Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Over production of acid in the stomach causes irritation and pain. In severe cases, ulcers are developed in the stomach. Until 1970, only treatment for acidity was administration of antacids, such as sodium hydrogencarbonate or a mixture of aluminium and magnesium hydroxide. However, excessive hydrogencarbonate can make the stomach alkaline and trigger the production of even more acid. Metal hydroxides are better alternatives because of being insoluble, these do not increase the pH above neutrality. These treatments control only symptoms, and not the cause. Therefore, with these metal salts, the patients cannot be treated easily. In advanced stages, ulcers become life threatening and its only treatment is removal of the affected part of the stomach. A major breakthrough in the treatment of hyperacidity came through the discovery according to which a chemical, histamine stimulates the secretion of pepsin and hydrochloricacid in the stomach. The drug cimetidine (Tegamet), was designed to prevent the interaction of histamine with the receptors present in the stomach wall. This resulted in release of lesser amount of acid. The importance of the drug was so much that it remained the largest selling drug in the world until another drug, ranitidine (Zantac), was discovered. What happens in serious case of over production of acid in the stomach ? |
| Answer» SOLUTION :In severe cases, ULCERS are PRODUCED in the stomach. | |
| 2. |
Over production of acid in the stomach causes irritation and pain. In severe cases, ulcers are developed in the stomach. Until 1970, only treatment for acidity was administration of antacids, such as sodium hydrogencarbonate or a mixture of aluminium and magnesium hydroxide. However, excessive hydrogencarbonate can make the stomach alkaline and trigger the production of even more acid. Metal hydroxides are better alternatives because of being insoluble, these do not increase the pH above neutrality. These treatments control only symptoms, and not the cause. Therefore, with these metal salts, the patients cannot be treated easily. In advanced stages, ulcers become life threatening and its only treatment is removal of the affected part of the stomach. A major breakthrough in the treatment of hyperacidity came through the discovery according to which a chemical, histamine stimulates the secretion of pepsin and hydrochloricacid in the stomach. The drug cimetidine (Tegamet), was designed to prevent the interaction of histamine with the receptors present in the stomach wall. This resulted in release of lesser amount of acid. The importance of the drug was so much that it remained the largest selling drug in the world until another drug, ranitidine (Zantac), was discovered. Which of the two treatments to heal acidity, hydrogencarbonate and metal hydroxide, is better and why ? |
| Answer» Solution :METAL hydroxides are better because they are INSOLUBLE and do not INCREASE PH beyond neutrality. | |
| 3. |
Outline the synthesis of each alcohol from the indicated starting materials (a) Allyl alcohol from propane (b) n - Butyl alcohol from acetylene (c ) 1 - Pentanol from 1 - bromopropane |
Answer» SOLUTION :
|
|
| 4. |
Over production of acid in the stomach causes irritation and pain. In severe cases, ulcers are developed in the stomach. Until 1970, only treatment for acidity was administration of antacids, such as sodium hydrogencarbonate or a mixture of aluminium and magnesium hydroxide. However, excessive hydrogencarbonate can make the stomach alkaline and trigger the production of even more acid. Metal hydroxides are better alternatives because of being insoluble, these do not increase the pH above neutrality. These treatments control only symptoms, and not the cause. Therefore, with these metal salts, the patients cannot be treated easily. In advanced stages, ulcers become life threatening and its only treatment is removal of the affected part of the stomach. A major breakthrough in the treatment of hyperacidity came through the discovery according to which a chemical, histamine stimulates the secretion of pepsin and hydrochloricacid in the stomach. The drug cimetidine (Tegamet), was designed to prevent the interaction of histamine with the receptors present in the stomach wall. This resulted in release of lesser amount of acid. The importance of the drug was so much that it remained the largest selling drug in the world until another drug, ranitidine (Zantac), was discovered. What happens in advanced cases to the patients of hyperacidity ? |
| Answer» SOLUTION :Ulcers in the stomach BECOME life-threatening and AFFECTED PART has to be removed. | |
| 5. |
Over production of acid in the stomach causes ________________ and ________________. |
| Answer» SOLUTION :IRRITATION, PAIN | |
| 6. |
Outline the principles of refining of metals by the following methods : (i) Zone refining (ii) Electrolytic refining (iii) Vapour phase refining. |
|
Answer» Solution :(ii) Electrolytic refining. This method is based UPON the phenomenon of electroysis. The CRUDE metal is made anode whereas the THIN sheet of pure metal is made cathode. Electrolyte is the solution of some salt of the metal to be refused. On passing electricity, the pure metal from the anode GOES into solution as ions due to oxidation while pure metal gets deposited at the cathode due to reduction of metal ions. The less electropositive impurities settle down below the anode as anode mud. At anode :`M(s) hArr M^(N+)(aq) + n e` At cathode: `M^(n+) (aq) + n e^(-) to M(s)` 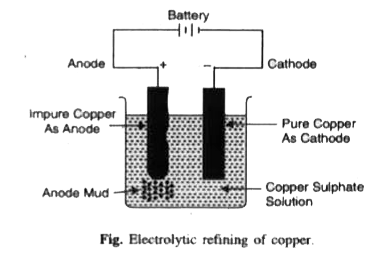 (iii) Vapour phase refining. Vapour phase refining is explained in brief review. |
|
| 7. |
Outline the synthesis of 2-mthytheptane starting with 1-bromopropane and 2-bromopropane. |
|
Answer» Solution :`(CH_3)_2CHBroverset((i)Lr)UNDERSET((ii) i - "BROMOPROPANE")RARR[(CH_3)_2CH]_2LiCuoverset(CH_3-CH_2-CH_2Br)rarr(CH_3)_2-CH-CH_2-CH_3` 2-methylpentane |
|
| 8. |
Outline the principles of refining metals by the following methods : (i) Zone refining (ii) Electrolytic refining (iii) Vapour phase refining |
|
Answer» Solution :(i) Zone REFINING : This process is based upon the principle that IMPURITIES are more soluble in the molten state of the metal than in the solid form. (II) Electrolytic refining : It is USED for less reactive metals like Cu, Ag, Al, etc. In it, anode is made by impure metal and a thin strip of pure metal acts as cathode. On passing electricity, metal dissolves form anode and pure metal gets deposited at cathode. At anod `MrarrM^(n+)+n e^(-)` `{:("Impure"),("Metal"):}` At cathode `M^(n+)+n e^(-)rarrM` Pure Metal Impurities settle down below anode in the form of anode mud. (III) Vapour phase refining : This method is based upon the following facts : (a) Metal to be refined should form a volatile compound, while impurities do not. (b) Volatile compound should be easily decomposable so that pure metal can be recovered easily. |
|
| 9. |
Outline the principles behind the refining of metals by the following methods : Zone refining method. |
| Answer» Solution :This method is based on the PRINCIPLE that the impurities are more SOLUBLE in the MELT than in the SOLID state of the metal. | |
| 10. |
Outline the principles behind the refining of metals by the following methods : Chromatographic method. |
| Answer» Solution :This METHOD is BASED on the principle that different COMPONENTS of a mixutre are differently adsorbed on an adsorbent. | |
| 11. |
Outline the principle of the method used for refining of Zirconium. |
|
Answer» Solution :van Arkel method is USED for the refining of zirconium `ZR +2I_(2) to ZrI_(4)` The metal iodide is decomposed on a TUNGSTEN filament, electrically heated to 1800K. The pure metal is deposited on the filament `ZrI_(4) to Zr + 2I_(2)`. |
|
| 12. |
Outline the principle of the method used for refining of Tin. |
| Answer» Solution :Liquation is used for refining tin. The melting tin is made to flow on a SLOPING surface. In this WAY, it is separated from higher melting IMPURITIES. | |
| 13. |
Outline the preparationof the following compounds using a nucliphileic subsituationr reaction. a. CH_(3)OC(CH_(3))_(3) b. CH_(3)C -= CCH_(2)CH_(3) c. CH_(3)CH_(2)CH_(2)N (CH_(3))_(2) d. C_(6) H_(5)CH_(2) OCOCH_(3) e. CH_(3)CH_(2)CH_(2)CH_(2)NO_(2) f. CH_(3)CH_(2)CH_(2)Cn g. C_(6) H_(5) CH_(2) N^(o+) -= C^(o-) h. CH_(3)CH_(2) - O - N = 0 |
Answer» SOLUTION :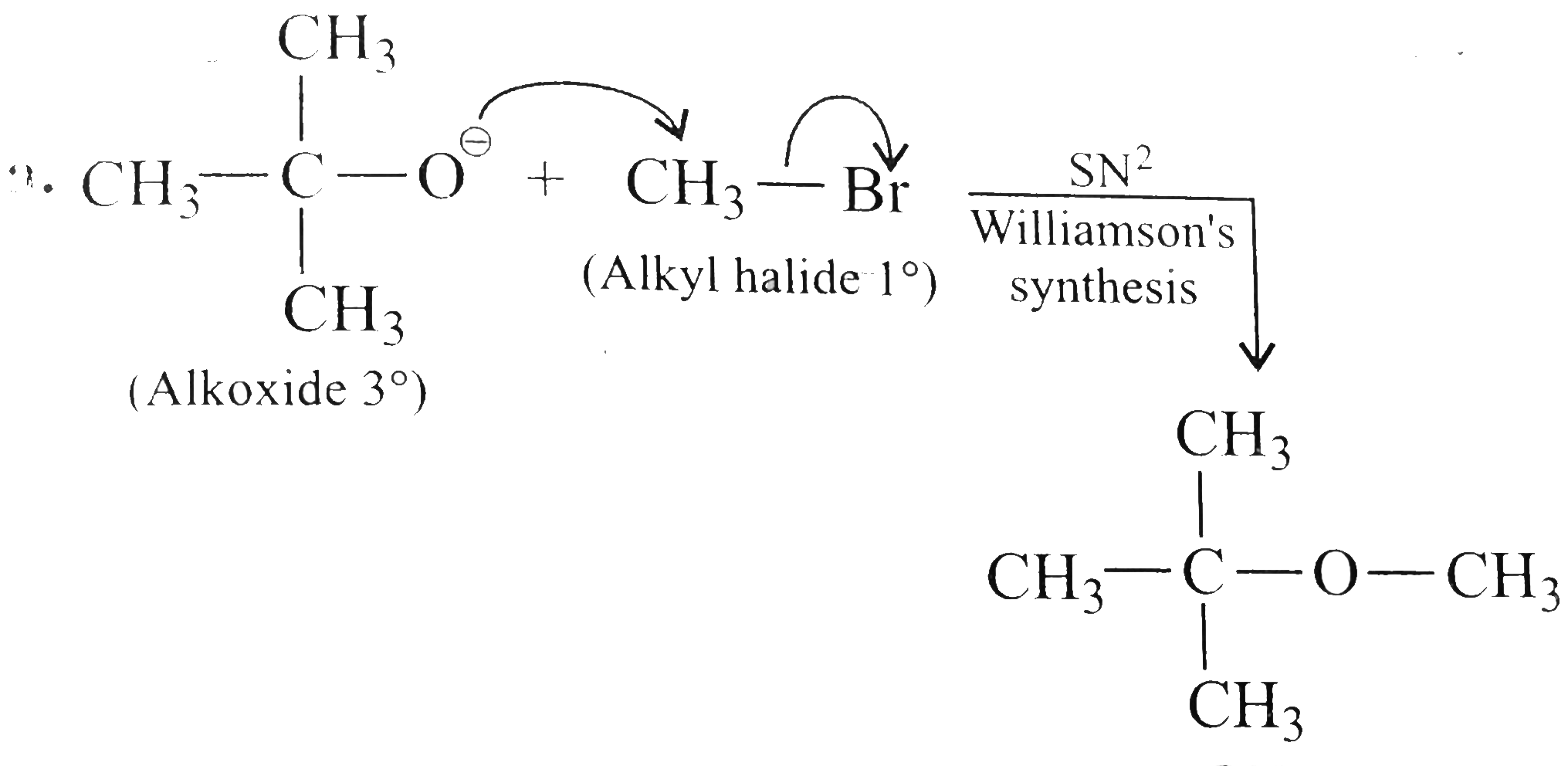 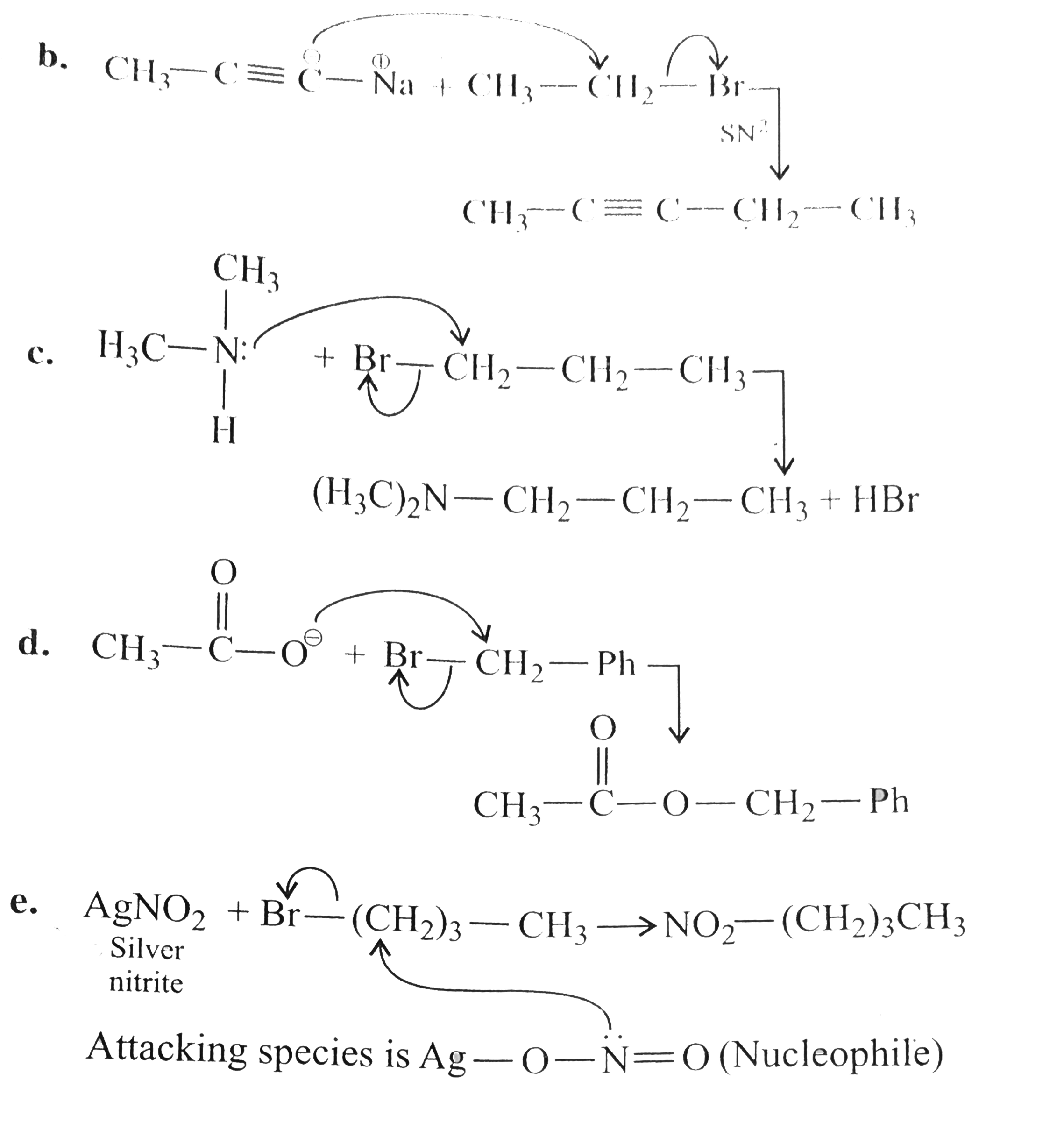
|
|
| 14. |
Outline the principle of the method used for refining of Nickel. |
|
Answer» SOLUTION :NICKEL is refined by vapour phase refining (Mond.s PROCESS) `{:(Ni+4CO overset(330-350K)(to)Ni(CO)_(4)),("Nickel carbonyl "):}` Nickel carbonyl is SUBJECTED to higher temperature when it decomposes to give pure nickel. `Ni(CO)_(4) overset(450-470K)(to) Ni + 4CO`. |
|
| 15. |
Outline the principle of refining of metals by (i) Zone refining. (ii) Electrolytic refining. |
|
Answer» Solution :(ii) ELECTROLYTIC refining. This method is based upon the phenomenon of electroysis. The crude metal is MADE anode whereas the thin SHEET of pure metal is made cathode. Electrolyte is the solution of some salt of the metal to be refused. On passing electricity, the pure metal from the anode goes into solution as IONS due to oxidation while pure metal gets deposited at the cathode due to reduction of metal ions. The less electropositive impurities settle down below the anode as anode mud. At anode :`M(s) hArr M^(n+)(aq) + n e` At cathode: `M^(n+) (aq) + n e^(-) to M(s)` 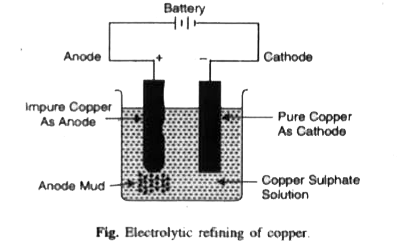 (iii) Vapour PHASE refining. Vapour phase refining is explained in brief review. |
|
| 16. |
Outline the preparation of (a) para nitroaniline from aniline, (b) tri bromo benzene from tribromo aniline. |
Answer» Solution :(i) p-nitroaniline is prepared from ANILINE in THREE STAGES as FOLLOWS :  (II) 
|
|
| 17. |
Outline the principels of refining of metals by the following methods : (i) Distillation (ii) Zone refining (iii) Electrolysis. |
|
Answer» Solution :DISTILLATION : It is used to remove the NON- volatile impurities from volatile METAL like zinc, cadmium and mercury. The impure metal is heated in a retory when the pure metal vaporises and condenses separately LEAVING behind the non-volatile impurities. Reactions in different zones in blast furnace during extraction of iron. (i) Zone of combustion `C(S) +O_2to CO_2` (ii) Zone of heat aborption. `CO_2+C(S)to 2CO(g)` (iii) Zone of slag formation : `CaCo_3(s) overset(1123K)to CaO(s)Co_2(g)` `CaO(s)+SiO_2(s) overset(1123K)to underset("Slag")(CaSiO_3(s) ` (iv) Zone of reduction `underset((s))(3Fe_2O_3)+underset((g))(CO)overset (573-673K)to 2Fe_3O_3(s) +CO_2(g)` `Fe_3O_4(s)+CO(g)overset(773-873K)to 3FeO(s) +CO_2(g)` `Fe_2O_3(s) +CO(g) overset(773-873K)2FeO(s)+CO_2(g)` `FeO(s) +CO9(g) overset(1123K)to Fe(s)+CO_2(g)` |
|
| 18. |
Outline the mechanism of Nitration of aniline. |
Answer» SOLUTION :Nitration of aniline is accompanied by OXIDATION. But a mixture of conc. `HNO_3` and conc. `H_2SO_4` gives m-nitroaniline also. Nitric acid is a strong acid. It protonates aniline forming anilinium ION `C_6H_5NH_3^+` because of positive CHARGE on nitrogen it is meta directing. 
|
|
| 19. |
Outline the mechanism of Acetylation of aniline. |
|
Answer» Solution :Aniline reacts with ACETYL chloride and acetic anhydride to form CORRESPONDING AMIDES called anilides. `{:(C_6H_5NH_2+ClCOCH_3 to C_6H_5NHCOCH_3+HCl),(" AnilineAcetylchlorideAcetanilide "):}` `{:(C_6H_5NH_2+CH_3COOCOCH_3 to C_6H_5NHCOCH_3+CH_3COOH),("ACETANILIDE "):}`. |
|
| 20. |
Outline the classification of carbohydrates giving example for each.(OR) How are carbohydrates classified? |
Answer» Solution :Carbohydrates are polyhydroxy aldehydes or polyhydroxy ketones. The carbohydrates may be summarised as below.  (i) Sugar : They are sweet crystalline substance and soluble in water. (a) Monosaccharides : They are polyhydroxy aldehydes (or) polyhydroxy ketones. Aldoses - Carbohydrates containing aldehyde group. Eg: Glucose Ketoses - Carbohydrates containing ketone group. Eg: Fructose. (B) Oligosaccharides : They are sugars that yield two to ten monosaccharide molecules on hydrolysis. Disaccharides : They are sugars which on hydrolysis give two molecules of same or different monosaccharides. Eg: Sucrose, maltose. `underset("Sucrose")(C_(12)H_(22))O_(11)+H_(2)Orarrunderset("Glucose")(C_(6)H_(12)O_(6))+underset("Fructose")(C_(6)H_(12)O_(6))` Trisaccharidcs: They are sugars which gives there molecucules of monosaccharides on hydrolysis Eg: Raffinose. `underset("Raffinose")(C_(18)H_(32)O_(16))+2H_(2)Orarrunderset("GALACTOSE")(C_(6)H_(12)O_(6))+underset("Glucose")(C_(6)H_(12)O_(6))+underset("Fructose")(C_(6)H_(12)O_(6))` (ii) Non-sugars (or) Polysaccharides: They are non-sugars which involve a large number of monosaccharide units linked to each other by OXIDE bridges. These bridges are called glycosidic linkages. Eg: starch, cellulose. `underset("Starch")(C_6H_10O_5)+nH_(2)Ooverset(H^(+))rarrunderset("Glucose")(nC_(6)H_(12)O_(6))` |
|
| 21. |
Outline a synthesis of each alcohol from the indicated starting materials. (a) Isopropyl alcohol from a hydrocarbon. (b) n-Butyl alcohol from acetylene. (c ) Ally alcohol from propene. (d) Glycerol from acetone or isopropyl alcohol or propene. (e ) Glycerol from carbon and hydrogen or acetylene. (f) n-Propyl alcohol from ethylene oxide. (g) tert-Butyl alcohol from acetone. (h) Isopropyl alcohol from acetaldehyde. (i) 2-methyl propan-1-ol from formaldehyde. (j) Cyclohexyl methanol from formaldehyde. (k) 1-Phenylethanol from styrene (vinyl benzene). (l) tert-Amyl alcohol from methyl magnesium iodide. (m) 3,5-Dimethylhexan-3-ol from butan-2-one. (n) Butan-1-ol from but-1-ene. (o) Butan-2-ol from but-1-ene. (p) Allylalcohol from propane. |
|
Answer» `(##GRB_ORG_CHM_P2_C10_E01_009_A02##)` |
|
| 22. |
Outline synthesis of DL-tyrosine. |
Answer» SOLUTION :
|
|
| 23. |
Outline a synthesis of 4-methylpentanamine using the Gabriel synthesis. |
Answer» SOLUTION :
|
|
| 24. |
Outline a mechanism to account for different isomer formed when reacts with CH_(3)OH in acidic and in basic medium. |
| Answer» Solution :In ACIDIC medium `Me_(2)-overset(OCH_(3))overset(|)(C)-UNDERSET(OH)underset(|)(C)H_(2) ` and In basic medium `Me_(2)C-overset(OCH_(3))overset(|)(C)H_(2)` | |
| 25. |
Outer shells of two eggs are removed. One of the egg is placed in purewater and the other is placed in saturated solution of NaCl. What will beobserved and why ? |
| Answer» Solution :In PURE water the egg SWELLS and in SATURATED solution of NACL it will shrinks. | |
| 26. |
Outer electronic configuration of the element Palladium is |
| Answer» Answer :D | |
| 28. |
Out of zinc and tin, whose coating is better to protect iron objects? |
| Answer» SOLUTION :TIN COATING is BETTER. | |
| 29. |
Out of Sn and Zn which one protects Fe better evenafter cracks ? |
| Answer» Solution :ZINC PROTECTS better because oxidation of zinc is greater but that of tin is LESS than that of IRON. | |
| 30. |
Out of zinc and tin which one protects iron better even after cracks and why? |
| Answer» Solution :ZINC protects better because oxidation POTENTIAL of zinc is GREATER but that of tin is LESS than that of iron. | |
| 31. |
Out of which one is more reactive towards S_(N)1reaction? |
Answer» SOLUTION : is more REACTIVE TOWARDS `S_(N)1` reaction. |
|
| 32. |
Out of TiF_(6)^(2-),CoF_(6)^(3-) , Cu_(2)Cl_(2) and NiCl_(4)^(2-)(ZofTi =22, Co=27 , Cu =29 , Ni = 28), the colourless species are |
|
Answer» `Cu_(2)Cl_(2)` and `NiCl_(4)^(2-)` |
|
| 33. |
Out of TiF_6^(2-), Cu_2Cl_2, NiCl_4^(2-) and CoF_6^(3-) (Z of Ti=22 , Co=27 , Cu=29, Ni=28) the colourless species are |
|
Answer» `CoF_6^(3-)` and `NiCl_4^(2-)` |
|
| 34. |
Out of three isomeric nitrophenols, the p-nitrophenol is most acidic. |
|
Answer» |
|
| 35. |
Out of these the correct match is |
|
Answer» BAYER's method `-Na_2CO_3` |
|
| 36. |
Out of these which colloidal solution is not a lyophilic colloid ? |
|
Answer» GOLD sol |
|
| 37. |
Out of the various possible isomers of C_(7)H_(7)Cl containing a benzene ring, suggest the structure with the weakest C-Cl bond. |
Answer» Solution :Four isomers are possible: their STRUCTURE are: 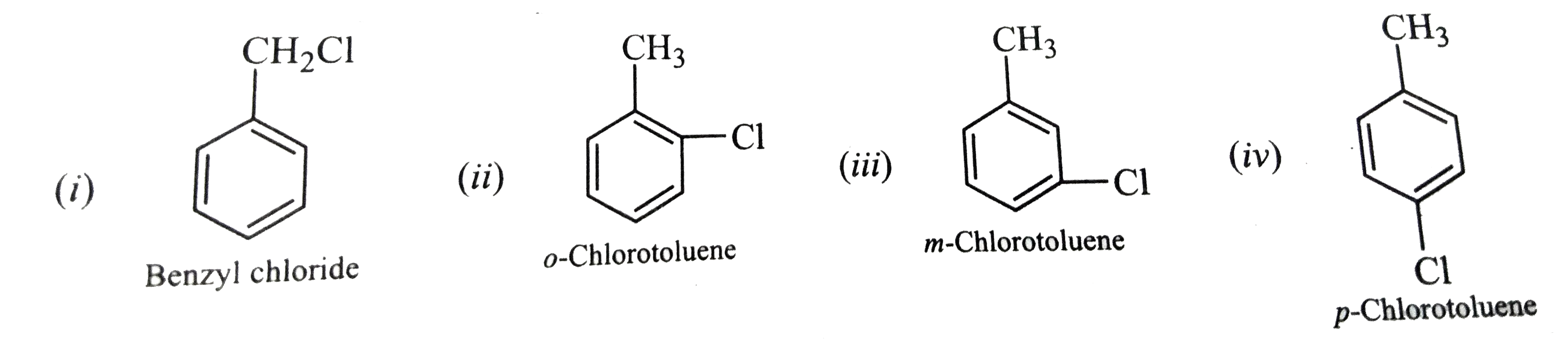 Just as in CHLOROBENZENE, the C-Cl bond is a pure single bond. Hence, out of the four isomers, the C-Cl bond is the weakest in benzyl chloride. Alternatively, the cleavage of C-Cl bond in benzyl chloride gives benzyl cation which is stabilized by RESONANCE. 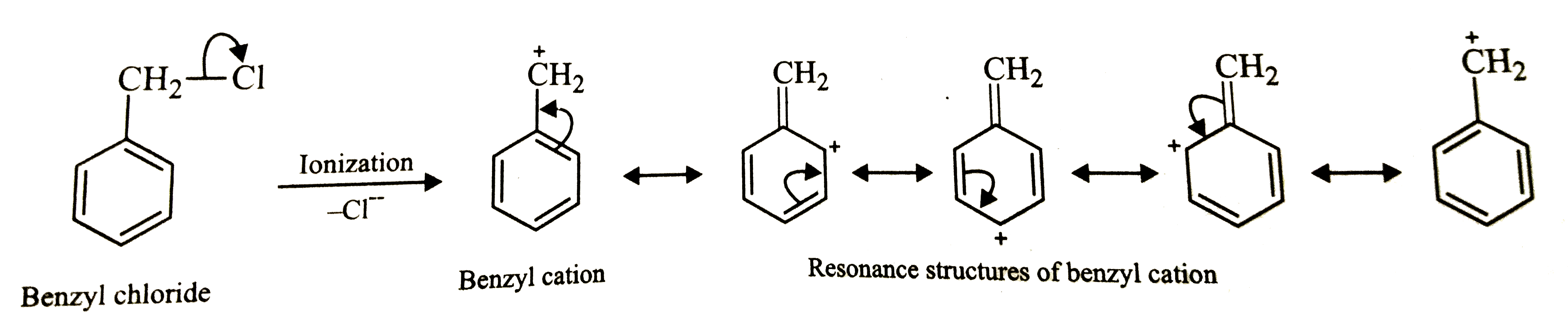 On the order hand, cleavage of C-Cl bond in o-, m- or p-chlorotoluenes gives toyl cation which is highly unstable. as a result, the energy of activation for the cleavage of C-Cl bond in benzyl chloride is much SMALLER that that of C-Cl bond in o-, m-or p-chlorotoluenes. CONSEQUENTLY, the C-Cl bond o- , m-or p-chlorotoluenes. Consequently, the C-cl bond is the weakest in benzyl chloride. |
|
| 38. |
Out of the various methods for determining the molar mass of the solute such as protein, which is considered to be the best? |
| Answer» SOLUTION :Osmotic pressure is CONSIDERED to be the best for determining the MOLAR MASS of the solute such as protein. | |
| 39. |
Out of the two compounds shown below, the vapour pressure of B at a particular temperature is expected to be : |
|
Answer» higher than that of A |
|
| 40. |
Out of the two bromoderivatives,C_(6)H_(5)CH(CH_(3))Br and C_(6)H_(5)CH(C_(6)H_(5))Br, which one is more reactive in S_(N^(1)) reaction and why? |
Answer» Solution : In the `S_(N^(1))` reaction, the REACTIVITY of an alkyl halide depends upon the stability of the CARBOCATION which is forme din Although both the carbocations are secondary in nature, the carbocation formed in the second reaction is more resonance STABILISED DUE to the presence of two phenyl groups that are involved in conjugation. therefore, the corresponding halogen DERIVATIVE is more reactive. |
|
| 41. |
Out of the two compound's shown below, the vapour pressure of B at a particular temperature is expected to be. |
|
Answer» HIGHER than that of A |
|
| 42. |
The p-isomer of dichlorobenzene has higher melting point than O-and M-isomer. Why ? |
| Answer» | |
| 43. |
Out of the ions Zn^(2+), Ni^(2+) and Cu^(3+) (At. Nos. Zn=30 Ni=28, Cr=24) |
|
Answer» only `Zn^(2+)` is COLOURLESS and `Ni^(2+)` and `Cr^(3+)`are coloured `Ni^(2+) and Cu^(3+)` have partly FILLED d - orbital thus coloured but `Zn^(2+)` has completely filled d- orbital, thus colourless. |
|
| 44. |
Out of the given values of temperature, which one is the highest? |
| Answer» Answer :C | |
| 45. |
Out of the two compounds below the vapour pressure of (B) at a particular temperature is |
|
Answer» lower than that of A |
|
| 46. |
Out of the following , which will NOT show geometrical isothermism ? |
|
Answer» `[PT(NH_3)_2(H_2O)_2]^(2+)` |
|
| 47. |
Out of the following which will not show geometrical isomerism ? |
|
Answer» `[CO(NH_(3))_(4)Cl_(2)]^(+)` |
|
| 48. |
Out of the following which reaction gives a colloidal solution ? |
|
Answer» `Cu+HgCl_2rarrCuCl_2+Hg` |
|
| 49. |
Out of the following which is Correct ? |
|
Answer» 1 Joule =1 Watt`xx` 1 COULOMB |
|
| 50. |
Out of the following which reaction give nonpolar product? |
|
Answer» `CH_(3)-C-=C-CH_(3)underset("Lead Acetate") overset (H_(2)-Pd//CaCO_(3))to` |
|