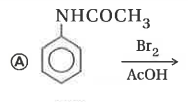
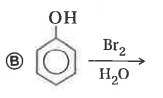
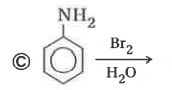
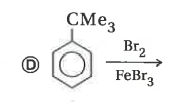
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Polymer obtained by condensation polymerisation is : |
|
Answer» Polythene |
|
| 2. |
Polymer obtained by condensation polymerisation is: |
|
Answer» POLYTHENE |
|
| 3. |
polymer obtainded by condensation polymerisation is : |
|
Answer» Polythene |
|
| 4. |
Polymer given below is [-HN-CH_2-CONH(CH_2)_5CO]_n |
|
Answer» NYLON -6 |
|
| 5. |
Polymer formation from monomers starts by |
|
Answer» CONDENSATION reaction between monomers |
|
| 6. |
Polymer formation from monomers starts by : |
|
Answer» condensation REACTION between MONOMERS |
|
| 7. |
Polymer fonnation from monomers starts by |
|
Answer» condensation reaction between monomers |
|
| 9. |
Polymer formation from monomers in step growth polymerisation starts by |
|
Answer» condensation REACTION between MONOMERS |
|
| 10. |
Polyhosphates are usedas water softening agents because they |
|
Answer» 1. formsoluble complexeswith anionic species `2CA^(2+)+Na_(4)[Na_(4)(PO_(3_6))rarr4Na^(2+)+Na_(2)Ca_(2)(PO_(3_6))` |
|
| 11. |
Polyethyleneglycols are used in the preparation of which type of detergents? |
|
Answer» CATIONIC detergents |
|
| 12. |
Polyethyleneglycols are used in the preparation of which\ type of detergents ? |
|
Answer» CATIONIC detergents |
|
| 13. |
Polythyleneglycols are used in the preparation of which type of detergents ? |
|
Answer» Cationic detergents |
|
| 14. |
Polyethyleneglycols are used in the preparation of which type of detergents ? |
|
Answer» CATIONIC detergents |
|
| 15. |
Polyethylene Glycols are used in the preparation of which type of detergents ? |
|
Answer» CATIONIC detergents |
|
| 16. |
Polyethylene is |
|
Answer» RANDOM copolymer `nCH_(2)=CH_(2) to (-CH_2-CH_(2)-)_(n)` |
|
| 17. |
Polyethene can be prepard by CaC_(2) by the following sequence of reactions . CaC_(2) + H_(2)O to CaO + C_(2)H_(2) C_(2)H_(2) + H_(2) to C_(2)H_(4) nC_(2)H_(4)to (C_(2)H_(4))_(n) "(Polythene)" The mass in kg of polythene that can be prepared by 20 kg CaC_(2) . |
|
Answer» 4.1 kg |
|
| 18. |
Polyethylene glycol stearate is a _______detergent |
|
Answer» |
|
| 19. |
Polyethylene glycols are used in the preparation of which type of detergent ? |
|
Answer» Cationic detergents |
|
| 20. |
Polyethylene glycol stearate is a |
| Answer» Answer :D | |
| 21. |
Polychlorinated biphenyls, PCBs, known to be dangerous environment pollutants , are a group of compounds with the general empirical formula C_12H_mCl_(10-m), where m is an integer. What is the value of m, if percentage of carbon atom in the compounds is 40 ? |
|
Answer» On SOLVING m=4 |
|
| 25. |
Pollution can be controlled by |
|
Answer» Sewage treatment |
|
| 26. |
Polonium crystallizes in a simple cubic unit cell. Its atomic mass is 209 and density is 91.5 kg m^(-3). What is the edge length of its unit cell? |
|
Answer» |
|
| 27. |
Poly Beta-hydroxybutyrate-co-Beta-hydroxy valerate (PHBV) is obtained by the copolymerisation of |
|
Answer» 2-hydroxybutanoic ACID and 3-hydroxypentanoic acid |
|
| 28. |
Polonium is a radioactive element. This element w discovered by |
|
Answer» Faraday |
|
| 29. |
Poly - beta- hydroxybutyrate - co - beta hydroxy valerate is an example of |
|
Answer» thermoplastic |
|
| 30. |
Polling process is used for the refining of (1) Iron (2) Copper (3) Tin (4) Lead |
|
Answer» 1, 2 |
|
| 31. |
Polling process us used in the purification of |
|
Answer» Ge |
|
| 32. |
Poling process is used ......... |
|
Answer» for removal of `Fe_2O_3` from FE. |
|
| 34. |
Polling process is used : |
|
Answer» For the REMOVAL of `Cu_2O` from Cu |
|
| 35. |
Polarisation of sigma - bond is caused by polarisation of adjacent sigma - bond is referred as- |
|
Answer» Mesomeric EFFECT |
|
| 36. |
Polarisation of electrons in acrolein may be written as : |
|
Answer» `OVERSET(delta-)(CH_(2))=CH-overset(delta+)(CH)=O` |
|
| 37. |
Polarisation of electrons in acrolein may be written as |
|
Answer» `OVERSET(-delta)(CH_(2))=CH-CH=O` |
|
| 38. |
Polarimeter is used to determine of compounds. |
|
Answer» D and L CONFIGURATION |
|
| 39. |
Polar covalent molecules exhibit dipole moment. Dipole moment is equal to the product of charge separation , q and the bond length d for the bond. Unit of dipole moment is debye. One debye is equal to 10^(-18) esu cm. Dipole moments is a vector quantity. It has both magnitude and direction. Hence,dipole moment of a molecule depends upon the relative orientation of the bond dipoles, but not on thepolarity of bonds alone. A symmetrical structure shows zero dipole moment. Thus, dipole moment helps to predict the geometry of a molecules. Dipole moment values can be distinguish between cis- and trans- isomers, ortho, meta and pare-forms of asubstance, etc. Q. mu of the AX_(4) type of molecule is zero. The geometry of it can be : |
|
Answer» tetrahedral |
|
| 40. |
Polar covalent molecules exhibit dipole moment. Dipole moment is equal to the product of charge separation , q and the bond length d for the bond. Unit of dipole moment is debye. One debye is equal to 10^(-18) esu cm. Dipole moments is a vector quantity. It has both magnitude and direction. Hence,dipole moment of a molecule depends upon the relative orientation of the bond dipoles, but not on thepolarity of bonds alone. A symmetrical structure shows zero dipole moment. Thus, dipole moment helps to predict the geometry of a molecules. Dipole moment values can be distinguish between cis- and trans- isomers, ortho, meta and pare-forms of asubstance, etc. Q. Arrange the following compounds in increasing order of dipole moments, toluene (I), o-dichlorobenzene (II), m-dichlorobenzene (III) and p-dichlorobenzene (IV) : |
|
Answer» `IVltIltIIltIII` 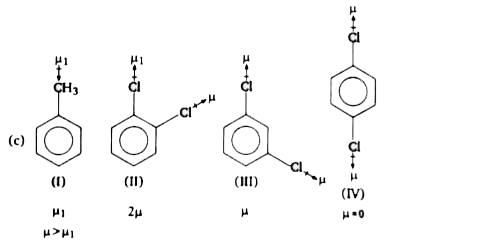
|
|
| 42. |
Polar covalent molecules exhibit dipole moment. Dipole moment is equal to the product of charge separation, q and the bond length d for the bond. Unit of dipole moment is Debye. One Debye is equal to 10^(-18) esu-cm. Dipole moment is a vector quantity. It has both magnitude and direction. Hence, dipole moment of a molecule depends upon the relative orientation of the bond dipoles, but not on the polarity of bonds alone. A symmetrical structure shows zero dipole moment. Thus, dipole moment helps to predict the geometry of a molecules. Dipole moment values can be used to disinguisd between cis- and trans- isomers, ortho-, meta - and para - forms of a substance, etc. Which of thefollowing pair of species have same diple moment value? |
|
Answer»
|
|
| 43. |
Poisonous gas 'Lewisite' is obtained by the reaction of |
|
Answer» `CH-=CH` and `AsCl_3` |
|
| 44. |
Poisonous gas present in the exhaust fumes of automobiles is : |
| Answer» Answer :B | |
| 46. |
Poison for platinum, a catalyst in contact process of H_2SO_4 is: |
|
Answer» S |
|
| 48. |
Point out wrong statement about protems |
|
Answer» These are POLYMERIC macromolecules |
|
| 49. |
Point out the wrong statement On moving horizontally from left to right across a period in the periodic table |
|
Answer» Metallic character decreases |
|