Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Predict the major products of acid catalysed dehydration of butan-1-ol |
Answer» SOLUTION :
|
|
| 2. |
Predict the major products of acid catalysed dehydration of 1-Methylcyclohexanol |
Answer» SOLUTION :
|
|
| 3. |
Predict the major product, when -2methyl but -2 - ene is converted into an alcohol in each of the following methods. (i) Acid catalysed hydration (ii) Hydroboration (iii) Hydroxylation using bayers reagent |
|
Answer» Solution :(i) Acid catalysed HYDRATION : `underset(("2-METHYL -but - 2- ENE"))(CH_3-underset(CH_3)underset(|)(C) = CH)-CH_3overset(H^(+)//H_2O)tounderset("(2-methyl-BUTAN - 2-ol)")(CH_3- underset(CH_3)underset(|)overset(OH)overset(|)C-CH_2-CH_3)` (ii) Hydroboration : `underset("2 - methyl -but - 2- ene")(6CH_3-underset(CH_3)underset(|)C=CH -CH_3) overset(H^(+)//H_2O)to2((CH_3)_2CH- overset(CH_3)overset(|)(CH)-)_3B` `((CH_3)_2CH-overset(CH_3)overset(|)CH-)_3B + 3H_2O_2 overset(OH^(Θ))tounderset("2 - methyl - butan - 2,3-diol")(3 CH_3 - underset(CH_3)underset(|)overset(OH)overset(|)C - overset(OH)overset(|)(CH) - CH_3)` `underset("(2 - methyl - but - 2ene)")(CH_3-underset(CH_3)underset(|)C=CH - CH_3)underset((O))overset(Cold KMnO_4//OH^(Θ))tounderset("(2- methyl - butan - 2,3-diol)")(3CH_3 -underset(CH_3)underset(|)overset(OH)overset(|)C - overset(OH)overset(|)CH - CH_3)` |
|
| 4. |
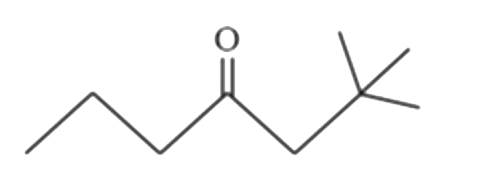
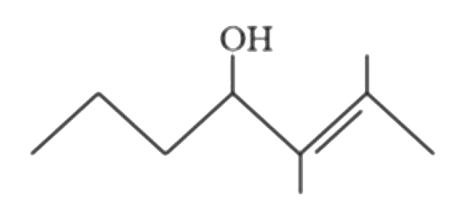
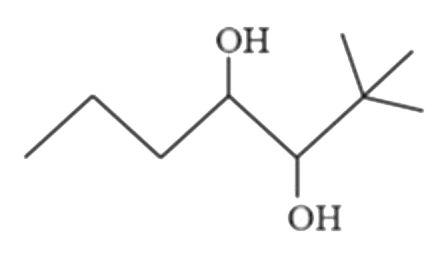
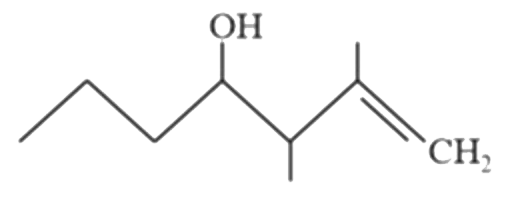
Predict the major products (A) and (B) (OMDM is oxymercuration demercuration) |
|
Answer»
|
|
| 5. |
Predict the major products formed from the following reactions : a) Dinitration of 3-methylphenol b) Mononitration of phenylmethanoate c) Mononitration of 3-methylphenol |
Answer» SOLUTION :
|
|
| 6. |
Predict the major product, when 2-methyl but-2-ene is converted into an alcohol in each of the following methods. (i) Acid catalysed hydration. (ii) Hydroboration (iii) Hydroxylation using Bayer's reagent. |
|
Answer» SOLUTION :(i) ACID catalysed hydration : `UNDERSET(("2- METHYL - but -2-ene"))(CH_3-underset(CH_3)underset(|)C-CH-CH_3)overset(H^+//H_2O)rarrCH_3-underset(("2- methyl - butan - 2- ol"))(underset(CH_3)underset(|)overset(OH)overset(|)C-CH_2-CH_3)` (ii) Hydroboration : `underset(("2- methyl - but -2-ene"))(6CH_3-underset(CH_3)underset(|)C=CH-CH_3+B_2H_6)rarr2((CH_3)_2CH-overset(CH_3)overset(|)(CH))_3B` `((CH_3)_2CH-overset(CH_3)overset(|)(CH))_3B+3H_2O_2overset(H^(o+))rarrunderset((3-"methyl - butane - 2- ol"))(3CH_3-overset(CH_3)overset(|)(CH)-overset(CH_3)overset(|)(CH)-OH+H_3BO_3)` (III) Hydroxylation using bayers reagent : `underset(("2 - methyl - but - 2 ene"))(CH_3-underset(CH_3)underset(|)C=CH-CH_3)underset((O))overset("Cold "KMnO_4//OH^(o+))rarrunderset(("2- methyl - butan 2,3-diol"))(3CH_3-underset(CH_3)underset(|)overset(OH)overset(|)C-overset(OH)overset(|)(CH)-CH_3)` |
|
| 7. |
Predict the major product that would be obtained on nitration of the following compounds. |
Answer» SOLUTION : 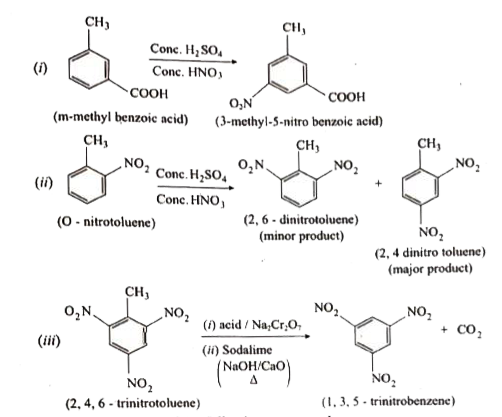
|
|
| 8. |
Predict the major product that would be obtained on nitration of the following compounds. |
Answer» SOLUTION :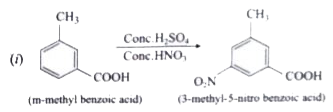 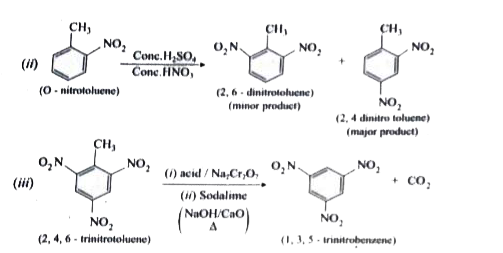
|
|
| 9. |
Predict the major product P in following reaction : |
|
Answer»
|
|
| 10. |
Predict the major product P in the following reaction. |
|
Answer»
|
|
| 11. |
Predict the major product of acid catalysed dehydration of (i) 1 - methylcyclohexanol and (ii) Butan-1-ol |
Answer» Solution :(i) Acid - catalysed dehydration of 1 - methylcyclohexanol can give tow products, VIZ, I and II. According to Saytzeff rule, I is the major product since it is more highly substituted.  (ii) Acid - catalysed dehydration of butan-1-ol produces but-2-ene as the major product and but-1-ene as the minor product. Now butan-1-ol being a `1^(@)` alcohol upon protonation and elimination of `H_(2)O` first gives a `1^(@)` carbocation (I) which is less stable rearranges to form the more stable `2^(@)` carbocation (II). This can then LOSE a proton in two DIFFERENT ways to form either but -2- ene or but-1- ene. According to Saytzeff rule, the preferred alkene is ONE which is more highly substituted (i.e., contains more alkyl GROUPS linked to the carbon - carbon double bond). Thus, but -2- ene is the major product. 
|
|
| 12. |
Predict the major product of acid -catalysed dehydration of Methylcyclohexanol |
Answer» SOLUTION :
|
|
| 13. |
Predict the major product of acid -catalysed dehydration of Butan-1-ol |
| Answer» SOLUTION :`CH_3 CH_2 CH_2 CH_2 OH to UNDERSET("But-2-ene (MAJOR)")(CH_3CH=CH-CH_3) + underset("But-1-ene (MINOR)")(CH_3CH_2CH=CH_2)` | |
| 14. |
Predict the major product of acid-catalysed dehydration of (i) 1-methylcyclohexanol and (ii) butan-1-ol. |
Answer» Solution :(i) Acid-catalysed dehydration of 1-methylcyclohexanol can, in principle, give two produces, VIZ., I and II. But since product (I) is more highly substituted, THEREFORE, according to Saytzeff rule, it is the major 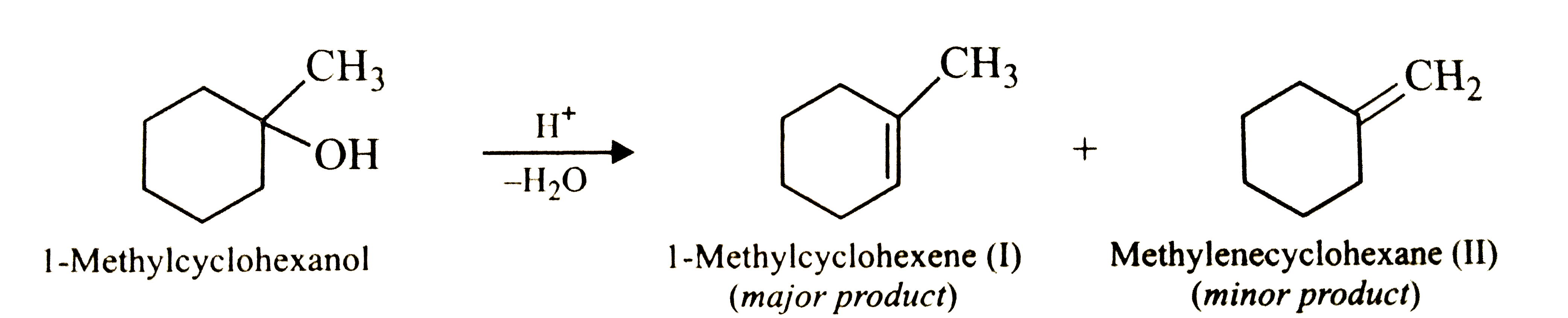 (ii) Acid-catalysed dehydration of butan-1-ol produces but-2-ene as the major product and but-1-ene as the minor product. the reason being the dehydration of alcohols occurs through carbocation intermediates. now butan-1-ol being a `1^(@)` alcohol upon PROTONATION and elimination of `H_(2)O` first gives a `1^(@)` carbocation (I) which being less stable rearrages to FORM the more stable `2^(@)` carbocation (II). this can then lose a proton in two different ways to form either but-2-ene or but-1-ene. Since buut-2-ene is more stable, therefore, according to Sayyttzeff rule, this is the major product. 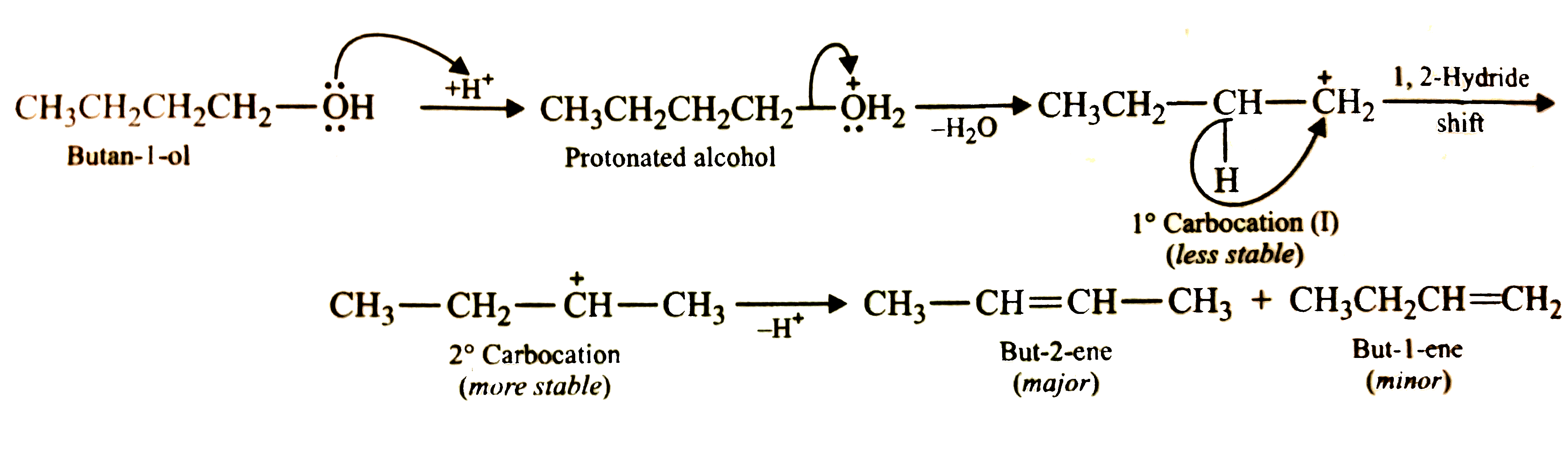
|
|
| 15. |
Predict the major product obtained by dehydrochlorination of |
Answer» SOLUTION :The major product is  . Newly formed double BOND is in conjugation with benzene ring and hence the PRODCUT is more stable . Newly formed double BOND is in conjugation with benzene ring and hence the PRODCUT is more stable
|
|
| 16. |
Predict the major product formed when HCl is added to isobutylene. Explain the mechanism involved. |
|
Answer» SOLUTION :`underset("Isobutylene")(CH_(3)-underset(CH_(3))underset("|")("C ")=CH_(2))+HCl rarr underset("(2-chloro-2-methylpropane)")(CH_(3)-overset("Cl")overset("| ")underset(CH_(3))underset("| ")("C ")-CH_(3))` Mechanism : STEP - 1 : FORMATION of Carbocation `CH_(3)-underset(CH_(3))underset("|")("C ")=CH_(2)overset(H^(+))rarr underset((3^(@)"-charbocation"))(CH_(3)-overset(o+)underset(CH_(3))underset("|")("C ")-CH_(3))+underset((1^(@)"-carbocation"))(CH_(3)-underset(CH_(3))underset("|")("C ")H-overset(o+)(C )H_(2))` Step - 2 : Attack of Nucleophile `CH_(3)-underset(CH_(3))underset("| ")overset(o+)(C )-CH_(3)overset(Cl^(-))rarr CH_(3)-overset("Cl")overset("| ")underset(CH_(3))underset("| ")("C ")-CH_(3)` |
|
| 17. |
Predict the major product formed when HCl is added to isobutylene. Explain the mechanism involved. |
Answer» Solution :MECHANISM: it is an electrophilic addition reaction which proceeds ACCORDING to Markownikov's rule. Two carocation INTERMEDIATES result in the slow step as a result of electrophile attack. However, TERTIARY carbocation is preferably formed since it is more stable. 
|
|
| 19. |
Predict the formula of the products in the following reactions. (i) CH_(3)COCH_(3)+HCNto? (ii) C_(6)H_(5)COCH+NH_(2)OHto |
Answer» SOLUTION :
|
|
| 20. |
Predict the direction in which Delta G^(0) for the equilibrium N_(2)(g)+3H_(2)(g) hArr 2NH_(3)(g),DeltaG_(298)^(0)=-33.32 kJ will change with increase in temperature. Calculate DeltaG^(0) at 500^(0)C, assuming that DeltaH and DeltaS do not change with temperature, that is DeltaH_(298)^(0)=Delta H_(793) and DeltaS_(298)^(0)=DeltaS_(793) DeltaH^(0)=-92.38kJ"and"DeltaS^(0)=-198.2 J//K |
|
Answer» |
|
| 21. |
Predict the correct intermediate and product in the following reaction H_(3)C-C-=H underset(HgSO_(4))overset(H_(2)O, H_(2)SO_(4))rarr "Intermediate (A)" rarr "Product (B)" |
|
Answer» `A : H_(3)C-underset(OH)underset(|)(C)=CH_(2), B : H_(3)C-underset(O)underset(||)(C)-CH_(3)` |
|
| 22. |
Predict the correct product when Cl_(2) passed through H-overset(18)O-overset(18)(O)-H solution |
|
Answer» `H^(+)+Cl^(-)+O_(2)` (both OXYGEN having 18) |
|
| 23. |
Predict the correct intermediate and product in the following reactionH_3C-C-=CH underset(HgSO_4)overset(H_2O, H_2SO_4)to underset"(A)""Intermediate" to underset"(B)""product " |
|
Answer» `A:H_3C-undersetunderset(OH)|C=CH_2 "" B:H_3C-undersetunderset(SO_4)|C=CH_2` 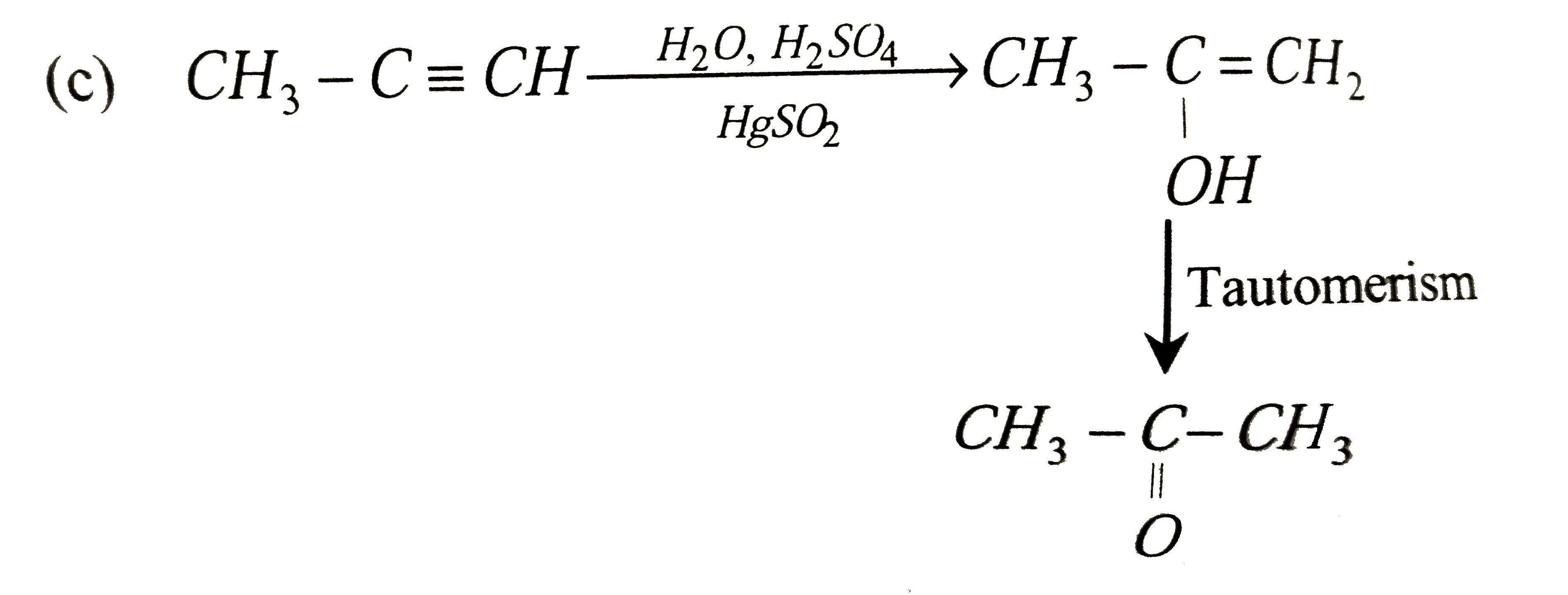
|
|
| 24. |
Predict the conditions under which (a)Aluminium might be expected to reduce magnesia (b)Magnesium could reduce alumina. (ii)Carbon monoxide is more effective reducing agent than carbon below 983 K but, above this temprature,the reverse is true -Explain. (iii)it is possible to reduce Fe_(2)O_(3) by coke at a temprature around 1200 K. |
|
Answer» Solution :(i)The CONDITIONS under which: (a)Ellingham diagram is used to PREDICT thermodynamic feasiblity of reduction of OXIDES of one metal by another metal.Any metal can reduce the oxides of other metals that are located above it in Ellingham diagram.In the Ellingham diagram,for the formation of magnesia (magnesium oxide) occupy lower than aluminium oxide.therefore aluminium cannot be used to reduce the oxides of magnesium (magnesia).Above 1623k,Al can reduce MgO to Mg,so that `Delta_(r)G^(@)` becomes negative an the process becomes thermodynamically feasible. `3MgO+2Aloverset(1623K)(to)Al_(2O_(3))+3Mg` (b) 1.`(4)/(3)Al+O_(2)to(2)/(3)Al_(2)O_(3)`2.`2Mg+O_(2) to 2MgO` At the point of intersection of the `Al_(2)O_(3)` and MgO curves in Ellingham diagram. `DeltaG^(@)` becomes zero for the reaction: `(2)/(3)al_(2)O_(3)2Mgto2MgO+(4)/(3)Al` Below that point magnesium can reduce alumina. (ii) From the Ellingham diagram,we find that at 983 K,the curves intersect. 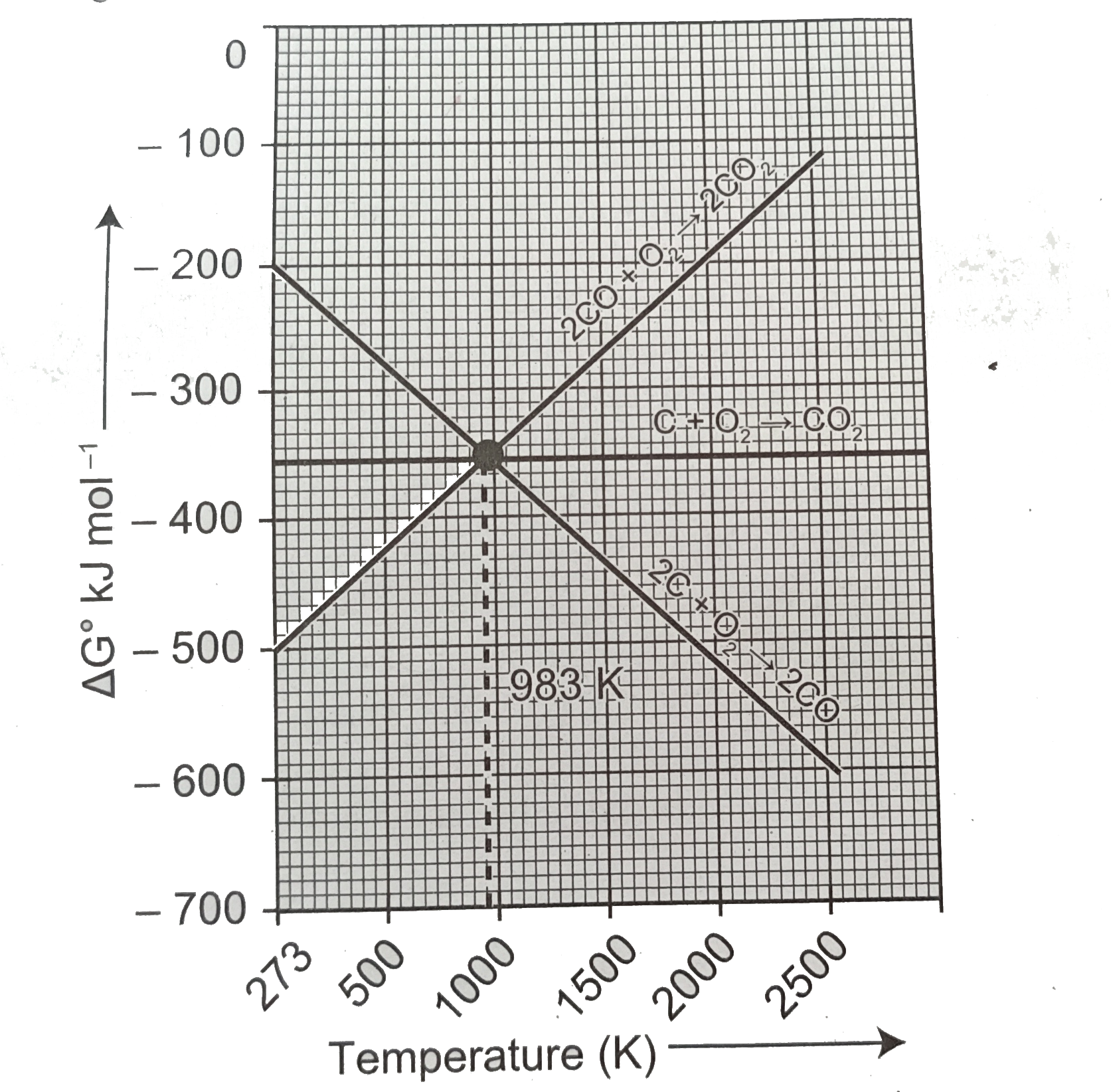 The value of `DeltaG^(@)` for change of C to `CO_(2)` is less than the value of `DeltaG^(@)` for change of CO to `CO_(2)`.therefore ,coke (C) is a better reducing agent than CO at 983K or above temprature.However below this temprature (e.g. at 673K), CO is more effective reducing agent than C. (iii)(a)Yes,it is possible to reduce `Fe_(2)O_(3)` by coke at a temprature arounf 1200K (b)In the Ellingham diagram ,carbon line cuts across the lines of many metal oxides and hence it can reduce all those metal oxides at sufficiently high temprature.Ellingham diagram for the formation of `Fe_(2)O_(3)` and CO intersects around 1000K.Below this temprature ,the carbon line lies above the iron line which indicates that `Fe_(2)O_(3)` is more stable than CO and hence at this temprature range the reduction is not line and hence we can use coke as a agent arounf 1200 K. (C)Around 1200 K,coke is better reducing agent because above 1000 K,Gibb,s free energy for the formation of `Fe_(2)O_(3)` is more than the formation of `CO_(2)` from C. |
|
| 25. |
Predictthe correctintermediateand product in thefollowingreaction: H_3 C-C-= CH underset(Hg SO_4) overset(H_2 O ,H_2 SO_4 ) to underset(( B)) underset("Product ") underset(darr ) underset((A)) ontermediate |
|
Answer» `A :H_3C-UNDERSET(OH)underset(| ) C=CH_2 "" B: H_3 C- underset(SO_4)underset(| )C=CH_2` |
|
| 26. |
Predict the conditions under which Al might be expected to reduce MgO. |
|
Answer» Solution :The Equations for the formation of two oxides are `(4)/(3)Al_((s))+O_(2_((g)))rarr(2)/(3)Al_(2)O_(3),2Mg_((s))+O_(2_((g)))rarr2MgO_((s))` from the Ellingham diagram the two curves of these oxides formation INTERSECT each otherat a certain point .The corresponding value of `DELTAG^(@)` becomes Zero for the reducction of MgO by aluminium metal, `2MgO_((s))+(4)/(3)Al_((s))hArr2Mg_((s))+(2)/(3)Al_(2)O_(3_((S)))` `rarr` From the above INFORMATION the REDUCTION of MgO by Al metal connot occur below this temperature (1665 K) instead , Mg can reduce `Al_(2)O_(3)` to Al below 1665 K. `rarr` Al-Metal can reduce MgO to Mg above 1665K becouse `DeltaG^(@)` for `Al_(2)O_(3)` is less compared to that of MgO. `3MgO_((s))+2Al_((s))overset(Tgt1665K)rarrAl_(2)O_(3(s))+2Mg_((s))` |
|
| 27. |
Predict the conditions under which (a) Aluminium might be expected to reduce magnesia. (b) Magnesium could reduce alumina. |
|
Answer» Solution :The conditions under which : Ellingham diagram is USED to predict thermodynamic feasibility of reduction of oxides of one metal by another metal. Any metal can reduce the oxides of other metals that are located above it in the Ellingham diagram. In the Ellingham diagram, for the fomation of magnesia (magnesium oxide) occupy lower position that a aluminium oxide. Therefore aluminium cannot be used to reduce the oxides of magnesium (magnesia). Above 1623K, AL can reduce MgO to Mg, so that `Delta_(r) G^(0)` becomes negative and the process becomes thermodynamically feasible. `3MgO + 2Al OVERSET(1623K)rarr Al_(2)O_(3) + 3Mg` (b) 1. `(4)/(3) Al + O_(2) rarr (2)/(3) Al_(2)O_(3)` 2. `2MG + O_(2) rarr 2MgO` At the point of intersection of the `Al_(2)O_(3)` and MgO curve in Ellingham diagram. `Delta G^(@)` becomes zero for the reaction: `(2)/(3) Al_(2)O_(3) + 2Mg rarr 2MgO + (4)/(3) Al` |
|
| 28. |
Predict the close packed structure of an ionic compound A^(+)B^(-) in which the radius of cation and anion are 148 pm and 195 pm respectively. |
|
Answer» |
|
| 29. |
Predictsigh ofworkdone in following reactions at constant pressure. |
|
Answer» (II)`DeltaV = + ve rArr W = -ve` (iii) `DeltaV = + ve rArr W = -ve` (IV)`DeltaV = + ve rArr W = -ve` |
|
| 30. |
Predict the acidic, basic or neutral nature of the following salts: NaCl,KBr,NaCN,NH_4NO_3,NaNO_2,KF. |
|
Answer» Solution :`NaCN,NaNO_2,KF` solutions are BASIC, as they are salts of strong base, WEAK acid. NACL, KBr solutions are neutral as they are salts of strong acid, strong base. `NH_4NO_3` solutions is acidic, as it is SALT of strong acid, weak base. |
|
| 31. |
Predict products of the following reactions |
Answer» SOLUTION :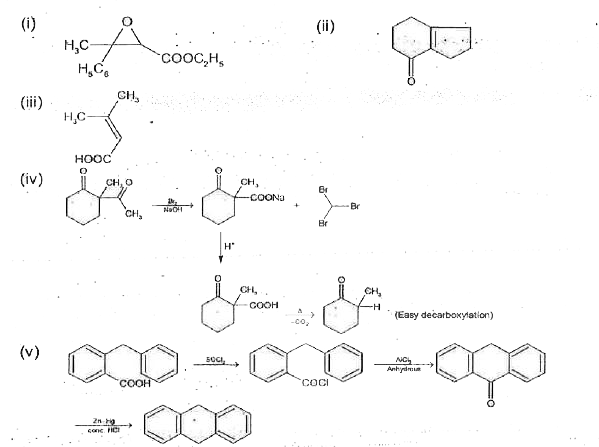
|
|
| 32. |
Predict products in the following reactions and write their structures c) (C_6H_5CH_2)_2Cd+2CH_3COCl |
Answer» SOLUTION : C) `CH_3COCH_2C_6H_5` |
|
| 33. |
Predict product formed in the following reaction: |
|
Answer»
|
|
| 35. |
Predict if the reaction between the followig is feasible: (i) Fe^(3+)(aq) and I^(-)(aq) (ii) Ag^(+)(aq) and Cu (iii) Fe^(3+) and Br^(-1)(aq) (iv) Ag(s) and Fe^(3+)(aq) (v) Br_(2)(aq) and Fe^(2+)(aq). Given standard electrode potential: E_(1//2I_(2),I^(-))^(@)=-0.541,E_(Cu^(2+),Cu)^(@)=0.34V E_(1//2Br_(2),Br^(-))^(@)=1.09V,E_(Ag^(+),Ag)^(@)=+0.80V,E_(Fe^(3+),Fe^(2+))^(@)=+0.77V |
|
Answer» Solution :A reaction FEASIBLE if EMF of the cell reaction is +ve <BR> (i) `Fe^(3+)(aq)+I^(-)(aq)toFe^(2+)(aq)+(1)/(2)I_(2)` i.E., `Pt|I_(2)|I^(-)(aq)||Fe^(3+)(aq)|Fe^(2+)(aq)|Pt` `thereforeE_(cell)^(@)=E_(Fe^(3+),Fe^(2+))^(@)-E_(1//2I_(2),I^(-))^(@)=0.77-0.54=0.23V` (Feasible) (ii) `Ag^(+)(aq)+CutoAg(s)+Cu^(2+)(aq),` i.e., `Cu|Cu^(2+)(aq)||Ag^(+)(aq)|Ag` `E_(cell)^(@)+E_(Ag^(+),Ag^(-))^(@)-E_(Cu^(2+),Cu)^(@)=0.80-0.34=0.46V` (Feasible) (III) `Fe^(3+)(aq)+Br^(-)(aq)toFe^(2+)(aq)+(1)/(2)Br_(2),E_(cell)^(@)=0.77-1.09=-0.32V` (Not feasible) (IV) `Ag(s)+Fe^(3+)(aq)toAg^(+)(aq)+Fe^(2+)(aq)E_(cell)^(@)=0.77-0.80=-0.03V` (Not feasible) (v) `(1)/(2)Br_(2)(aq)+Fe^(2+)(aq)toBr^(-)+Fe^(3+),E_(cell)^(@)=1.09-0.77=0.32V` (Feasible) |
|
| 36. |
Predict giving reasons, the order of basicity of the following compounds in (i) gaseous phase and in (ii) aqueous solutions (CH_(3))_(3)N, (CH_(3))_(2)NH, CH_(3)NH_(2), NH_(3). |
|
Answer» Solution :(i) In gaseous phase : The order of basicity is : `""(CH_(3))_(3)N gt (CH_(3))_(2)NH gt CH_(3)NH_(2) gt NH_(3)` This is explained on the INDUCTIVE effect (+I) of the alkyl groups. (ii) In AQUEOUS phase : The order of basicity in aqueous solution is : `""(CH_(3))_(2)NH gt CH_(3)NH_(2) gt (CH_(3))_(3)N gt NH_(3)` Due to steric hindrance, the lone pair of electrons on N in `(CH_(3))_(3)N` is not readily availlable. Therefore, `(CH_(3))_(3)N` is LESS basic than `(CH_(3))_(2)NH" and "CH_(3)NH_(2)`. The inductive effect, solvation effect, H-bonding and steric hindrance together decide the basicity of amines in aqueous solutions. |
|
| 37. |
Predict, giving reasons, the order of basicity of the following compounds in gaseous phase, (i) (CH_(3))_(3)N,(CH_(3))_(2)NH,CH_(3)NH_(2),NH_(3) (ii) C_(2)H_(5)NH_(2),(C_(2)H_(5))_(2)NH,(C_(2)H_(5))_(3),N,CH_(3)NH_(2) |
|
Answer» Solution :DUE to +I-effect of the alkyl groups, the electron density on the N-atom of `1^(@),2^(@) and 3^(@)` AMINES is higher than that on the N-atom in `NH_(3)`. Therefore, all amines are more basic than `NH_(3)`. (i) In gaseous phase, solvation effect are absent and hence the relative basicity of amines depends only on the +I-effect of the alkyl groups. now since the +I-effect increases in going from `1^(@) ` to `2^(@)` to `3^(@)` amine, therefore, the relative basicity of amines decreases in the order: `3^(@)` aminegt`2^(@)` amine `gt1^(@)` amine. in other words, in the gaseous phase, the basicity of methylamines relative to `NH_(3)` decreases in the order: `(CH_(3))_(3)N gt (CH_(3))_(2)NH gtCH_(3)NH_(2)gtNH_(3)` (II) I the gas phase, solvent EFFECTS are absent and hence the relative basicity of amines depends only on the +I-effect of the alkyl groups. now the +I-effect of the `C_(2)H_(5)` group is more than that of `CH_(3)` group and the +I-effect increases as the number of alkyl groups increases, therefore, overall basicitiy of the four amines increases in the order: `CH_(3)NH_(2) lt CH_(2)H_(5)NH_(2) lt (C_(2)H_(5))_(2)NH lt (C_(2)H_(5))_(3)N` |
|
| 38. |
Predict, giving reasons the order of basicity of the following compounds: (i) gaseous phase (ii) in aqueous solution. (CH_(3))_(3)N, (CH_(3))_(2)NH, CH_(3)NH_(2),NH_(3) |
|
Answer» Solution :(i) In gaseous phase, basic character of amines increases with the INCREASE in the number of electron the releasing alkyl groups due to +I effect the TREND of basic character is `3^(@)gt2^(@)gt1^(@)gtNH_(3)` Therefore `(C_(3))_(3)Ngt(CH_(3))_(3)NHgtCH_(3)NH_(2)gtNH_(3)` (ii) In aqueous phase, solvatiion of ammonium cation OCCURS by water molecules, greater the size of ion, lesser will be the solvation and lesser will be the stability of ion. So on combining +I effect and solvation effect. In aqueous phase trend changes to `2^(@)gt1^(@)gt3^(@), (CH_(3))_(2)NHgtCH_(3)NH_(2)gt(CH_(3))_(3)NgtNH_(3)` |
|
| 39. |
Predict correct statement. |
|
Answer» `E_(EA) gt 0 (E_(ea) `= ELECTRON affinity)implies that electron attachment is exothermic |
|
| 40. |
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene :(i) 1-Bromo-1-methylcyclohexane(ii) 2-Chloro-2-methylbutane(iii) 2,2,3-Trimethyl-3-bromopentane. |
Answer» SOLUTION :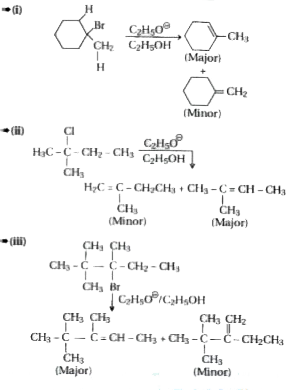
|
|
| 41. |
Predict conditions under which Al might be expected to reduce MgO. |
|
Answer» Solution :Above 1623K, AL can REDUCE MgO to Mg. Refer to Ellingham diagram Fig. 6.1. Below 1623 K, `triangleG*vs T` CURVE for the FORMATION of MgO lies below the similar curve for the formation of `Al_(2)O_(3)`. That means below 1623 K, Mg can reduce `Al_(2)O_(3)`. But above 1623K, `triangleG vs T` curve for the formation of MgO lies above the CORRESPONDING curve for the formation of `Al_(2)O_(3)`. That means above 1623K, Al can reduce MgO to Mg. |
|
| 42. |
Predict and explain the reactions: (a) Kl+Cl_(2)to (b) KClO_(3)+l_(2)to |
|
Answer» Solution :(a) `2Kl+Cl_(2)to2KCl+l_(2)` (b) `KClO_(3)+l_(2)toKlO_(3)+Cl_(2)` Because in the FIRST CASE `Cl_(2)` is more powerful oxidizing agent than `l_(2)` and in second case `CIO_(3)^(-)` is more powerful oxidizing agent than `lO_(3)^(-)[Cl^(5+)]` being reduced to `Cl^(0)` and `L^(0)` being OXIDIZED to `l^(5+)`] |
|
| 43. |
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene : 2,2,3- trimethyl -3-Bromopentane |
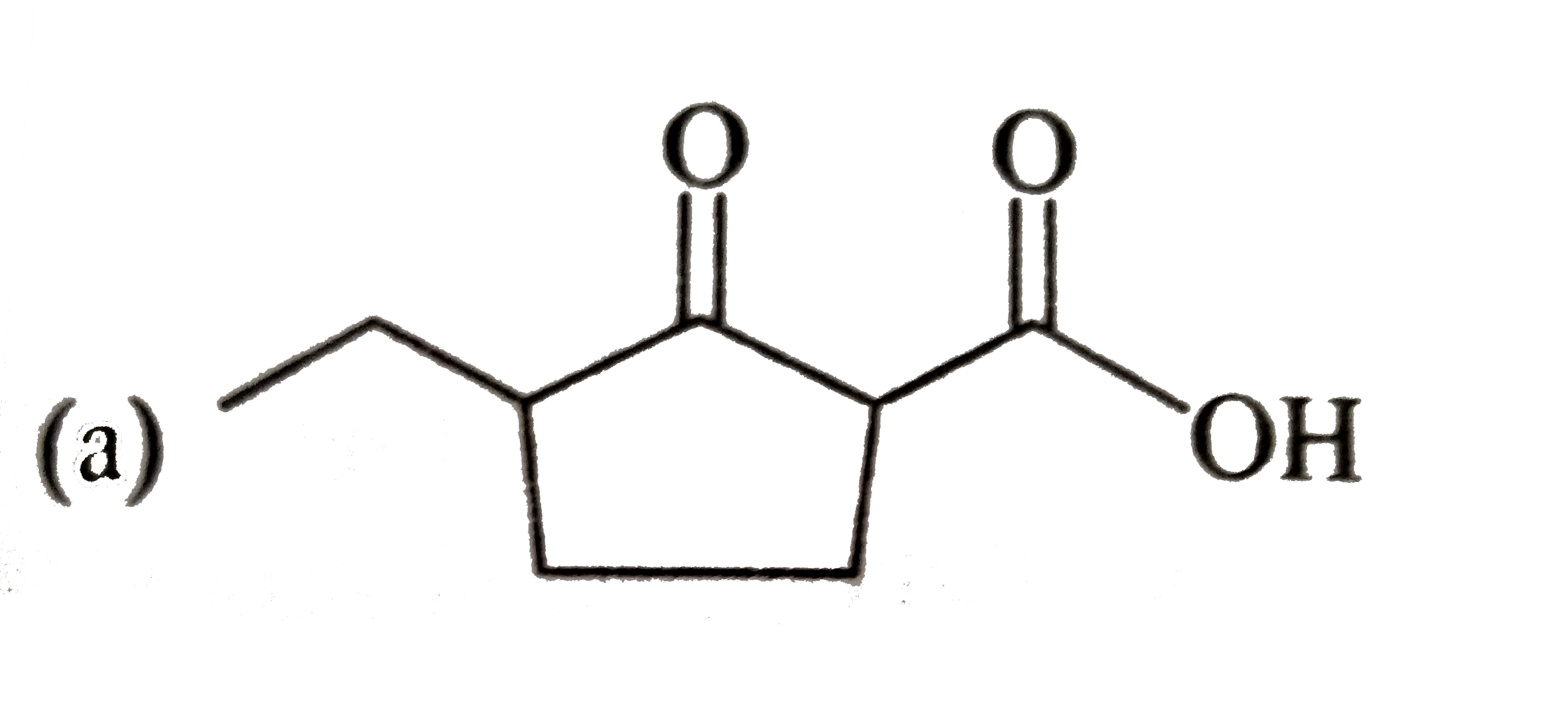
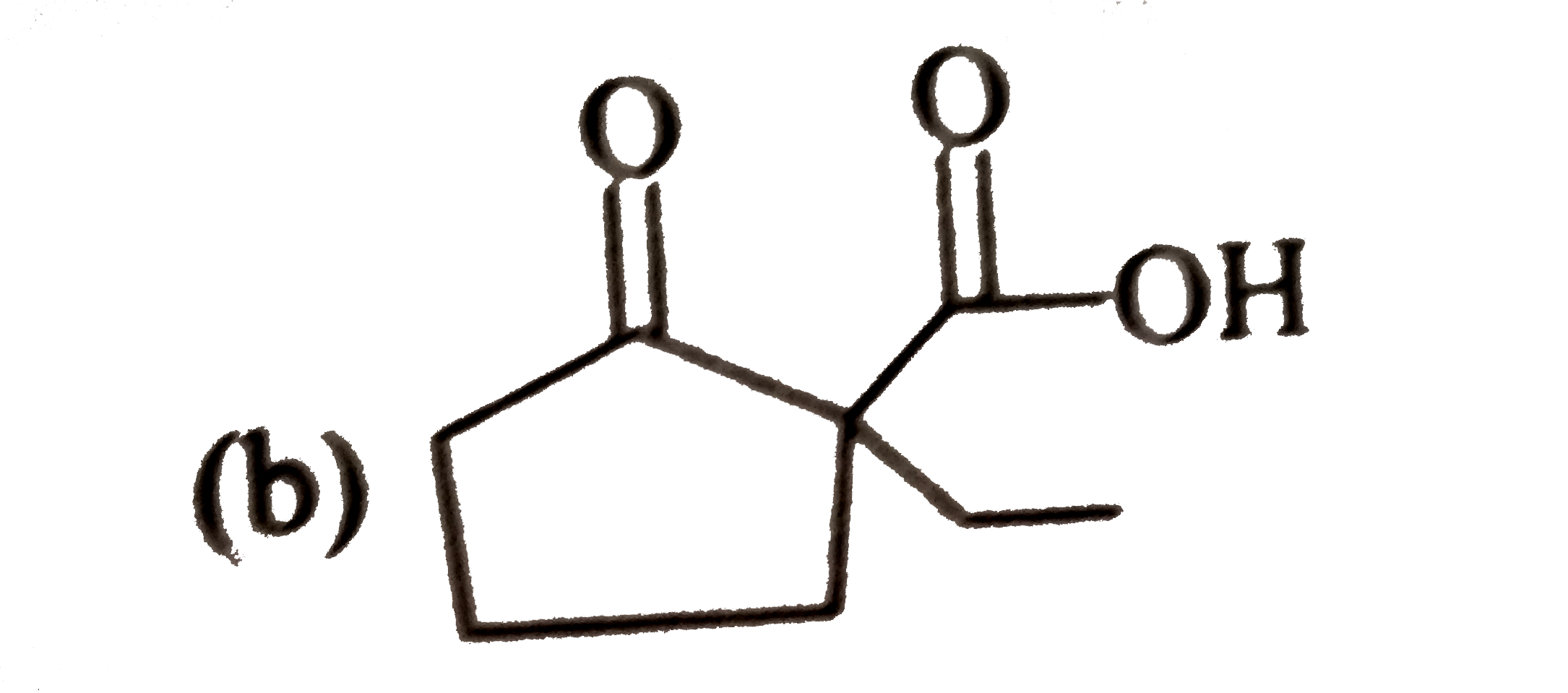
Answer» Solution :3-Bromo-2, 2, 3-trimethylpentane has two DIFFERENT SETS of b HYDROGENS. In principle, it can give two alkenes (I and II). According to Saytzeff rule, more HIGHLY SUBSTITUTED alkene (II) is more stable and is the major product. 
|
|
| 44. |
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene : 2-chloro -2- methylbutane |
|
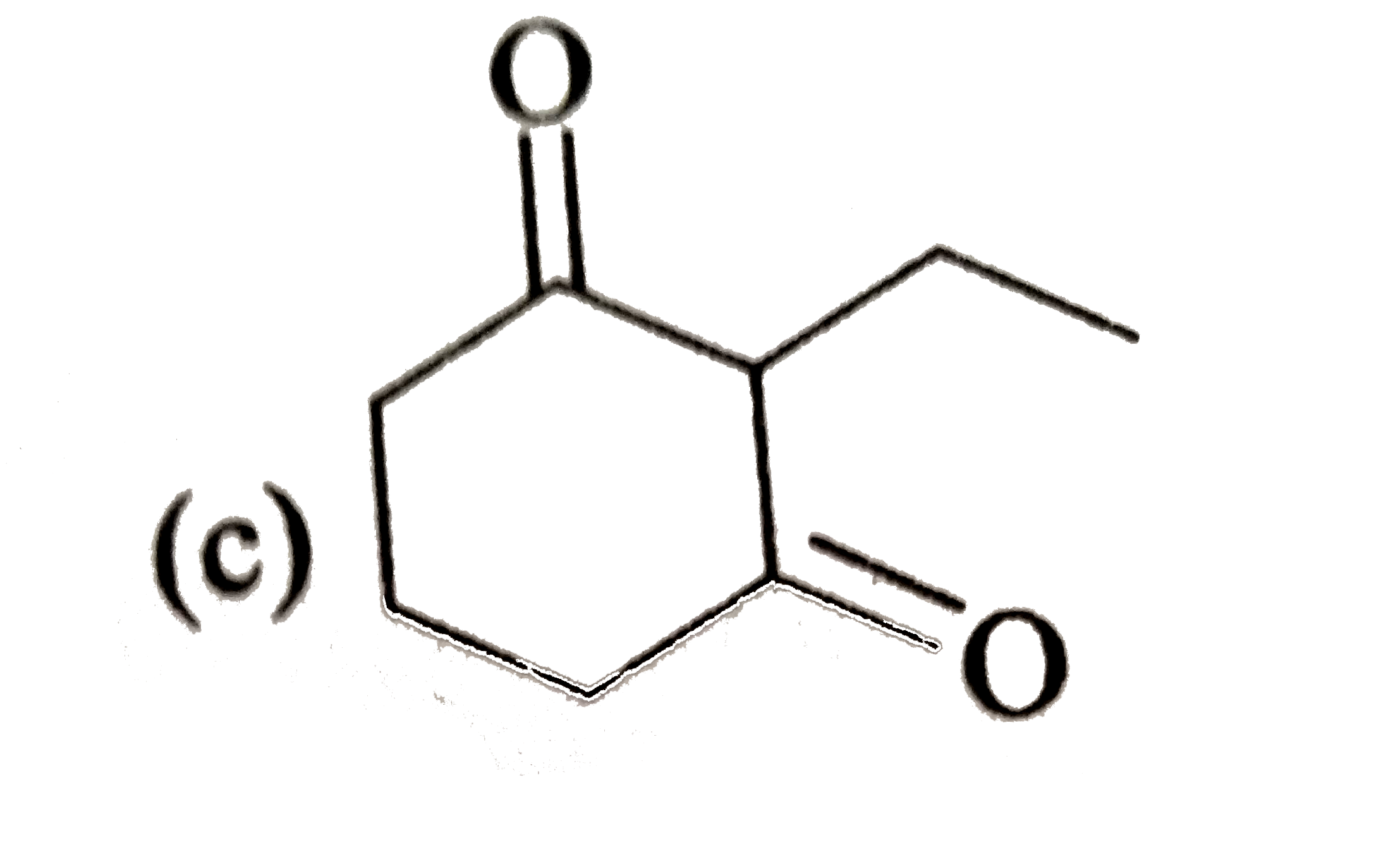
Answer» Solution :2-Chloro-2-methylbutane has TWO different sets of equivalent b-hydrogens. In principle, it can give two alkenes (I and II). ACCORDING to Saytzeff rule, more highly substituted ALKENE (II) is more stable and is the major product. `underset("2-Chloro -2-methylbutane") (overset(beta)CH_(3) - underset(Cl)underset(|)overset(beta)overset(CH_(3))overset(|)C- overset(beta)CH_(2)CH_(3)) underset(-(HCl))overset(C_(2)H_(5)ONa//C_(2)H_(5)OH) to underset("Less stable, Minor product ")underset("2-Methylbut -2- ENE (I)")(CH_(2) =overset(CH_(3))overset(|)C - CH_(2)CH_(3)) + underset(("More stable, Major product ")) underset("2-Methylbut -2-ene (II)") (CH_(3) - overset(CH_(3))overset(|)C = CHCH_(3))` |
|
| 45. |
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with NaOEt in ethanol and identify the major product obtained in each case. I-bromo-1-methycyclohexane. |
|
Answer» Solution :According to Saytzeff rule, ALKYL halides undergo dehydrohalogenation to give more highly substituted alkene as the major PRODUCT. Thus, the HALIDE (1),(2) and (3) undergo dehydrohalogenation in the PRESENCE of `NaOEt//EtOH` to YIELD 3,4,4-trimethylpent-2-ene(III) and 1-methycyclohexana (V) repectively as more products. (iii) 
|
|
| 46. |
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene : 1-Bromo -1- methylcyclohexane |
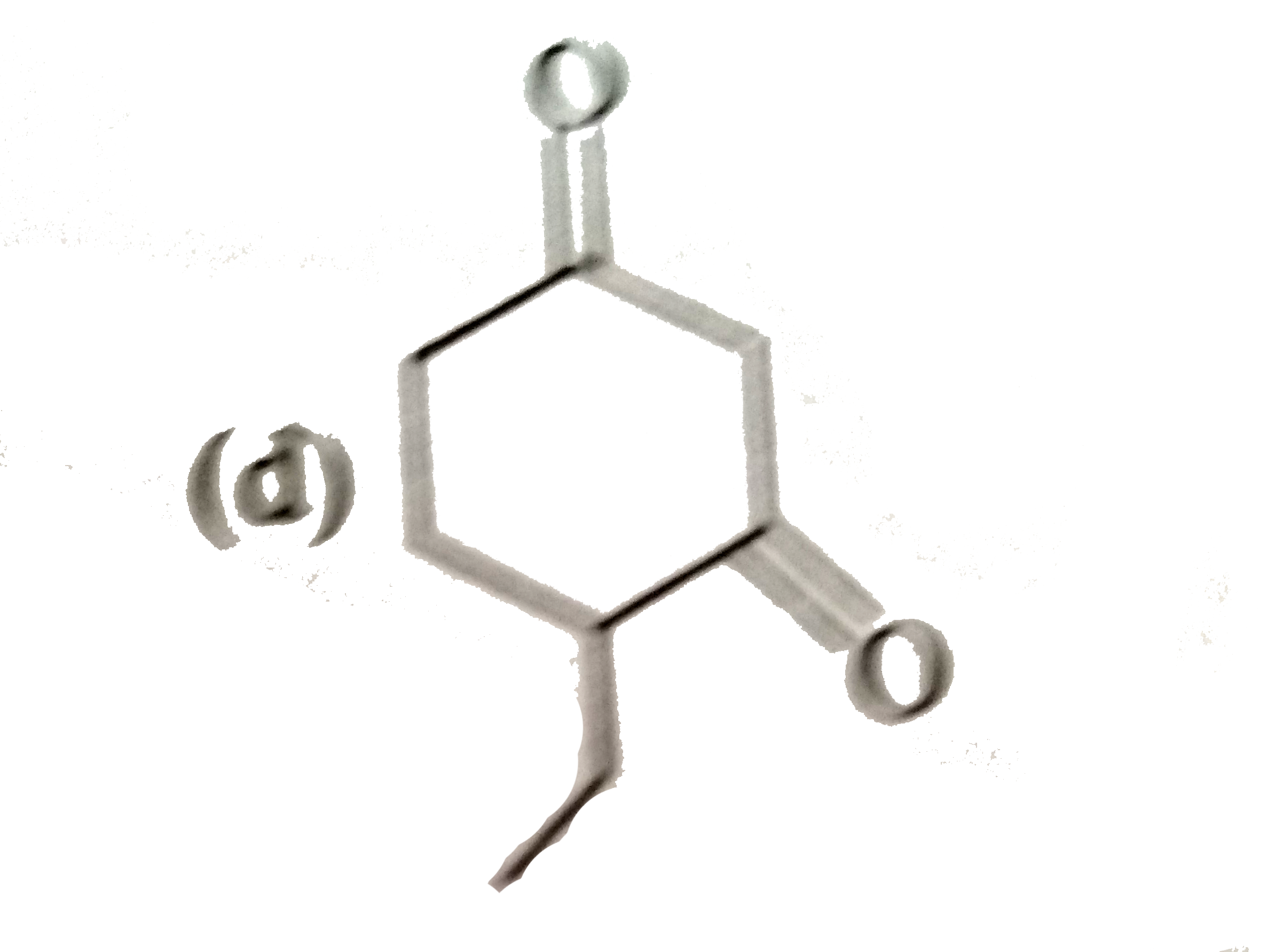
Answer» Solution : In 1-bromo- 1 methylcyclohexane, the b-hydrogens on either side of the Br atom are EQUIVALENT, THEREFORE, only 1-alkene is FORMED. 
|
|
| 47. |
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with NaOEt in ethanol and identify the major product obtained in each case. (ii) 2-chloro-2-methybutane |
|
Answer» Solution :ACCORDING to Saytzeff rule, alkyl halides undergo dehydrohalogenation to give more highly SUBSTITUTED ALKENE as the major product. Thus, the halide (1),(2) and (3) undergo dehydrohalogenation in the presence of `NaOEt//EtOH` to yield 3,4,4-trimethylpent-2-ene(III) and 1-methycyclohexana (V) repectively as more products. (ii) 
|
|
| 48. |
Predict all order of reactivity of the following compounds in dehydrohalogenation. a,. I. CH_(3)CH_(2)CH_(2)CH_(2)Cl II. (CH_(3))_(2)CHCH_(2)Cl III. (CH_(3))_(2)CH - CH_(2)Br IV. CH_(3)CH(Br)CH_(2)CH_(3) V. (CH_(3))_(3) C - Br b. I. CH_(3)CH(Br)CH_(3) II. CH_(3)CH_(2)CH_(2)Br III. (CH_(3))_(2) CH - CH_(2) Br IV. (CH_(3))_(3)C - CH_(2) Br |
Answer» Solution :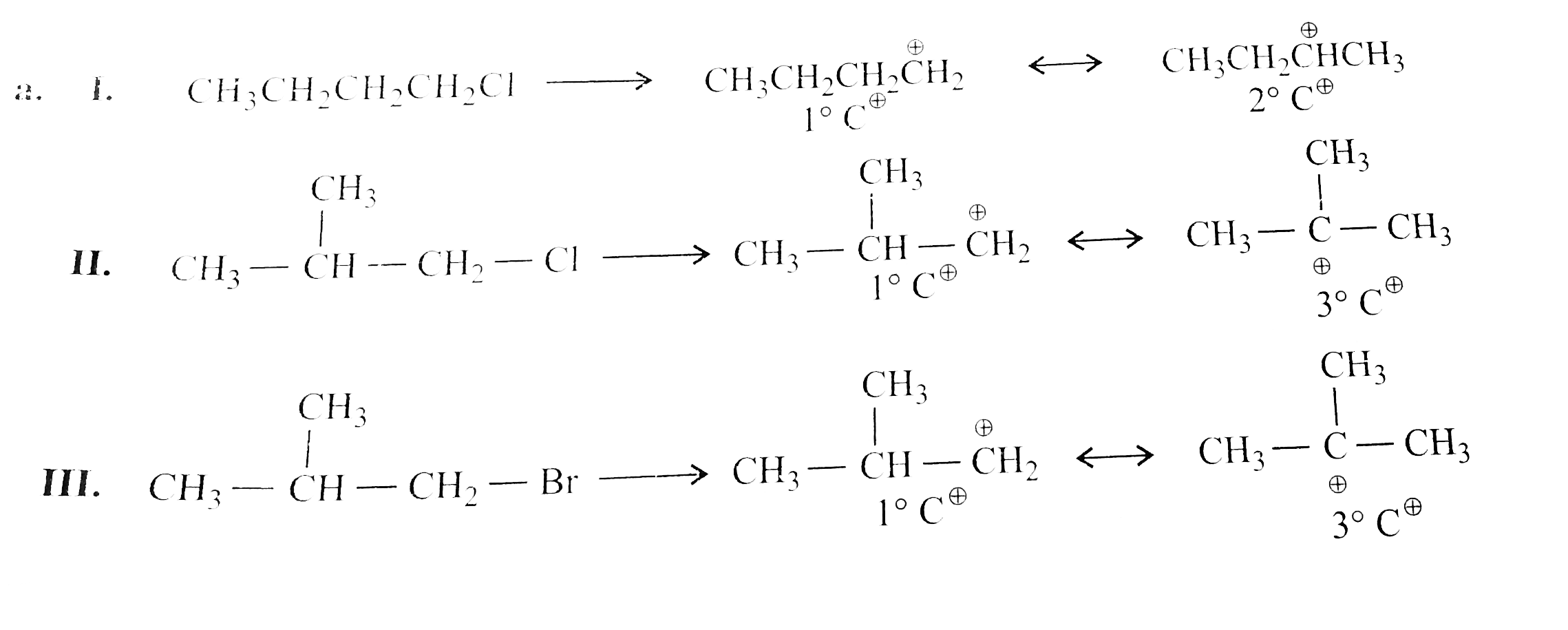 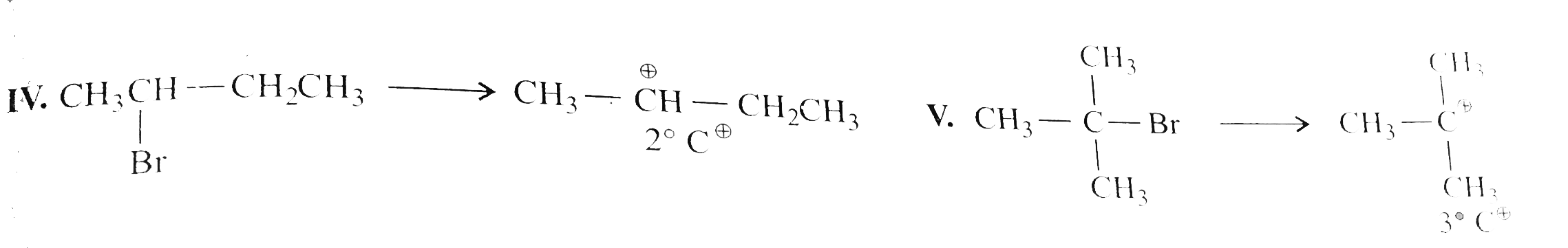 Ease FO dehdrohalogenation is DUE to the formation of carbocation and the stability of carbocation is `3^(@) gt 2^(@) gt 1^(@)`, and CASE of fornation of carbocation is `(R-1) gt (R-Br) gt (R-Cl)` So the order of dehydrogenation is `(V)gt (III) gt (II) gt (IV) gt (I)`. |
|
| 49. |
Predict all the alkenes that would be formed by dehydrohalogenation of following alkyl halides with sodium ethoxide in ethanol. (i) 1-Bromo-1-methylcyclohexane (ii) 2-Chloro-2-methylbutane (iii). 3-Bromo-2,2,3-trimethylpentane. |
|
Answer» Solution :(i). 1-Bromo-1-methylcyclohexane has two `BETA`-hydrogen atoms. This will give a mixture of two alkenes as a RESULT of dehydrohalogenation. Since alkene (B) is more SUBSTITUTED according to Saytzeff's rule, it is more stable and will be the major product. the same rule applies to the other alkyl HALIDES also. (ii). The compound has two sets of `beta`-hydrogen atoms. therefore, two elimination products are formed. HOWEVER, more substituted alkene is formed in greater proportion as compared to less substituted alkene. The explanation is similar. More substituted alkene is formed in preference to less substituted alkene.    . .
|
|
| 50. |
Predict all the alkenes that would be formed by dehydrohalogenation of the following halides with NaOEt in ethanol and identify the major product obtained in each case. (i) 3-bromo-2, 2,3-trimethylpentane. |
|
Answer» Solution :According to Saytzeff rule, ALKYL halides undergo DEHYDROHALOGENATION to give more highly substituted alkene as the MAJOR product. Thus, the halide (1),(2) and (3) undergo dehydrohalogenation in the presence of `NaOEt//EtOH` to yield 3,4,4-trimethylpent-2-ene(III) and 1-methycyclohexana (V) REPECTIVELY as more products. (i) 
|
|




<br><img src=)
</p>Solution :<img src=)