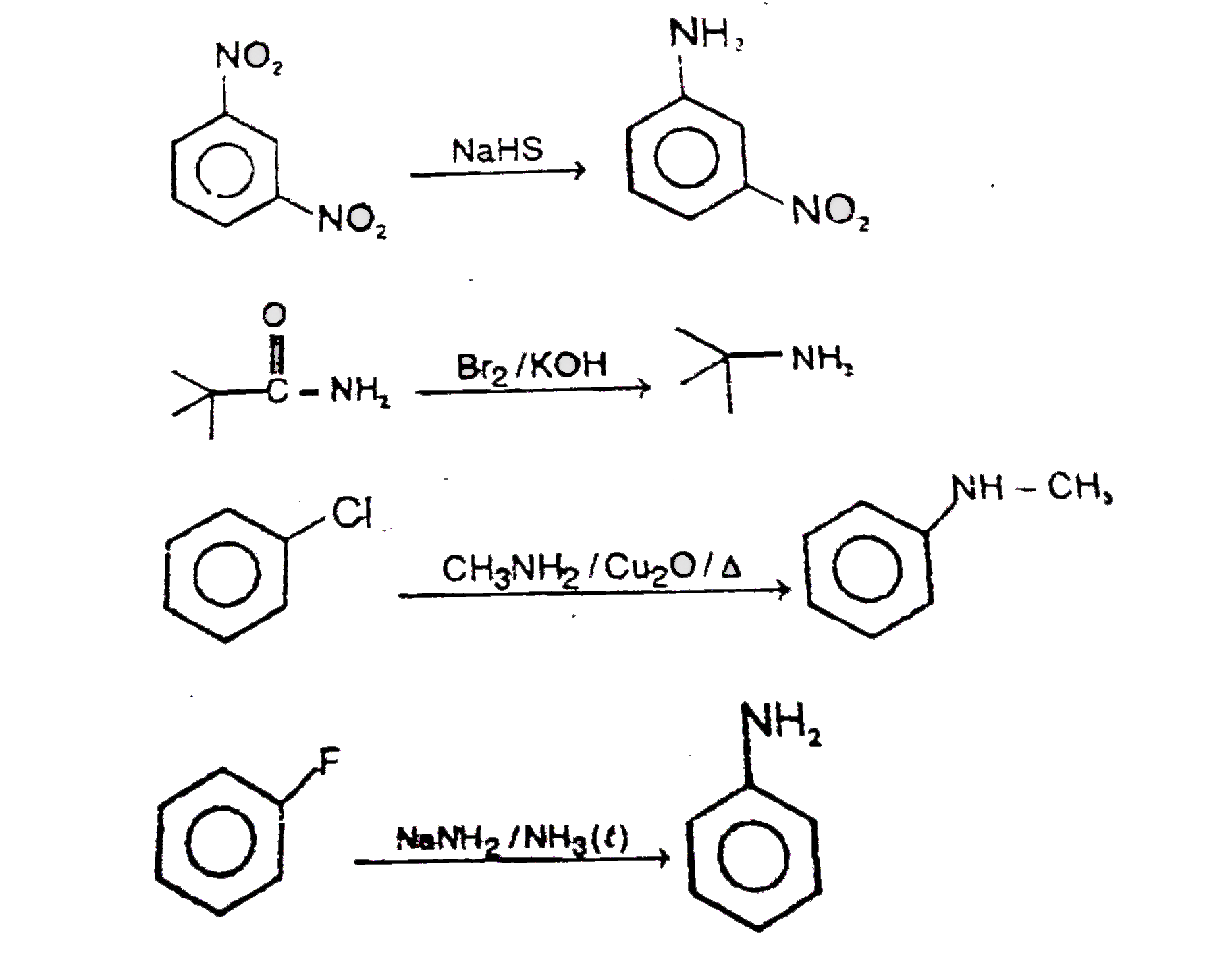
|
|
| 37. |
Preteins when heated with conc. NHO_3 give a yellow colour. This is |
|
Answer» OXIDISING test Protein + conc. `HNO_3 to ` Yellow Colour |
|
| 38. |
Pressure-volume (PV) work done by an ideal gaseous system at constant volume is (where E is internal energy of the system) |
|
Answer» `-DeltaP//P` `DeltaE=q+w. "Now w = "PDeltaV. "for "DeltaV=0` w=0 |
|
| 39. |
Pressure volume (PV) work done by an ideal gas system at constant volume is |
| Answer» Answer :B | |
| 40. |
Pressure remaining the same, the volume of a given mass of an ideal gas increase for every degree centigrade rise in temperature by a definite fraction of its volume at : |
|
Answer» ZERO DEGREE centigrade |
|
| 41. |
Pressure over 1 literof s liquid isgraduallyincreasefrom1 vbar to 100 1 bar under adiabaticcondition. If the final volumeof theliquidis 990 ml , calculate DeltaE andDelta H of theprocess assurimg linearvariation of volumewith pressure. |
|
Answer» <P> Solution :Assuminglinervariationof volumewithpressueW= areaunder the show line = `(10 xx 10^(-6) m^(3)) xx (10 ^(5) N//m^(2)) +(1)/(2)(1000xx 10^(5)N//m^(2))xx (10 xx 10^(-6) m^(3))` ` 1 J + 500 J = 501 J "JOULE"` `DeltaE= 501 "Joule"` `DeltaH=DeltaE+P_(1)V_(2)-P_(1)V_(1)` `=501J +(1001+xx10^(5)N//m^(2))xx(990xx10^(6)m^(3))-(1xx10^(5)N//m^(2))` `xx(1000xx10^(-6)m^(3))` `=501 J +(99xx1001J)-(100J)` `=99.5K J` 
|
|
| 42. |
Pressure over 1000 mlof a liquid is gradually increases from1 barto 1001 bar underadiabiticconditions. Ifthe finalvolumeof the liquid is 990 ml, calculateDeltaU and DeltaH of the process, assuminglinear variationof volume with pressure . |
|
Answer» `=(1)/(2) xx(1000 xx 10 xx 10^(-3))+ 10 xx 10^(-3)` `= 5.01 "bar"-L"" = 501 J` `= 501 J` `Q=0` `Delta U = 501 J""(DELTAU= q +W)` DeltaH = DeltaU + Delta(PV) = DeltaU + (P_(2)V_(2)- P_(1)V_(1)) ` = 501+[1001 xx 990 xx 10^(-3) - 1 xx 10^(-3)xx 10^(-3)] xx 100= 501 + 98999 J` `DeltaH = 99.5 KJ` 
|
|
| 43. |
Pressure over ideal binary liquid mixture containing 10 moles each of liquid A and B is gradually decreased isothermally. If P_A^@ = 200 mm Hg and P_B^@ = 100 mm Hg, find the pressure at which half of the liquid is converted into vapour. |
|
Answer» |
|
| 44. |
Pressure of a mixture of 4 g of O_(2) and 2g of H_(2) confind in a bulb of 1 litre at 0^(@)C is |
|
Answer» 25.215 atm No. of moles of `H_(2)=(2)/(2) =1` Total no. of moles` = 1+0.125= 1.125` `P= (nRT)/(V)= (1.125 XX 0.0821 xx 273)/(1)=25.21 atm` |
|
| 45. |
Pressure of a mixture of 4 g of O_2and 2 g of H_2confined in a bulb of 1 litre at 0^@Cis |
|
Answer» 25.215 ATM |
|
| 46. |
Pressure of a gas in a vessel can be measured by |
|
Answer» Barometer |
|
| 47. |
Pressure of 1 g of an ideal gas A at 27^(@)C is found to be 2 bar. When 2 g of another ideal gas B is introduced in the same flask at same temperature, the pressure becomes 3 bar. Find the relationship between molecular masses. |
|
Answer» Solution :Suppose MOLECULAR masses of A and B are `M_(A) and M_(B)` RESPECTIVELY. Then their number of moles will be `n_(A) =(1)/(M_(A)), n_(B)=(2)/(M_(B))` `P_(A) =2" bar ," P_(A) +P_(B) =3` bar i.e, `P_(B)=1` bar Applying the relation PV =nRT `THEREFORE (P_(A))/(P_(B))=(n_(A))/(n_(B))=(1//M_(A))/(2//M_(B))=(M_(B))/(2M_(A))` or `(M_(B))/(M_(A))=2xx(P_(A))/(P_(B))=2xx(2)/(1)=4` or `M_(B) =4 M_(A)` |
|
| 48. |
Pressure of 0.210 bat at 37^(@)C. The vapour pressure of pure liquid (vapour pressure of water at 37^(@)C=0.198 bar in bar is |
|
Answer» 0.448 Mole fraction of water, `chi_(w)=1.0-0.048=0.952` `0.210=P_(t)^(@)chi_(l) + P_(w)^(@)chi_(w)` `0.210=P_(l)^(@) XX 0.048 + 0.198 xx 0.952` `P_(l)^(@)=(0.210-0.198 xx 0.952)/0.048 =0.448 "BAR"` |
|
| 49. |
Pressure is determined as force per unit area of the surface. The S.I. unit of pressure, pascal, is 1Pa=1Nm^(-2) If mass of air at sea level is 1034gcm^(-2), calculate the pressure in pascal. |
|
Answer» Solution :Pressure is the forece (i.e., weight) acting per UNIT area But weight = MG `therefore"Pressure"="Weight per unit area"=(1034gxx9.8ms^(-2))/(cm^(2))` `=(1034gxx9.8ms^(-2))/(cm^(2))XX(1kg)/(1000g)xx(100CM)/(1m)xx(100cm)/(1m)xx(1N)/("KG ms"^(-2))xx(1Pa)/("1 N m"^(-2))` `=1.01332xx10^(5)Pa` |
|
| 50. |
Pressure cooker reduces cooking time for food because |
|
Answer» Heat is more evenly DISTRIBUTED in the cooking space |
|