Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Predict the product Z in the following series of reactions Ethanoic acid overset(PCl_5)(to) X underset("anhydrous"AlCl_3)overset(C_6H_6)(to)Y underset((ii) H_3O^+)overset((i) CH_3MgBr)(to) Z |
|
Answer» `(CH_3)_2C (OH)C_6H_5` 
|
|
| 2. |
Predict the product Z in the following series of reactions Ethanoic acid overset(PCl_5)rarrX underset("Anhydrous "AlCl_3)rarrYunderset((ii)H_3O^+)overset((i)CH_3MgBr)rarrZ |
|
Answer» `(CH_3)_2C(OH)C_6H_5` |
|
| 3. |
Predict the product reaction of aniline with bromine in non-polar solvent such as CS_(2). |
Answer» SOLUTION :In non-polar solvent, the resonance STRUCTURE involving separation of +ve and -ve CHARGES are not stabilized by dipole-dipole interaction with the non-polar solvent. As a RESULT activating EFFECT of the `-NH_(2)` is reduced and hence monosubstitution occus only at o- and p-positions giving amixture of 2-bromoaniline (minor) and 4-bromoaniline (major). 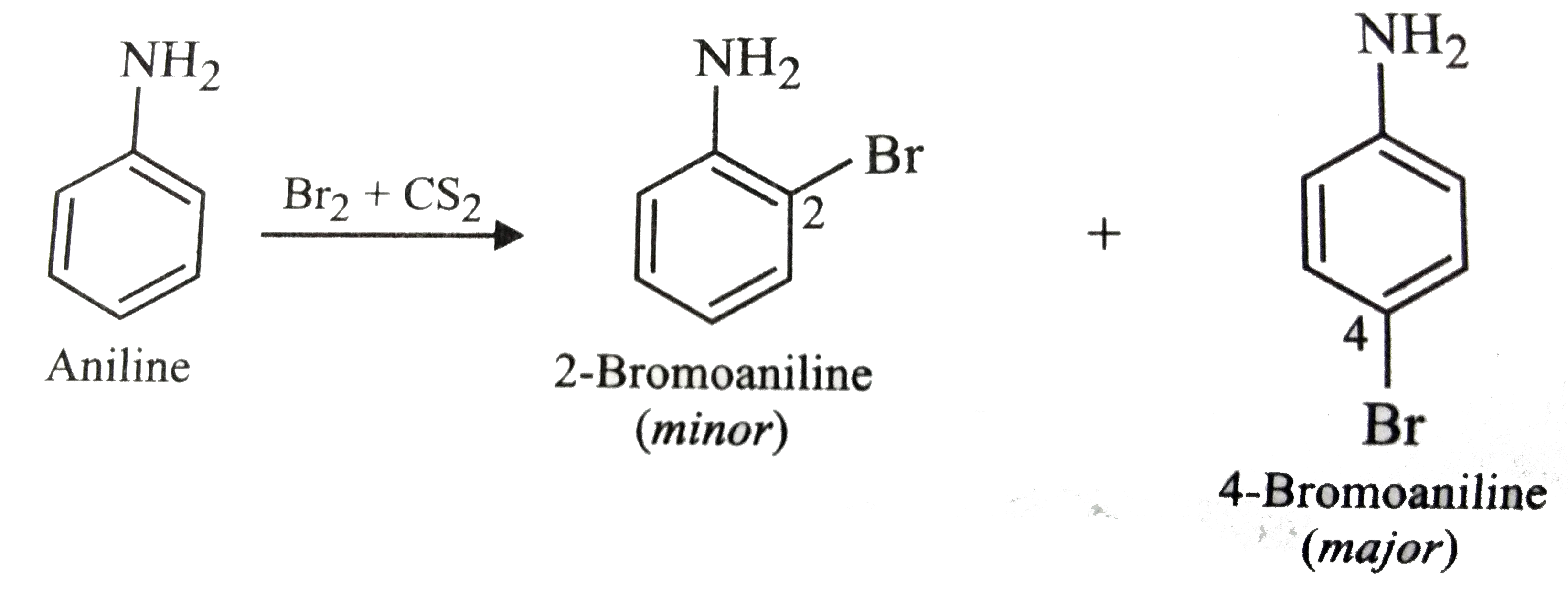
|
|
| 4. |
Predict the product X in the sequence of the reaction HC -= CH underset(H_2SO_4)overset(HgSO_4)(to) A underset(NaOH)overset(40%)(to) X |
|
Answer» `CH_3COONa` |
|
| 5. |
Predict the product when calcium ethanoate and calcicum methanote are dry distilled. Explain the reaction. |
Answer» SOLUTION :The PRODUCT OBTAINED is ETHANOL. 
|
|
| 6. |
Predict the product R in the reaction |
|
Answer» 1-Methylcyclopentanol 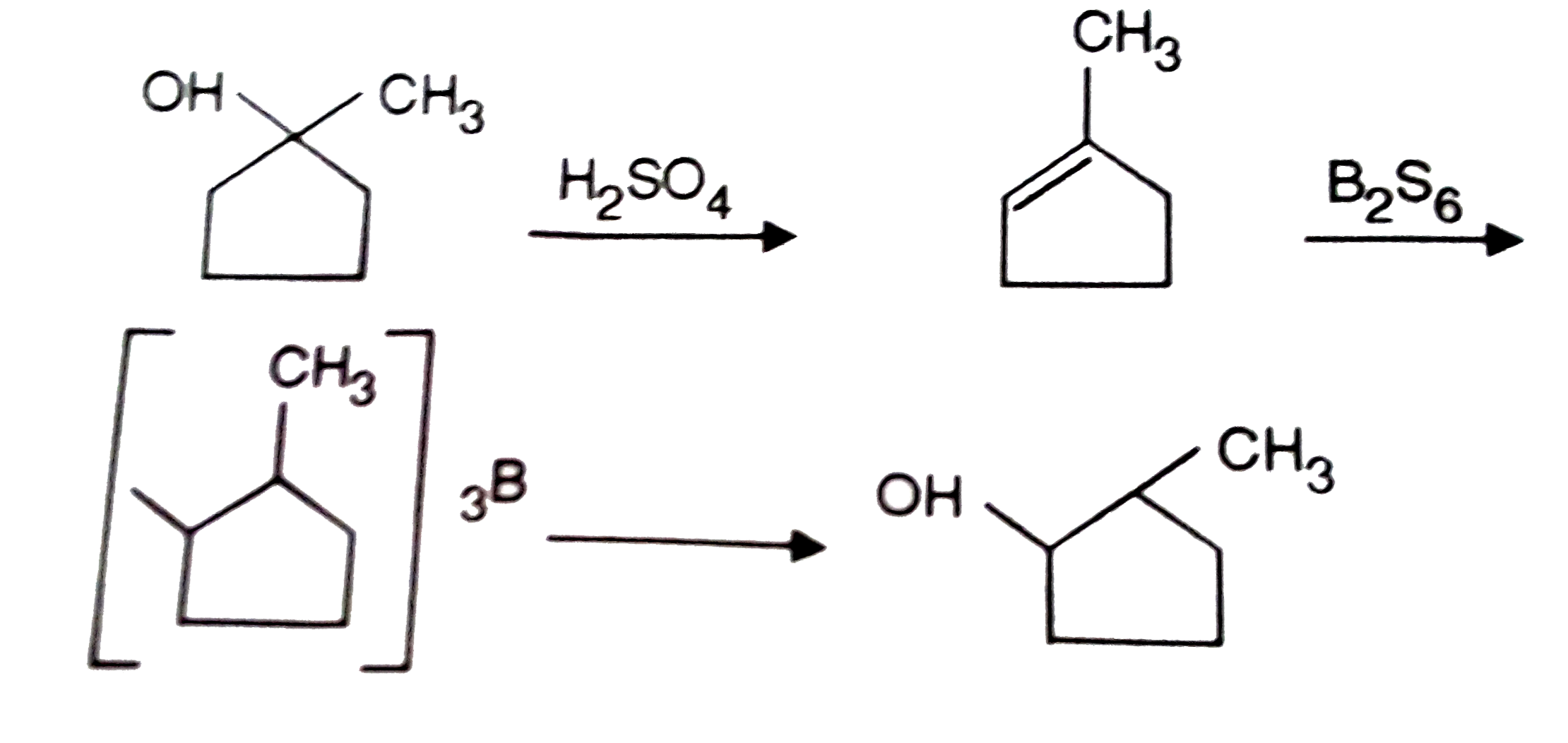
|
|
| 7. |
Predict the product of the reaction of propanal with each of the following (a) Lithium aluminum hydride (b) Sodium borohydride (c) Hydrogen(nickel catalyst) (d) Methylmagnesium iodide, followed by dilute acid. (e) Sodium acetylide, followed by dilute acid (f) Phenyllithium, followed by dilute acid (g) Methanol containing dissolved hydrogen chloride (h) Ethylene glycol, p-toluenesulfonic acid, benzene (i) Aniline (C6H5NH2) (j) Dimethylamine, p-toluenesulfonic acid, benzene (k) Hyderoxylamine (l) Hydrazine (m) Product of part(l) heated in triethylene glycol with sodium hydroxide (n) p- Nitrophenylhydrazine (o) Semicarbazide (p) Ethylidenetriphenylphosphorane [(C_(6) H_(5) )_(3) overset(+)(P)–bar(ddotC)HCH_(3) ] (q) Sodium cyanide with addition of sulfuric acid (r) Chromic acid |
|
Answer» <P> Solution :(a) `CH_(3)—CH_(2)—CH_(2)—OH"" "(b)" CH_(3)—CH_(2)—CH_(2)—OH"""(c)" CH_(3)—CH_(2)—CH_(2)—OH`(d) `CH_(3)—CH_(2)—overset(OH)overset(|)(CH)—CH_(3)"""(e)" CH_(3) —CH_(2)—overset(OH)overset(|)(CH)- C equiv CH"" "(F)" CH_(3) —CH_(2)—overset(OH)overset(|)(CH)— PH` (g)` CH_(3)—CH_(2)—underset(OCH_(3))underset(|)(CH) —OCH_(3)""" (h)"` `(i)CH_(3)—CH_(2)—CH= N —Ph""" (j)" CH_(3) —CH_(2)—overset(OH)overset(|)(CH)—underset(CH_(3))(N)—CH3""" (k)"` C—C—C=N—OH (l)` CH_(3)—CH_(2)—CH=N—NH_(2)"""(m)" CH_(3)—CH_(2)—CH_(3_"" "(n) P-Nitro PHENYL HYDRAZONE"` (o) semi carbazane`"" "(p)" CH_(3)—CH_(2)—CH=CH—CH_(3)""" (q)" CH_(3)—CH_(2)—overset(OH)overset(|)(CH)—CN ` (r)` CH_(3)—CH_(2)—CO_(2)`H 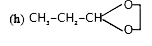
|
|
| 8. |
Predict the product of the reaction CH_3-CH_2-CH_2 - O-CH_3 + HBr to |
| Answer» Solution :`CH_3 CH_2 CH_2 O CH_3 + HBR OVERSET(373K)to underset("Propan-1-ol")(CH_3CH_2CH_2OH) + underset("Bromomethane")(CH_3Br)` | |
| 9. |
Predict the product of the reaction (CH_(3))_(2)CHCHClCH_(2)CH_(3)+KOH overset(alc) to |
|
Answer» `(CH_(3))C=CHCH_(2)CH_(2)CH_(3)` `(CH_(3))CHCHCl CH_(2)CH_(2)CH_(2)CH_(3) +KOH OVERSET(alc) to (CH_(3))_(2)C= CHCH_(2)CH_(2)CH_(3)+KCl+H_(2)O` |
|
| 10. |
Predict the product of the reaction (CH_3)_3C-OC_2H_5 overset(HI)to |
| Answer» Solution :`(CH_3)_3 C - OC_2 H_5 + HI to CH_2 - UNDERSET(CH_3)underset(|)OVERSET(CH_3)overset(|)C- I + CH_3 CH_2 OH` | |
| 11. |
Predict the product of the following reactions: |
Answer» SOLUTION :
|
|
| 12. |
Predict the product of the following reaction CH_(3)COONa underset(Delta)overset(NaOH//CaO)to? |
Answer» Solution :SODIUM salts of carboxylic acids on heating with soda-lime `(NaOH//CaO)` undergo DECARBOXYLATION to GIVE an alkane with ONE carbon atom less than the starting carboxylic acids salts. 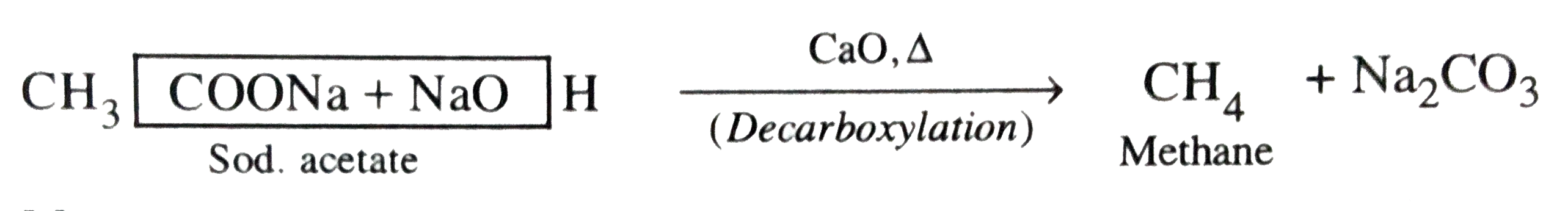 . .
|
|
| 13. |
Predict the product of the following reaction CH_(3)NO_(2)+C_(6)H_(5)CHO overset(KOH (alc.))to |
|
Answer» Solution :DUE to the strong electron-withdrawing effect of the `NO_(2)` group, the H-atom of `CH_(3)NO_(2)` are weekly acidic. Therefore, in presence of KOH (alc.) it forms a carbanion (I) which adds to gtC=O group of `C_(6)H_(5)CHO` to form ALDOL (II) which SUBSEQUENTLY undergoes dehydration to form `beta`-nitrostyrene (or `beta`-nitrostyrene) `underset("Nitramethane")(CH_(3)NO_(2)) underset(-H_(2)O)overset(OH^(-)) tounderset("Carbanion (I)")(.^(-)CH_(2)-NO_(2)) overset(C_(6)H_(5)CH=O)to [underset((II))(C_(6)H_(5)-overset(OH)overset(|)(C)H-CH_(2)NO_(2))] underset(-H_(2)O)to underset("1-Nitro-2-phenylethene "(beta-"Nitrostyrene"))(C_(6)H_(5)overset(alpha)(C)H=overset(beta)(C)HNO_(2))` |
|
| 14. |
Predict the product of the following reactions : |
Answer» Solution :`CH_(3)-CH_(2)-CH_(2)-O-CH_(3)+HBr overset(373 K) to underset("Propane-1-ol") (CH_(3)CH_(2)CH_(2))-OH+ underset("Bromomethane")(CH_(3)Br)` Both the alkyl groups attched to O atom are primary and therefore, ATTACK of `Br^(-)` ION occurs on the smaller methyl GROUP. 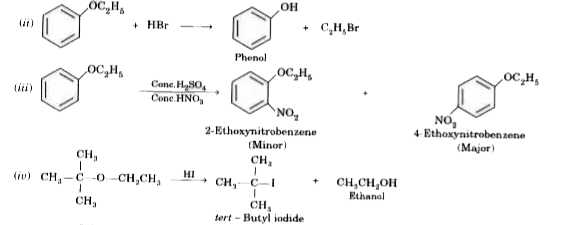
|
|
| 15. |
Predict the product of the following reaction: C_(6)H_(5)-CO-CH_(3) overset(NaOH//I_(2))to?+? |
|
Answer» Solution :`NaOH//I_(2)` i.e., sodium hypoiodite (NaIO) oxidises methyl ketones to `-COO^(-)Na^(+)+CHI_(3)-`IODOFORM reaction. `UNDERSET("Acetophenone")(C_(6)H_(5)-CO-CH_(3)) underset(("Iodoform reaction"))overset(NaOH//I_(2))to underset("Sod. BENZOATE")(C_(6)H_(5)-COO^(-)Na^(+))+underset("Iodoform")(CHI_(3))`. |
|
| 16. |
Predict the product of reaction of aniline with bromine in non-polar solvent such as CS_(2). |
|
Answer» |
|
| 17. |
Predict the product in the following series of reactions : CH_(3)-COOHoverset (PCl_(5))to Aoverset(C_(6)H_(6))underset(Anhyd.AlCl_(3))to Boverset(CH_(3)MgBr)toC |
|
Answer» `CH_(3)CH_(2)CH(OH)C_(6)H_(5)` 
|
|
| 18. |
Predict the product for the following reaction C_(6)H_(5)COCH_(3)+Br_(2)to? |
|
Answer» m-nitro acetophenone |
|
| 19. |
Predict the product for he following reaction |
|
Answer»
|
|
| 20. |
Predict the product 'C' obtained in the following reaction of butyne-1 CH_(3)CH_(2) - C -= CH + HCl rarr B overset(HI)rarr C |
|
Answer» `CH_(3)CH_(2) - underset(Cl)underset(|)overset(I)overset(|)(C) - CH_(3)` |
|
| 21. |
Predict the product expected to be obtained when HCl is added to isobutylene. Explain the mechanism. |
Answer» Solution :ISOBUTYLENE `(Me_(2)C = CH_(2))` undergoes addition with HCl according to the Markownikoff's rule and teri-butyl chloride is obtained as the only product. The other product (1-chloro-2-methylpropane) is not obtained because it is EXPECTED to be obtained through the formation of a very UNSTABLE `1^(@)` carbocation. 
|
|
| 22. |
Predict the product for each of the following reactions. |
Answer» 
|
|
| 23. |
Predict the product C obtained in the following reaction of butyne-1 CH_3CH_2-C-=CH+HCl to B overset"HI"to C |
|
Answer» `CH_3-undersetunderset(CL)|CH-CH_2CH_2I` |
|
| 24. |
Predict the product .C in the following series of reactions: CH_3 - COOH overset(PCl_5)to A underset("Anhyd AlCl_3)overset(C_6 H_6)to B overset(CH_3 MgBr)to C |
|
Answer»

|
|
| 25. |
Predict the product 'C' in the following reaction of but-1-ene. underset((A))(CH_(3)-CH_(2)-C-=CH+HCltoBoverset(HI)toC) |
|
Answer» `CH_(3)CH_(2)CH_(2)-underset(CL)underset(|)OVERSET(I)overset(|)(C)-H` 
|
|
| 26. |
Predict the product 'C' in the following series of reactions : CH_3 - COOH overset(PCl_5)to A underset("Anhyd. " AlCl_3)overset(C_6H_6)to B overset(CH_3MgBr)toC. |
|
Answer»
|
|
| 27. |
Predict the product (B) in the following sequence of reactions: Ethylbenzene underset(Delta)overset(KMnO_(4)-KOH)to A overset(H_(3)O^(+))toB |
|
Answer» benzaldehyde |
|
| 28. |
Predict the product 'B' in the sequence of reaction CH-=CHunderset(HgSO_(4))overset(30%G_(2)SO_(4))rarrAunderset(10^(@)C)overset(30% H_(2)SO_(4))rarrB |
|
Answer» `CH_(3)COONA` |
|
| 29. |
Predict the product A,B,X and Y in the following sequences of reaction underset(Cu//573 K) underset(X) underset(darr) ("butan"-2-ol) overset(SOCl_(2)) to A overset(Mg) underset("ether") B overset(X) to Y |
Answer» SOLUTION : A- 2-chloro BUTANE B- MAGNESIUM chloride C - 2-(sec BUTYL) -2-propanol |
|
| 30. |
Predict the product |
|
Answer» |
|
| 31. |
Predict the product A,B,X and Y in the following sequence of reaction |
Answer» SOLUTION :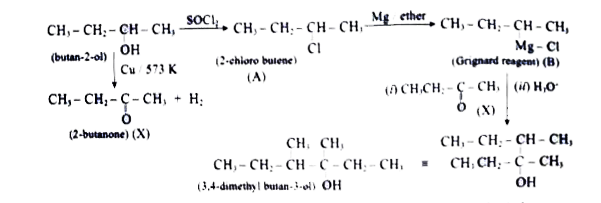
|
|
| 32. |
Predict the principal organic product of each of the following reactions. Specify stereochemistry where appropriate. |
Answer» Solution : (C). `Ph-CH(Cl)-CH_(2)OH` (d). `H_(3)C-CH_(2)-CH_(2)-CH_(2)-S-CH_(2)-(CH_(2))_(16)-CH_(3)` |
|
| 33. |
Predict the percentage of space filled by particles in bcc |
| Answer» Solution :The cubic close packed (cpp)is also called face central cubic(fcc) lattice. In both of these PACKINGS `74%` SPACE is filled The remaining space is present in the form of two typesof voids-octahedral voids and tetrahedral voids.Other type of packing of particles. While in body- centred cubic lattice (BCC)`68%` space is filled, in simple cubic lattice only `52.4%` space is filled. | |
| 34. |
Predict the order of reactivity of the following compounds in S_(n^(1)) and S_(N^(2)) reactions : The four isomeric bromobutanes, |
|
Answer» Solution :`CH_(3)CH_(2)CH_(2)CH_(2)BR LT (CH_(3))_(2)CHCH_(2)Br lt CH_(3)CH_(2)CH(Br)CH_(3) lt (CH_(3))_(3)CBr (S_(N^(1)))` `CH_(3)CH_(2)CH_(2)CH_(2)Br gt (CH_(3))_(2) CHCH_(2)Br gt CH_(3)CH_(2)CH(Br)CH_(3) gt (CH_(3))_(3)CBr (S_(N^(2)))` |
|
| 35. |
Predict the organic products of the following reactions : . |
Answer» SOLUTION : . .
|
|
| 36. |
Predict the percentage of space filled by particles in fcc |
| Answer» Solution :The cubic close packed (CPP)is also called FACE central cubic(fcc) lattice. In both of these packings `74%` space is filled The REMAINING space is present in the form of TWO TYPESOF voids-octahedral voids and tetrahedral voids.Other type of packing of particles. While in body- centred cubic lattice (bcc)`68%` space is filled, in simple cubic lattice only `52.4%` space is filled. | |
| 38. |
Predict the order of reactivity of the following compounds in S_(N^(1)) and S_(N^(2)) reactions :(i) The four isomeric bromobutanes(ii) C_(6)H_(5)CH_(2)Br, C_(6)H_(5)CH(C_(6)H_(5))Br, C_(6)H_(5)CH(CH_(3))Br, C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br |
|
Answer» Solution :(i)`CH_(3)CH_(2)CH_(2)CH_(2)BR lt (CH_(3))_(2)CHCH_(2)Br lt CH_(3)CH_(2)CH(Br)CH_(3)lt (CH_(3))_(3)CBr(S_(N^(1)))` `CH_(3)CH_(2)CH_(2)CH_(2)Br gt (CH_(3))_(2)CHCH_(2)Br gt CH_(3)CH_(2)CH(Br)CH_(3)gt (CH_(3))_(3)CBr(S_(N^(2))` Of the two primary bromides, the CARBOCATION intermediate derived from `(CH_(3))_(2) CHCH_(2)Br` is more stable than derived from `CH_(3)CH_(2)CH_(2)CH_(2)Br` because of greater electron donating inductive effect of `(CH_(3))_(2)CH-` group. Therefore, `(CH_(3))_(2)CHCH_(2)Br` is more reactive than `CH_(3)CH_(2)CH_(2)CH_(2)Br` in `S_(N^(1))` reactions. `CH_(3)CH_(2)CH(Br)CH_(3)` is a secondary bromide and `(CH_(3))_(3)C-Br` is a tertiary bromide. Hence the above order is followed in `S_(N^(1))`. The reactivity in `S_(N^(2))` reactions follows the reverse order as the steric hinderance AROUND the ELECTROPHILIC carbon increases in that order. (ii)`C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br gt C_(6)H_(5)CH(C_(6)H_(5))Br gt C_(6)H_(5)CH(CH_(3))Br gt C_(6)H_(5)CH_(2)Br(S_(N^(1))` `C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br lt C_(6)H_(5)CH(C_(6)H_(5))Br lt C_(6)H_(5)CH(CH_(3))Br lt C_(6)H_(5)CH_(2)Br (S_(N^(2)))` Of the two secondary bromides, the carbocation intermediate obtained from `C_(6)H_(5)CH(C_(6)H_(5))Br` is more stable than obtained from `C_(6)H_(5)CH(CH_(3))Br` because it is stabilised by two phenyl groups due to resonance. Therefore, the former bromide is more reactive than the latter in `S_(N^(1))` reactions. A phenyl group is BULKIER than a methyl group. Therefore, `C_(6)H_(5)CH(C_(6)H_(5))Br` is less reactive than `C_(6)H_(5)CH(CH_(3))Br` in `S_(N^(2))` reactions. |
|
| 39. |
Predict the order of reactivity of the following compounds in S_(N)1 and S_(N)2 reactions: (i) the four isomeric bromobutanes (ii) C_(6)H_(5)CH_(2)Br,C_(6)H_(5)CH(C_(6)H_(5))Br,C_(6)H_(5)CH(CH_(3))Br,C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br. |
|
Answer» Solution :(i) In `S_(N)1` reactions, the order of reactions, the order of reactivity depends upon the stability of the intermediate CARBOCATIONS, therefore, `(CH_(3))_(3)CB r` which gives a `3^(@)` carbocations, i.e., `(CH_(3))C^(+)` is the most reactive. `CH_(3)CH_(2)CH(Cl)CH_(3)` gives a `2^(@)` carbocation, i.e., `CH_(3)CH_(2)overset(+)(C)HCH_(3)` and hence is less reactive that `(CH_(3))_(3)CB r`. Out of the `CH_(3)CH_(2)CH_(2)CH_(2)^(+)` due to greater +I-effect of `(CH_(3))_(2)CHCH_(2)`-group w.r.t. `CH_(3)CH_(2)CH_(2)CH_(2)`-group and increasing reactivity of the four ISOMERIC bromobutanes towards `S_(N)1` reaction follows the order: `CH_(3)CH_(2)CH_(2)CH_(2)Br lt (CH_(3))_(2) CHCH_(2) Br lt CH_(3)CH_(2)CH(Br)CH_(3) lt (CH_(3))_(3) CB r` The reactivity in `S_(N)2` reactions, however, follows the reverse order, i.e., `CH_(3)CH_(2)CH_(2)CH_(2)Br gt (CH_(3))_(2)CHCH_(2)Br gt CH_(3)CH_(2)CH(Br)CH_(3) gt (CH_(3))_(3)CB r`. Since the steric hindrance around the electrophilic carbon (i.e., `ALPHA`-carbon) increase in the order (ii) Since the reactivity in `S_(N)1` reactions increases as the stability of the intermediate carbocations increases, therefore, `(C_(6)H_(5))C(CH_(3))(C_(6)H_(5))Br` which gives `3^(@)` carbocation, i.e., `(C_(6)H_(5))_(2)overset(+)(C)(CH_(3))` is the most reactive. Of the two `2^(@)` bromides, the carbocation intermediate derived from `C_(6)H_(5)overset(+)(C)HCH_(3)` obtained from `C_(6)H_(5)CH(CH_(3))Br` because it is stabilized by two phenyl groups due to resonance. the fourth alkyl BROMIDE, i.e., `C_(6)H_(5)CH_(2)Br` which gives the `1^(@)` carbocation, i.e., `C_(6)H_(5)CH_(2)^(+)` is, however, the least reactive. THUS, the overall reactivity of these alkyl bromides towards `S_(N)1` reactions follows the order: `C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br gt C_(6)H_(5) CH(C_(6)H_(5))Br gt C_(6)H_(5)CH(CH_(3))Br gt C_(6)H_(5)CH_(2)Br` In `S_(N)2` reactions, it is the steric hindrance which determines the reactivity. since a phenyl group is much bulkier than a methyl group, therefore, `C_(6)H_(5)C(CH_(3))(C_(6)H_(5))` Br is the least reactive followed by `C_(6)H_(5)CH(C_(6)H_(5))Br` and `C_(6)H_(5)CH(CH_(3))Br` while `C_(6)H_(5)CH_(2)Br` is the most reactive. thus, the overall reactivity of these alkyl bromides towards `S_(N)2` reactions follows the order: `C_(6)H_(5)CH_(2)Br gt C_(6)H_(5)CH(CH_(3))Br gt C_(6)H_(5)CH(C_(6)H_(5))Br gt C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br`. |
|
| 40. |
Predict the order of reactivity of the following compounds in S_N1 and S_N2 reactions: (i) The four isomeric bromobutanes, (ii) C_6H_5CHBr, C_6H_5(C_6H_5)Br, C_6H_5CH(CH_3)Br and C_6H_5C(CH_3)(C_6H_5)Br. |
|
Answer» Solution :i) `CH_3CH_2CH_2CH_2Br < (CH_3)_2 CHCH_2Br < CH_3CH_2CH(BR)CH_3 < (CH_3)_3 CBR (S_N1)` `CH_3CH_2CH(Br)CH_3 < (CH_3)_(2) CHCH_2Br > CH_3CH_2CH(Br)CH_3 < (CH_3)_(3) CBr (S_N2)` ii) `C_6H_5C(CH_3)(C_6H_5)Br > C_6H_5CH(C_6H_5)Br > C_6H_5CH(CH_3)Br > C_6H_5CH_2Br (S_N1)` `C_6H_5C(CH_3)(C_6H_5)Br < C_6H_5CH(C_6H_5)Br < C_6H_5CH(CH_3)Br < C_6H_5CH_2Br (S_N2)` |
|
| 41. |
Predict the order of reactivity of the compounds in S_(N)1 and S_(N) 2 reactions . C_(6)H_(5)CH_(2) Br, C_(6)H_(5) CH (C_(6)H_(5)) Br , C_(6) H_(5) CH(CH_(3)) Br , C_(6)H_(5) C (CH_(3)) (C_(6)H_(5)) Br |
|
Answer» Solution :`C_6 H_5 C(CH_3) (C_6 H_5) Bt gt C_6 H_5 CH (C_6 H_5) BR gt C_6 H_5 CH(CH_3)Br gtC_6 H_5CH_2 Br(S_(N^(1)))` `C_6 H_5 C (CH_3)(C_6H_5) Br ltC_6H_5 CH (C_6 H_5) Br ltC_6 H_5 CH(CH_3)Br lt C_6 H_5 CH_2Br(S_(N^(2)))` |
|
| 42. |
Predict the order of reactivity of the folloiwingcompoundss in SN^(1) and SN^(2) reacations. a.The four isomeric bromolbutanes b. I. C_(6)H_(5)CH_(2)Br II. C_(6)H_(5) CH (C_(6) H_(5)) Br III. C_(6)H_(5)CH (CH_(3) Br IV. C_(6)H_(5)C(CH_(3)(C_(6) H_(5)) Br |
Answer» SOLUTION :a. 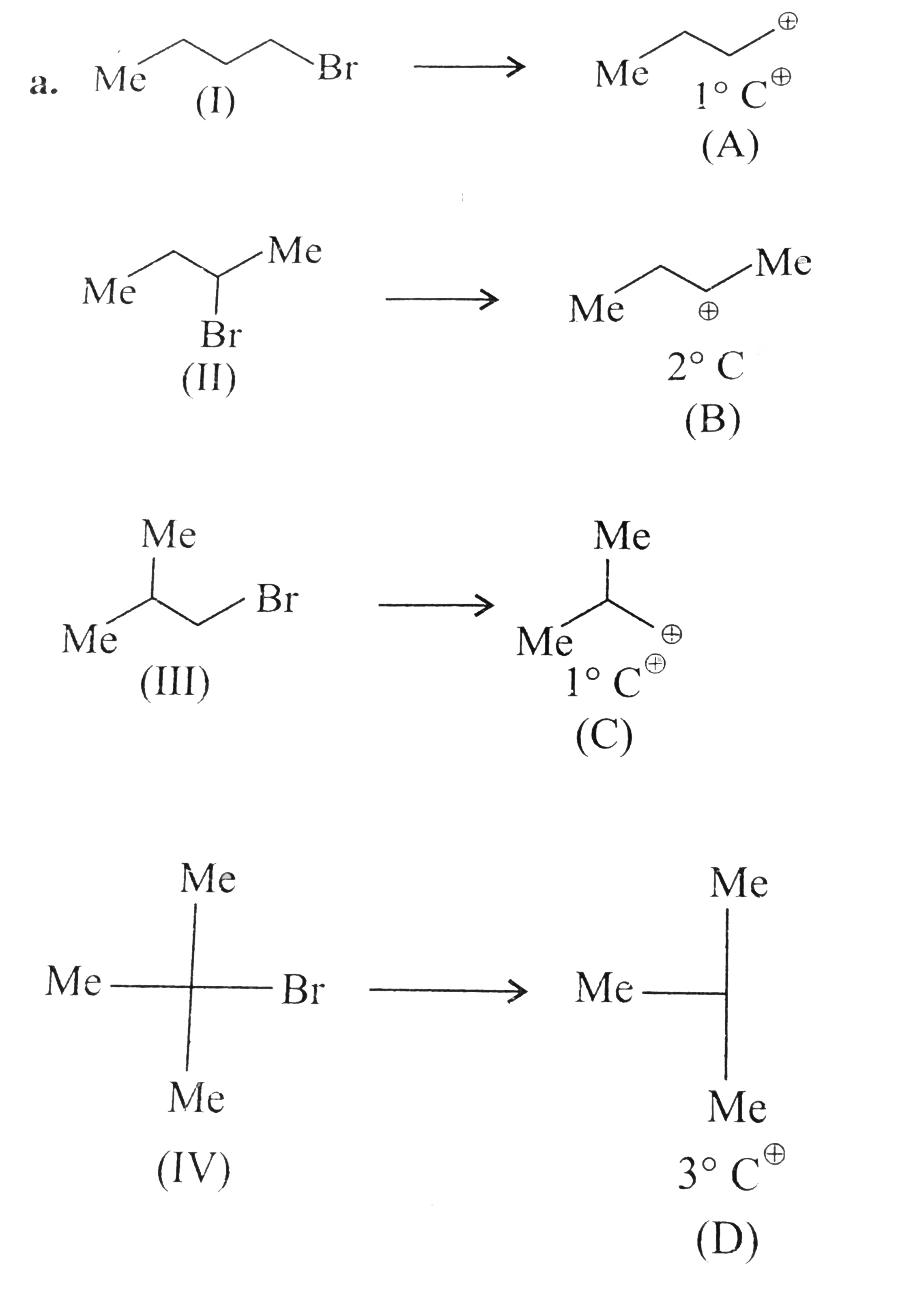 Out of `1^(@) C^(o+) (A)` and `(C),(C)` is more stable due to `+I` effect of TWO `(Me)` groups. Therefore, `(III)` is more reactive than `(I)` in `SN^(1)` reaction. `SN^(1) implies )I) gt (III) gt (II) gt (IV)` `SN^(2) implies (I) gt (III) gt (II) gt (IV)` b. `SN^(1) implies C_(6) H_(5) C(CH_(3)) (C_(6)H_(5))Br (IV)` `gt C_(6) H_(5) CH (C_(96) H_(5)) - Br gt C_(6) H_(5) CH(CH_(3)) Br gt C_(6) H_(5) CH_(2)Br` `gt (II) gt (III) gt (I)` `SN^(2) implies C_(6)H_(5)C(CH_(3))(C_(6)H_(5))Br lt C_(6) H_(5) CH(C_(6) H_(5)()-Br lt` `(IV) lt (II)` `C_(6)H_(5)CH(CH_(3))Br lt C_(6)H_(5)H_(2)Br` `(III) lt (I)` Of the two secondary bromides, the carbocation intermediate obtainedfrom `(II)` is more stable than thatobtained FORM`(III)` becuase it is stablised byu two phenyl groupsdue to resonacne. Therefore the foremer bormide is more reactive than the latter in `SN^(-1)` reacations. A phenyl group is bulkier than a methuyl group . THerefore `(II0` is less reactive than `(III)` in `SN^(2)` reacations. |
|
| 43. |
Predict the order of reacitivity of the following compounds in S_(N)1 and S_(N)2 reactions: (2) C_(6)H_(5)CH_(2)Br C_(6)H_(5)(CH_3)Br, C_(6)H_(5)C(CH_3)(C_(6)H_5)Br. |
|
Answer» SOLUTION :The order of stabilty of the CARBOCATIONS derived from the given alkyl broamides is : `C_(6)H_(5)overset(color(red)(o+))(C)H_(2) < C_(6)H_(5)overset(color(red)(o+))(C)H(CH_3) < C_(6)H_(5)overset(color(red)(o+))(C)H(C_(6)H_5)< C_(6)H_(5)overset(color(red)(o+))(C)(CH_3)(C_(6)H_(5))`. so in `S_(N)1` pathway reactivity is `C_(6)H_(5)CH_(2)Br < C_(6)H_(5)CH(CH_(3))Br < C_(6)H_(5)CH(C_(6)H_(5))Br < C_(6)H_(5)C(CH_3)(C_(6)H_(5))Br`. In `S_(N)1` pathway, reactivity of the alkly HALIDES decreases as the steric hindrance around the electrophilic carbon (i.e. `alpha`-carbon) incrases. Since `C_(6)H_(5)` group is larger than `CH_(3)` group, the reactivity follows the sequence: `C_(6)H_(5)C(CH_3)(C_(6)H_(5))Br < C_(6)H_(5)CH(C_(6)H_(5))Br < C_(6)H_(5)CH(CH_3)Br < C_(6)H_(5)CH_(2)Br`. |
|
| 44. |
Predict the order of reactivity of following compound in S_(N^(1)) reactions. |
|
Answer» SOLUTION :Except for CHLOROCYCLOHEXANE which is a secondary halide, the other compound are tertiary halides in nature. We know that tertiary halides are more REACTIVE than secondary halides in `S_(N^(1))` reactions. Moreover, the order of the cleavage of `C-X` bonds is: `C-I gt C-Br gt C-Cl`  Based on these GUIDELINES the increasing order of reactivity of the cyclic compounds listed above is: |
|
| 45. |
Predict the order of reacitivity of the following compounds in S_(N)1 and S_(N)2 reactions: (1) The four isomeric bromobutanes. |
|
Answer» Solution :The four isomericc bromobutances are as follows: (a) `CH_(3)CH_(2)CH_(2)CH_(2)Br`, (b)  (c) `CH_(3)CH_(2)CH(Br)CH_(3)` and (d) `(CH_3)_(3)C-Br`. In `S_(N)1` pathway, reactivity of the alkly halides increases as the stability of the corresponding carbocations increases. since stability of the order : `CH_(3)CH_(2)overset(color(red)(o+))(C)H_(2)(1^(@)) < (CH_(3))_(2)CHoverset(color(red)(o+))(C)H_(2)(1^(@))< CH_(3)CH_(2)overset(color(red)(o+))(C)H_(3)(2^(@)) < (CH_(3))_(3)overset(color(red)(o+))(C)(3^(@))`. [REMEMBER that electoron DONATING +I effect of `(CH_3)_(2)CH` - is greater than that of `CH_(3)CH_(2)CH_(2)`-], the reactivity increases in the order (a) < (b) < (c) < (d). |
|
| 46. |
Predict the number of unpaired electrons in the square planar [Pt(CN)_(4)]^(2-) ion. |
Answer» Solution :`._(78)Pt` lies in Group 10 with the configuration `5d^(9)6S^(1)`. Hence, `Pt^(2+)` has the configuration `d^(8)`  For square planar shape, the hybridisation is `dsp^(2)`. Hence, the unpaired ELECTRONS in 5d PAIR up to make one d-orbital empty for `dsp^(2)` hybridization. Thus, there is no unpaired ELECTRON. |
|
| 47. |
Predict the number of unpaired electrons in [CoCl_(4)]^(2-) ion on the basis of VBT. |
|
Answer» Solution :`[CoCl_(4)]^(2-)` Electronic configuration of `Co` atom Electronic configuration of `Co^(2+)` ion Hybridisation and FORMATION of `[CoCl_(4)]^(2-)` complex `CL^(-)` isweak field ligand , THEREFORE no electrons pairing occurs. `sp^(3)` hybridisation It has `3` unpaired electrons hence it isparamagnetic Magentic moment, `mu_(s)=sqrt(N(n+2))` `=sqrt(3(3+2))=sqrt(15)=3.87BM` It has tetrahedral geometry. 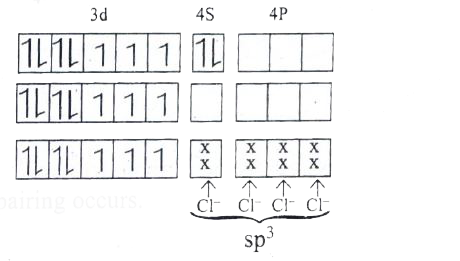
|
|
| 48. |
Predict the number of unpaired electrons in the squar planar [Pt(CN)_4]^(2-) ion. |
| Answer» Solution :The PUTER electronic CONFIGURATION of Pt is `5d^96s^1`.in `[Pt(CN)_4]^2-`the oxidation state of Pt is +2 and has the configuration `5d^8` for squar planar shape the hybridisation of Pt must be `dsp^2` hence under the influence of CN- the d ELECTRONS of Pt get paired to make one d orbital empty so THER are no unpairedelectrons in the complex ion. | |
| 49. |
Predict the number of ions produced per formula unit in an aqueous solutin of [Co(en)_(3)]Cl_(3) |
|
Answer» 4 |
|
| 50. |
Predict the number of unpaired electrons in a square planar d^(8) ion. |
Answer» 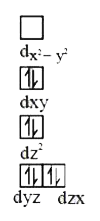 NUMBER of unpaired electron = 1. NUMBER of unpaired electron = 1.
|
|