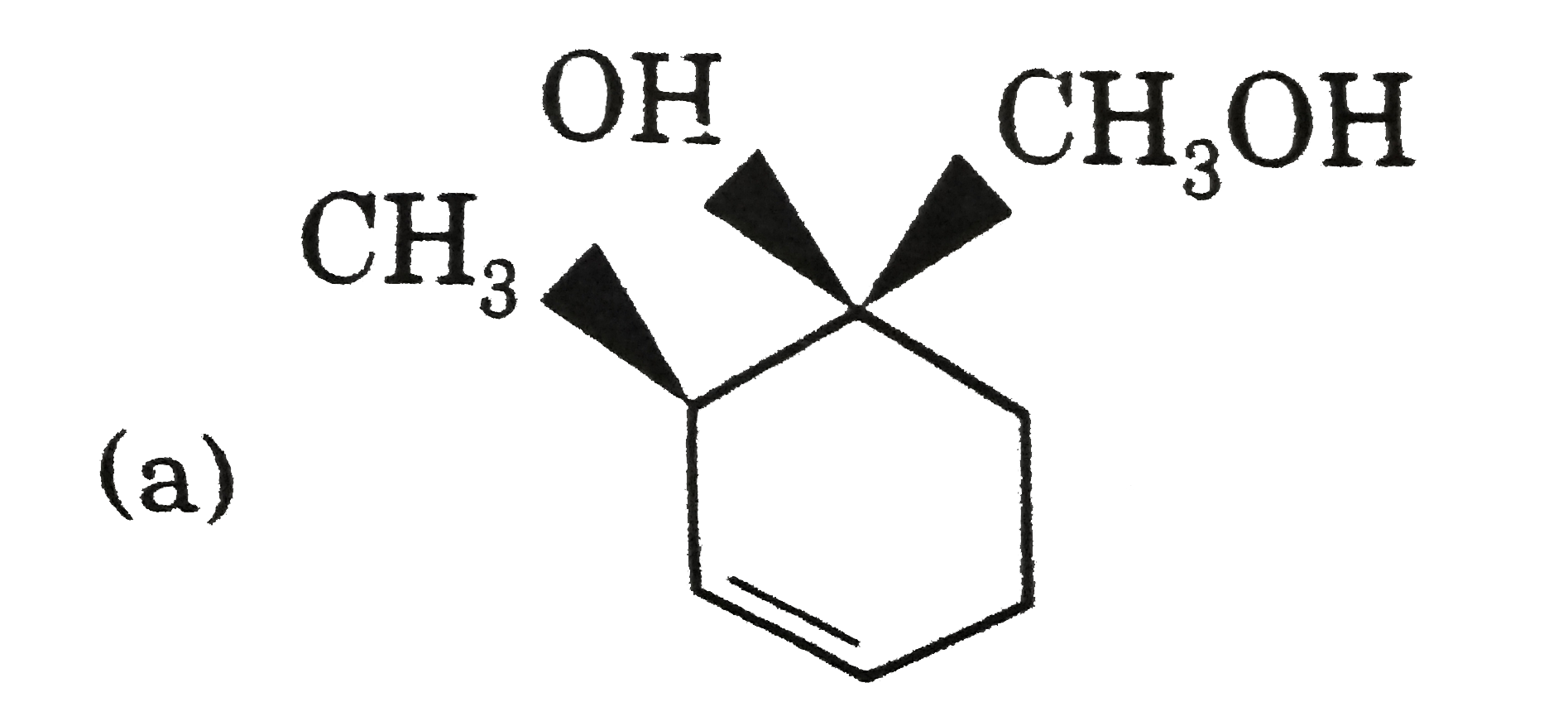
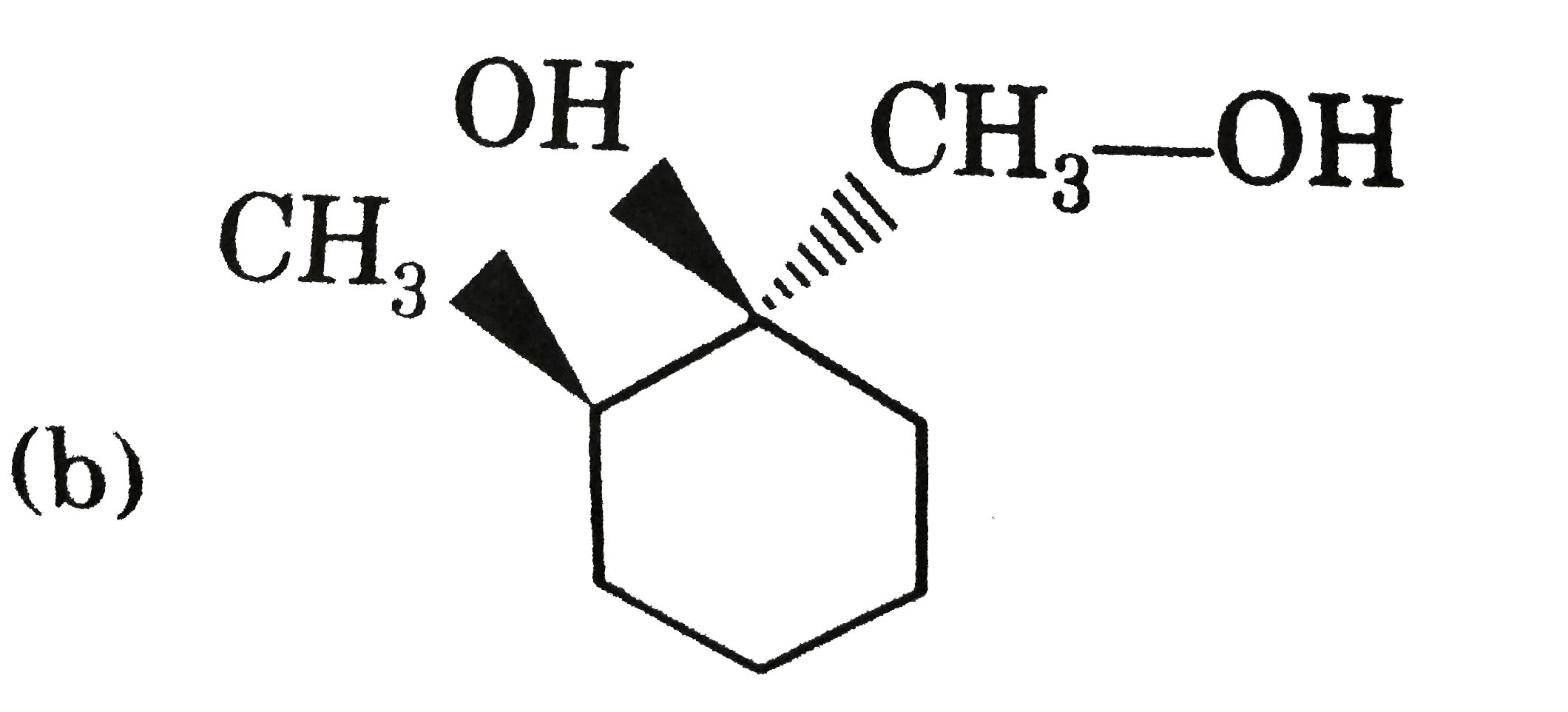
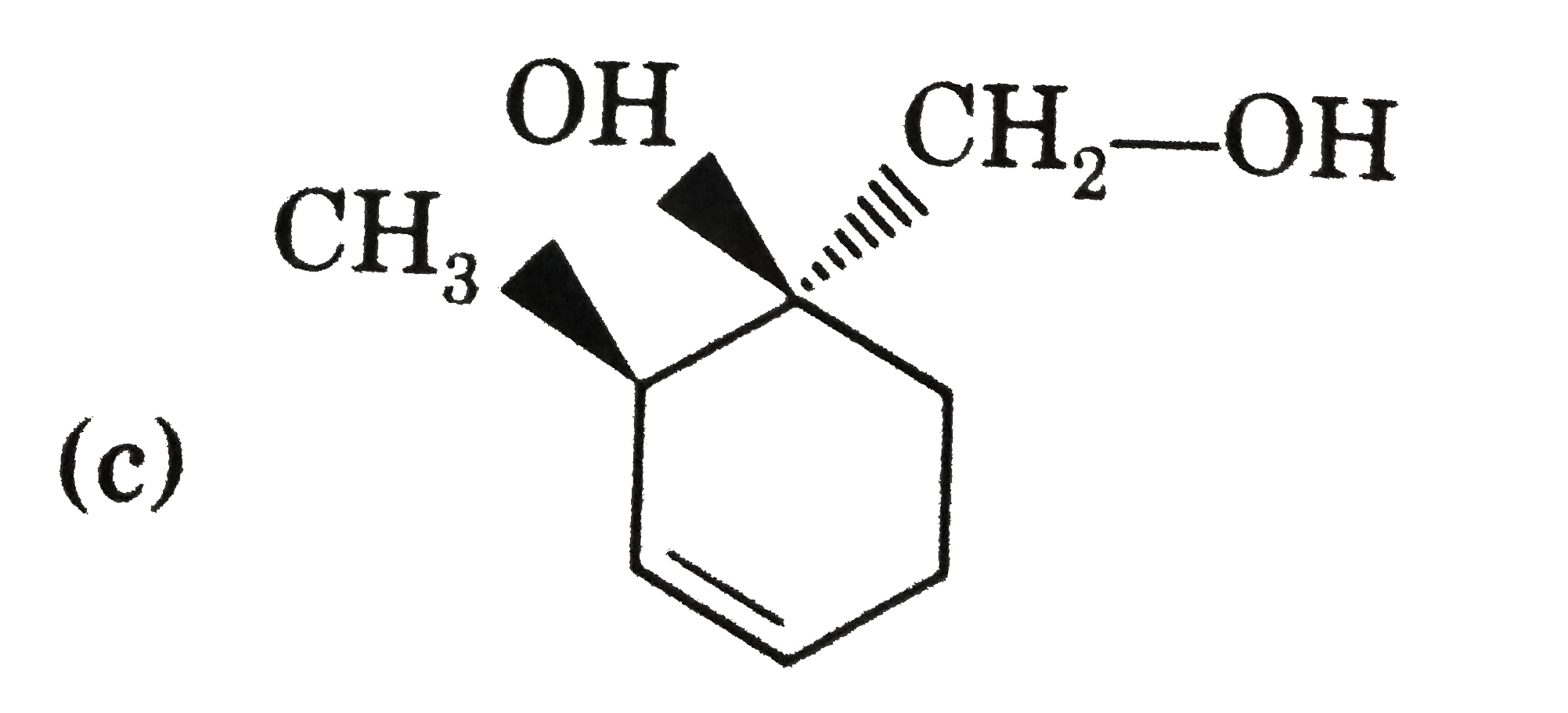
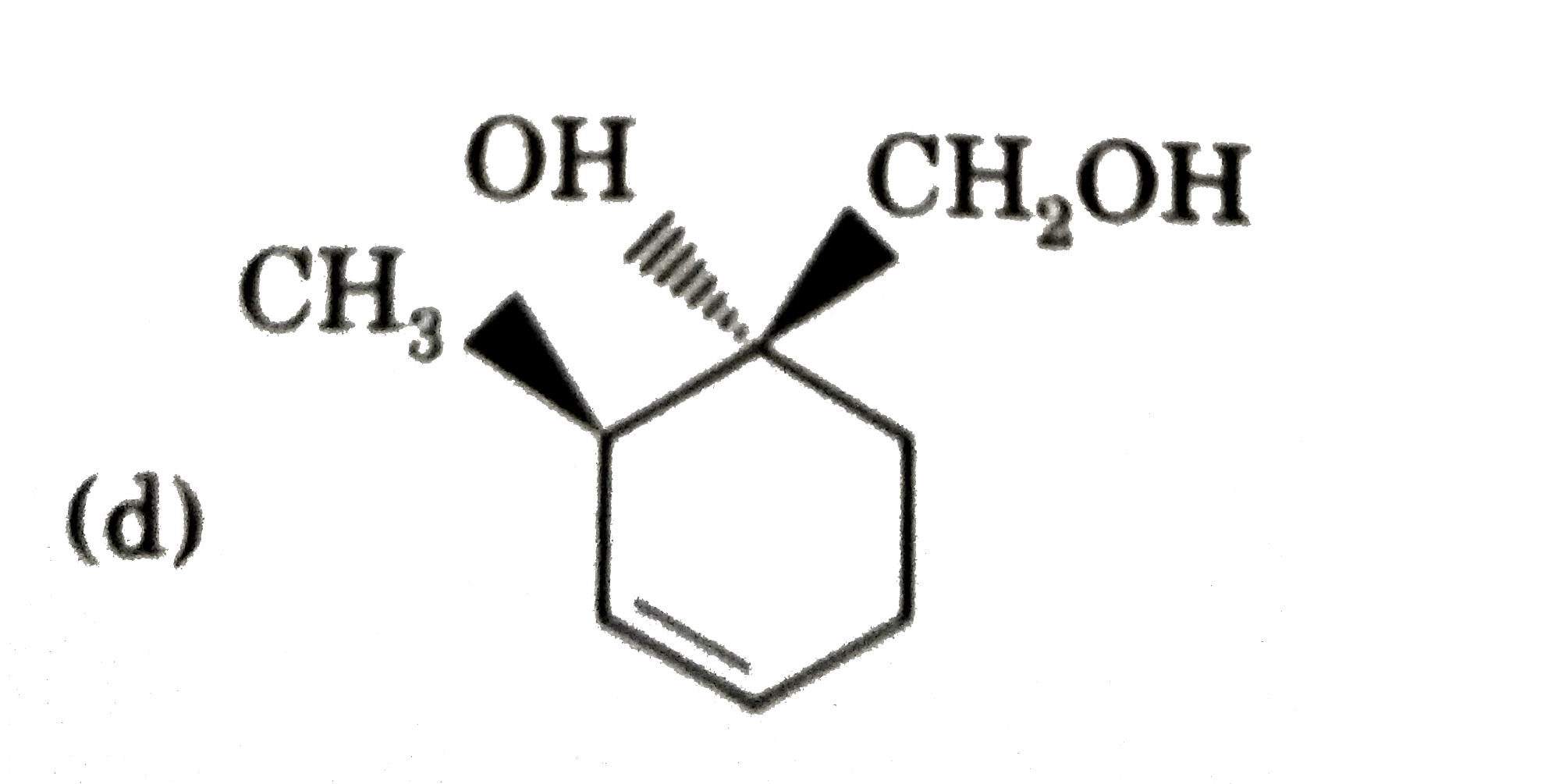
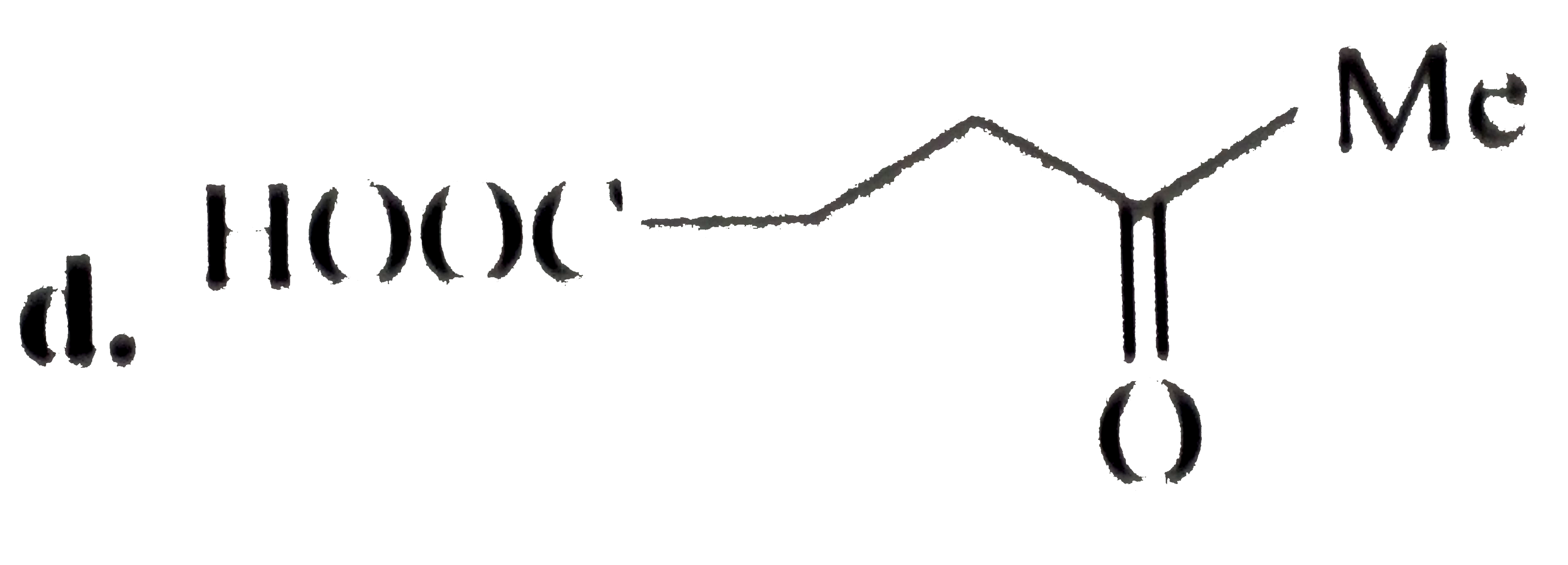Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Oxidation in absorbed by molten Ag, which is evolved on cooling and the silver particles are scattered, this phenomenon is known as: |
|
Answer» SILVERING of mirror |
|
| 2. |
Oxidatio potential of Al, Cu,Zn and Ag are given and series of reaction . Identify the correct group pf reaction : E_(Al)^(0) = 1.66 V, E_(Cu)^(0) = -0.34 V, E_(Zn)^(0) = 0.76 V, E_(Cu)^(0) = -0.8 V |
|
Answer» `AlCl_(3) + 3Ag to 3AgCl + Al`, |
|
| 3. |
Oxidation and reduction take place in a cell, then its electromotive force will be |
|
Answer» Positive |
|
| 4. |
(A): Oxidation ability increases from HOCI to HCIO_4 (R): Oxidation number of chlorine increases from HOCI to HCIO_4. |
|
Answer» Both (A) and (R ) are TRUE and (R ) is the CORRECT EXPLANATION of (A) |
|
| 5. |
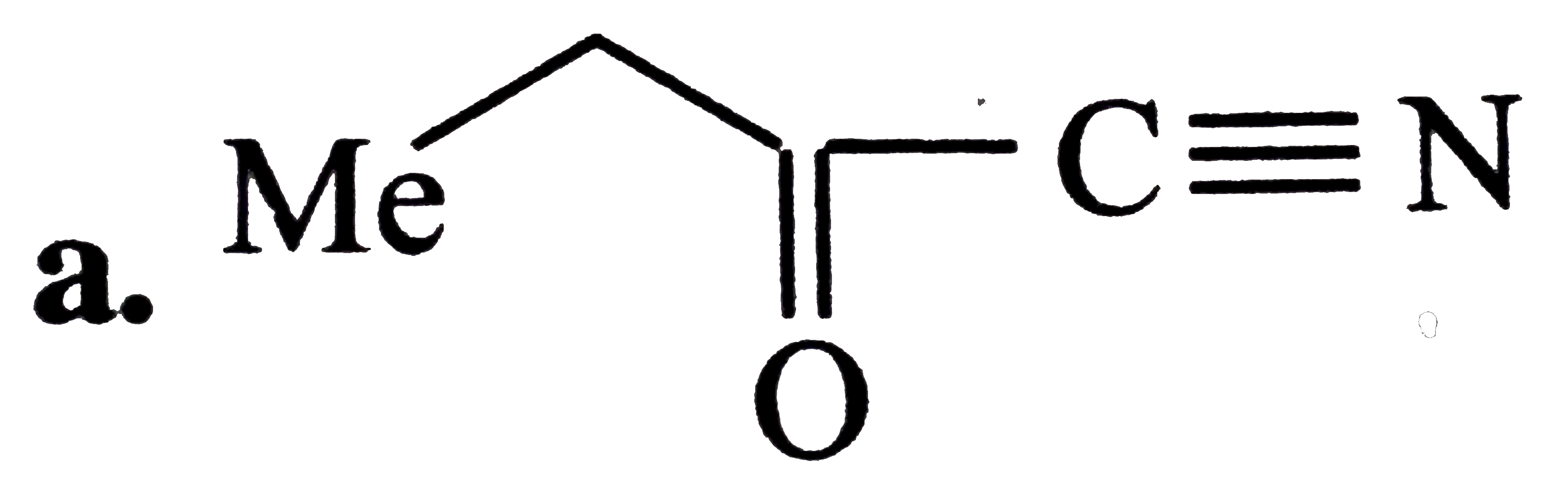
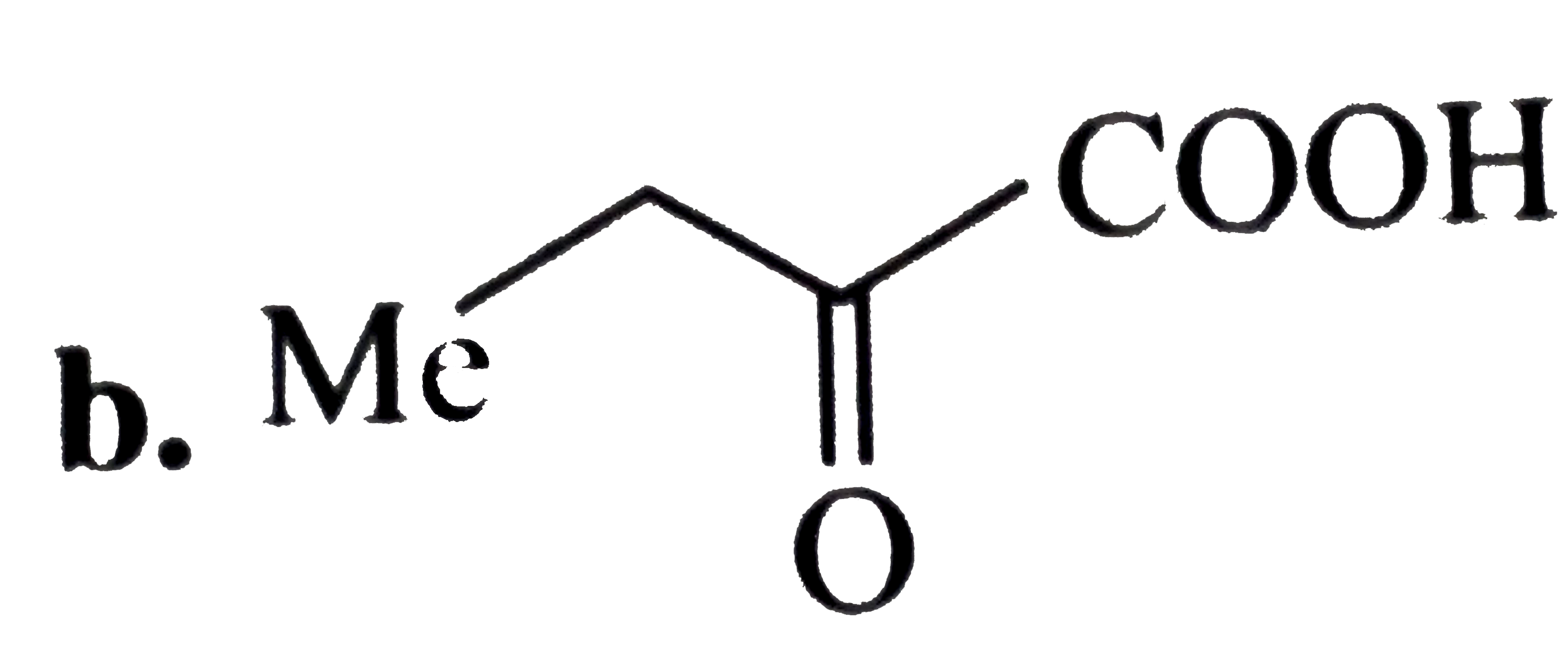
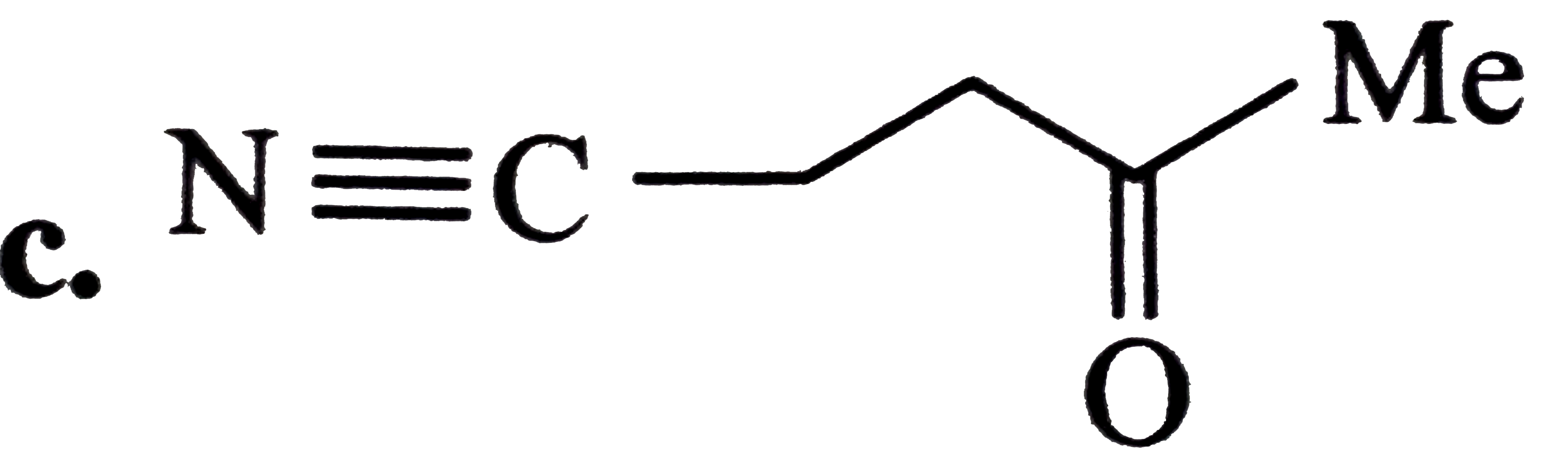
Oxidatinio of acetaldoxime with ……………….. Gives 1- nitro ethane. |
|
Answer» |
|
| 7. |
Oxiacid are compounds in which X-OH bond is present where X is a non-metal generally. Which of the following oxy-salt does not exist? |
|
Answer» SODIUM HYDROGEN phosphate |
|
| 9. |
Oxalic (ethanedioic acid ) acid is prepared on a large scale by : |
|
Answer» Hydrolysis of `C_2H_4` |
|
| 10. |
Oxalic acid when reduced with zinc and H_(2)SO_(4) gives |
|
Answer» Glyoxallic acid |
|
| 11. |
Oxalic acid reacts with con. H_2SO_4 to produce |
|
Answer» `SO_2` |
|
| 12. |
Oxalic acid reacts with NaOH according to the given reaction. (COOH)_2 + 2NaOH to (COONa)_2 + 2H_2 O If 0.816 g of oxalic acid dihydrate. (COOH)_2 2H_2 O, is dissolved in 1 L of water and titrated with 0.120 M NaOH solution, what volume of NaOH will be needed? |
|
Answer» 108 ml |
|
| 13. |
Oxalic acid on being heated upto 90^(@)C with conc. H_(2)SO_(4) forms |
|
Answer» `HCOOH+CO_(2)` |
|
| 14. |
Oxalic acid when heated with conc.H_2SO_4 gives: |
|
Answer» `CO+H_2O_2` |
|
| 15. |
Oxalic acid may be distinguished from tartaric acid by |
|
Answer» SODIUM BICARBONATE solution |
|
| 16. |
Oxalic acid may be distinguished from tartaric acid by : |
|
Answer» `NaHCO_3` |
|
| 17. |
Oxalic acid, malonic acid and succinic acid can be distinguished by : |
|
Answer» Heat |
|
| 18. |
(A) OX^(-) on heating in the presence of OH^(-) gives X^(-)andXO_(3)^(-)(X=Cl,Br,I,) (R ) Conversion of OX^(-) to XandXO_(3)^(-) is called disproportionation |
|
Answer» Both (A) and (R ) are TRUE and (R ) is the correct EXPLANATION of (A) |
|
| 20. |
overset(V) (Cr) O_(4)^(3-)in acidic mediumundergoes disproportionation to form "…................" and "…...................". |
| Answer» Solution :`OVERSET(VI)(Cr) O_(4)^(2-) , overset(III) (Cr^(3+))` | |
| 21. |
overset("SOCl"_(2))(rarr) (A) overset("KCN")(rarr) (B) overset(H^(+))(rarr)(C ), the compound (C ) is |
|
Answer»
|
|
| 22. |
overset(Sn-HCl)toA overset(NaNO_(2)-HCl)underset(0-5^(@)C)toB overset(Cu_(2)Cl_(2)//KCl)toC The compound 'C' in the above sequence is |
|
Answer»

|
|
| 23. |
overset(NaNO_(2)+HCI)rarr (B) overset((i)Ac_(2)O)underset((ii)H_(2)O^(o+))rarr (C ) The final product (C ) is: |
|
Answer»
|
|
| 24. |
overset(OH^(-))underset(/_\)toX Total number of pi bonds in X is |
|
Answer» |
|
| 25. |
overset("Monochlorination")(to) how many enantiomeric pairs can be formed. |
Answer» 
|
|
| 26. |
overset(I)overset("|")(PhCHBr) overset(PhC equiv CPh)underset("EtOK")(rarr)A Compound 'A' is : |
|
Answer»
|
|
| 27. |
overset(HlO_(4))to product is |
|
Answer»
|
|
| 28. |
overset(KCN)rarr(A)overset(H_(3)O^(+))rarr(B)overset(Delta)rarr(C) Identify the correct statement(s) about the above sequence of reactions : |
|
Answer» Compound (A) is formed through `S_(N)` reaction |
|
| 29. |
overset((i)LiAIH_(4))underset((ii)H_(2)O)to(A) Product (A) of above reaction is : |
|
Answer»
|
|
| 30. |
overset(HlO_(4)(to) x underset(/_\) overset(Conc. barO H)to A+B (A contains 3 oxygen atom per molecule) Aunderset(/_\) overset("acidified" with H^(+))toC DBE of C is |
Answer» 
|
|
| 31. |
overset"HOCl"to A overset"HOCl"to B overset(-H_2O)to O Product 'C' is :- |
|
Answer» `CH_3-CH_2-CH=O` 
|
|
| 32. |
overset("Heat")toAoverset(NH_(3))toBoverset(NaOH)toCunderset(KOH)overset(Br_(2))toDoverset(H^(+))toE,the product E is : |
|
Answer» SALICYLIC acid 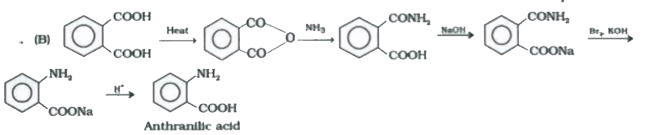
|
|
| 33. |
overset(HBr)underset((Cs_(2)))rarrB (halogen compound) A & B respectively are |
| Answer» ANSWER :A | |
| 34. |
overset(CH_(3)(1eq.))rarr overset(Ag_(2)O(moist.))rarr overset(Delta)rarrP ("Major product") +3^(@) amine Write the number of carbon atoms in P (major product) in given reaction? |
|
Answer» Ist step is `S_(N)2`, reaction and IIND step is Hofman eliminatio. |
|
| 35. |
overset(CH_(2)-OH)overset(|)underset(CH_(2)-OH)( )overset(PI_(1))to________? |
|
Answer» ETHANE |
|
| 36. |
overset(CH_(3)MgBr)toA overset(H_(3)O^(+))toB. The compound ‘B' is |
|
Answer»

|
|
| 37. |
overset("Alo KOH")to Possible product are : |
|
Answer»
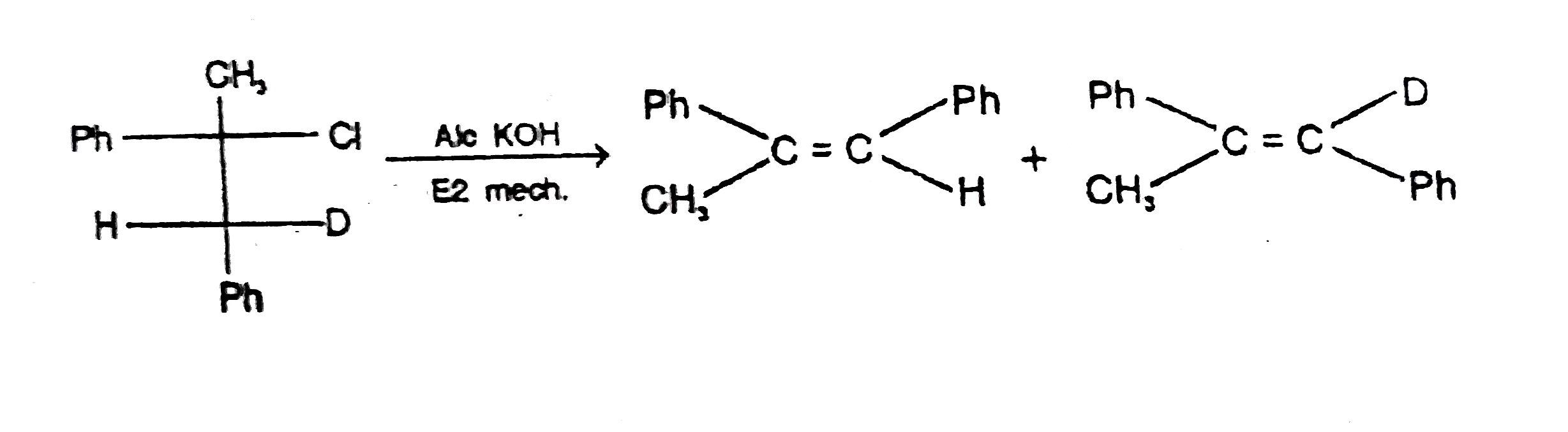
|
|
| 38. |
overset(CH_(2)-CN)overset(|)underset(CH_(2)-CN)underset(|)((CH_(2))_(7)) overset(H_(3)O^(+)Delta)toA The number of oxygen atoms in A |
|
Answer» |
|
| 40. |
overset(AlCl_(2))rarrAunderset(HCl)overset(Zn(Hg))rarrBoverset(SOCl_(2))rarrCoverset(AlCl_(3))rarr Doverset(LiAlH_(4))rarrEoverset(H^(+)//Delta)rarrFoverset(NBS)rarrGoverset("alcoholic" KOH)rarrH Identify A to H. |
|
Answer» |
|
| 41. |
overset(AlCl_(3))rarrA, What is A ? |
|
Answer» |
|
| 42. |
overset(AICI_(4))(to) A underset(130^(@)C)overset(O_(3))(to)(B) overset(H^(+))(to) Phenol + C Identify ‘C’ in the following is: |
|
Answer» Water |
|
| 43. |
overset(+)NH_(3)-CH(R )-CO_(2)H hasits Ka_(1)=10^(-7). The isoelectric point of the a-amino acidoccur at pH = 8.2 hence pKa_(2) of the conjugate acid of the alpha -amino acid is equal to : |
|
Answer» 9.4 `:.""8.2=(7+pK_(a)^(2))/(2)` `rArrpK_(a)^(2)=16.4-7=9.4` |
|
| 44. |
overset(14)(C)H_(2)=CH-CH_(3)overset(NBS)underset(C Cl_(4),Delta)(to)overset(Na)underset("Dry ether",Delta)(to)"Products". Products can be : |
|
Answer» `CH_(2)=CH-overset(14)(C)H_(2)-CH_(2)-CH=overset(14)(C)H_(2)` |
|
| 45. |
overset(-)(M_n)and overset(-)(M_w)of a synthetic polymer are related as |
|
Answer» `OVERSET(-)(M_n) LT overset(-) (M_W) ` |
|
| 46. |
overset ("NBS") underset ("hv") to " P is " |
|
Answer»
|
|
| 47. |
overset("CHO")underset("CHO")("|")overset(NaOH)toX The product (X) will be |
|
Answer» `overset("CH"_(2)ONa)underset("COONA")("|")` |
|
| 48. |
Over production of acid in the stomach causes irritation and pain. In severe cases, ulcers are developed in the stomach. Until 1970, only treatment for acidity was administration of antacids, such as sodium hydrogencarbonate or a mixture of aluminium and magnesium hydroxide. However, excessive hydrogencarbonate can make the stomach alkaline and trigger the production of even more acid. Metal hydroxides are better alternatives because of being insoluble, these do not increase the pH above neutrality. These treatments control only symptoms, and not the cause. Therefore, with these metal salts, the patients cannot be treated easily. In advanced stages, ulcers become life threatening and its only treatment is removal of the affected part of the stomach. A major breakthrough in the treatment of hyperacidity came through the discovery according to which a chemical, histamine stimulates the secretion of pepsin and hydrochloricacid in the stomach. The drug cimetidine (Tegamet), was designed to prevent the interaction of histamine with the receptors present in the stomach wall. This resulted in release of lesser amount of acid. The importance of the drug was so much that it remained the largest selling drug in the world until another drug, ranitidine (Zantac), was discovered. Name two drugs that have helped check acidity without causing damage to the stomach. |
| Answer» SOLUTION :TEGAMET and Zantac. | |
| 49. |
overser(H^(+))rarr? Product is |
|
Answer»
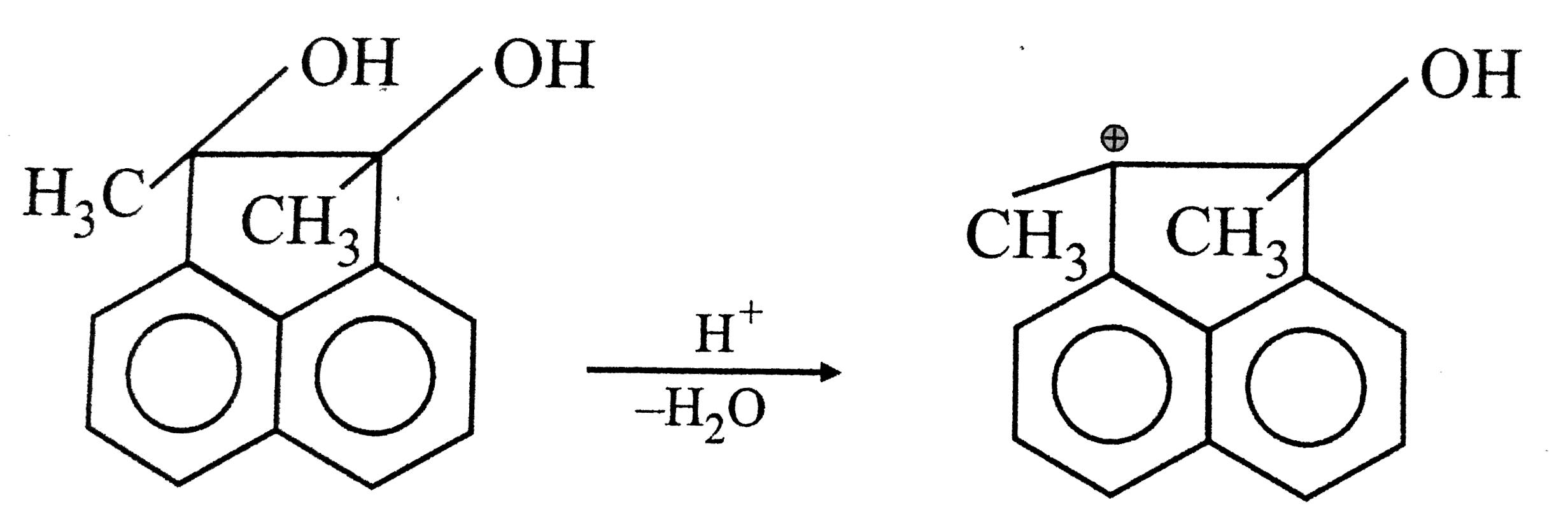 (II) But in this carbocation EVERY atom has COMPLETE octet. Hence (II) is more STABLE than (I). 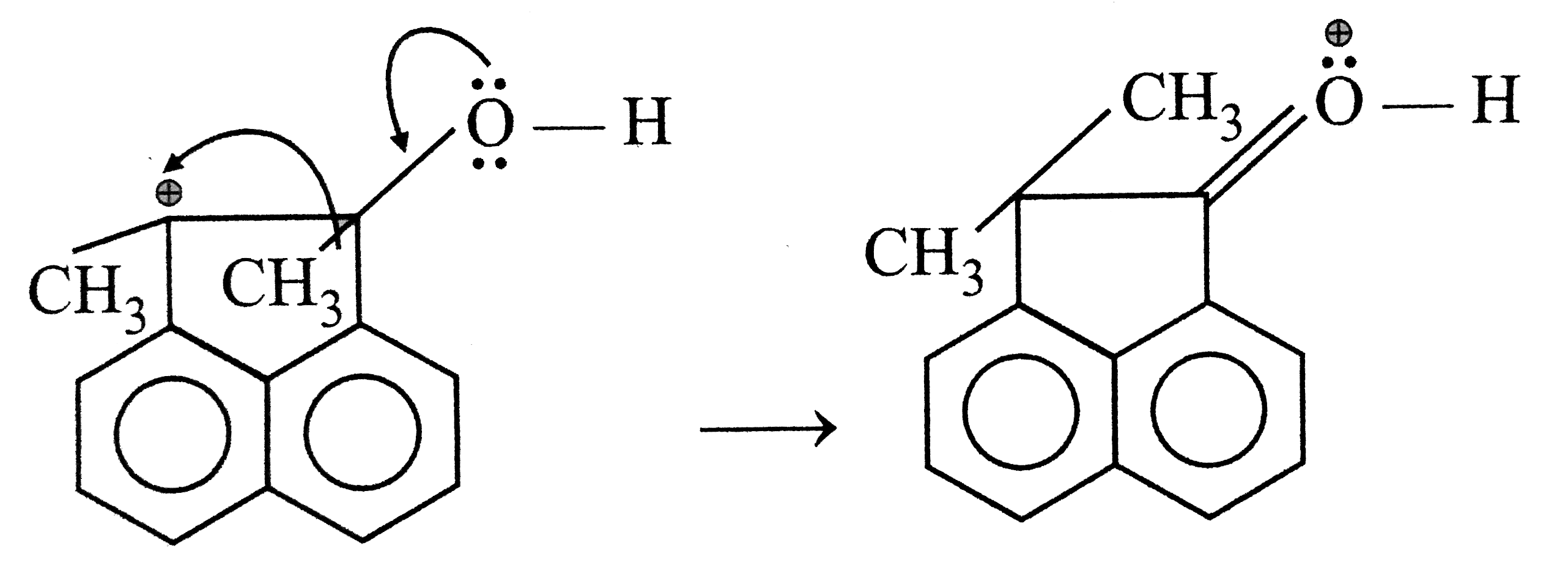
|
|
| 50. |
Over production of acid in the stomach causes irritation and pain. In severe cases, ulcers are developed in the stomach. Until 1970, only treatment for acidity was administration of antacids, such as sodium hydrogencarbonate or a mixture of aluminium and magnesium hydroxide. However, excessive hydrogencarbonate can make the stomach alkaline and trigger the production of even more acid. Metal hydroxides are better alternatives because of being insoluble, these do not increase the pH above neutrality. These treatments control only symptoms, and not the cause. Therefore, with these metal salts, the patients cannot be treated easily. In advanced stages, ulcers become life threatening and its only treatment is removal of the affected part of the stomach. A major breakthrough in the treatment of hyperacidity came through the discovery according to which a chemical, histamine stimulates the secretion of pepsin and hydrochloricacid in the stomach. The drug cimetidine (Tegamet), was designed to prevent the interaction of histamine with the receptors present in the stomach wall. This resulted in release of lesser amount of acid. The importance of the drug was so much that it remained the largest selling drug in the world until another drug, ranitidine (Zantac), was discovered. Why is acidity caused in the stomach ? |
| Answer» SOLUTION :ACIDITY is CAUSED by histamine which STIMULATES the RELEASE of pepsin and hydrochloric Ans. acid. | |