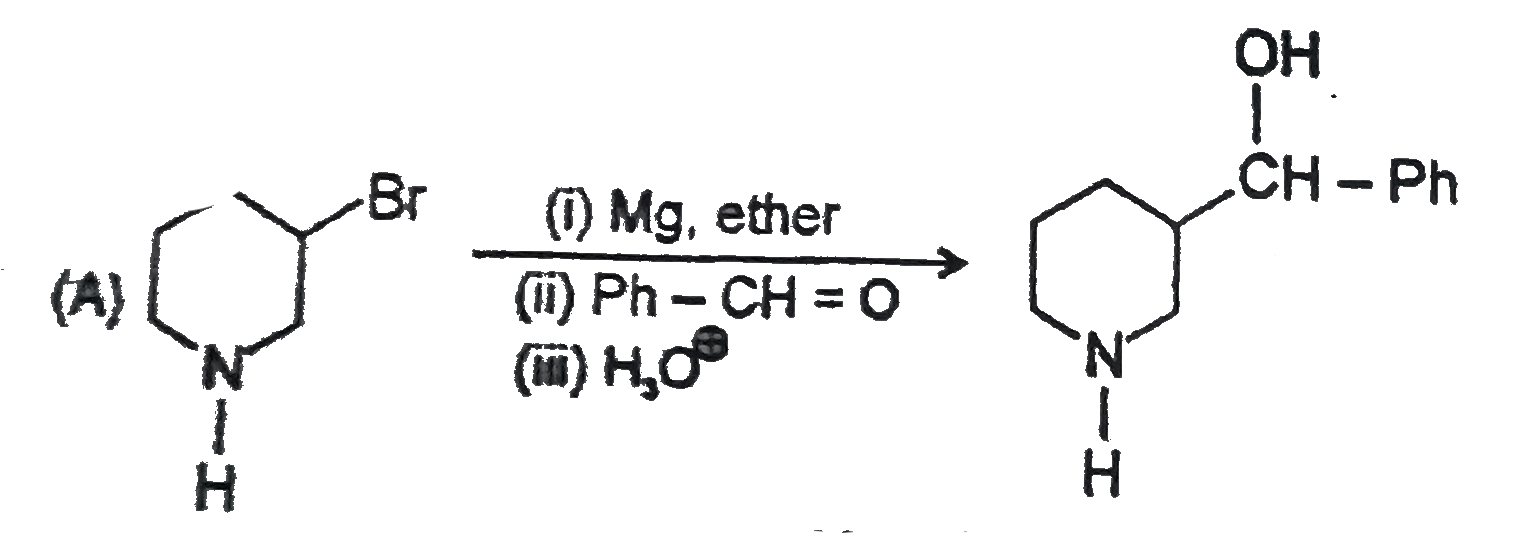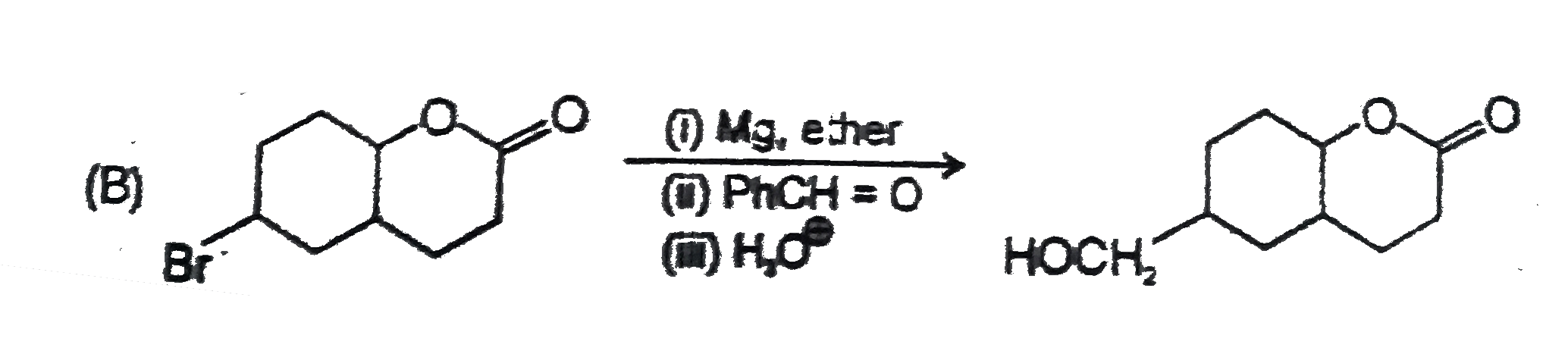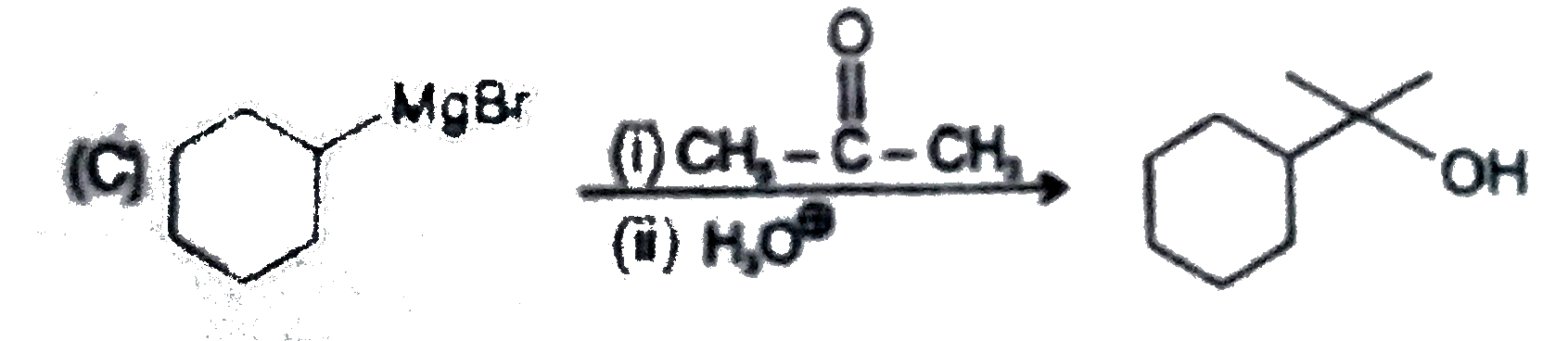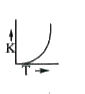Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Point out the wrong statement in relation to enthalpy |
|
Answer» It is a state function |
|
| 2. |
Point out wrong statement about protein |
|
Answer» They are nitrogenous organic compound with high MOLECULAR weight |
|
| 3. |
Point out the wrong statement, in a given period of the periodic table, the s-block elemtns has, in general, a lower value of |
|
Answer» electronegativity |
|
| 4. |
Point out the wrong statement In a given period of the periodic table the s-block element has, in general, a lower value of |
|
Answer» IONISATION energy |
|
| 5. |
Point out the non - existing molecule out of the following ? |
|
Answer» `CBr_(4)` |
|
| 6. |
Point out the incorrect statement regarding Be (Group IIA ) |
|
Answer» It forms an ionic carbide |
|
| 7. |
Point out the wrong statement: For a first order reaction |
|
Answer» time for half-change (`t_(1 // 2)`) is independent of initial concentration |
|
| 8. |
Point out the incorrect statement: |
|
Answer» RATE law is an EXPERIMENTAL value |
|
| 9. |
Pointout theflase statement . |
|
Answer» Colloidal sols are homogeneous |
|
| 10. |
Point out the false statment . |
|
Answer» colloidal sols are homogeneous. |
|
| 11. |
Point out the false statement - |
|
Answer» Colloidal sols are homogenous |
|
| 12. |
Point out the difference between ionic product and solubility product. (ii) The solubility of AgCl in water at 298 K is 1.06 times 10^-5 mole per litre. Calculate is solubility product at this temperature. |
Answer» Solution : (ii) The solubility equilibrium in the saturated solution is `AgCl(s) LEFTRIGHTARROW Ag^(+) (aq)+CL^-(-) (aq)` The solubility of AgCl is `1.06 TIMES 10^-5` MOLE per litre. `[Ag^(+)(aq)]=1.06 time 10^-5 mol L^-1` `[Cl^(-) (aq)]=1.06 times 10^-5 mol L^-1` `K_(sp)=[Ag^+(aq)][Cl^(-) (aq)]` `=(1.06 times 10^-5 mol L^-1) times (1.06 times 10^-5 mol L^-1)` `=1.12 times 10^-2 mol^2 L^-2` |
|
| 14. |
Point out the difference between addition reactions in alkenes and carbonyl compounds. |
Answer» SOLUTION :
|
|
| 15. |
Point out the correct statements/s |
|
Answer» Rate law is an experimental fact whereas law of mass action is a theoretical porposal. |
|
| 16. |
Point out the correct statement about proteins: |
|
Answer» They are nitrogenous ORGANIC COMPOUNDS |
|
| 17. |
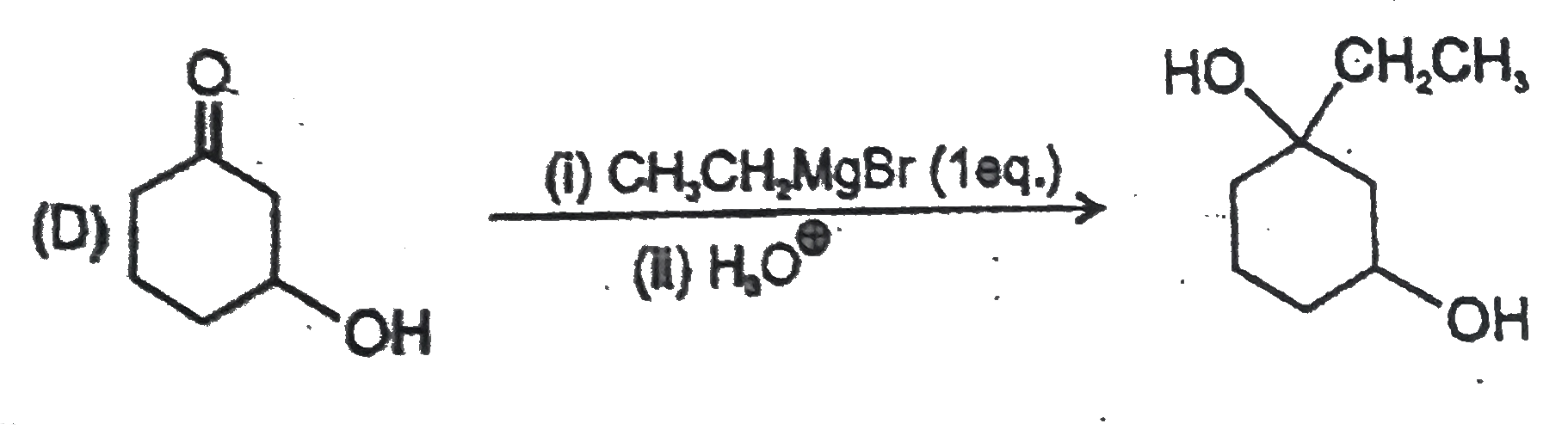
Point out of the following Incorrect Griganard synthesis. |
|
Answer»
|
|
| 18. |
Point out of the wrong statement about proteins: |
|
Answer» They are nitrogenous organic COMPOUNDS of high MOLECULAR mass |
|
| 19. |
Point out the central ion ligand in the complex K_2[Cd(CN)_4]. |
|
Answer» `CD^+, Cn^(1-)` |
|
| 20. |
Point defects are present in |
|
Answer» IONIC solids |
|
| 21. |
PMMA is the polymer of: |
|
Answer» METHYL methacrylate |
|
| 22. |
Pentan-3- one does not undergo iodoform reaction. Give reason. |
| Answer» Solution :Pentan - 3 - one does not UNDERGO IODOFORM REACTION because it is not a METHYL ketone. | |
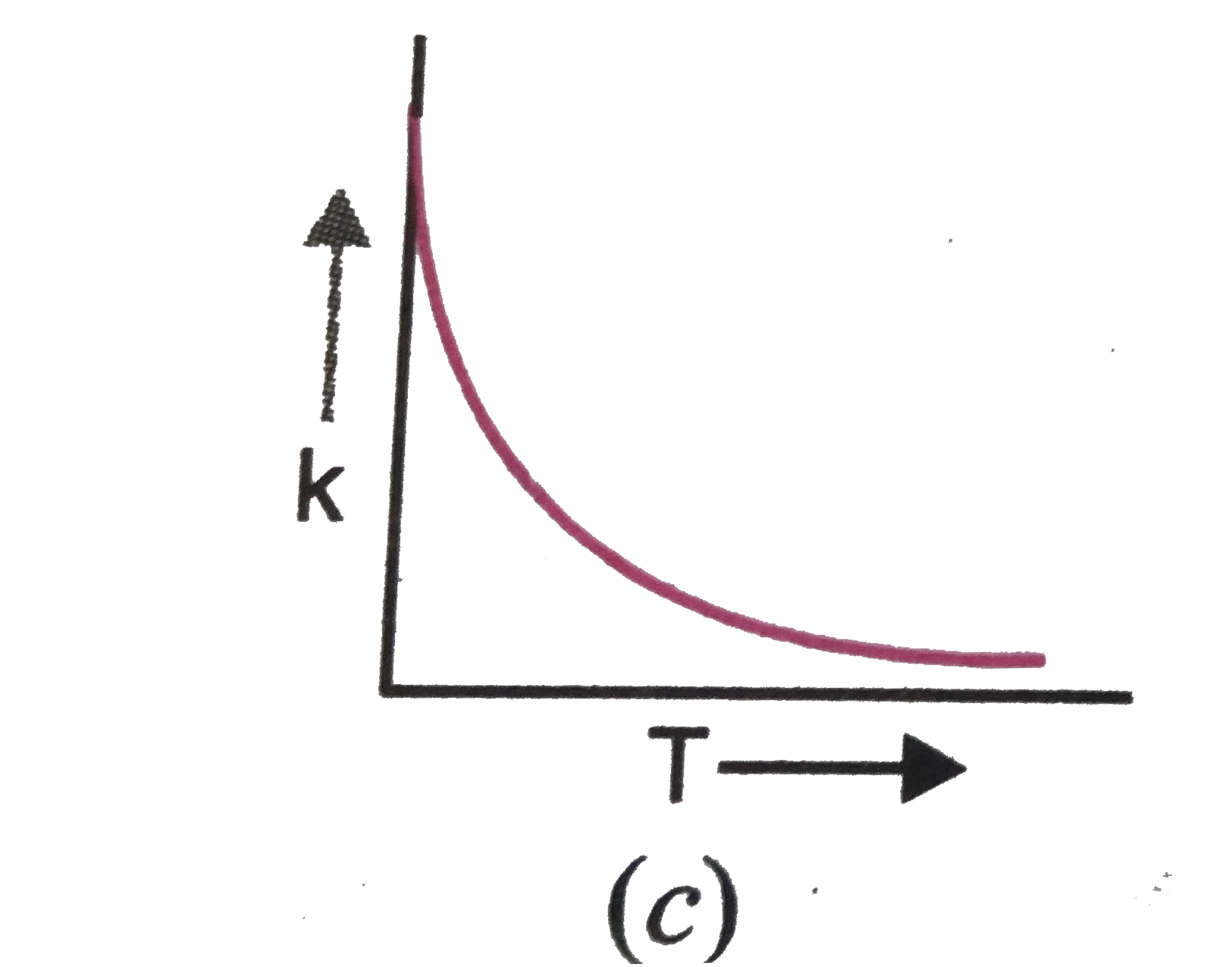
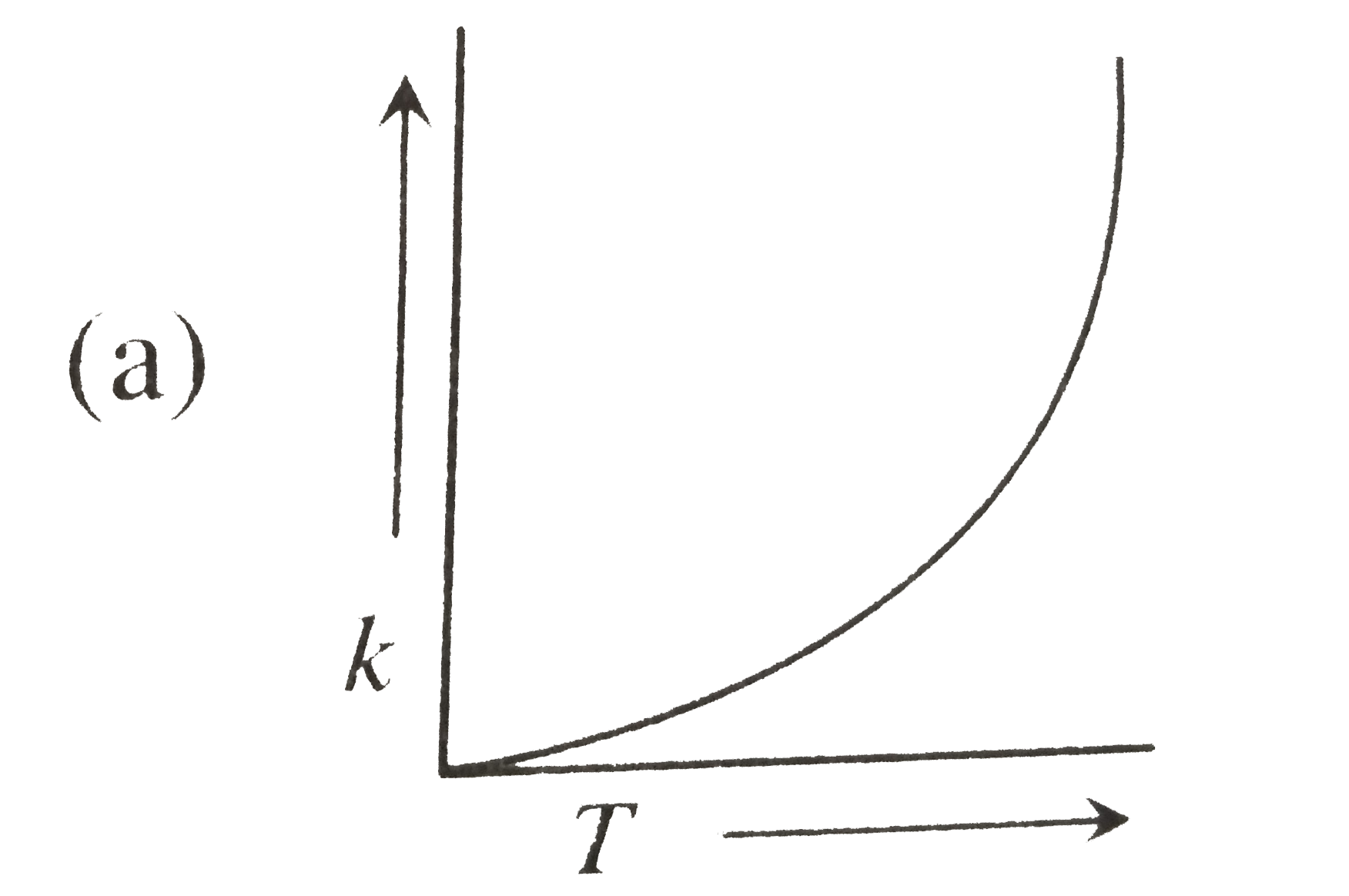
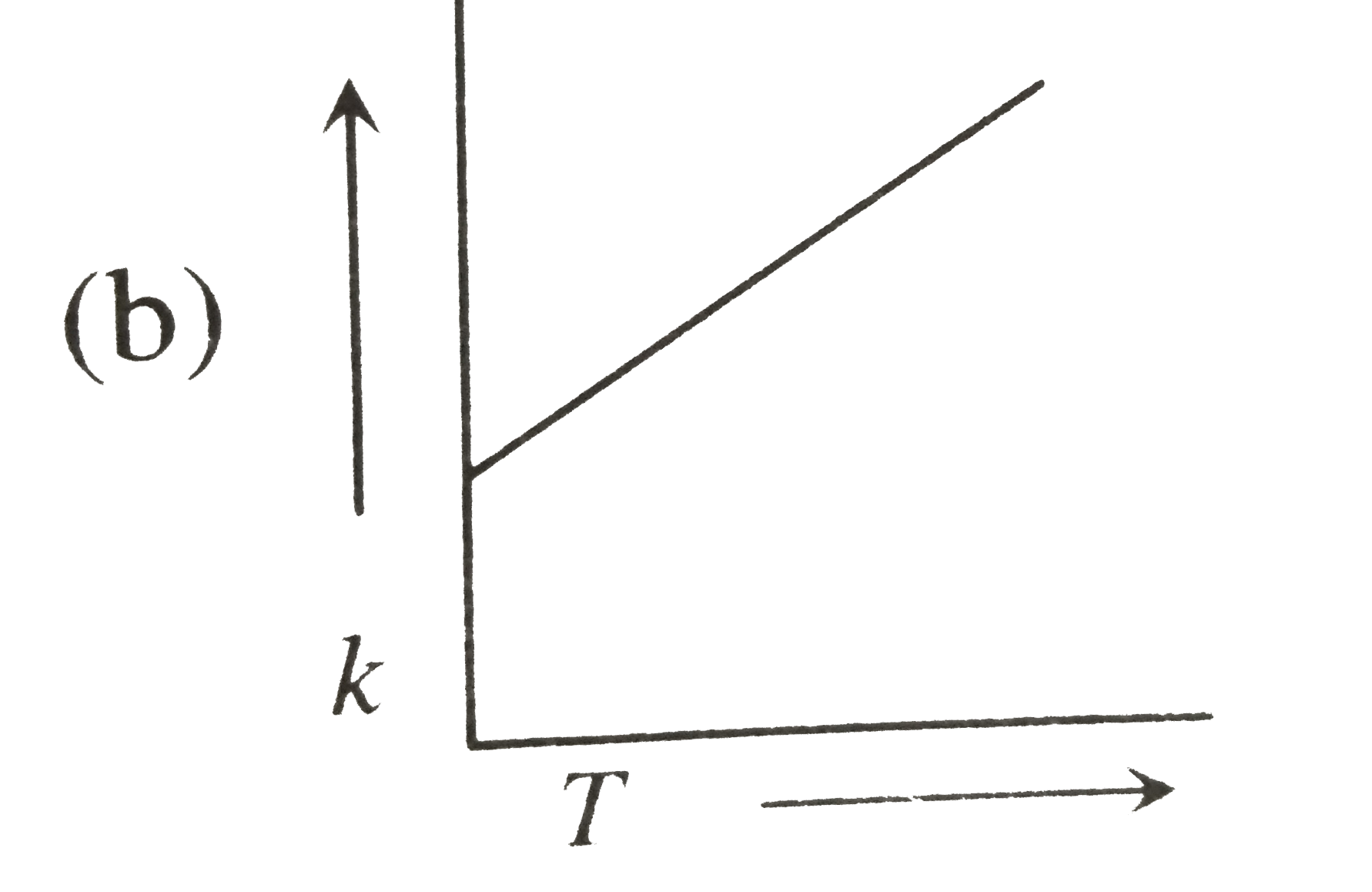
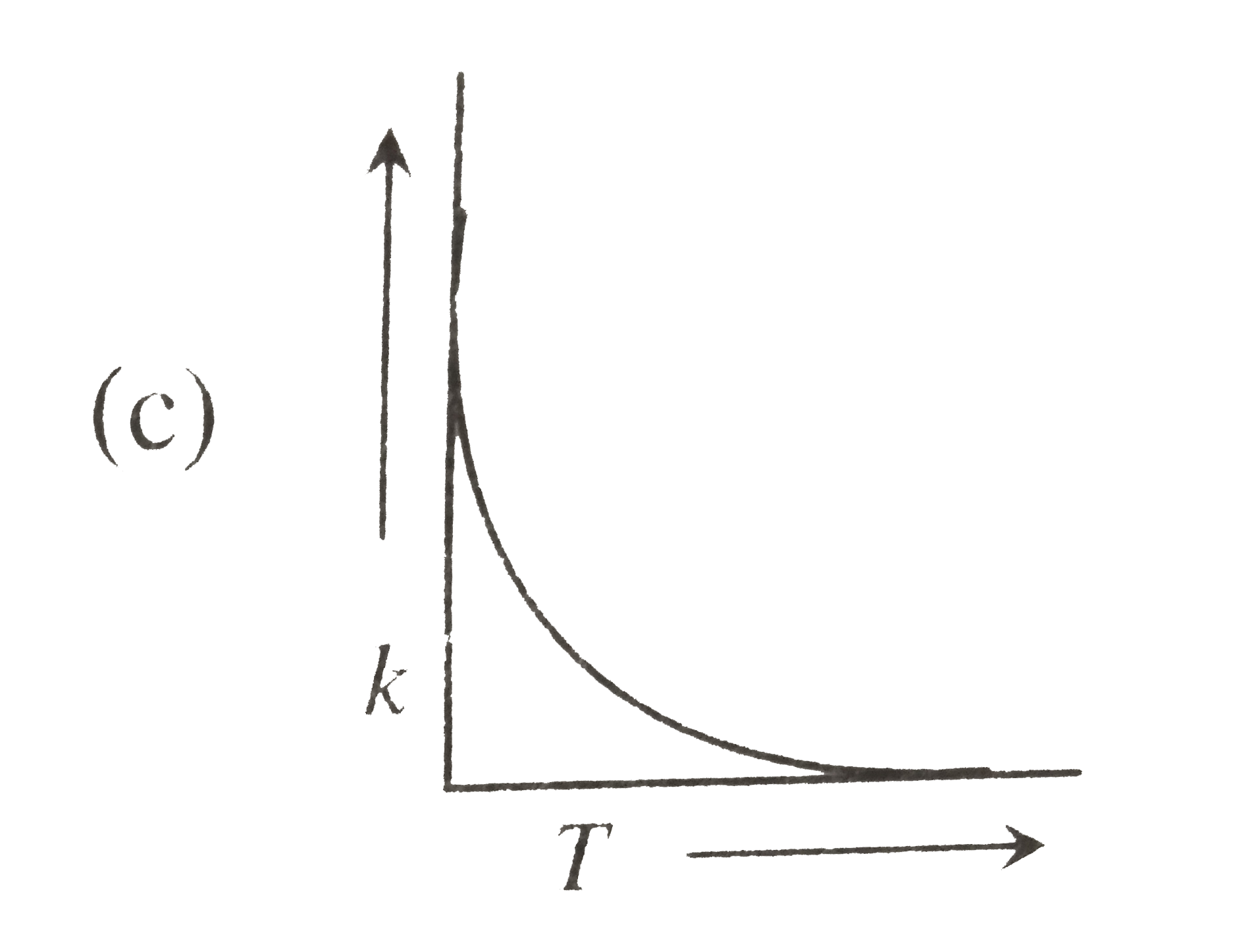
| 23. |
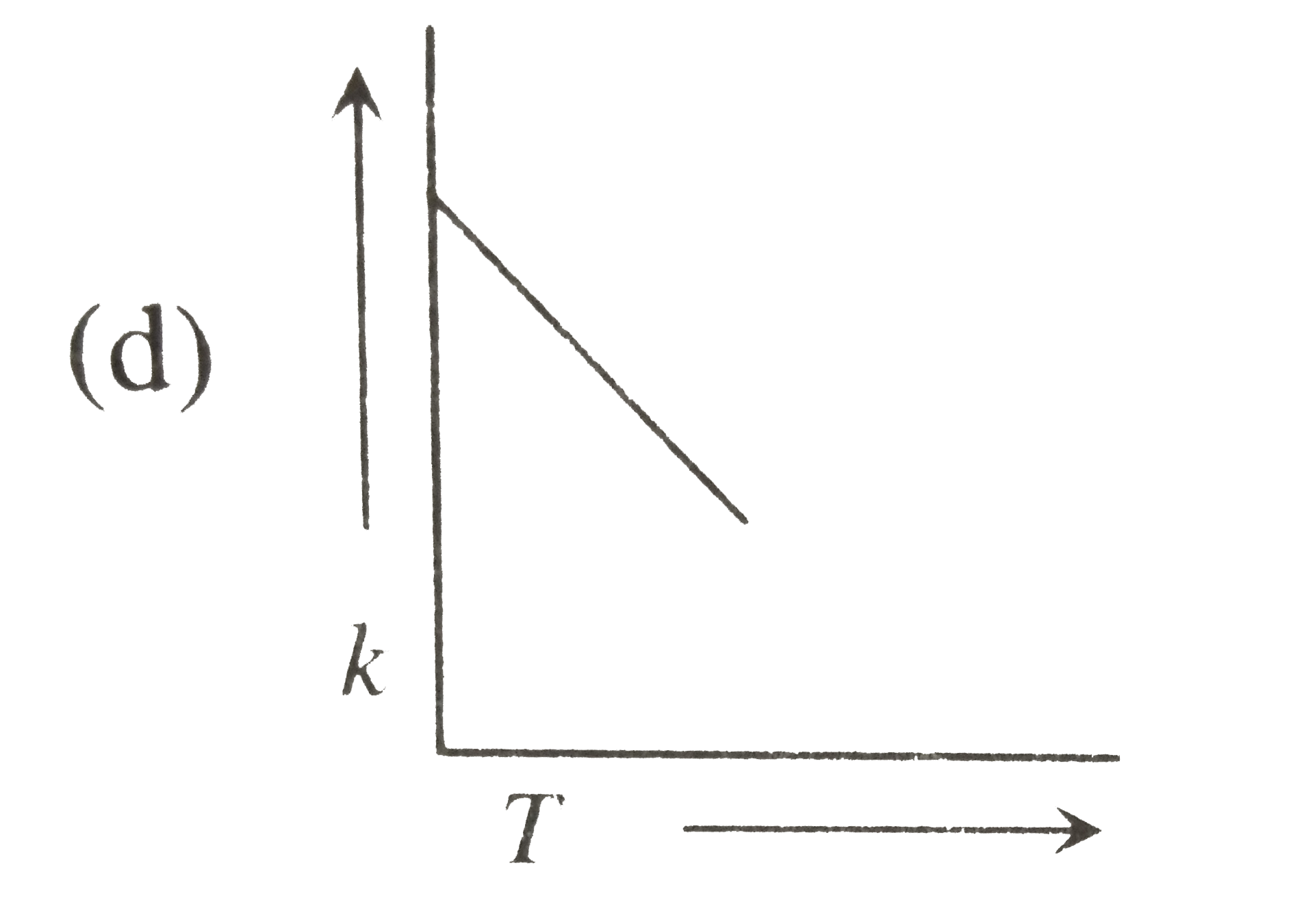
Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrhenius equation is |
|
Answer»
|
|
| 24. |
Plumbo-solvency refers to: |
|
Answer» Oxidation of lead to lead oxide |
|
| 25. |
Plots showing the variation of the rate constant (k) with temperature (T) are given below . The plot that follows Arrhenius equation is |
|
Answer»
So , VARIATION will be 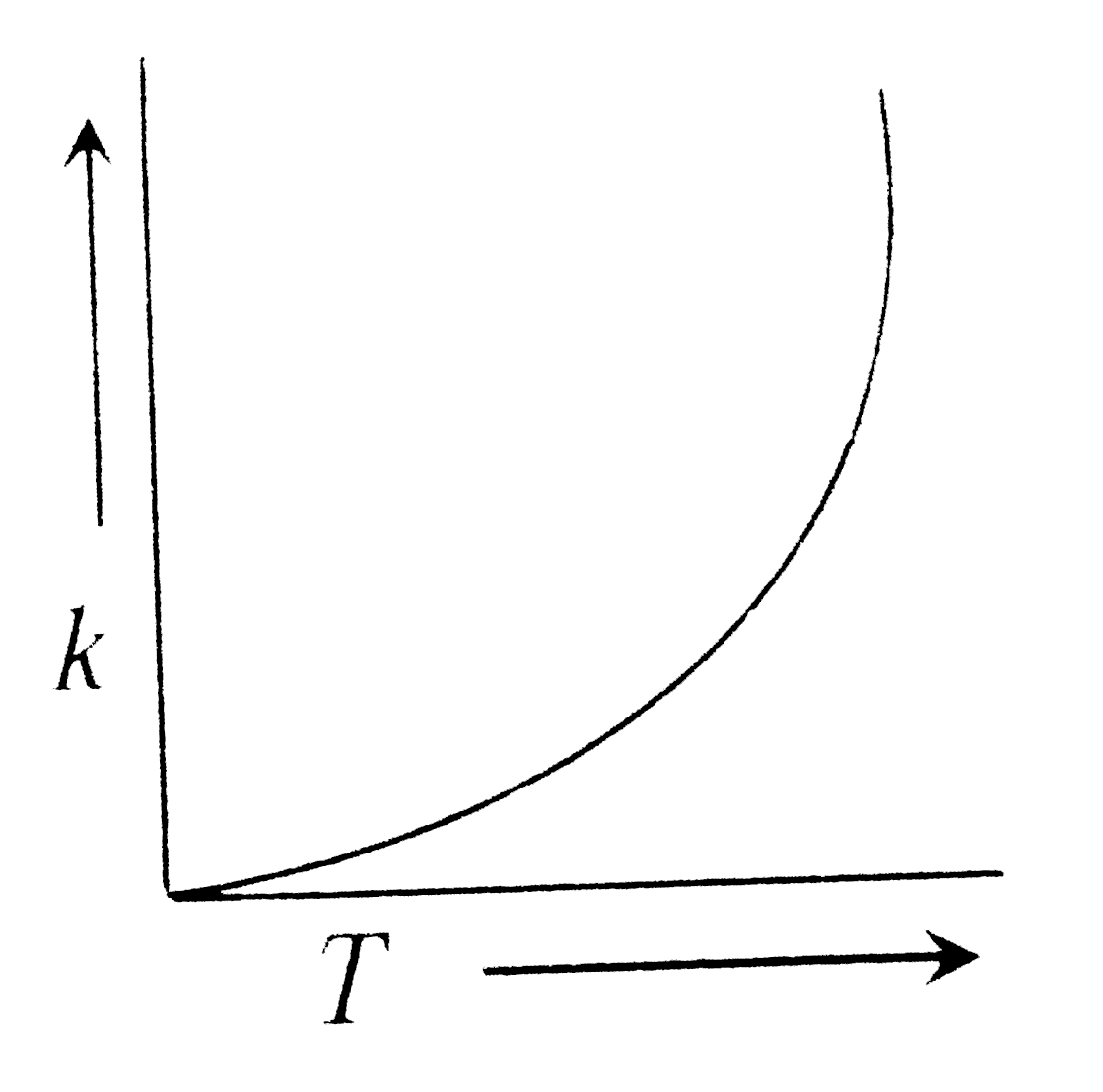
|
|
| 26. |
Plots showing the variation of the rate constant (k) with temperature (T) are given below. The plot that follows Arrhenius equations is |
|
Answer»
|
|
| 27. |
Plots showing the variation of the rate constant (k) with temperature (T) ar given below. The plot that follows arrhenius equation is |
|
Answer»
|
|
| 28. |
Plot of logx/m against logp is straight line inclined at an angle of 45^@.. When the pressure is 0.5 atm and Freundlich parameter, k is 10, the amount of solute adsorbed per gram of adsorbent will be: (log 5 = 0.6990) |
|
Answer» 1g |
|
| 29. |
Plot of Maxwell's distribution of velocities is given below : Whichof the following is correct about this plot ? |
|
Answer» `f_(1) GT f_(2)` `T_(1) gt T_(2)` `V_(1) gt V_(2)` `f_(2) gt f_(1)` |
|
| 30. |
Plot of "log" x/m against log p is a straight line inclined at an angle of 45^@. When the pressure is 0.5atm and k value is 10, the amount of solute adsorbed per gram of adsorbent will be |
|
Answer» Solution :`"log" x/m = log K + 1/n "log P"` `tan 45^@ , 1/n = 1 , x/m = K.P.^(1/n) , 10 XX 0.5= 5 
|
|
| 31. |
Plot of log (a- x) vs time t is straight line. This indicates that the reaction is of: |
|
Answer» SECOND order |
|
| 32. |
Plexsi glassis a commercial name of |
|
Answer» glyptal |
|
| 33. |
Plexi glass is |
|
Answer» `(-CH_2-underset(COOCH_3)underset(|)OVERSET(CH_3)overset(|)C-)_n` |
|
| 34. |
Plexiglass is a commercial name of : |
|
Answer» GLYPTAL |
|
| 36. |
Platinum , palladium , iridium , etc are called noble metals because: |
|
Answer» Alfred Nobel DISCOVERED them |
|
| 37. |
Platinum, Palladium, Indium, etc. are called noble metals because: |
|
Answer» Alfred nobel discovered them |
|
| 38. |
Platinum metal can be dissolved in : |
|
Answer» Hot concentrated HYDROCHLORIC ACID |
|
| 39. |
Platinum is used as a catalyst in : |
|
Answer» OXIDATION of AMMONIA to from NITRIC acid |
|
| 40. |
Platinum is not used as a catalyst in the : |
|
Answer» Oxidation of `CH_3OH` to `HCHO` |
|
| 41. |
Platinum dissolves in aqua regia to form |
|
Answer» `PtCl_(4)` |
|
| 42. |
'Platinum black' is - |
|
Answer» a mixture of Pt and `MnO_(2)` |
|
| 43. |
Platinum black is : |
|
Answer» Platinum metal MIXED with `MnO_2` |
|
| 44. |
Platinised asbestos helps in the formation of SO_(3) from SO_(2) and O_(2). But, if even a small amount of As_(2)O_(3) is present the platinised asbestos does not help in the formation of SO_(3), As_(2)O_(3) acts here as a//an. |
|
Answer» CATALYTIC promoter |
|
| 45. |
Platinised asbestos used as a catalyst in manufacture of H_(2)SO_(4) is an example of |
|
Answer» HOMOGENOUS CATALYSIS |
|
| 46. |
Platelet aggregation is inhibited by |
|
Answer» BORIC ACID |
|
| 47. |
Plat of Maxwell.s distribution of velocities is gives below : Which of the following is correct about this plot ? |
|
Answer» `V_1 lt V_2` `T_1 gt T_2` `V_1 gt V_2` ` f_2 gt f_1` |
|
| 48. |
Plastic is : |
|
Answer» Natural POLYMER |
|
| 49. |
Plaster of paris of |
|
Answer» `CaSO_(4)2.H_(2)O` |
|