Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Predict whether the reaction CO_((g)) + H_2O_((g)) to CO_(2(g)) + H_(2(g)) is spontaneousor not. The standard free energies of formation of CO_((g)), H_2O_((g)) and CO_(2(g)) are – 137.27, –228.6 and –394.38 kJ mole"^(-1) respectivley. |
| Answer» SOLUTION :`Delta_r^@ = - 28.51 KJ` | |
| 2. |
Predict whether the entropy change of the system in each of the following processes is positive or negative. (a) CaCO_(3)(s) to CaO(s)+CO_(2)(g) (b) N_(2)(g)+3H_(2)(g) to 2NH_(3)(g) (c ) N_(2)(g)+O_(2)(g) to 2NO(g) (d) HCl(g)+NH_(3)(g) to NH_(4)Cl(s) (e) 2SO_(2)(g)+O_(2)(g) to 2SO_(3)(g) (f) Cooling of N_(2)(g) from 20^(@)C to -50^(@)C |
|
Answer» Solution :GASEOUS substances generally POSSESS more ENTROPY than solids. So whenever the products CONTAIN more moles of a gas than the reactants, the entropy change is probably positive. And hence, DeltaS is (a) positive (B) negative (c ) small, the sign of DeltaS is impossible to predict (d) negative (e ) negative (f) negative |
|
| 3. |
Predict whether the following reactions occur under standard state conditions : (a) Oxidationof Ag_((S)) by Cl_(2(g)).E_("Ag")^(0) = 0.8V, E_("Cell")^(0) = 1.36V (b) Reduction of Fe^(3+) to Fe^(2+) by Au_((s)) E_(Fe^(3+),Fe^(2+))^(0) = 0.77 V, E_(("Au"))^(2) = 1.4V |
|
Answer» Solution :(a) SINCE THESTANDARD reduction potentialof `Cl_(2(g))` ishigher`(E_(cl^(2+)//cl^(-0))^(0) - = 1.36V) `than thatof `Ag,(E_(Ag^(2+)//Ag)^(0) = 0.8V)`,chlorine will oxidise `Ag_((S))` to `Ag^(+)` . The redox reactionwill be , `2Ag_((s)) + Cl_((2)) to2Ag^(+) + 2Cl_((aq))^(-)` (B) Sincethestandardreductionpotential of Auis higher`(E_(Cl^(2+)//Cl^(-))^(0) = - 1.4V)` thanthat of `Fe^(3+)`, Fe^(2+)`couple`(E_(Fe^(3+),Fe^(2+))^(0) = 0.77V)` , Auwillnot reduce `Fe^(3+)`, to `Fe^(2+)` but Au can oxidise `Fe^(2+)` to `Fe^(3+)` sinceAu has moretendency to accpet electrons. |
|
| 4. |
Predict whether the following reaction (s) is (are) feasible or not (i) Fe+Zn^(2+)toFe^(2+)+Zn,E_(Zn)^(@)=-0.76,E_(Fe)^(@)=-0.44V (ii) Zn+2Ag^(+)toZn^(2+)+2Ag,E_(Zn)^(@)=-0.76V,E_(Ag)^(@)=-0.80V |
| Answer» SOLUTION :(i) `E_(CELL)^(@)=E_(Fe//Fe^(2+))^(@)+E_(Zn^(2+)//Zn)^(@)=+0.44+(-0.76)=-0.32V` LTBRGT (ii) `E_(cell)^(@)=E_(Zn//Zn^(2+))^(@)+E_(Ag^(+)//Ag)^(@)=+0.76+0.80=1.56V`. | |
| 5. |
Predict whether the reaction 2Ag_((s))+Zn_((aq))^(2+)rarr 2Ag_((aq))^(+)+Zn_((s)) is feasible or not. |
|
Answer» Solution :The cell half REACTIONS are `{:("Anode",:,2Ag_((s))rarr2Ag_((AQ))^(+)+2e^(-),E^(@)=0.80V),("Cathode",:,Zn_((aq))^(+)+2e^(-)rarrZn_((s)),E^(@)=-0.763V):}` `E_("cell")^(@)=E_("cathode")^(@)-E_("anode")^(@)` `E_("cell")^(@)=-0.763V-0.80V` `=-.1563V` Since `E_("cell")^(@)`is NEGATIVE, the given reaction is not feasible. |
|
| 6. |
Predict whether a spontaneous reaction will occur: |
|
Answer» When a piece of silver wire is DIPPED in a `ZnSO_4` SOLUTION |
|
| 7. |
Predict whether Ag^(+) can oxidize Pb to Pb^(2+) under standard state conditions. E_(Ag)^(@)=0.799V and E_(Pb)^(@)=-0.126V. |
|
Answer» SOLUTION :(a) (i) Since the standard reduction potential of Ag under standard CONDITIONS is higher `(E_(Ag^(+)//Ag)=0.799V)` than that of `Pb(E_(Pb^(2+)//Pb)^(@)=-0.126V), Ag^(+)` have higher TENDENCY to gain electrons than Pb. (ii) Hence `Ag^(+)` acts as a STRONGER oxidising AGENT and oxidises Pb to `Pb^(2+)` while `Ag^(+)` gets reduced to Ag. The redox reaction will be, `Pb_((s))+2Ag_((aq))^(+) rarr Pb_((aq))^(2+)+2Ag_((s))` |
|
| 8. |
Predict titration curve for titration of Na(2)CO_(3) & HCl |
|
Answer» Solution :`Na_(2)CO_(3)+HClrarrNaHCO_(3)+NaCl` `pH_(1)="Half equivalence point with respect to" 1^(st)` neutralisation =`pK_(a_(2))`(:.solution will be a buffer consisting of equal amount of `CO_(3)^(2-)&HCO_(3)^(-))` `pH_(1^(st eqpt))=(pK_(a_(1))+pK_(a_(2)))/(2)( :."AMPHIPROTIC ANION")` `NaHCO_(3)+NaClrarr H_(2)CO_(3)+NaCl` `pH_(2)`=half equivalence point with respect to `2^(nd)`neutralisation =`pK_(a_(2))` ( :.solution will be a buffer) consisting of equal amount of `HCO_(3)^(-)&H_(2)CO_(3))` `pH_(2^(ndaqpt))=1//2(pK_(a_(1))-LOGC)(":.weak acid")` 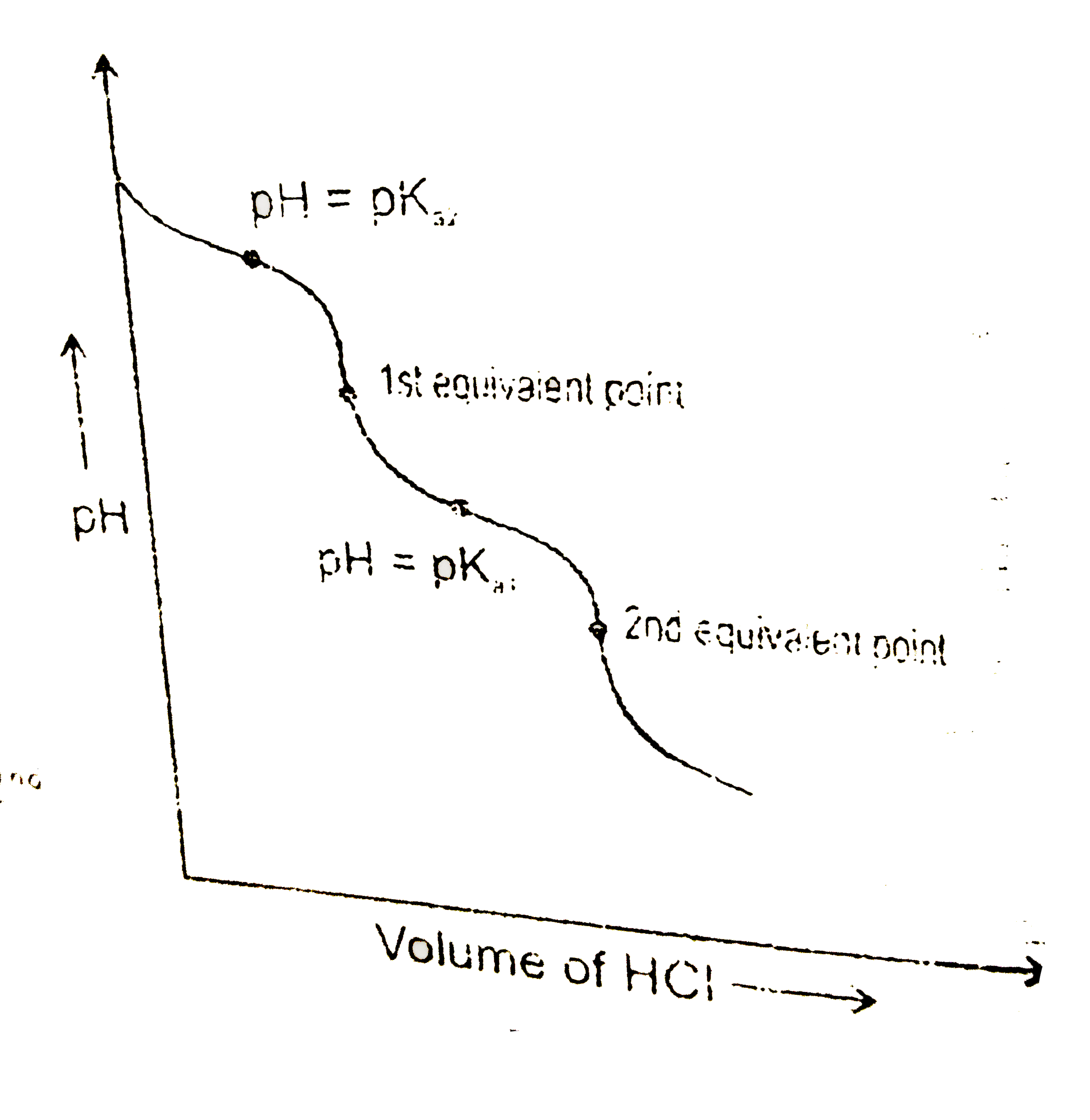
|
|
| 9. |
Predict the type of radioactive decay process that is likely for each of the following nuclides (a) ""_(92)^(228)U (b) ""_(5)^(8)B (c ) ""_(29)^(68)Cu |
| Answer» SOLUTION :(a) `alpha` (B) `BETA^(+)` or K-capture (c ) `beta` EMISSION | |
| 10. |
Predict the temperature for the carbon reduction of Cu_(2)O from the Ellingham diagram. |
| Answer» SOLUTION :500 - 600 K temperature is ideal for the reduction of `Cu_(2)O`. | |
| 11. |
Predict the tendency of undergoing possible mechanism for the athyl, isopropyl and t-butyl halides. (ii) Complete the following equations showing major product with reaction mechanism (S_(N)1,S_(N)2,E1,E2). |
Answer» Solution :When `S_(N)1` mechanism favoured it means that the activation ENERGY for this path is less than that for the `S_(N)2`. Practically, more stable the lon, the lower is the activation energy and more favoured will be the mechanism. As we KNOW, the order of stability of carbonium ion is `1^(@)LT2^(@)lt3^(@)`and hence the TENDENCY for the `S_(N)1` mechanism to operate will be `t-BuXgt" ios PROPYL "XgtEtX`. The tendency willbe reverse for the `S_(N)2` mechanism, 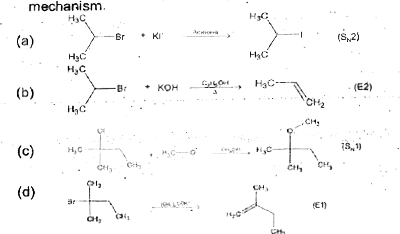
|
|
| 12. |
Predict the structure of MgO crystal and coordination number of its cation in which the cation and anion radii are equal to 65 pm and 140 pm respectively. |
|
Answer» Solution :Radius of cation (`Mg^(2+)`) = 65 pm. Radius of anion (`O^(2-)`) = 140 pm Radius ratio, `(r_(+))/(r_(-))=(r_(Mg^(2+)))/(r_(O^(2-)))=(65)/(140) =0.464` The radius ratio lies between 0.414-0.732. HENCE the EXPECTED structure is octahedral with coordination number of SIX. |
|
| 13. |
Predict the structure of propane -1,2 diol |
|
Answer» `CH_(2)(OH) -CH_(2)CH_(2)OH` |
|
| 14. |
Predict the state of the solute in the solution in the following situations : When 'I' is found to be more than one. |
|
Answer» SOLUTION :When .i. is FOUND to be more than ONE. The SOLUTE is DISSOCIATED in the solution. |
|
| 15. |
Predict the state of the solute in the solution in the following situations : When 'I' is found to be less than one. |
|
Answer» SOLUTION :When .i. is FOUND to be LESS than ONE. The SOLUTE is associated in the solution. |
|
| 16. |
Predict the species produced during heterolytic fission of the following: |
|
Answer»
|
|
| 17. |
Predict the sign/value of DeltaS^(@) for the following reaction2H_(2)S_((g))+3O_(2(g))rarr2H_(2)O_((g))+2SO_(2(g)) |
|
Answer» `+ve` |
|
| 18. |
Predict the sign of DeltaS in the following processes. Give reasons for your answer. (a) N_(2(g))+3H_(2(g))rarr2NH_(3(g)) (b) CO_(2(g))rarrCO_(2(s)) |
|
Answer» Solution :`N_(2(G))+3H_(2(g))rarr2NH_(3(g))` In the reaction, 4 moles of gaseous reactants form 2 moles of gaseous products `(DELTA n LT 0)`. Therefore disorder decreases and hence entropy decreases, `DeltaS lt 0.` |
|
| 19. |
Predict the sign of DeltaS in the following processes. Give reasons for your answer : N_(2_((g)))+3H_(2_((g))) to 2NH_(3_((g))) |
|
Answer» Solution :`N_(2_((g)))+3H_(2_((g))) to 2NH_(3_((g)))` In the reaction, 4 moles of GASEOUS reactants form 2 molesgaseous products `(Deltan lt 0)`. Therefore disorder decreases and HENCE entropy decreases, `DeltaS lt 0.` |
|
| 20. |
Predict the sign of DeltaS in the following processes. Give reasons for your answer : Fe_(2)O_(3_((s)))+3H_(2_((g))) to 2Fe_((s))+3H_(2)O_((g)) |
|
Answer» SOLUTION :`Fe_(2)O_(3_((s)))+3H_(2_((g)))to 2Fe_((s))+3H_(2)O_((g))` In the REACTION number of moles of gaseous reactants and PRODUCTS are same, hence `DeltaS=0` |
|
| 21. |
Predict the sign of Delta S forthe following process: H_(2)O_((g)) rarr H_(2)O_((l)) |
| Answer» SOLUTION :NEGATIVE or - ve. | |
| 22. |
Predict the shape of ClF_3 on the basis of VSPER theory. |
| Answer» Solution :`ClF_3` has T- shapestructure with 3 BOND PAIRS and 2 lone pairs. | |
| 23. |
Predict the shape and the asked angle (90^(@)" or more or less") in the following case SO_(3)^(2-) and the angle O-S-O. |
Answer» SOLUTION :PYRAMIDAL : 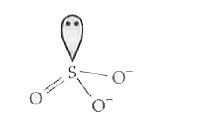 ANGLE : `O-S-O` more than `90^(@)` |
|
| 24. |
Predict the products of the following reactions : (i) CH_(3)-CH_(2)-CH_(2)-O-CH_(3)+HBr rarr (ii) (iv) (CH_(3))_(3)C-OC_(2)H_(4)overset(HI)rarr |
|
Answer» Solution :(i) Both the alkyl groups ATTACHED to the oxygen atom are primary, therefore, attack of `Br^(-)` ion occurs on the smaller methyl GROUP giving propan-1-ol and bromomethane as the products. `underset("N-Propyl methyl ether")(CH_(3)-CH_(2)-CH_(2)-O-CH_(3))+HBr overset(373K)rarr underset("Propan-1-ol")(CH_(3)-CH_(2)-CH_(2)-OH)+underset("Bromomethane")(CH_(3)-Br)` (ii) `C_(6)H_(5)-O` bond has some double bond character DUE to resonance and hence is STRONGER than `O-C_(2)H_(5)` bond. Therefore, the cleavage takes place at the weaker `O-C_(2)H_(5)` bond to yield phenol and bromoethane as the products.  (iii) `-OC_(2)H_(5)` group is activating as well as o-, p - directing. Therefore, nitration of ethoxybenzene will give a mixture of 2- and 4- ethoxynitrobenzene in which 4 - nitroethoxybenzene dominates due to steric hindrance at 2 - position.  (iv) The reaction follows `S_(N)1` MECHANISM leading to the formation of tert - butyl iodide and ethanol. tery - butyl carbocation is much more stable than ethyl carbocation. Step 1 : 
|
|
| 25. |
Predict the products of the following reactions : (i) CH_(3) - underset(CH_(3))underset(|)C=O overset((i) H_(2)N-NH_(3))underset((ii)KOH//"Glycol",Delta) to ? (ii) C_(6)H_(5)-CO-CH_(3) overset(NaOH//I_(2)) to ?+ ? (iii) CH_(3)COONa overset(NaOH//CaO)underset(Delta) to ? |
|
Answer» Solution :(i) `CH_(3)-underset(CH_(3))underset(|)C = O overset((i) H_(2)N, NH_(2))underset((ii) KOH//"GLYCOL",Delta) to underset("Propane")(CH_(3)CH_(2)CH_(3))` (ii) `C_(6)H_(5) - CO - CH_(3) overset(NaOH//I_(2))to underset("Sod.benzoate")(C_(6)H_(5)COONa) + underset("Iodoform")(CHI_(3))` (III) `CH_(3)COONa overset(NaOH//CaO)underset(Delta)to underset("METHANE")(CH_(4))` |
|
| 26. |
Predict the products of the following reactions : (iii) R-CH=CH-CHO+NH_2-overset(O)overset(||)(C )-NH-NH_2overset(H^+)(rarr) (iv) |
Answer» Solution : (ii) `NH_2` attached to `NH` is more nucleophilic than `NH_2`, attached to `C=O`group, therefore, reaction OCCURS through `NH_2` attached to NH to GIVE the CORRESPONDING semicarbozone, i.e., `R-CH=CH-CH= O + H_2 NNHCONH_2 RARR R -CH=CH-CH= NNHCONII_2 + H_2O` (IV) 
|
|
| 27. |
Predict the products of the following reactions CH_3-CH_2-CH_2-O-CH_3+HBr rarr |
| Answer» SOLUTION :`CH_3-CH_2-CH_2-OH` and `CH_3Br` | |
| 28. |
Predict the products of the following reactions. |
|
Answer» |
|
| 29. |
Predict the products of the following reactions. |
|
Answer» |
|
| 30. |
Predict the products of the following reactions |
Answer» SOLUTION :
|
|
| 31. |
Predict the products of the following reactions. |
|
Answer» |
|
| 32. |
Predict the products of the following reactions. |
|
Answer» |
|
| 33. |
Predict the products of the following reactions |
Answer» SOLUTION :
|
|
| 34. |
Predict the products of electrolysis of the following: (i) An aqueous solution of AgNO_(3) with silver electrodes (ii) An aqueous solution of AgNO_(3) with platinum electrodes. (iii) A dilute aqueous solution of H_(2)SO_(4) with platinum electrodes. (iv) An aqueous solution of CuCl_(2) with platinum electrodes. (Given E_(Ag^(+)//Ag)^(@)=+0.80V, E_(Cu^(2+)//Cu)^(@)=+0.34V) |
|
Answer» Solution :(i) Electrolysis of aqueous solution of `AgNO_(3)` with silver electrodes. `AgNO_(3)(s)+aqtoAg^(+)(aq)+NO_(3)^(-)(aq)` `H_(2)OhArrH^(+)+OH^(-)` At cathode: `Ag^(+)` ions have lower discharge potential than `H^(+)` ions. Hence, `Ag^(+)` ions will be deposited as Ag in preference to `H^(+)` ions. Alternatively, we have stanard reduction potential potentials as `Ag^(+)(aq)+e^(-)toAg(s),E^(@)=+0.80V` `H^(+)(aq)+e^(-)to(1)/(2)H_(2)(g),E^(@)=0.00V` As `Ag^(+)` ions hae HIGHER standard reduction potential than that of `H^(+)` ions, hence `Ag^(+)` ions will be reduced more easily and deposited as Ag. At anode: As Ag anode is attached by `NO_(3)^(-)` ions, Ag of the anode will dissolve to form `Ag^(+)` ions in the solution. `AgtoAg^(+)+e^(-)` Alternatively, out of the three possible oxidation reactions occurring at the anode, i.e., `AgtoAg^(+)+e^(-),2OH^(-)toH_(2)O+(1)/(2)O_(2)+e^(-) and NO_(3)^(-)toNO_(3)+e^(-)`, Ag has highest oxidation potential. hence, Ag of anode is oxidized to `Ag^(+)` ions which pass into the solution. (ii) Electrolysis of aqueous solution of `AgNO_(3)` using platinum electrodes. At cathode: Same as above. ltBrgt At anode: As anode is not attackable, out of `OH^(-) and NO_(3)^(-)` ions, have lower discharge potential. hence, `OH^(-)` ions will be DISCHARGED in preference to `NO_(3)^(-)` ions, which then decompose to give out `O_(2)`. `OH^(-)(aq)toOH+e^(-),4OHto2H_(2)O(L)+O_(2)(g)` (iii) Electrolysis of dilute `H_(2)SO_(4)` with platinum electrodes. `H_(2)SO_(4)(aq)to2H^(+)(aq)+SO_(4)^(2-)(aq)` `H_(2)OhArrH^(+)+OH^(-)` At cathode: `H^(+)+e^(-)toH,H+HtoH_(2)(g)` At anode: `OH^(-)toOH,e,4OHto2H_(2)O+O_(2)(g)` Thus, `H_(2)` is LIBERATED at the cathode and `O_(2)` at the anode. (iv) Electrolysis of aqueous solution of `CuCl_(2)` with platinum electrodes. `CuCl_(2)(s)+aqtoCu^(2+)(aq)+2Cl^(-)(aq)` `H_(2)OhArrH^(+)+OH^(-)` At cathode: `Cu^(2+)` ios will be reduced in preference to `H^(+)` ions `Cu^(2+)+2e^(-)toCu` At anode: `Cl^(-)` ions will be oxidized in preference to `OH^(-)` ions `Cl^(-)toCl+e^(-),Cl+CltoCl_(2)(g)` Thus, Cu will be deposited on the cathode and `Cl_(2)` will be liberated at the anode. |
|
| 35. |
Predict the products of the following reactions |
Answer» SOLUTION :
|
|
| 36. |
Predict the products of electrolysis obtained at the electrodes in each case when the electrodes used are of platinum : (i) An aqueous solution of AgNO_3. (ii) An aqueous solution of H_2SO_4. |
|
Answer» Solution :(i) `AgNO_(3)(aq) to Ag^(+) (aq) + NO_(3)^(-) (aq)` `H_(2)O (l) to H^(+) (aq) + OH^(-) (aq)` At cathode: `Ag^(+) (aq) + E^(-) to O_(2) + 2H^(+) (aq) +4e^(-)` `H_(2)O(l) to H^(+) (aq) + OH^(-) (aq)` At cathode: `2H^(+) (aq) + 2e^(-) to O_(2) + 2H^(+) (aq) + 4e^(-)` or `2H_(2)O (l) to O_(2) + 4H^(+) (aq) + 4e^(-)` Hydrogen gas is produced at the cathode and oxygen at the anode. |
|
| 37. |
Predict the products of the following reactions. |
|
Answer» |
|
| 38. |
Predict the products of electrolysis of an aqueous solution of CuBr_(2) . |
|
Answer» Solution :The probable reactions at CATHODE are : `(i) CU^(2+) (aq) + 2e^(-) to Cu (s) , "" E^(Theta) = 0.34 V` (ii) `2 H_(2) O (l) + 2e^(-)to H_(2) (g) + 2 OH^(-) (aq) , E^(Theta) = -0.83` V Since the reduction potential of `Cu^(2+)` ions is higher than that of water , copper will be reduced preferably at cathode , The probable reactions at anode are : `2 Br^(-) (aq) to Br_(2) (g) + 2e^(-) "" E^(Theta) = 1.08 V` `H_(2) O(l) to (1)/(2) O_(2) (g) + 2 H^(+) + 2e^(-) E^(Theta) = 1.23` V Since the reduction potential of `Br^(-)` is LESS than water , it will be readily oxidised to `Br_2` at anode. Therefore , the reactions are : At cathode : `Cu^(2+) (aq) + 2 e^(-) to Cu (s)` At anode : `2 Br^(-) (aq) to Br_(2) (g) + 2 e^(-)` |
|
| 39. |
Predict the products of electrolysis obtained at the electrodes in each case by using platinum electrodes : (i) An aqueous solution of AgNO_(3) using platinum electrodes (ii) An aqueous solution of CuSo_(4) using attackable electrodes. |
|
Answer» SOLUTION :(i)Ag is DEPOSITED at cathode. `O_(2)` is evolved at anode. (II)`Cu^(2+)` IONS are deposited at cathode. Equivalent amount of Cu from anode goes into the solution. |
|
| 40. |
Predict the products of electrolysis in each of the following: (i) An aqueous solution of AgNO_3 with silver electrodes. (ii) An aqueous solution of AgNO_3 with platinum electrodes. (iii) A dilute solution of H_2SO_4 with platinum electrodes. (iv) An aqueous solution of CuCl_(2) , with platinum electrodes. |
|
Answer» Solution :Electrolysis of AQUEOUS solution of `AgNO_(3)` with silver electrodes: The ions produced by the dissociation are as under: `AgNO_(3) (s) + aq to Ag^(+) + NO_(3)^(-3) (aq)` `H_(2)O At anode : As Ag anode is attacked by `NO_(3)^(-)`ions, Ag of the anode will dissolve to form `Ag^+` ions in the solution. `Agto Ag^(+) + E^(-)` (ii) Electrolysis of aqueous solution of `AgNO_3` using platinum electrodes : At cathode : Same as in (i). At anode : Out of `OH^(-)`and `NO_3^(-)`ions, `OH^(-)`ions have lower discharge potential. Hence, `OH^(-)` ions will be discharged in preference to `NO_3^(-)` ions. OH ions decompose to give out `O_(2)`as under: `OH^(-)(aq) to OH + e^(-), 4OH to 2H_(2)O (l) +O_(2)(g)` (III) Electrolysis of dilute `H_(2)SO_(4)` with platinum electrodes: `H_(2)SO_(4)` and `H_(2)O` ionise as under: `H_(2)SO_(4)(aq) to 2H^(+) (aq) + SO_(4)^(2-) (aq)` `H_(2)O At cathode: `OH^(-) to OH +e^(-), 4OH to 2H_(2)O + O_(2)(g)` (because of lower discharge potential of `OH^(-)`) (iv) Electrolysis of aqueous solution of `CuCl_2` with platinum electrodes : `CuCl_(2)(s) + aq to Cu^(2+) (aq) + 2Cl^(-)(aq)` `H_(2) O `Cu^(2+) + 2e^(-) to Cu` At anode: `Cl^(-)` ions will be oxidised in preference to `OH^(-)` ions. `Cl^(-) to Cl + e^(-), Cl + Cl to Cl_(2)(g)` THUS, Cu will be deposited on the cathode and `Cl_2` will be liberated at the anode. |
|
| 41. |
Predict the products of electrolysis in each of the following: (i) An aqueous solution of AgNO_(3) with silver electrodes. (ii) An aqueous solution of AgNO_(3) with platinum electrodes. |
|
Answer» Solution :(i) Electrolysis of aqueous solution of `AgNO_(3)` using silver electrodes : `AgNO_(3)(s)overset("water")toAg^(+)(aq)+NO_(3)^(-)(aq)` `H_(2)OhArrH^(+)OH^(-)` cathode. `Ag^(+)` ions have lower discharge potential than `H^(+)` ions. Hence `Ag^(+)` ions will be deposited as silver (in preference to `H^(+)` ions) At ANODE. Since silver electrode is attacked by `NO_(3)^(-)` ions, Ag anode will DISSOLVE to form `Ag^(+)` ions in the solution. `Ag""toAg^(+)+e^(-)` (ii) Electrolysis of aqueous solution of `AgNO_(3)` using platinum electrodes : At cathode. Same as above. At anode. Since silver is not attacked, out of `OH^(-)` and `NO_(3)^(-)` ions, `OH^(-)` ions have lower discharge potential and hence `OH^(-)` ions will be discharged in preference to `NO_(3)^(-)`. The `OH^(-)` will decompose to give `O_(2)`. `OH^(-)(aq)toOH+e^(-)` `4OHto2H_(2)O(l)+O_(2)(g)` |
|
| 42. |
Predict the product(s) of (A) Friedel-Crafts acylation of anisole (methoxybenzene) with acetyl chloride in the presence of one equivalent of AlCl_(3) follwed by H_(2)O. (B) Friedel-Crafts alkylation of a large excess of ethylbenzene with chloromethane in the presence of AlCl_(3). |
|
Answer» |
|
| 43. |
Predict the products in the given reaction 2HCHO overset(conc NaOH)(to) |
|
Answer» `HCOOH + CH_3O^(-) NA^(+)` `2HCHO underset(Delta)overset(conc NAOH)(to) CH_3OH+HCOO^(-) Na^(+)` |
|
| 44. |
Predict the products: (i) CH_(3)CH_(2)OH overset(K_(2)Cr_(2)O_(7))underset(H_(2)SO_(4)) to A overset(SOCl_(2)) to B overset(NH_(2)) to C overset(NaOBr) to D (ii) CH_(3)CH_(2)Cl overset(KCN) to A overset(OH^(-))underset(H_(2)O_(2)) to B overset(P_(2)O_(5)) to C (iii) CH_(3)COOHoverset(C_(2)H_(5)OH)underset("conc"H_(2)SO_(4)) to A overset(CH_(3)MgBr)underset(H^(+)//H_(2)O) to B overset(CH_(3)MgBr)underset(H_(2)O//H^(+)) to C |
|
Answer» Solution :The PRODUCTS A, B, C, D are identified with the help of the following reactions : (i)`CH_(3)CH_(2)OH overset(K_(2)Cr_(2)O_(7))UNDERSET(H(2)SO_(4)) to CH_(3)COOH overset(SOCl_(2))to CH_(3)COCl overset(NH_(3))to CH_(3)CONH_(2) overset(NaOBr)to CH_(3)NH_(2)` (ii) `CH_(3)CH_(2)CL overset(KCN) to underset(A)(CH_3)CH_(2)C-= N overset(OH^(-))underset(H_(2)O_(2)) to CH_(3)CH_(2) - underset(B)overset(O)overset(|\)C-NH_(2) overset(P_(2)O_(3))underset("Dehydration")to underset( C)(CH_(3)CH_(2)C -= N + H_(2)O)` (iii) `CH_(3)COOH overset(C_(2)H_(5)OH)underset("conc "H_(2)SO_(4)) to underset(A)(CH_(3)COOC_(2)H_(5)) overset(CH_(3)MgBr)underset(H_(2)O//H^(+)) to CH_(3) -underset(B)overset(O)overset(||)C-CH_(3) overset(CH_(3)MgBr)underset(H_(2)O//H^(+)) to CH_(3) - underset(CH_(3))underset(|)overset(OH)overset(|)C-CH_(3)` |
|
| 45. |
Predict the products in the following sequence of reactions. |
Answer» SOLUTION :
|
|
| 46. |
Predict the products formed when cyclohexanecarbaldehyde reacts with following reagents : (i) PhMgBr and then H_3O^(+) "" (ii) Tollens' reagent (iii) Semicarbazide and weak acid "" (iv) Excess ethanol and acid (v) Zinc amalgam and dilute hydrochloric acid. |
|
Answer»
 
|
|
| 47. |
Predict the products formed when cyclohexanecarbaldehyde reacts with following reagents: (i) PhMgBr and then H_(3)O^(+) (ii) Tollens' reagent (iii) Semicarbazide and weak acid (iv) Excess ethanol and acid (v) Zinc amalgam and dilute hydrochloric acid. |
Answer» SOLUTION :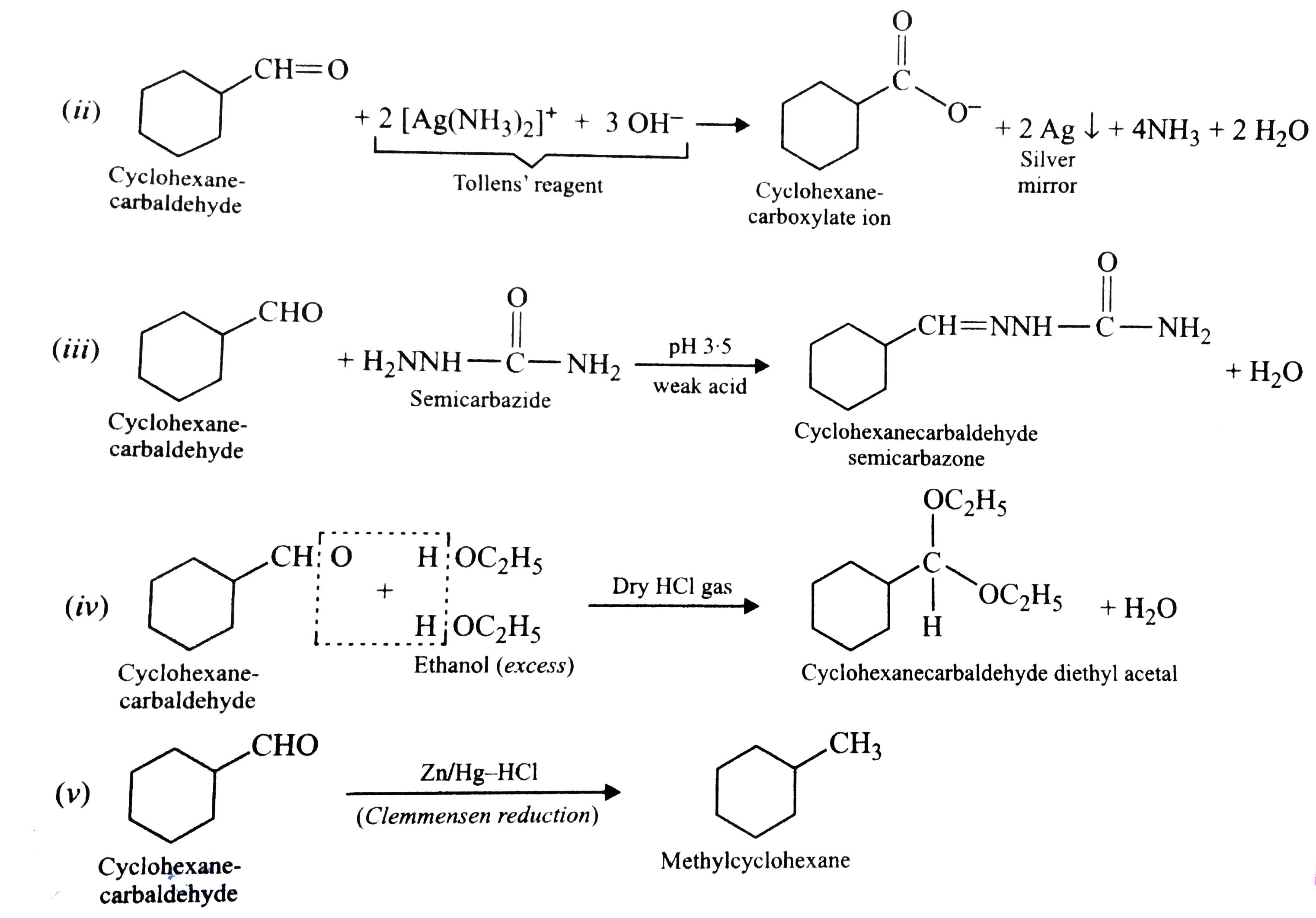 . .
|
|
| 48. |
Predict the products formed when cyclohexanecarbaldehyde reacts with following reagents: (a) PhMgBr and then H_(3)O^(+). (b) Tollens' reagent. |
Answer» SOLUTION : 
|
|
| 49. |
Predict the products expexted of the following reactions: |
Answer» SOLUTION :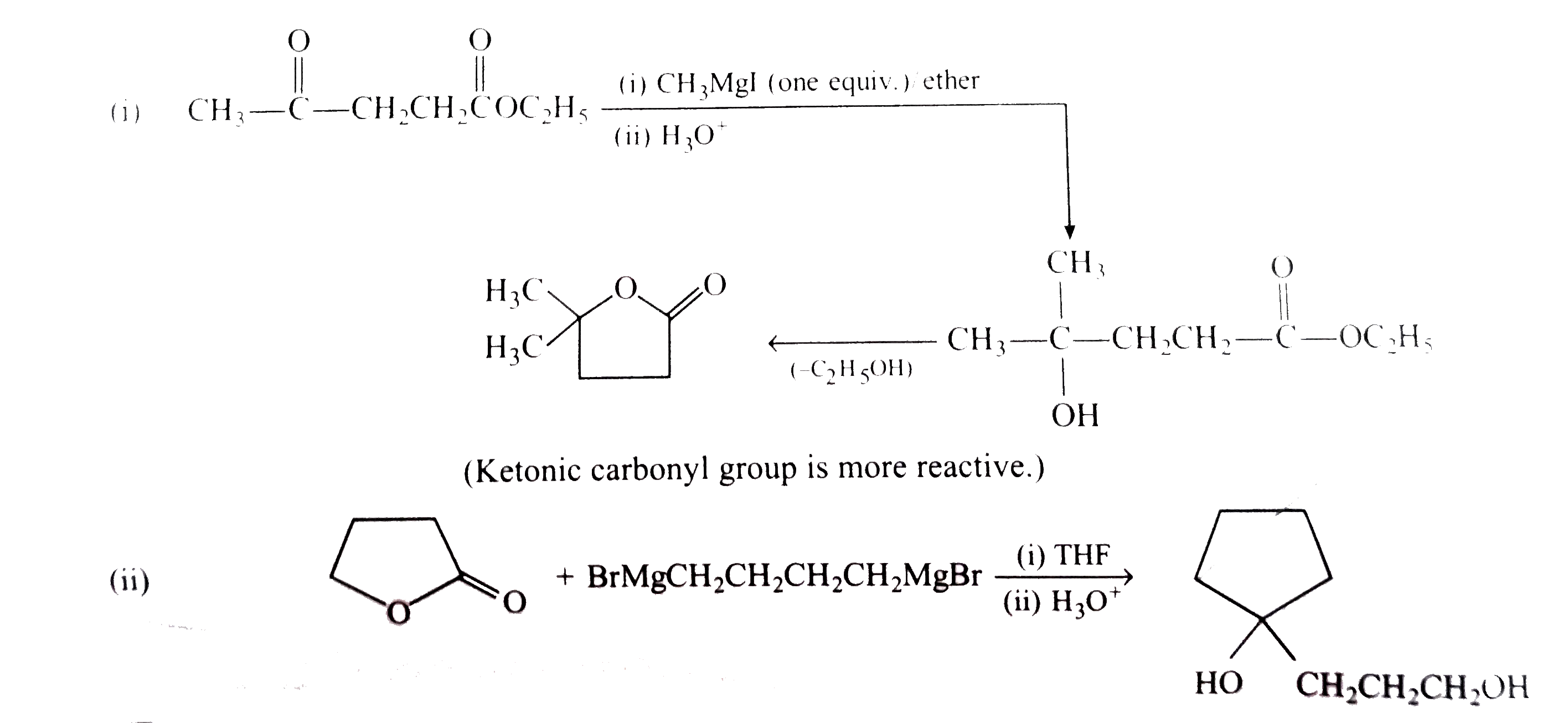
|
|
| 50. |
Predict the products formed in the following cases: (i) (A) reacts with PhMgBr and then hydrolysed. (ii) (A) reacts with hydrazine and is then heated with KOH and ethylene glycol. |
|
Answer» SOLUTION :(i) `underset((A))underset(CH_(3))underset(|)underset(CH_(2))underset(|)(CH_(3)CH_(2)C)=O+PhMgBr to CH_(3)CH_(2)-underset(CH_(3))underset(|)underset(CH_(2))underset(|)overset(Ph)overset(|)C-OMgBr overset(H_(2)O)to underset("3-Phenylpentane-3-ol")(CH_(3)CH_(2)-underset(CH_(3))underset(|)underset(CH_(2))underset(|)overset(Ph)overset(|)C-OH)` (ii) `underset((A))(CH_(3)CH_(2)COCH_(2)CH_(3)) underset({:(KOH//"Ethylene glycol"),(" (Wolff-Kishner)"),("reduction"):})overset(NH_(2)NH_(2))to underset(N-"PENTANE")(CH_(3)CH_(2)CH_(2)CH_(2)CH_(3))` |
|