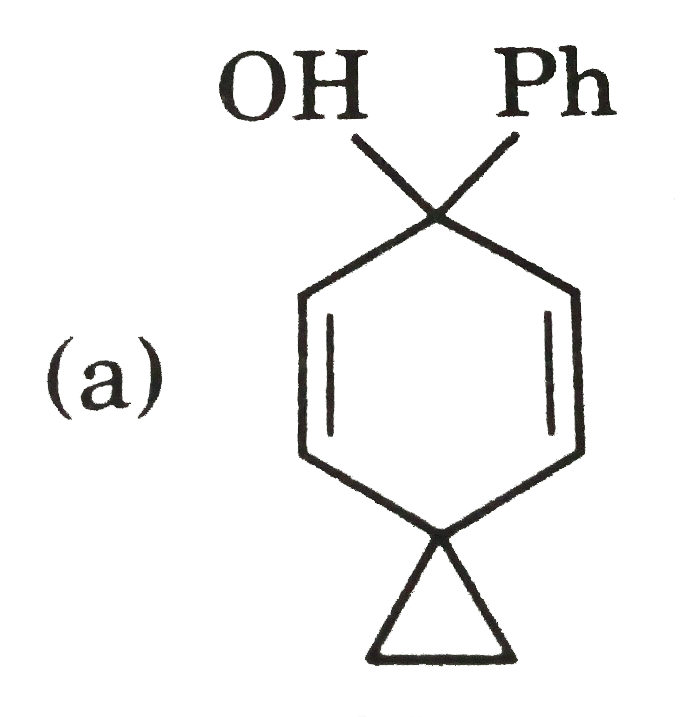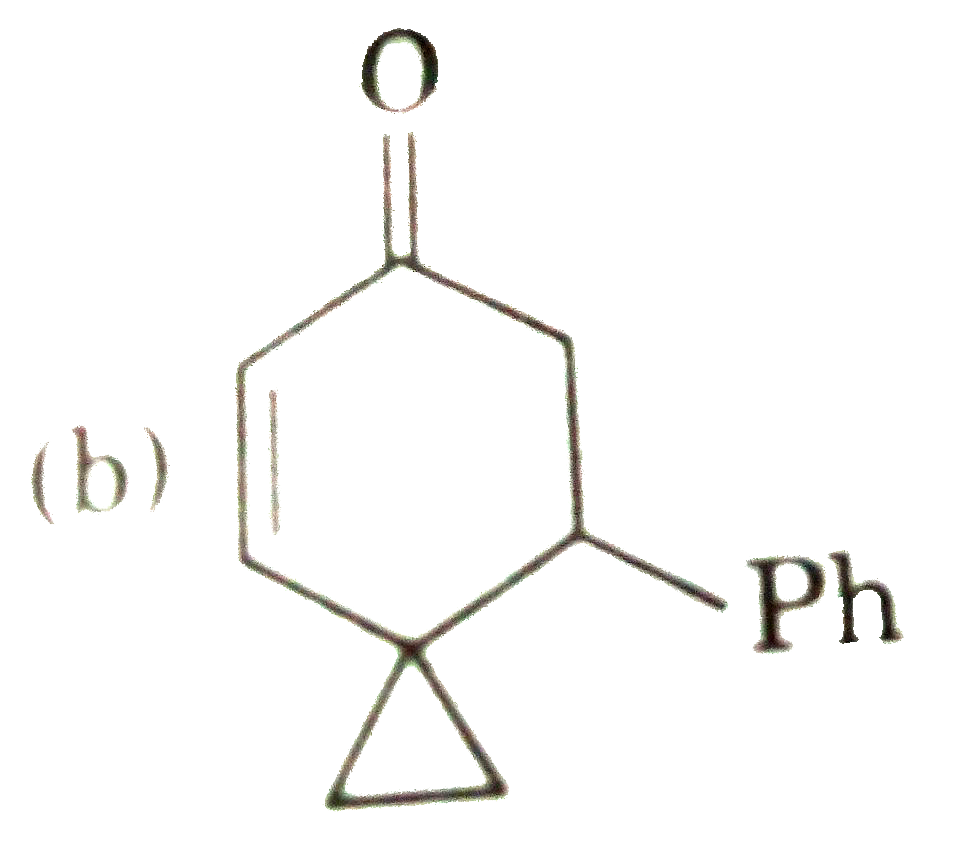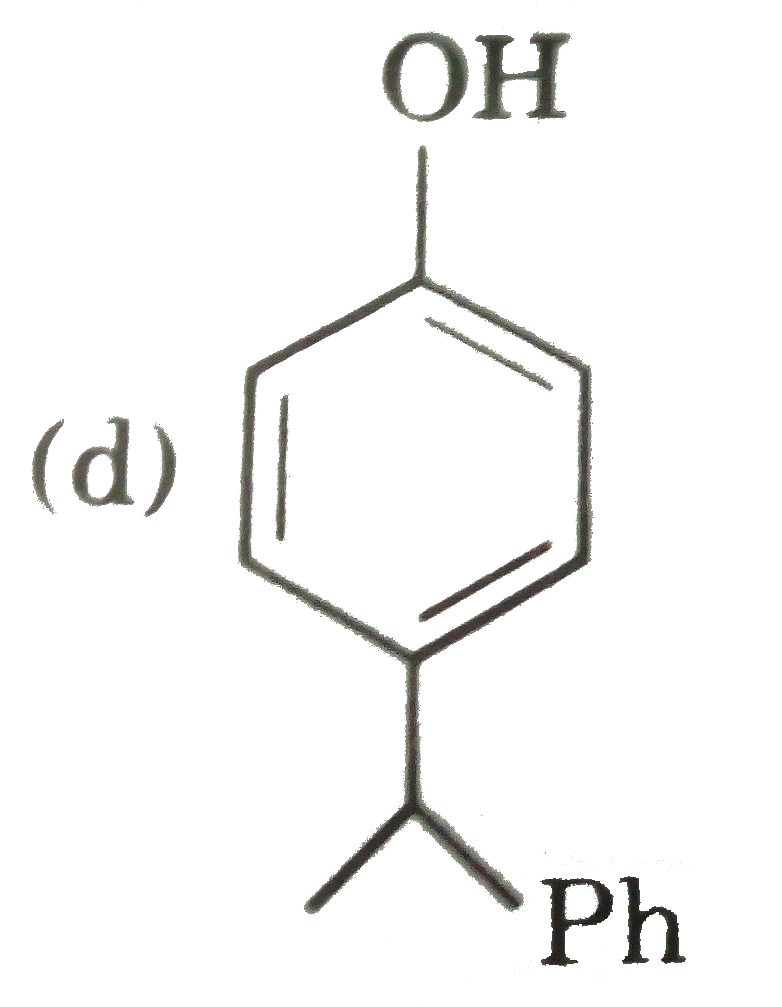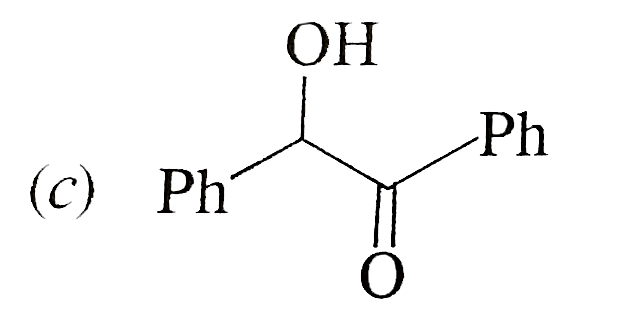Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Potassium bromide, KBr containss, 32.9% by mass of potassium. If 6.40 g of bromine reacts with 3.60 g of potassium, calculate the number of moles of potassium which combine with bromine to form KBr. |
|
Answer» SOLUTION :In KBr, 32.9 g of K are combined with 67.1 g of Br `THEREFORE"3.6 g of K will combine with "Br_(2)=(67.1)/(32.9)xx3.6g=7.34" g which is not PRESENT"` `"or6.4 g or "Br_(2)" will combine with K"=(32.9)/(67.1)xx6.4=3.14g` Thus, `Br_(2)` is the limiting reactant. `"K reacted= 3.14 g"=(3.14)/(36)" MOLE = 0.08 mole"` (K left unreacted `=(3.60-3.14)g=0.46g`) |
|
| 2. |
Potash solution is not recommended in a hydrocarbon - oxygen fuel cell. Why ? |
| Answer» SOLUTION :The combustion products of a hydrocarbon are `CO_2` and `H_2O.CO_2` is a non-metallic OXIDE and acidic. It is absorbed in the electrolyte if KOH is used. To avoid this, phosphoric ACID is used as electrolyte in hydrocarbon-oxygen fuel cell. | |
| 3. |
Potash solution is not recommended in a hydrocarbon-oxygen fuel cell. Why? |
|
Answer» Solution :The combustion products of a HYDROCARBON are `CO_(2)` and `H_(2)O.CO_(2)` is a non-metallic oxide and acidic. It is ABSORBED in the electrolyte if KOH is used. To avoid this, phosphoric acid is used as electrolyte in hydrocarbon-oxygen fuel cell. |
|
| 4. |
Potash alum is water soluble and ionises in aqueous solution to give : |
|
Answer» ONE TYPE of ions |
|
| 5. |
Potash alum is K Al(SO_4)_2. 12H_2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO_4^(2-). The solution is acidic because of the hydrolysis of Al^(3+), but not so acidic as might be expected, because the SO_4^(2-) can sponge up some the H_3O^+ by forming HSO_4^-. Given a solution made by dissolving 11.85 gm of KAl(SO_4)_2.12H_2O in enough water to make 100cm^3 of solution. Match the Column |
|
Answer» |
|
| 6. |
Potash alum is K Al(SO_4)_2. 12H_2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO_4^(2-). The solution is acidic because of the hydrolysis of Al^(3+), but not so acidic as might be expected, because the SO_4^(2-) can sponge up some the H_3O^+ by forming HSO_4^-. Given a solution made by dissolving 11.85 gm of KAl(SO_4)_2.12H_2O in enough water to make 100cm^3 of solution. Only Al3^+ is hydrolysing and its first hydrolsis constant is 1.4 xx 10^(-5)M |
| Answer» Answer :A | |
| 7. |
Potash alum is K Al(SO_4)_2. 12H_2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO_4^(2-). The solution is acidic because of the hydrolysis of Al^(3+), but not so acidic as might be expected, because the SO_4^(2-) can sponge up some the H_3O^+ by forming HSO_4^-. Given a solution made by dissolving 11.85 gm of KAl(SO_4)_2.12H_2O in enough water to make 100cm^3 of solution. Both Al^(3+) and SO_4^(2) are hydrolysing |
| Answer» Answer :A | |
| 8. |
Potash alum is K Al(SO_4)_2. 12H_2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO_4^(2-). The solution is acidic because of the hydrolysis of Al^(3+), but not so acidic as might be expected, because the SO_4^(2-) can sponge up some the H_3O^+ by forming HSO_4^-. Given a solution made by dissolving 11.85 gm of KAl(SO_4)_2.12H_2O in enough water to make 100cm^3 of solution. What is [H3O^+] of the solution if 27. None of the ion is hydrolysing |
| Answer» Answer :A | |
| 9. |
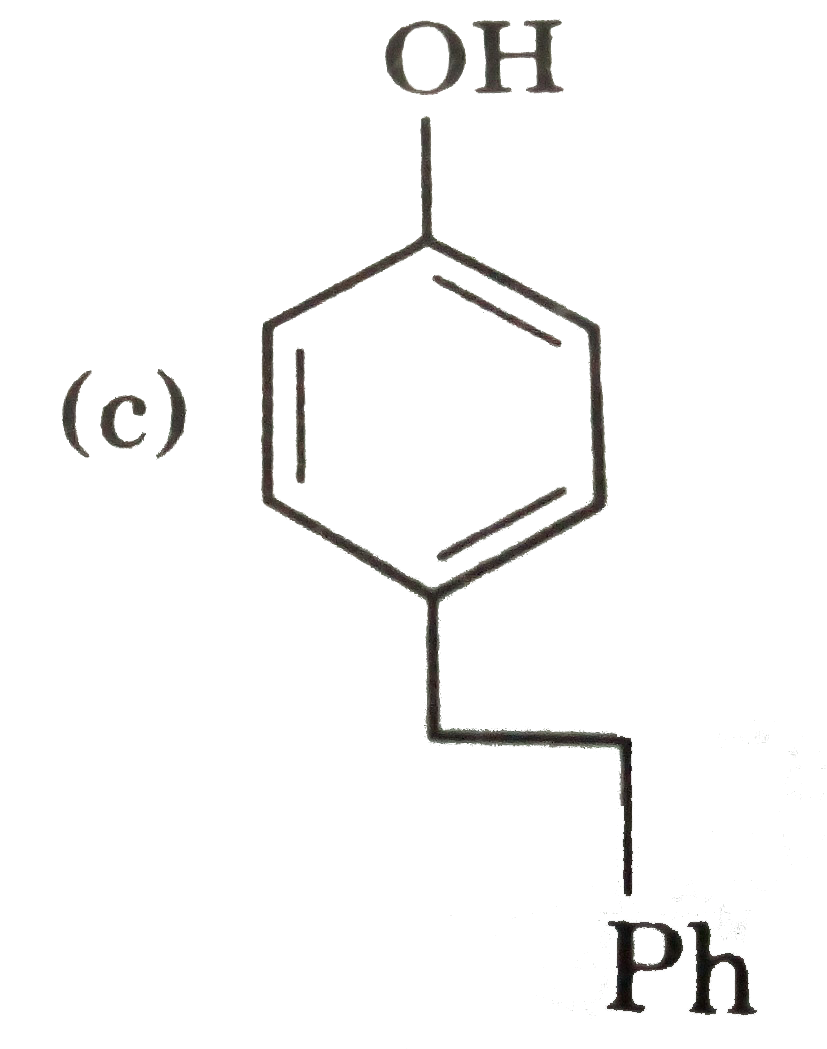
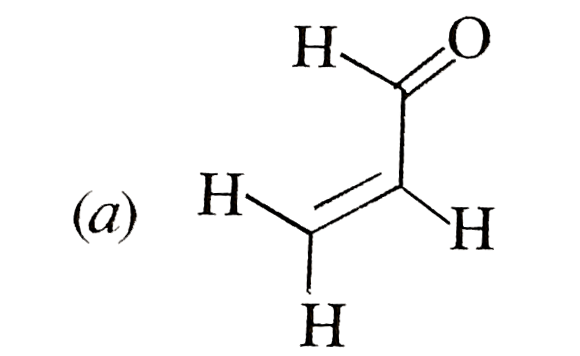
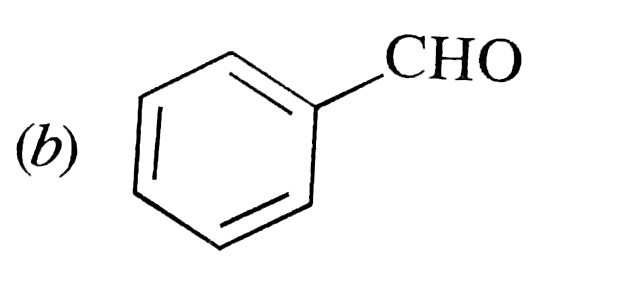
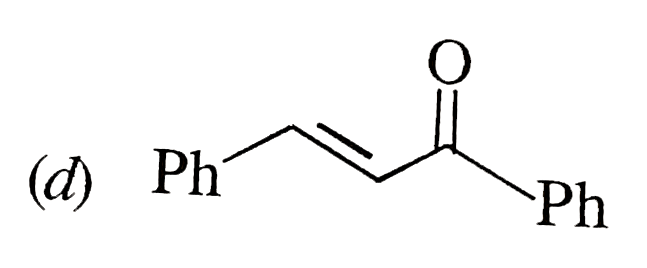
possible product can be : |
|
Answer»
(B) Minor product by 1,4--adentical. |
|
| 10. |
Potash alum is |
|
Answer» `K_(2)SO_(4).Al_(2)(SO_(4))_(3).24H_(2)O` |
|
| 12. |
Possible monochloro derrivative product (excluding stereo isomer) :- |
|
Answer» 4 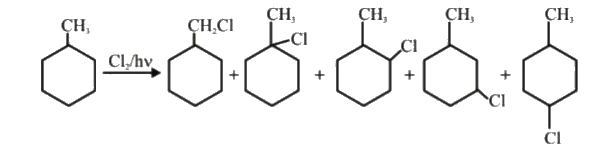 . .
|
|
| 13. |
Possible isomers of monohydric phenol having molecular formula C_(7)H_(8) are ………… |
|
Answer» 3 |
|
| 14. |
Possible isomers of monohydric phenol having molecular formula C_(7) H_(8)Oare .. |
|
Answer» 3 |
|
| 15. |
Positron has nearly the same mass as that of: |
|
Answer» `ALPHA`-particle |
|
| 16. |
Positively charged colloidal solution is |
| Answer» Answer :A | |
| 17. |
" Positronium " is the name given to an aotm like combination formed between : |
|
Answer» A POSITRON and a proton |
|
| 18. |
Positron emission results from the transformation of one nuclear proton into neutron. The isotope thus produced possesses |
|
Answer» Same mass number |
|
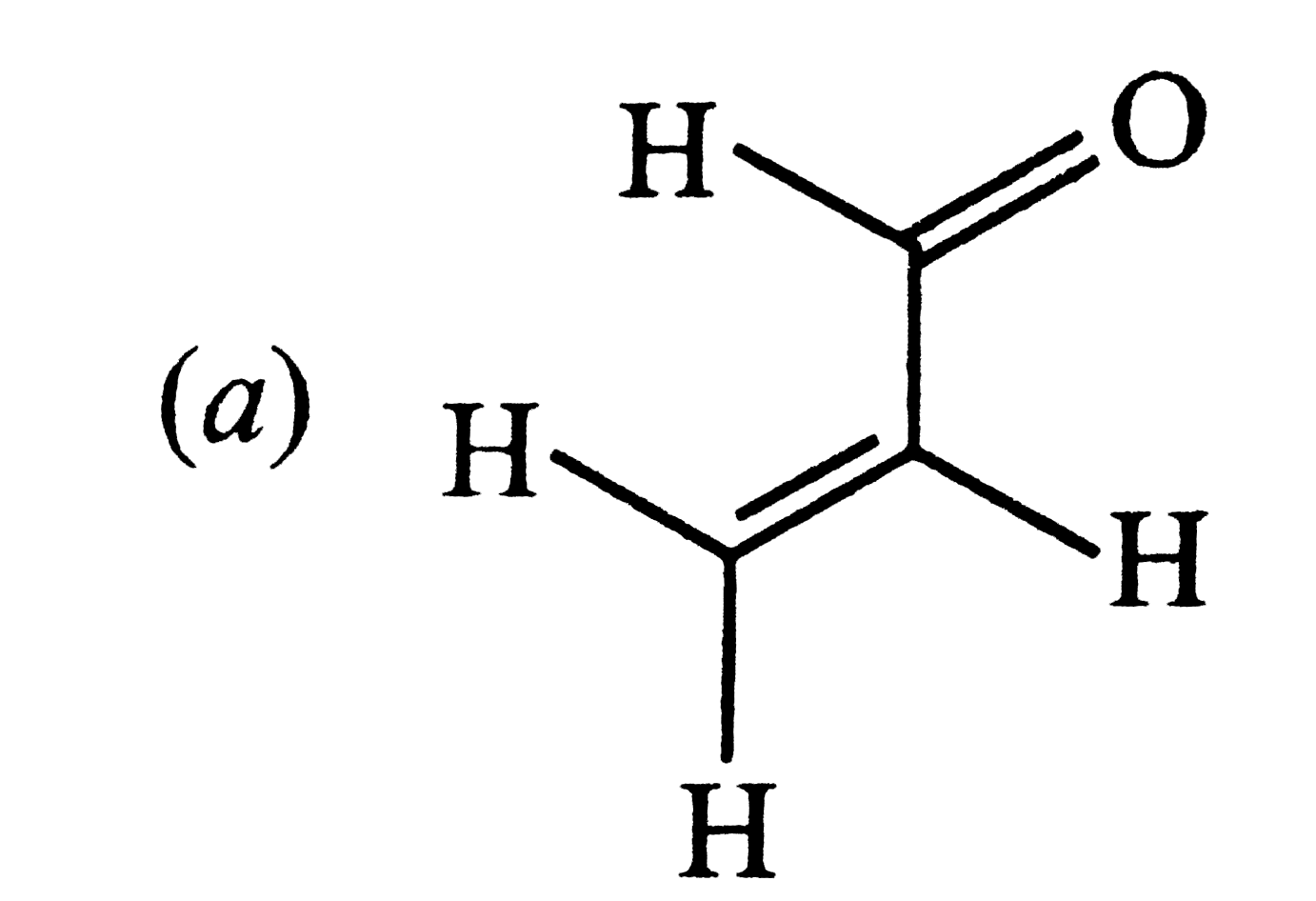
| 19. |
Positive Tollen'stest is observed for |
|
Answer»
 tollen.s TESTIS GIVENBY compoundshavingaldehydicgroupand ` ALPHA ` 0- hydroxycarbonylcompounds |
|
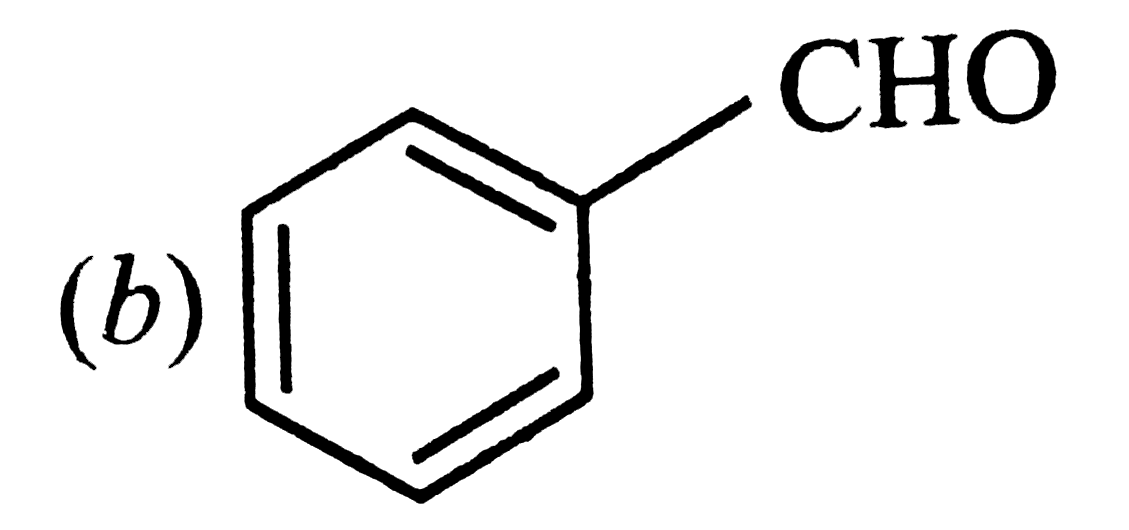
| 20. |
Positive Tollens' test is observed from |
|
Answer»
|
|
| 21. |
Positive value of electron gain enthalpy (Delta H_("eg")) among the following will be of: |
| Answer» ANSWER :D | |
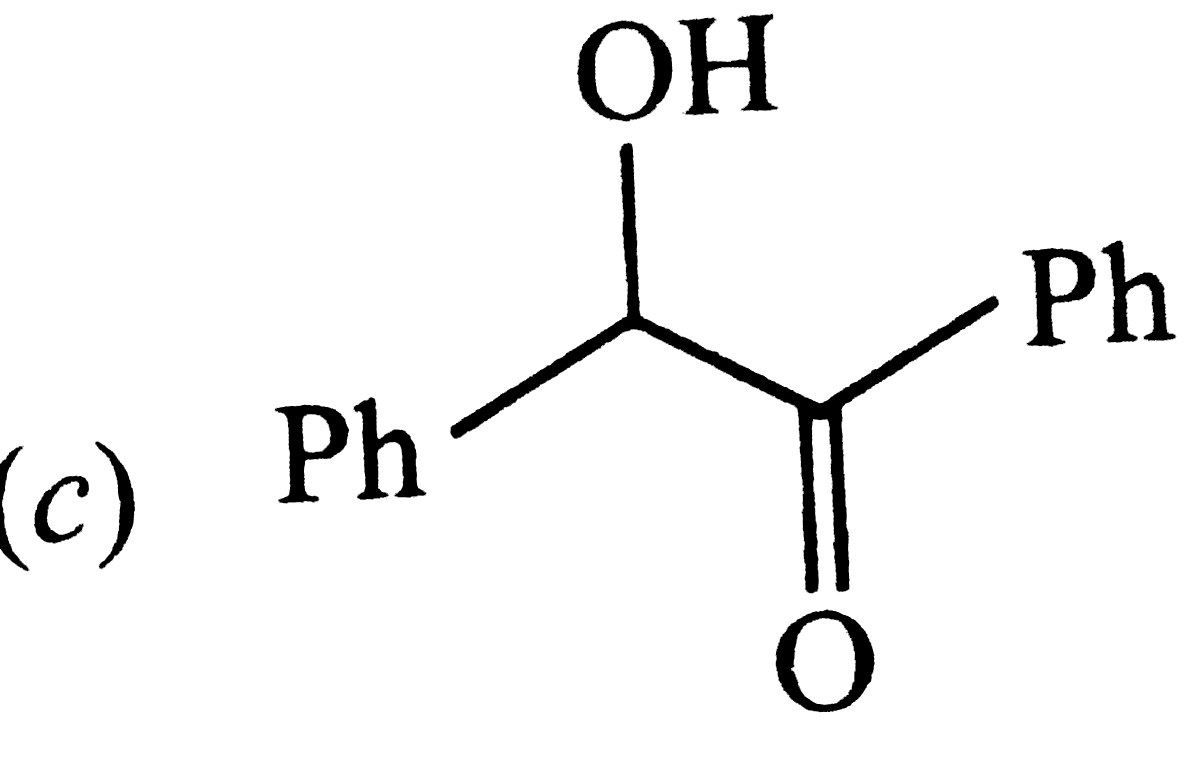
| 22. |
PositiveTollen's test is observed for |
|
Answer»
|
|
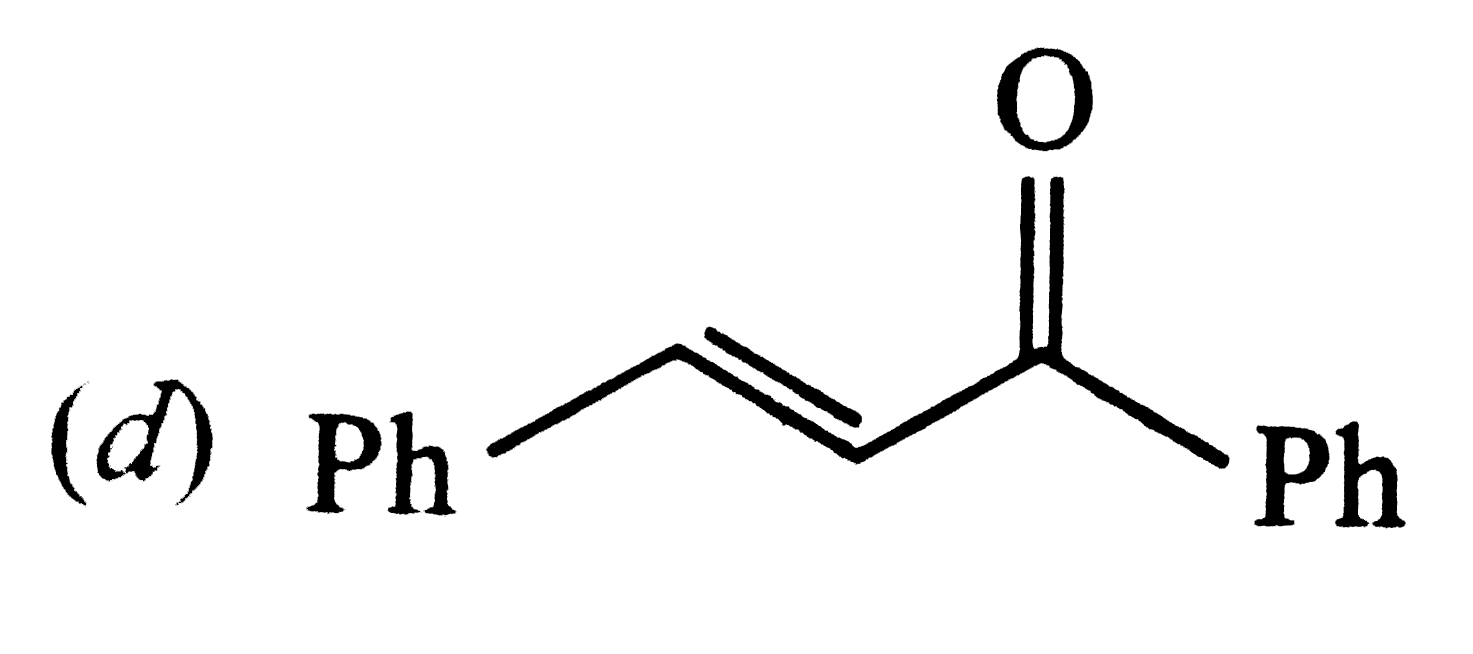
| 23. |
Positive Tollen's test is observed for |
|
Answer»
|
|
| 24. |
Positive pollution of soil is due to: |
|
Answer» EXCESSIVE USE of fertilizers |
|
| 25. |
Positive deviations from ideal behaviour take place because of : |
|
Answer» MOLECULAR interactions between atoms and PV/nRT `lt` 1 |
|
| 26. |
Positive carbylamine test is shown by |
|
Answer» N,N-dimethylaniline |
|
| 27. |
Positive Beilstein test shows that : |
|
Answer» HALOGENS are SURELY present |
|
| 28. |
Position of double bond in an organic compound is determined by |
|
Answer» Ozonolysis `CH_3-OVERSET(H)overset(|)C=O+O=oversetoverset(H)(|)C-CH_3 to CH_3 -UNDERSET"2-Butene"(CH=CH)-CH_3` |
|
| 29. |
What are non-polar and polar covalent bonds? Give examples. |
|
Answer» Polar on OUTER SURFACE and non polar on inner surface |
|
| 30. |
Portland cement is: |
|
Answer» CALCIUM alumino silicate |
|
| 31. |
P–O–P bond is absent in :- |
|
Answer» <P>Dimetaphosphoric ACID  (ii) 2 mole `H_(3)PO_(3) to H_(4)P_(2)O_(5)`  pyrophosphorous acid (iii)  (iv)Hypophosphorous acid - 
|
|
| 33. |
Polyvenyl alcohol is prepared using which starting monomer ? |
|
Answer» Venyl ALCOHOL |
|
| 34. |
Polyvinyl choride is ….. Polymer |
|
Answer» elastomer |
|
| 35. |
Polyvinylalcohol can be prepared by |
|
Answer» polymerization of vinyl alcohol |
|
| 36. |
Polyurethanes: |
|
Answer» <P>Have structure features of both an ester andan amide. |
|
| 37. |
Polythene, PVC, Bakelite, polystrene. |
| Answer» Solution :BAKELITE. It is thermosetting PLASTIC WHEREAS OTHERS are THERMOPLASTIC. | |
| 38. |
Polythene is prepared by the polymerisation of ethane at 350-570 K at high pressure in the presence of traces of__________. |
| Answer» SOLUTION :PEROXIDES | |
| 39. |
Polysubstitution occurs forwhich of the following reactions- |
|
Answer» `C_6H_6+CH_3CH_2CH_2Cl overset("ANHYDROUS" AlCl_3)to` |
|
| 40. |
Polythene is a resin obtained by polymerisation of |
|
Answer» Butadiene |
|
| 41. |
Polythene, PVC are _____. |
| Answer» SOLUTION :HOMOPOLYMER | |
| 42. |
polythene is a\an____. |
|
Answer» ADDITION copolymer |
|
| 43. |
Polythene is a ................homopolymer while bakeline is a ..........copolymer. |
|
Answer» |
|
| 45. |
Polystyrene is an example of _______ polymer. |
|
Answer» CONDENSATION |
|
| 46. |
Polystyrene is of which type polymer ? |
|
Answer» NATURAL |
|
| 47. |
Polystyrene is not an example of addition polymer. |
| Answer» SOLUTION :NYLON is not an EXAMPLE of ADDITION POLYMER. | |
| 48. |
Polystyrene is a : |
|
Answer» Addition POLYMER |
|
| 49. |
Polystyrene , dacron and orlon are classified respectively as |
|
Answer» CHAIN GROWTH, STEP growth , step growth |
|
| 50. |
Polysaccharides have … linkages. |
|
Answer» GLYCOSIDIC |
|