Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Predict al the alkenes that would be formed by dehyddrohalogenation of the following halides with sodium ethoxide in ethanol and identify the major alkene. (i) 1-Bromo-1-methylcyclohexane (ii) 2-Chloro-2-methylbutane (iii) 3-Bromo-2,2,3-trimethylpentane. |
Answer» SOLUTION :(i) In 1-bromo-1-methylcyclohexane, the `beta`-hydrogens on either side of the Br atom are equivalent, THEREFORE, only 1-alkene is formed.  (ii) 2-Chloro-2-methylbutane has two different sets of equivalent `beta`-hydrogens and hence, in PRINCIPLE, can GIVE two alkenes (I and II). but according to Sayzeff rule, more highly substituted alknene (II), being more stable, is the major product. `underset("2-Chloro-2-methylbutane")(overset(beta)(C)H_(3)-underset(Cl)underset(|)overset(overset(beta)(C)H_(3))overset(|)(C)-overset(beta)(C)H_(2)CH_(3)) underset(-(HCl))overset(C_(2)H_(5)ON a//C_(2)H_(5)OH)to underset("2-Methylbut-1-ene (I) (Less stable, minor product)")(CH_(2)=overset(CH_(3))overset(|)(C)-CH_(2)CH_(3))+underset("2-Methylbut-2-ene (II) (More stable, major product)")(CH_(3)-overset(CH_(3))overset(|)(C)=CHCH_(3))` (iii) 3-Bromo-2,2,3-trimethylpentane has two different sets of `beta`-hydrogens and hence, in principle, can give two alkenes (I and II)). but according to Saytzeff rule, more highly substituted alkene (II), being more stable, is the major product. 
|
|
| 2. |
Predict A,B, C and D for the following reaction |
Answer» SOLUTION :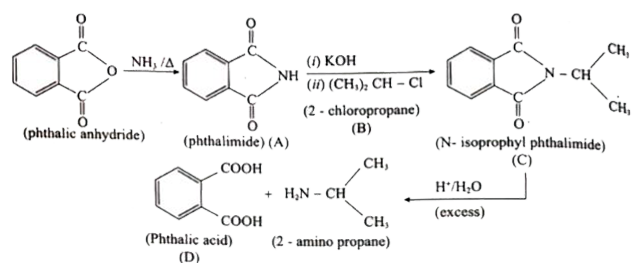
|
|
| 3. |
Predic whichof the followingwillcolouredin aqueoussolutionTi^(2+), V^(3+) Sc^(4+) Cu^(+) ,Sc^(3+) ,Fe^(3+) ,Ni^(2+)and Co^(3+) |
|
Answer» Solution :(I ) Onlythe ionsthat haveunpairedelectronsin d-orbitaland in whichd- dtransitionis possiblewill becoloured. (ii) The ionsin whichd - orbitalsareemptyor COMPLETELY filledwill be colourlessas nod-dtransitionis possiblethoseconfiguratin Fromthe aboveions , it canbe easilyobservedthat only`SC^(3+)`hasan EMPTYD- ORBITAL and `Cu^(+)` hascompletely filledd- orbital. (iv)All otherions except `Sc^(3+)`and `Cu^(+)`will be COLOURED inaqueoussolutionbecauseofd-d transition. |
|
| 4. |
Precision refers to the closeness of a set of values obtained for identical measurement of a quantity. Precision depends on the limitations of measuring devices and the skills with which it is used. However, accuracy refers to the closeness of a single measurement to its true value. The digits in a properly measurement are known as significant figures. these are meaningful digits in a measured or calculated quantity. The greater the number of significant figures in a reported result, smaller is the uncertainly and greater is the precision. the zeros at the beginning are not counted. the zeros to the right of a decimal point are counted. in the numbers that do not contain a decimal point. "trailing" zeros may or may not be significant . the purpose of zeros at the end of anumber is to convey the correct range of uncertainly. If repeated measurements give values close to one another, the number is: |
|
Answer» SURELY PRECISE |
|
| 5. |
Precision refers to the closeness of a set of values obtained for identical measurement of a quantity. Precision depends on the limitations of measuring devices and the skills with which it is used. However, accuracy refers to the closeness of a single measurement to its true value. The digits in a properly measurement are known as significant figures. these are meaningful digits in a measured or calculated quantity. The greater the number of significant figures in a reported result, smaller is the uncertainly and greater is the precision. the zeros at the beginning are not counted. the zeros to the right of a decimal point are counted. in the numbers that do not contain a decimal point. "trailing" zeros may or may not be significant . the purpose of zeros at the end of anumber is to convey the correct range of uncertainly. In which of the following numbers, all the zeros are not significant? |
|
Answer» 0.001 |
|
| 6. |
Precision refers to the closeness of a set of values obtained for identical measurement of a quantity. Precision depends on the limitations of measuring devices and the skills with which it is used. However, accuracy refers to the closeness of a single measurement to its true value. The digits in a properly measurement are known as significant figures. these are meaningful digits in a measured or calculated quantity. The greater the number of significant figures in a reported result, smaller is the uncertainly and greater is the precision. the zeros at the beginning are not counted. the zeros to the right of a decimal point are counted. in the numbers that do not contain a decimal point. "trailing" zeros may or may not be significant . the purpose of zeros at the end of anumber is to convey the correct range of uncertainly. If we add 296.2 and 2.256, we get the answer as 298.456g. The number of significant figures in the result are: |
|
Answer» 6 |
|
| 7. |
Precision refers to the closeness of a set of values obtained for identical measurement of a quantity. Precision depends on the limitations of measuring devices and the skills with which it is used. However, accuracy refers to the closeness of a single measurement to its true value. The digits in a properly measurement are known as significant figures. these are meaningful digits in a measured or calculated quantity. The greater the number of significant figures in a reported result, smaller is the uncertainly and greater is the precision. the zeros at the beginning are not counted. the zeros to the right of a decimal point are counted. in the numbers that do not contain a decimal point. "trailing" zeros may or may not be significant . the purpose of zeros at the end of anumber is to convey the correct range of uncertainly. In the number 2.4560, there are 5 significant digits. which one is the least significant digit? |
|
Answer» 2 |
|
| 8. |
Precision refers to the closeness of a set of values obtained for identical measurement of a quantity. Precision depends on the limitations of measuring devices and the skills with which it is used. However, accuracy refers to the closeness of a single measurement to its true value. The digits in a properly measurement are known as significant figures. these are meaningful digits in a measured or calculated quantity. The greater the number of significant figures in a reported result, smaller is the uncertainly and greater is the precision. the zeros at the beginning are not counted. the zeros to the right of a decimal point are counted. in the numbers that do not contain a decimal point. "trailing" zeros may or may not be significant . the purpose of zeros at the end of anumber is to convey the correct range of uncertainly. The number of significant figures in a measured number contained how many uncertain number of digits? |
|
Answer» Zero |
|
| 9. |
Precipitation takes place when the producty of concentrationof ions : |
|
Answer» equals the solubility PRODUCT |
|
| 10. |
Precipitation occurs when |
|
Answer» Salt is able to form SUPERSATURATED solution |
|
| 11. |
Precipitate of group IV cations takes place when H_(2)S is |
|
Answer» HIGHLY ionised |
|
| 12. |
Precipitates are converted into colloids by addition of electrolyte is called .. . |
|
Answer» hydrolysis |
|
| 13. |
Precipitateof Na_(2)CO_(3) extract is made for acidradicalanalysdis because |
|
Answer» All ANIONS react with NA to give WATER soluble compound |
|
| 14. |
Precipitate of Cd^(2+) and Cu^(2+) takes place in presence of (a)____by (b)____. |
|
Answer» |
|
| 15. |
Precipitate of AgCl is soluble in liquid NH_(3) the compound forms |
|
Answer» `Ag(NH_4)_2OH` |
|
| 16. |
Precentage of argon in air is about: |
| Answer» Answer :D | |
| 17. |
Precentagecompositionof an organic compounds is as follows, C = 10.06 , H = 0.84, Cl = 89.10. Which of the following corresponds to its molecular formula if the vapour density is 60.0 |
| Answer» Solution :Molecular of weight of `CHCl_(3)` is `120`. | |
| 18. |
Percentage of nitric acid obtained in Ostwald's process is |
| Answer» ANSWER :2 | |
| 19. |
Pre -exponential factorfor areaction is 8.4xx 10^(21)molL^(-1) s^(-1) . Whatwill be the specific rateattemperatureof one million degree ? |
|
Answer» Solution :ARRHENIUS equation for the specific rate, `k=Ae^(-Ea//RT)` Since temperature T is very large, `T^(-1)` tends to ZERO and `e^(@)` becomes equal to `1`. Specific rate of the reaction, `k=A=8.4xx10^(21)"mol"L^(-1)s^(-1)`. |
|
| 20. |
Pr, Nd, Tb & Dy show +4 oxidation state in the form of - |
|
Answer» CHROMATES & HALIDES |
|
| 21. |
P,Q, R and S are four metals whose typical reaction are given below (I) (II) When Q is added to a solution containing the ions of the other metals, metallic P, R, and S are formed (III) P reacts with concentrated HNO_(3) but S does not The correct order of their reducing character is |
|
Answer» <P>`S LT P lt R lt Q` |
|
| 22. |
ppi-ppi bondingis possible in |
|
Answer» N |
|
| 23. |
Power alcohol is a mixtureof |
|
Answer» `80% ` petrol + 20% BENZENE + SMALL quantity of ethanol |
|
| 24. |
Power alcohol is a mixture of petrol and alcohol in the ratio : |
|
Answer» `4:1` |
|
| 25. |
Power alcohol is a mixture of |
|
Answer» 80 % Petrol + 20 % Benzene + Small quantity of ETHANOL |
|
| 26. |
Powdered silver oreis treated with NaCN solution and air is bubbled through the mixture to give : |
| Answer» ANSWER :D | |
| 27. |
Powdered silver ore is rated with NaCN solution and all bubled through the mixture to give |
|
Answer» `AgCN` `4g_(2)+4NaCN hArr 2NA[Ag(CN)_(2)]+Na_(2)S` `4Na_(2)S+SO_(2)+2H_(2) to 2Na_(2)+4NaOH+2S` |
|
| 28. |
Poverset(PhMgBr)underset(H^(oplus)//H_(2)O)rarrCH_(3)-overset(OH)overset(|)underset(Ph)underset(|)C-Ph Identify structures of P : |
|
Answer» `CH_(3)-OVERSET(O)overset("||")C-Cl` |
|
| 29. |
potochemicalchlorinationof alkaneis initiated byprocessof : |
|
Answer» pyrolysis |
|
| 30. |
Potential of electrochemical cell does not depend on which of the following ? |
|
Answer» CONCENTRATION of SOLUTION |
|
| 31. |
Potential of a hydrogen electrode at one atmosphere in 0.48 V if the P^(H) value is _________ |
| Answer» Solution :`((-E_("RP"))/(0.0591)) = - log[H^(+)] = P^(H) , P^(H) = (0.48)/(0.0591) = 8.121 = 8` | |
| 32. |
Potato starch is less stable than gelatin. Why? |
|
Answer» SOLUTION :(i) The gold number of gelatin is 0.005 - 1 and the gold number of potato starch is 25. (II) Smaller the gold number, greater the protective power. |
|
| 33. |
Potassium superoxide ,KO_(2), is utilised in closed system breathing apparatus. Exhaled air contains CO_(2) and H_(2)O ,both of which are removed and the removal of water generates oxygen for breathing by the reaction 4KO_(2)(s) + 2H_(2)O(l) to 3O_(2)(g) + 4KOH(s) The potassium hydroxide removes CO_(2) from the apparatus by the reaction : KOH(s) + CO_(2)(g)to KHCO_(3)(s) (a) What mass of KO_(2) generates 24 gm of oxygen ? (b) What mass of CO_(2) can be removed from from the apparatus by 100 gm of KO_(2) ? |
|
Answer» |
|
| 34. |
Potassium superoxide,KO_(2), is used in rebreathing gas masks to generate oxygen : ""KO_(2)(s) + H_(2)O(I) to KOH +O_(2)(g) If a reaction vessel contains 0.158 mol KO_(2) and 0.10 mol H_(2)O , how many moles of O_(2) can be produced? |
|
Answer» |
|
| 35. |
Potassium when heated strongly in oxygen, it forms: |
|
Answer» K_2O` |
|
| 36. |
Potassium superoxide, KO_2 , is used in rebreathing gas masks to generate oxygen. 4KO_2(s) + 2H_2O(l) to 4KOH(s) + 3O_2(g) If a reaction vessel contains 0.15 mole of KO_2and 0.10 mole of H_2O , what is the limiting reactant? How many moles of oxygen can be produced? |
| Answer» SOLUTION :`KO_2 , 0.1125` MOLE | |
| 37. |
Potassium salt of isophthalic acid on acid hydrolysis gives |
|
Answer» PHTHALIC ACID |
|
| 38. |
Potassium sulphate is a strong electrolyte which dissociates completely in aqueous solution. Urea is a nonelectrolyte substance. A 0.01 molar solution (aq) of potassium sulphate depressed the f.p. of water by 0.0558^@C . What will be the depression caused by a 0.01 molar solution of urea? |
| Answer» SOLUTION :`0.0186^@C` | |
| 39. |
Potassium pthalimide on reaction wiith (A) followed by hydrolysis forms isopentyl amine. Compound (A) is |
|
Answer» `CH_(3)-CH_(3)underset(|)(C)Hoverset(BR)overset(|)(C)HCH_(2)` |
|
| 40. |
Potassium phthalimidereact withA whichon hydrolysis given2- methyl propan -1- amine . Whatis 'A' ? |
|
Answer»
|
|
| 41. |
Potassium permangante on heating gives : |
|
Answer» `K_(2)MnO_(4), MnO_(2), O_(2)` |
|
| 42. |
Potassium phthalimideis reactedwithethyl halideandfollowedby acidhydrolysis gives |
|
Answer» ethanol |
|
| 43. |
Potassium permanganate titrations are not performed in the presence of hydrochloric acid because |
|
Answer» HCL is not available in pure form. |
|
| 44. |
Potassium permanganate is prepared from the mineralpyrolusite, MnO_(2). Its crystals have deep purple colour. It acts as an oxidizing agentin the neutral, alkaline as well as acidicmedium. In acidicmedium , it is used in volumetric analysis for estimationof ferrous salts, oxalates etc. The titrations are carried out in presence of H_(2)SO_(4). However ,before using it as a titrant, it is first standized with standard oxalic acid solution or Mohr salt solution. In one of the experiments on titration , 13.4 g of dry pure sodium oxalate ( molar mass =134 g mol^(-1) ) was dissolvedin 100mLof distilled waterand then 100 mL of 2MH_(2)SO_(4) were added. The solution, was cooledto 25.30^(@) C. Now to this solution , 0.1 M KMnO_(4) solution was added till a very faint pink colour persisted. If of 0.1 M K_(2)Cr_(2)O_(7) solution were usedin place 0.1 M KMnO_(4) in presenceof H_(2)SO_(4) in each case, the volume of 0.1 MK_(2)Cr_(2)O_(7) solution used would be |
|
Answer» 200 mL `Cr_(2)O_(7)^(2-) + 16 H^(+) + 3C_(2)O_(4)^(2-) rarr 2Cr^(3+) + 7H_(2)O + 6CO_(2)` Moles of `K_(2)Cr_(2)O_(7)` required for 0.1 molof sodium oxalate `= (1)/(3) xx 0.1= 0.03333`.Hence , volume of 0.1 M `K_(2)Cr_(2)O_(7)` solution required `= 33.3mL` |
|
| 45. |
Potassium permanganate is used as a ul"reducing agent" in acidic medium. |
| Answer» SOLUTION :REDUCING AGENT | |
| 46. |
Potassium permanganate is prepared from the mineralpyrolusite, MnO_(2). Its crystals have deep purple colour. It acts as an oxidizing agent in the neutral, alkaline as well as acidicmedium. In acidicmedium , it is used in volumetric analysis for estimationof ferrous salts, oxalates etc. The titrations are carried out in presence of H_(2)SO_(4). However ,before using it as a titrant, it is first standized with standard oxalic acid solution or Mohr salt solution. In one of the experiments on titration , 13.4 g of dry pure sodium oxalate ( molar mass =134 g mol^(-1) ) was dissolvedin 100mLof distilled waterand then 100 mL of 2MH_(2)SO_(4) were added. The solution, was cooledto 25.30^(@) C. Now to this solution , 0.1 M KMnO_(4) solution was added till a very faint pink colour persisted. Mohr salt, FeSO_(4). (NH_(4))_(2) SO_(4). 6 H_(2)O is preferred over FeSO_(4). 7 H_(2)O for standardization of KMnO_(4) solution because. |
|
Answer» Mohr salt is a DOUBLE salt while ferrous sulphate is a single salt. |
|
| 47. |
Potassium permanganate is prepared from the mineralpyrolusite, MnO_(2). Its crystals have deep purple colour. It acts as an oxidizing agentin the neutral, alkaline as well as acidicmedium. In acidicmedium , it is used in volumetric analysis for estimationof ferrous salts, oxalates etc. The titrations are carried out in presence of H_(2)SO_(4). However ,before using itas a titrant, it is first standized with standard oxalic acid solution or Mohr salt solution. In one of the experiments on titration , 13.4 g of dry pure sodium oxalate ( molar mass =134 g mol^(-1) ) was dissolvedin 100mLof distilled water and then 100 mL of 2MH_(2)SO_(4) were added. The solution, was cooled to 25.30^(@) C. Now to this solution , 0.1 M KMnO_(4) solution was added till a very faint pink colour persisted. Th evoluem of KMnO_(4) solution that must have been added to obtain the faint pink colour at the end point must be |
|
Answer» 100mL `2MnO_(4)^(-)+ 16 H^(+) + 5CrO_(4)^(2-) rarr 2Mn^(2+) + 8 H_(2)O + 10 CO_(2)` `:. ` Moles of `KMnO_(4)` used `= ( 2)/( 5) xx 0.1= 0.04 ` MOL As`KMnO_(4)` solution used is `0.1 M` , i.e., 0.1 mol are present in 1000 mL, therefore , 0.04mol will be present in 400 mL. |
|
| 48. |
Potassium permanganate is prepared from the mineralpyrolusite, MnO_(2). Its crystals have deep purple colour. It acts as an oxidizing agent in the neutral, alkaline as well as acidicmedium. In acidicmedium , it is used in volumetric analysis for estimationof ferrous salts, oxalates etc. The titrations are carried out in presence of H_(2)SO_(4). However ,before using it as a titrant, it is first standized with standard oxalic acid solution or Mohr salt solution. In one of the experiments on titration , 13.4 g of dry pure sodium oxalate ( molar mass =134 g mol^(-1) ) was dissolvedin 100mL of distilled water and then 100 mL of 2MH_(2)SO_(4) were added. The solution, was cooledto 25.30^(@) C. Now to this solution , 0.1 M KMnO_(4) solution was added till a very faint pink colour persisted. Which pyrolusite is fused with KOH andKClO_(3), we get |
|
Answer» `KMnO_(4)` |
|
| 49. |
Potassium permanganate is prepared from the minera lpyrolusite, MnO_(2). Its crystals have deep purple colour. It acts as an oxidizing agent in the neutral, alkaline as well as acidicmedium. In acidicmedium , it is used in volumetric analysis for estimationof ferrous salts, oxalates etc. The titrations are carried out in presence of H_(2)SO_(4). However ,before using itas a titrant, it is first standized with standard oxalic acid solution or Mohr salt solution. In one of the experiments on titration , 13.4 g of dry pure sodium oxalate ( molar mass =134 g mol^(-1) ) was dissolvedin 100mL of distilled waterand then 100 mL of 2MH_(2)SO_(4) were added. The solution, was cooled to 25.30^(@) C. Now to this solution , 0.1 M KMnO_(4) solution was added till a very faint pink colour persisted. If instead of H_(2)SO_(4), HCl or HNO_(3) of suitable concentration were used, the volume ofKMnO_(4) soltuionused would have been |
|
Answer» less in case of HCL but more in case of `HNO_(3)` |
|
| 50. |
Potassium permanganate is prepared from the mineral pyrolusite, MnO_(2). Its crystals have deep purple colour. It acts as an oxidizing agent in the neutral, alkaline as well as acidicmedium. In acidicmedium , it is used in volumetric analysis for estimationof ferrous salts, oxalates etc. The titrations are carried out in presence of H_(2)SO_(4). However ,before using itas a titrant, it is first standized with standard oxalic acid solution or Mohr salt solution. In one of the experiments on titration , 13.4 g of dry pure sodium oxalate ( molar mass =134 g mol^(-1) ) was dissolvedin 100mLof distilled water and then 100 mL of 2MH_(2)SO_(4) were added. The solution, was cooled to 25.30^(@) C. Now to this solution , 0.1 M KMnO_(4) solution was added till a very faint pink colour persisted. The purple colour of KMnO_(4)is due to |
|
Answer» incompleted-subhsell |
|



