Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Plaster of paris is hardened by: |
|
Answer» GIVING off `CO_2` |
|
| 2. |
Plaster of paris is : |
|
Answer» `CaSO_(4)` |
|
| 3. |
Plaster of paris in contact with water sets into a hard mass. The composition of hard mass is: |
|
Answer» `CaSO_4.H_2O` |
|
| 4. |
Plants synthesise …………………… and ………………………… to protect then from being eaten away by insects and other animals. |
|
Answer» |
|
| 5. |
Platinized asbestos used as a catalyst in the manufacture of H_2SO_4 is an example of: |
|
Answer» HETEROGENEOUS catalysis |
|
| 7. |
Plane polarized light. |
| Answer» SOLUTION :A RAY of monochromatic light in which vibrations of the radiation WAVES take place only in one plane is called plane POLARIZED light. | |
| 8. |
Placedo' is often given to patients. It is |
|
Answer» A tonic |
|
| 9. |
Placedo' is often given to patients. It is : |
|
Answer» An ANTIDEPRESSANT |
|
| 10. |
Placedo. is often given to patients. It is : |
|
Answer» An ANTIDEPRESSANT |
|
| 11. |
Place the following alcohols in decreasing order of rate of dehydration with conc. H_2SO_4 . (1)CH_3CH_2CH(OH)CH_2CH_2CH_3 (2)(CH_3)_2C(OH)CH_2CH_2CH_3 (3)CH_3CH_2CH_2CH_2CH_2CH_2-OH (4)(CH_3)_2C(OH)CH(CH_3)_2 |
|
Answer» 4 gt 2 gt 1 gt 3 |
|
| 12. |
Placebo is often given to patients it is |
|
Answer» A SUGAR PILL |
|
| 13. |
pK_(a1) and pK_(a_(2)) of H_(2)CO_(3) are 6.38 and 10.26 respectively. The pH of 1 M and 0.1 M NaHCO_(3) are |
|
Answer» 8.32,7.32 |
|
| 14. |
pK_(a) values of two acids A and B are 4 and 5. The strengths of these two acids are related as |
|
Answer» ACID A is 10 times STRONGER than acids B We KNOW that `pK_(a) = -log K_(a)` `:.` Acid A `K_(a) = 10^(-4)`, Acid `B K_(a) = 10^(-5)` Hence A is ten times stronger than that of B. |
|
| 15. |
pk_a values of two acids A and B are 4 and 5. The strength of these two acids are related as |
|
Answer» acid A is 10 TIMES STRONGER than acid B `K_a"of"B=10^(-5)` i.e,A is10timesstrongerthanB. |
|
| 16. |
P^(ka) values of different hydro halic acids in random is 3.2, 9.5 -10 and -7. So In aq. Solution HCl has an Ka value |
|
Answer» `10^(6)` |
|
| 17. |
pK_(a)value of 4-nitrobenzoic acid is lower than that of benzoic acid. Why ? |
| Answer» Solution :`pK_(a)`VALUE of 4-nitrobenzoic acid is lower than benzoic acid is DUE to `e^(–)`withdrawing nature of —`NO_(2)`attach at PARA position of BENZENE due to which tendency to loose ` H^(+)`ion increases and acidic character increases. | |
| 19. |
pK_(a) value of the strongest acid among the following is |
|
Answer» `3.0` |
|
| 20. |
pK_(a) of weak acid (HA) and pK_(b) of a weak base (BOH) are 3.2 and 3.4, respectively. The pH of their salt (AB) solution is |
|
Answer» Solution :Salt of weak ACID and weak base `PH = (1)/(2)(pk_(w)+ pK_(a) - pK_(b))` `= (1)/(2)(14 + 3.2 - 3.4)` = 6.9 |
|
| 21. |
pKa of waek acid is 3.0. Effective range of a buffer of HA and A^(-) is about pH :- |
| Answer» Answer :4 | |
| 22. |
pK_(a) of chloroacetic acid is lower than pK_(a) of acetic acid. Explain. |
|
Answer» Solution :`pK_(a)` of chloroacetic acid is lower than `pK_(a)` of acetic acid. This MEANS, chloroacetic acid is a stronger acid than acetic acid. This may be explained on the basis of the following two reasons: (i) DUE to -I-effect of Cl atom, the electron density in the O-H bond in chloroacetic acid is much lower than due to +I-effect of `CH_(3)` GROUP in acetic acid. as a result, O-H bond in chloroacetic acid is much weaker than in acetic acid and hence loses a proton more easily acid. 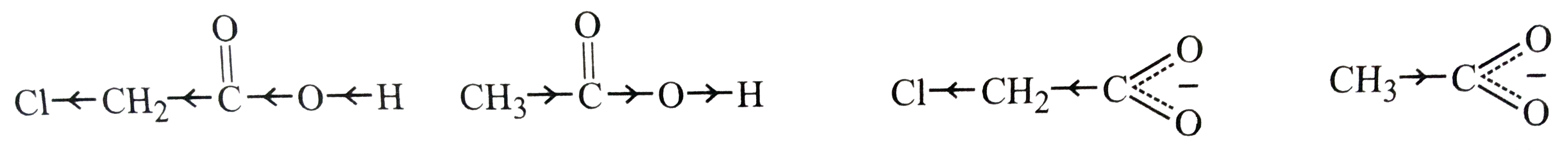 (ii) Due to -I-effect of Cl, dispersal of the -ve charge occurs in chloroacetate ion but due to +I-effect of `CH_(3)` group, intensification of -ve charge occurs in acetate ion. in other words, chloroacetate ion is much more stable than acetate ion. |
|
| 23. |
pK_(a) of acetic acid is 4.74. The concentration of CH_(3)COONa is 0.01 M. The pH of CH_(3)COONa is |
|
Answer» 8.37 `= 7 + (1)/(2)[4.74 -2] = 7+ (2.74)/(2) = 8.37` |
|
| 24. |
pK_(a) of a weak acid is defined as |
|
Answer» `log_(10)K_(a)` |
|
| 25. |
pK_(a) for acetic acid is 4.74. What should be the ratio of concentration of acetic acid and acetate ions to has a solution with pH = 5.74 ? |
|
Answer» `1 : 10`  , (RETENTION of CONFIGURATION) , (RETENTION of CONFIGURATION)
|
|
| 26. |
pK_(a) and K_(a) of an acid are connected by the relation....... |
|
Answer» |
|
| 27. |
pK_a for acetic acid is 4.74. The concentration of CH_3COONa is 0.01 M. The pH of CH_3COONa is……. |
|
Answer» 3.37 `pH=-LOG(K_a.C)^(1/2)` `=1/2[-log K_a-log c]` `=1/2[4.74-log 10^-2]` `=1/2[4.74+2]=3.37` `pH=3.37` |
|
| 28. |
pK_(a) and K_(a) of an acid are connected by the relation ______. |
| Answer» SOLUTION :`pK_(a)=-logK_(a)`. | |
| 29. |
pKa values of two acids A and B are 4 and 5. The strengths of these two acids are related as |
|
Answer» the strengths of the TWO acid cannot be compared `pK_(a (B)) = - log K_(a) (B) = 10^(-5)` By comparing, `K_(a(A)) = 10K_(a(B))` |
|
| 30. |
pK, value of trifluoroacetic acid, benzoic acid and acetic acid are respectively |
|
Answer» 4.76, 4:19 and 0:23 |
|
| 31. |
Piperidine is a secondary amine, which is subjected to Hofmann elimination. The alkene formed as a final product is |
|
Answer» 1, 3-butadiene 
|
|
| 32. |
Pithchblends is the main source of : |
|
Answer» U |
|
| 33. |
Piperidine is |
|
Answer» HOMOCYCLIC COMPOUND  , PIPERIDINE is heterocyclic, alicylic compound. , PIPERIDINE is heterocyclic, alicylic compound.
|
|
| 34. |
Pinacol is converted in to pinacolone by |
|
Answer» REARRANGMENT |
|
| 35. |
Pinacole is: |
|
Answer» 2,3-dimethyl-2,3-butanediol |
|
| 36. |
Pig iron is manufactured using : |
|
Answer» An ELECTRIC FURNACE |
|
| 37. |
Pig iron is manufactured by: |
|
Answer» An ELECTRIC furnace |
|
| 38. |
Pig iron is converted into steel by decreasing the amount of carbon contained in it, in a: |
|
Answer» BLAST furnace |
|
| 39. |
Pig iron__________. |
|
Answer» is the iron containing the carbon and other impurities |
|
| 40. |
Pig iorn has more or less same composition as that of |
|
Answer» CAST iron |
|
| 41. |
Pig iron : |
|
Answer» Contains CARBON and other impuriies |
|
| 42. |
Picric acid is (at 25^(@) C) |
|
Answer» A WHITE solid |
|
| 43. |
Pieces of wood burn faster than a log of wood of the same mass because |
|
Answer» surface AREA of log of wood is larger and needs more time to burn |
|
| 44. |
Picric acid is obtained by the nitration of |
|
Answer» cumene |
|
| 45. |
Picric acid is… |
|
Answer» Trinitro TOLUENE  Trinitro phenol (PICRIC ACID) |
|
| 46. |
Picric acid is- |
|
Answer» 2-ntirophenol |
|
| 47. |
Picric acid contain |
|
Answer» 2-nitro groups |
|
| 48. |
Pickout the correct statements: |
|
Answer» Negative `BETA`-decay decreases the proportion of neutrons and INCREASES the proportion of proton |
|
| 49. |
Pickup the correct formula of potash alum. |
|
Answer» `K_(2)SO_(4). Al_(2)(SO_(4))_(3).24H_(2)O` |
|
| 50. |
Pickles have a long shelf life and do not get spoiled for months, why? |
| Answer» Solution :Plenty of salt and cover of OIL act as preservatives. These food preservatives prevent spoilage of food DUE to microbial growth. Salt and oil do not allow moisture and AIR to ENTER the material and hence bacteria cannot THRIVE on them. Therefore, pickles do not set spoiled for month together. | |



