Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Potassium permanganate and potassium dichromate are two transition metal compounds. Write any two uses of potassium permanganate. |
| Answer» SOLUTION :`KMnO_4` is USED as an OXIDISING agent in the laboratory. It is used as a DISINFECTANT for water. | |
| 2. |
Potassium permanganate acts as an oxidant in neutral, alkaline as well as acidic media. The final products obtained from it in the three conditions are resectively |
|
Answer» `MnO_(4)^(2-),MN^(3+) and Mn^(2+)` |
|
| 3. |
Potassium permanganate acts as an oxidant in neutral, alkaline as well as acidic media. The final product obtained from it in three condition are respectively: |
|
Answer» `MnO_(2),NnO_(2),MN^(2+)` `2KMnO_(4)+3MnSO_(4)+2H_(2)OrarrK_(2)SO_(4)+2H_(2)SO_(4)+5MnO_(2)` In alkaline medium, `2KMnO_(4)+H_(2)Orarr 2MnO_(2)+2KOH+3O` In acidic medium, `2KMnO_(4)+3H_(2)SO_(4)rarrK_(2)SO_(4)+2overset(+2)MnSO_(4)+5H_(2)O+5O` |
|
| 7. |
Potassium metal crystallizes in a face-centred arrangement of atoms where the edge of the unit cell is 0.574 ran, What is the shortest separation of any two potassium nuclei? [Hint: Calculate diameter of K] |
|
Answer» |
|
| 8. |
Potassium manganite (K_(2)MnO _(3)) is formed when |
|
Answer» chlorine is passe dinto aqueous `KMnO_(4)` solution |
|
| 9. |
Potassium meta bisulphite is used as_____. |
|
Answer» FOOD PRESERVATIVE |
|
| 10. |
Potassium manganate (K_(2)MnO_(4))is formed when |
|
Answer» chlorine is passed into aqueous `KMnO_(4)`solution |
|
| 11. |
Potassium manganate (K_2MnO_4) is formed . When: |
|
Answer» Cuprous oxide reacts with alkaline `KMnO_4` solution (B)`2MnO_2 + 2K_2CO_3 + O_2 ("from" KNO_3) to 2K_2MnO_4 + 2CO_2` (C )`HCHO + 2KMnO_4 + 2K_2MnO_4 + H_2O +HCOOH` (D)`4KMnO_4+4KOH oversetDeltato4K_2MnO_4 +2H_2O + O_2` |
|
| 12. |
Potassium manganate (K_2MnO_4) is formed when: |
|
Answer» Chlorine is passed into AQUEOUS `KMnO_4` solution |
|
| 13. |
Potassium manganate (K_2MnO_4) is formed when : |
|
Answer» `Cl_2` is passed into an aqueous `KMnO_4` solution |
|
| 14. |
Potassium is kept in : |
|
Answer» kerosene |
|
| 15. |
Potassium hexacyanoferrate (II) is: |
| Answer» Answer :A | |
| 16. |
Potassium hexachloroplatinate, K_(2)[PtCl_(6)] adopts anti-fluorite crystal structure. The correct informations about ideal K_(2)[PtCl_(6)] crystal is/ are: |
|
Answer» `K^(+)` ions occupy all the tetrahedral voids. |
|
| 17. |
Potassium having atomic mass = 39.1 u contains 93.10 mass % ""^(39)K, having atomic mass 38.96371 u, 0.0118 mass % ""^(40)K, which has mass of 40.0 u and is radioactive witht_(1//2) = 1.3 xx 10^(9) y and 6.88 mass % ""^(41)K having a mass of 40.96184 u. Calculate the specific activity of naturally occurring potassium. |
|
Answer» |
|
| 18. |
Potassium has a bcc structure with nearest neighbor distance 4.52Å. Its atomic weight is 39. its density will be ……………… . |
|
Answer» `"915 kg m"^(-3)` `a=(4r)/(SQRT3)=(2xx4.52xx10^(-10))/(sqrt3)=5.21xx10^(-10)` `rho=(2xx39)/((5.21xx10^(-10))^(3)XX(6.023xx10^(23)))""rho="915 kg m"^(-3)N_(A)` |
|
| 19. |
Potassium hexachloroplatinate (IV) is given with the chemical formula |
|
Answer» `PT[K(Cl)_6]` |
|
| 20. |
Potassium ferrocyanide is used for testing |
|
Answer» `Cu^(2+)` and `ZN^(2+)` (C )`Ag_(4)[Fe(CN)_(6)],K_(2)Zn_(3)[Fe(CN)_(6)]`, (D)`Th[Fe(CN)_(6)],Cu_(2)[Fe(CN)_(6)]` |
|
| 21. |
Potassium ferrocyanide is used in the detection of |
|
Answer» `Cu^(2+)` ions `2CuSO_(4)+K_(4)[Fe(CN)_(6)] to underset("Chocolate ppt.")(Cu_(2)[Fe(CN)_(6)]+2K_(2)SO_(4)` `4FeCl_(3)+3K_(4)[Fe(CN)_(6)] to underset("(Prussian BLUE)")underset("ferriferro cyanide")(Fe_(4)[Fe(CN)_(6)])+12KCl` |
|
| 22. |
Potassium dicharomate solution is used to test drunker driver? Discuss. |
| Answer» Solution :Aqueous solution of potassium dichromate is ORANGE. The driver is ASKED to breath into the solution. If the solution changes to green, the driver is in DRUNKEN state. This is because dichromate oxidises ethanol to acetic ACID and itself is reduced to green chromic SALT in solution. | |
| 23. |
Potassium dichromate on heating with conc. H_2SO_4 andNaCl gives yellow coloured gas known as ........... which is used for the detection of ............. ions. |
| Answer» SOLUTION :CHROMYL CHLORIDE ,`CL^(-)` | |
| 24. |
Potassium dichromate is used in quantitative analysis for the estimation of …….. And ………. |
| Answer» SOLUTION : IRON COMPOUNDS, IODIDES | |
| 25. |
Potassium dichromate is used as'lab reagent and dichromate ion can be used as a primary standard in volumetric analysis.(1)/(2)Cr_(2)O_(7)^(2-)+7H^(+)+3e^(-)iffCr^(3+)+3(1)/(2)H_(2)O,E^(@)=1.33V WhenH_(2)O_(2) is added to an acidified' solution of dichromate, a complicated reaction occurs. The formation of product depends on the pH value and concentration of dichromate ions. A deep-blue violet peroxo compound X is formed which decomposes rapidly in aqueous solution. This compound can be extracted in ether, where it reacts with pyridine forming the adduct PyCro(O_(2))_(2) In alkaline solution, K_(2)Cr_(2)O_(7) with 30% H_(2)O_(2) a red-brown compound Y (K_(3) CrO_(8) is formed. In ammonia solution the dark red-brown compound. Z [(NH_(3)),CrO_(4)] is formed. In alkaline solution, dichromate ion changes to chromate ion which is also used as reagent in quantitative analysis of cationsQ0.1 M Cr_(2),O_(7)^(2-) will be a better oxidizing agent in which of the following solution |
|
Answer» `10 ml of 1 M CH_(3)COOH + 10 ml of 1 M NH_(4) OH` |
|
| 26. |
Potassium dichromate is used as'lab reagent and dichromate ion can be used as a primary standard in volumetric analysis.(1)/(2)Cr_(2)O_(7)^(2-)+7H^(+)+3e^(-)iffCr^(3+)+3(1)/(2)H_(2)O,E^(@)=1.33V WhenH_(2)O_(2) is added to an acidified' solution of dichromate, a complicated reaction occurs. The formation of product depends on the pH value and concentration of dichromate ions. A deep-blue violet peroxo compound X is formed which decomposes rapidly in aqueous solution. This compound can be extracted in ether, where it reacts with pyridine forming the adduct PyCro(O_(2))_(2) In alkaline solution, K_(2)Cr_(2)O_(7) with 30% H_(2)O_(2) a red-brown compound Y (K_(3) CrO_(8) is formed. In ammonia solution the dark red-brown compound. Z [(NH_(3)),CrO_(4)] is formed. In alkaline solution, dichromate ion changes to chromate ion which is also used as reagent in quantitative analysis of cationsQWhich of the following cations can be identified by using Na_(2) CrO_(4) solution? Ba^(2+)Pb^(2+) Ag^(+)(IV) Sr^(2+) |
|
Answer» (I), (II) PbCrO_(4)-yellow precitate` Ag_(2)CrO_(4)-red precitate |
|
| 27. |
Potassium dichromate is used as'lab reagent and dichromate ion can be used as a primary standard in volumetric analysis.(1)/(2)Cr_(2)O_(7)^(2-)+7H^(+)+3e^(-)iffCr^(3+)+3(1)/(2)H_(2)O,E^(@)=1.33V WhenH_(2)O_(2) is added to an acidified' solution of dichromate, a complicated reaction occurs. The formation of product depends on the pH value and concentration of dichromate ions. A deep-blue violet peroxo compound X is formed which decomposes rapidly in aqueous solution. This compound can be extracted in ether, where it reacts with pyridine forming the adduct PyCro(O_(2))_(2) In alkaline solution, K_(2)Cr_(2)O_(7) with 30% H_(2)O_(2) a red-brown compound Y (K_(3) CrO_(8) is formed. In ammonia solution the dark red-brown compound. Z [(NH_(3)),CrO_(4)] is formed. In alkaline solution, dichromate ion changes to chromate ion which is also used as reagent in quantitative analysis of cationsQThe shape of compound (Z) is |
|
Answer» butterfly shape 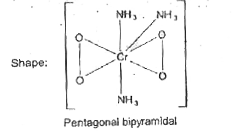
|
|
| 28. |
Potassium dichromate is used as'lab reagent and dichromate ion can be used as a primary standard in volumetric analysis.(1)/(2)Cr_(2)O_(7)^(2-)+7H^(+)+3e^(-)iffCr^(3+)+3(1)/(2)H_(2)O,E^(@)=1.33V WhenH_(2)O_(2) is added to an acidified' solution of dichromate, a complicated reaction occurs. The formation of product depends on the pH value and concentration of dichromate ions. A deep-blue violet peroxo compound X is formed which decomposes rapidly in aqueous solution. This compound can be extracted in ether, where it reacts with pyridine forming the adduct PyCro(O_(2))_(2) In alkaline solution, K_(2)Cr_(2)O_(7) with 30% H_(2)O_(2) a red-brown compound Y (K_(3) CrO_(8) is formed. In ammonia solution the dark red-brown compound. Z [(NH_(3)),CrO_(4)] is formed. In alkaline solution, dichromate ion changes to chromate ion which is also used as reagent in quantitative analysis of cationsQThe number of moles of Cr_(2))O_(7)^(2-)required in acidic medium to oxidize 1 mol of ferric oxalate is |
|
Answer» `10 MOL` |
|
| 29. |
Potassium dichromate is used as'lab reagent and dichromate ion can be used as a primary standard in volumetric analysis.(1)/(2)Cr_(2)O_(7)^(2-)+7H^(+)+3e^(-)iffCr^(3+)+3(1)/(2)H_(2)O,E^(@)=1.33V WhenH_(2)O_(2) is added to an acidified' solution of dichromate, a complicated reaction occurs. The formation of product depends on the pH value and concentration of dichromate ions. A deep-blue violet peroxo compound X is formed which decomposes rapidly in aqueous solution. This compound can be extracted in ether, where it reacts with pyridine forming the adduct PyCro(O_(2))_(2) In alkaline solution, K_(2)Cr_(2)O_(7) with 30% H_(2)O_(2) a red-brown compound Y (K_(3) CrO_(8) is formed. In ammonia solution the dark red-brown compound. Z [(NH_(3)),CrO_(4)] is formed. In alkaline solution, dichromate ion changes to chromate ion which is also used as reagent in quantitative analysis of cationsQWhat is the oxidation state of Cr in red-brown compound Y |
| Answer» Answer :C | |
| 30. |
Potassium dichromate is an orange colour crystal and is an important compound used as an oxidant in many reactions How will you account for the colour of potassium dichromate crystals? |
| Answer» Solution :Orange colour of dichromate ion and potassium dichromate is DUE to charge transfer (i.e., momentary transfer of charge from oxygen to metal). It is not,due to d-d transition because `CR^(+6)` does not CONTAIN any ELECTRON in tZ-orbital. | |
| 32. |
Potassium dichromate is a good oxidizing agent, in acidic medium the oxidation state of chromium changes by |
|
Answer» 2 |
|
| 34. |
Potassium dichromate. |
|
Answer» Solution :The uses of potassium dichromate, `K_(2)Cr_(2)O_(7)` are as follows : (1) As a strong oxidising agent. (2) In the preparation of CHROMIUM compounds LIKE lead chromate `PbCrO_(4)`, chromate alum, etc. (3) In tanning leather. (4) In dyeing (5) In the manufacture of inks and pigments. (6) In the volumetric analysis to determine FERROUS ammonium sulphate (Mohr's salt) and iodine. (7) In the DETECTION of `C^(-)` ions by chromly choride test in the qualitative analysis. |
|
| 35. |
Potassium cyanide is a deadly poison. Acidified solution of KCN is carefully heated after adding MnO_(2). The product(s) obtained is (are): |
|
Answer» cyanogens, `(CN)_(2)` |
|
| 36. |
Potassium crystallizes with b.c.c. lattice and has a density of 0.856 xx 10^3 kg m^(-3). What is the length of the side of the unit cell, a, and what is the distance between (2 0 0), (1 1 0), and (2 2 2) planes? What is the closest distance between atoms, and what is the potassium atom radius, r? |
|
Answer» |
|
| 37. |
Potassium cyanide is used for separating |
|
Answer» `Co^(2+)` and `Ni^(2+)` |
|
| 38. |
Potassium crystallizes in a body-centred cubic unit cell. What is the unit-cell content (Z) for potassium? |
|
Answer» Solution :One-eighth of each corner atom and the ENTIRE body-centred atom are contained within the UNIT cell of K. Thus, `Z =8(1/8) + f(1) =2` |
|
| 39. |
Potassium crystallizes in "body centred cubic" arrangement and the surfaces of adjacent atom along the edges of unit cells are 71.4 picometre apart. Determine the density of metal. Atomic weight of K = 39. |
|
Answer» Solution :In BCC, the relationship between edge LENGTH and radius of atom is `4r=SQRT(3)a` Also,gicen `a-2r=71.4xx10^(-10) cm` `IMPLIES`"" a-(sqrt(3))/(2)a=a[(2-sqrt(3))/(2)]` `=71.4xx10^(-10)cm` `implies ""a=5.329xx10^(-10) cm` `impliesDensity (rho)=(2xx39)/(6.023xx10^(23)(5.329xx10^(-8))^(3))` `=0.855 g/cm^(3)` |
|
| 40. |
Potassium crystallizes in a body centred cubic lattice. Calculate the number of unit cells in 1 g potassium. Atomic mass of potassium=39 u. |
|
Answer» SOLUTION :Gram atomic mass of potassium = 39 G `:.` 39G of potassium contains atoms = `6.023 xx 10^(23)` 1g of potassium contains atoms = `(6.023 xx 10^(23))/(39) = 1.54 xx 10^(22)` NUMBER of atoms in a body-centred unit cell = 2 `:.` Number of unit cells in 1 g of potassium = `(1.54 xx 10^(22))/(2) = 7.7 xx 10^(21)` |
|
| 41. |
Potassium crystallizes in a body-centered cubic lattice with edge length, a = 5.2 Å (a) What is the distance between nearest neighbours? (b) What is the distance between next-nearest neighbours? (c) How many nearest neighbours does each K atom have ? (d) How many next-nearest neighbours does each K atom have ? (e) What is the calculated density of crystalline potassium ? |
|
Answer» |
|
| 42. |
Potassium combines with two isotopes of chlorine (.^(35)Cl "and" .^(37)Cl) respectivel to form two samples of KCl. Their formation follows the law of: |
|
Answer» CONSTANT PROPORTIONS |
|
| 43. |
Potassium crystallises in a body centred cubic unit cell. The mass of one unit cell is |
|
Answer» `1.29 xx10^(-23)` GM |
|
| 44. |
Potassium chromate is isomorphous to potassium sulphate (K_(2)SO_(4)). It is found to contain 26*78 % chromium. Calculate the atomic mass of chromium (K = 39*10). |
|
Answer» `58` `= 2xx39.1+A+64 = 142.2 +A` Percentage of`Cr = (A)/(142.2+A)xx100` `26.78 = (A)/(142.2+A)xx100` On solving, A = 52.00 |
|
| 45. |
Potassium chromate solutionis added to an aqueoiussolution of a metal chloride.Theprecipitatethusobatainareinsoluble in acetic acid .These are subjectedto flame test , the colour of the flame is |
|
Answer» Lilac |
|
| 46. |
Potassium chromate is isomorphous to potassium sulphate (K_(2)SO_(4)) and is found to contain 26.78% chromium. Calculate the atomic mass of chromium (K=39.10) |
|
Answer» Solution :Since, the formula of potassium SULPHATE is `K_(2)SO_(4)`, so the formula of potassium chromate should be `K_(2)CrO_(4)` as it is isomorphous to `K_(2)SO_(4)`. If the atomic mass of chromium is A, then formula mass of potassium chromate should be `=2xx39.1+A+64=(142.2+A)` % of chromium `=(A)/((142.2+A))xx100` So, `(100A)/((142.2+A))=26.78` `100A=26.78(142.2+A)` or `A=(26.78xx142.2)/(73.22)=52.00` |
|
| 47. |
Potassium chlorate on heating with conc H_2SO_4 gives: |
| Answer» Answer :D | |
| 48. |
Potassium carbonate cannot be prepared by Solvay process, Why? |
|
Answer» SOLUTION :Potassium carbonate cannot be prepared by Solvay process because potassium BICARBONATE being more soluble than SODIUM bicarbonate does not get precipitated when CO, is passed through a concentrated solution of KCl saturated with ammonia. KCI+ `CO_(2) + NH_(3) + H_(2)O rarrKHCO_(3) + NH_(4)CI` |
|
| 49. |
Potassium bromide solution. |
| Answer» Solution :`2KBr_((aq))+Cl_(2_((G))) to 2KCl_((aq))+Br_(2_((L)))` | |