Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Optical isomerism is shown by : |
|
Answer» Butanol-1 |
|
| 2. |
Optical isomerism is exbibited by (ax= oxalate anion, en=ethylenediamine) |
|
Answer» cis `[CrCl_(2)("ox")_(2)]^(3-)` |
|
| 3. |
Optical isomerism is exhibited by a complex with |
|
Answer» coordination NUMBER 4, with a bidentate LIGAND |
|
| 4. |
Optical isomerism arises due to the presence of: |
|
Answer» An ASYMMETRIC carbon atom |
|
| 5. |
Optical isomer of molcular formula C_4H_10O on alpha-elimination gives |
|
Answer» butanal |
|
| 6. |
Optical activity of a substance is due to |
|
Answer» PRESENCE of ALDEHYDE group |
|
| 7. |
Optical activity is measured by : |
|
Answer» REFRACTOMETER |
|
| 8. |
Optical activityis expected for . |
|
Answer» 2-methylpropanoicacid |
|
| 9. |
Optical active compounds have same |
|
Answer» REFRACTIVE index |
|
| 10. |
Optical active amineof molecularformulaC_(4)H_(11)N on reaction withnitrousacid gives |
|
Answer» butan -1-ol `CH_(3) - underset(NH_(2))(CH)- CH_(2) - CH_(3) + HNO_(2) overset("Cold")toCH_(3)- underset(OH) (CH) - CH_(2) - CH_(3) - N_(2) + H_(2) O` |
|
| 11. |
O=overset(H)overset(|)(C)-underset(H)underset(|)(C)=Ooverset(overset(ө)(O)H)to O=underset(.^(ө)O)underset(|)(C)-underset(H)underset(|)overset(H)overset(|)(C)-OH Given reaction is cannizaro reaction at normal concentration of base. Predict the order of above reaction. |
|
Answer» 1 |
|
| 12. |
Oppenauer oxidation is the reverse process of : |
|
Answer» Wolff Krishner's REDUCTION |
|
| 13. |
Opening and closing of flower is controlled by …….. |
|
Answer» CAPILLARY action |
|
| 14. |
Oppanauer oxidation is the reverse process of |
|
Answer» Wolff Kisner REDUCTION |
|
| 15. |
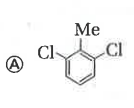
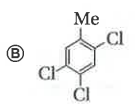
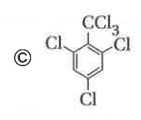
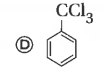
Only which oneof the following compounds is obtained when excess of Cl_2(g) is passed through boiling toluene- |
|
Answer»

|
|
| 16. |
Only two isomeric monochloro derivatives are possible for : |
|
Answer» 2- METHYLPROPANE |
|
| 17. |
Only two isomeric monochloro derivatives are possible for :- |
|
Answer» `N`-PENTANE |
|
| 18. |
Only two isomeric monochloro derivative are possible for |
|
Answer» n-hexane 1 `(CH_(3))CHCH_(2)Cl`- isobutyl chloride 2. `(CH_(3))_(3) C Cl` t-butyl choride |
|
| 19. |
Only two isomeric monochloro derivaties are possible from : |
|
Answer» n-Pentane |
|
| 20. |
Only two isomeric monochloro derivatives are possible for: |
|
Answer» n-butane |
|
| 21. |
Only those atoms which four covalent bonds produce a repetitive three dimesional structure using only covalent bonds., e.g. , diamond structure.The latter is based on a FCC lattice where lattice points are occupied by carbon atoms.Every atom in this structure is surrounded tetrahedrally by four others.Germanium, silicon and grey tin also crystallize in the same way as diamond. (Given :N_A=6xx10^(23) sin 54^@44'=0.8164) Total number of diamond unit coils in 1.2 gm of diamond sample is |
|
Answer» `6.0xx10^21` |
|
| 22. |
Only those atoms which four covalent bonds produce a repetitive three dimesional structure using only covalent bonds., e.g. , diamond structure.The latter is based on a FCC lattice where lattice points are occupied by carbon atoms.Every atom in this structure is surrounded tetrahedrally by four others.Germanium, silicon and grey tin also crystallize in the same way as diamond. (Given :N_A=6xx10^(23) sin 54^@44'=0.8164) If the edge length is 3.60Å, density of diamond crystal is |
|
Answer» 3.92 GM/cc |
|
| 23. |
Only those atoms which four covalent bonds produce a repetitive three dimesional structure using only covalent bonds., e.g. , diamond structure.The latter is based on a FCC lattice where lattice points are occupied by carbon atoms.Every atom in this structure is surrounded tetrahedrally by four others.Germanium, silicon and grey tin also crystallize in the same way as diamond. (Given :N_A=6xx10^(23) sin 54^@44'=0.8164) If edge length of the cube is 3.60Å, then radius of carbon atom is |
|
Answer» 0.78Å |
|
| 24. |
Only the surface atoms in an adsorbent, play an active role in adsorption. These atoms posses some residual forces like vander Waal's forces and chemical forces. In the process of adsorption, weak adsorbate is substituted by strong adsorbate. Activated charcoal used in gas mask is already exposed to the atmospheric air, so the gases and water vapours in air are adsorbed on its surface. When the mask is exposed to chlorine atmosphere, the gases are displaced by chlorine. Porous and finely powdered solids, e.g., charcoal and Fuller's earth adsorb more as compared to the hard non-porous material. It is due to this property that the powdered characoal is used in gas masks. In general, easily liquefiable gases like CO_2, NH_3, Cl_2, and SO_2 etc., are adsorbed to a greater extent than the elemental gases, eg., H_2, N_2, O_2, He, etc. Which of the following gases will substitute O_2 from adsorbed charcoal |
|
Answer» `H_2` |
|
| 25. |
Only the surface atoms in an adsorbent, play an active role in adsorption. These atoms posses some residual forces like vander Waal's forces and chemical forces. In the process of adsorption, weak adsorbate is substituted by strong adsorbate. Activated charcoal used in gas mask is already exposed to the atmospheric air, so the gases and water vapours in air are adsorbed on its surface. When the mask is exposed to chlorine atmosphere, the gases are displaced by chlorine. Porous and finely powdered solids, e.g., charcoal and Fuller's earth adsorb more as compared to the hard non-porous material. It is due to this property that the powdered characoal is used in gas masks. In general, easily liquefiable gases like CO_2, NH_3, Cl_2, and SO_2 etc., are adsorbed to a greater extent than the elemental gases, eg., H_2, N_2, O_2, He, etc. Gas mask contains |
|
Answer» CHARCOAL granules |
|
| 26. |
Only temporary hardness of water is removed by |
|
Answer» CALGON's method |
|
| 27. |
Only the chiral molecule show the phenomenon of |
|
Answer» OPTICAL ISOMERS |
|
| 28. |
Only the surface atoms in an adsorbent, play an active role in adsorption. These atoms posses some residual forces like vander Waal's forces and chemical forces. In the process of adsorption, weak adsorbate is substituted by strong adsorbate. Activated charcoal used in gas mask is already exposed to the atmospheric air, so the gases and water vapours in air are adsorbed on its surface. When the mask is exposed to chlorine atmosphere, the gases are displaced by chlorine. Porous and finely powdered solids, e.g., charcoal and Fuller's earth adsorb more as compared to the hard non-porous material. It is due to this property that the powdered characoal is used in gas masks. In general, easily liquefiable gases like CO_2, NH_3, Cl_2, and SO_2 etc., are adsorbed to a greater extent than the elemental gases, eg., H_2, N_2, O_2, He, etc. Which of the following gases will be most easily adsorbed by the charcoal in the gas mask |
|
Answer» `H_2` |
|
| 29. |
Only S_N1 mechanism is observed in : |
|
Answer» <P>(I)(i)(R) 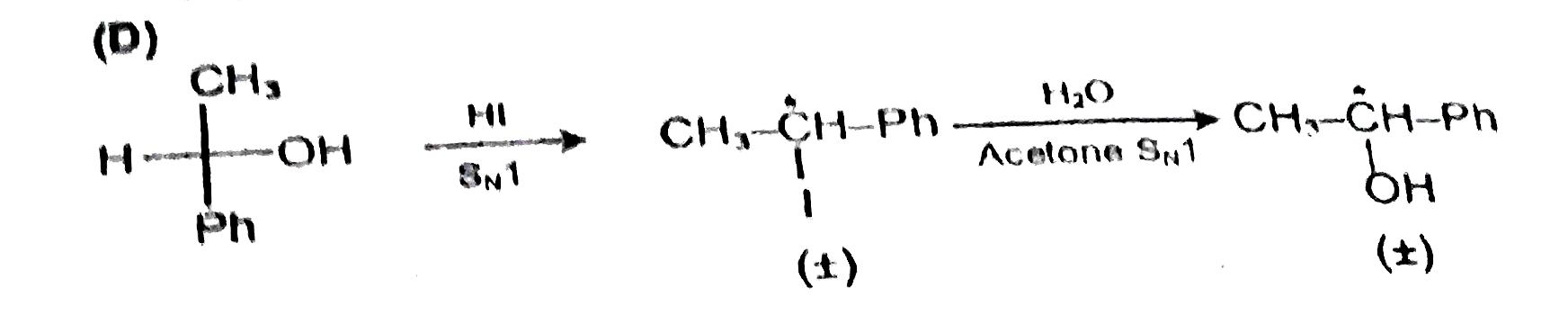
|
|
| 30. |
Only one of the following amines will lose its nitrogen atom as trimethyl amine by repeated Hofmann elimination reactions |
|
Answer»

|
|
| 31. |
Only N_(2) and CO_(2) gases remain after 15.5 g of carbon is treated with 25 litres of air at 25^(@)C and 5.5 atm pressure. Assume air of composition: O_(2)-19%,N_(2)-80% and CO_(2)-1% (by volume). Calculate the heat evolved under constant pressure Given, C+O_(2) to CO_(2),DeltaH=-94.05"kcal/mole" C+(1)/(2)O_(2) to CO,DeltaH=-26.41"kcal/mole" |
|
Answer» SOLUTION :Moles of `C=(15.5)/(12)=1.292` Moles of `O_(2)=(pV)/(RT)=(5.5xx(0.19xx25))/(0.0821xx298)=1.068` `C+O_(2) to CO_(2)` x moles (say) x moles `C+(1)/(2)O_(2) to CO` `(1.292-x)"moles" (1.292-x)"moles"` As `O_(2)` is fully consumed, number of moles of O before reaction = number of moles of O after reaction =moles of O in `CO_(2)+"moles"` of O in CO or `2xx1.068=2x+(1.292-x)` or `x=0.844` MOLE of `CO_(2)=0.844` Mole of `CO=1.292-0.844=0.448`. Total HEAT evolved `=0.844(-94.05)+0.448(-26.41)=-91.2"kcal"` |
|
| 32. |
Only………………..of the available volume is occupied by the spheres in simple cubic packing. |
| Answer» SOLUTION :`52.31%` | |
| 33. |
Only N_2,CO and CO_2 gases remain after 0.72 gm of carbon is treated with one litre of air at 27^@C and 4.92 atm pressure.Assume air composition O_2=20%,N_2=79%,and CO_2=1% (by volume).The heat evolved (in Kcal) under constant pressure is : Given : C+OtoCO_2, DeltaH=-100 Kcal/mol C+1/2O_2toCO, DeltaH=-25 Kcal/mol |
|
Answer» `n_(O_2)=0.2xx0.2=0.04 mol, n_c=0.72/12` `{:(,C" "+,1/2O_2,toCO),("initial MOLE",0.06,0.04,"" 0),("final mole",0,0.01,""0.06):}` `{:(,CO" "+,1/2O_2,toCO_2),("initial mole",0.06,0.01,"" 0),("final mole",0.04,0,""0.02):}` Heat evolved =`0.04xx25+0.02xx100=1+2=3 kcal` |
|
| 34. |
What happens when CHCl_3 reacts with oxygen in presence of sunlight? |
|
Answer» propane |
|
| 35. |
Only iodine forms hepta -fluoride IF_(7), but chlorine and bromine give penta -fluorides.The reason for this is |
|
Answer» low electron affinity of iodine |
|
| 36. |
Only an aldehyde having….can undergo the aldol condensation |
|
Answer» At LEAST one beta H atom |
|
| 37. |
One would come across the terms isotactic,syndiotactic,and atactic in connection with the chemistry of: |
|
Answer» POLYMERS |
|
| 39. |
One way of writing the equation of state for real gases is pbarV=RT[1+B/(barV)+...] The constant B is equal to |
|
Answer» `(a-b/(RT))` |
|
| 40. |
One weak acid (like CH_(3)COOH) and its strong base together with salt (like CH_(3)COONa) is a buffer solution. In which pair this type of characteristic is found. |
|
Answer» HCL and NaCl |
|
| 41. |
One way of writing the equation of state for real gases is PV=RT[1+(B)/(V)+….] where B is a constant. Derive an approximate expression for B in terms of the van der Waals constants a and b. |
|
Answer» Solution :The van der Waals equation for 1 MOLE of a gas is `(p+(a)/(bar(V)^(2)))(bar(V) - b) = RT` where `bar(V)` is the molar volume. or `(p+(a)/(bar(V)^(2))) = (RT)/((bar(V) - b))` or `(p bar(V) + (a)/(bar(V))) = (bar(V))/((bar(V) - b)) RT` or `(p bar(V))/(RT) + (a)/(bar(V)RT) = (bar(V))/(bar(V) - b)` `(p bar(V))/(RT) = (bar(V))/(bar(V) - b) - (a)/(RT bar(V))` `= (1)/((1-(b)/(bar(V)))) -(a)/(RT bar(V)) = (1-(b)/(bar(V)))^(-1) - (a)/(RT bar(V))` At LOW pressures, `(b)/(bar(V)) lt lt 1` so that we can expand the first term using `(1-x)^(-1) = 1 + x + x^(2) +`........... . This YIELDS the virial equation in terms of volume : `(PBAR(V))/(RT) = [1+(b)/(bar(V)) + ((b)/(bar(V)))^(2) +.....] - (a)/(RTbar(V))` `= 1 + (b-(a)/(RT))(1)/(V) + ((b)/(bar(V)))^(2) +.....` or `p bar(V) = RT [1 + (b-(a)/(RT)) (1)/(bar(V)) + ((b)/(bar(V)))^(2) +....]` Comparing with the given equation, we have `B = (b-(a)/(RT))`. |
|
| 42. |
One way of naming amines is to name in alphabetical order the alkyl groups attached to the nitrogen atom, using the prefixes di- and tri- if the groups are the same. An example is isopropylamine whose formula is shown above. What are names for (a), (b), (c), and (d)? |
| Answer» SOLUTION :(a) Butylisopropylamine, (B) tripropylamine, (E) N-methylbenzene: (d) N,N-dimethylbenzene. | |
| 43. |
One volume of a gaseous organic compound of C, H and N on combustion produced 2 volumes of CO_(2), 3.5 volumes of H_(2)O vapour and 0.5 volume of N_(2) under identical conditions of temperature and pressure. The molecular formula of the compound is |
|
Answer» `C_(2)H_(7)N` |
|
| 44. |
One volume of hydrogen combines with sulphur to produce one volume of a gas (A). If the vapour density of (A) be 17. what is its molecular formula? The volumes have been measured under same temperature and pressure. |
|
Answer» Solution :Hydrogen `rarr` A 1 vol1 VOL N molecules n molecules 1 molecule 1 molecule 2 ATOMS 1 molecule. So, the formula of `A=H_(2)S_(x+)` where x is the number of sulphur atoms. Mol. Mass of `A=2xx1+x xx 32=2V D=2xx17=34`. or `2+32x=34` or `x=1` Hence, the molecule formual of A is `H_(2)S`. |
|
| 45. |
One volume of a gaseous compound of carbon, hydrogen and nitrogen on combustion produced 2 volumes of CO_(2), 3.5 volumes of H_(2)O and 0.5 volume of nitrogen under identical conditions of temperature and pressure. Find the empirical and molecular formulae of the compound. |
|
Answer» Solution :Avogadro.s principle states that equal volumes of gases contain the same number of molecules or moles `{:("Compound",rarr,CO_(2),+,H_(2)O,+,N_(2)),(1"vol",,2"vol",,3.5"vol",,0.5"vol"),(1 "mole",,2"mole",,3.5"mole",,0.5"mole"):}` Moles of C in `CO_(2)=1 xx` moles of `CO_(2)` `=1 xx 2=2` Moles of H in `H_(2)O=2 xx` moles of `H_(2)O = 2 xx 3.5= 7` Moles of N in `N_(2)=2 xx` moles of `N_(2)= 2XX 0.5=1` Hence the empirical FORMULA is `C_(2)H_(7)N` Now we see that 1 mole of the compound contains 2 moles of `CO_(2)` and 1 mole of `CO_(2)` contains 1 mole of carbon atoms, so 1 mole of the compound should contain 2 moles of carbon atoms which is shown by the empirical formula. Hence, the molecular formula is the same as the empirical formula. |
|
| 46. |
One unpaired electron in atom contributes a magnetic moment of 1.1 BM. Calculate the magnetic moment of Cr. [Atomic number = 24] |
| Answer» Solution :As Cr has six unpaired ELECTRONS, its MAGNETIC MOMENT is `6xx1.1 = 6.6` BM. | |
| 47. |
One unpaired electron in an atom contributes a magnetic moment of 1.1 BM. The magnetic moment for chromium (Z=24)is |
|
Answer» 4.4 BM MAGNETIC MOMENT `=1.1xx66=6.6 B.M.` |
|
| 48. |
One unpaired electron in an atom contributes a magnetic moment of 1.1 B.M. Calculate the magnetic moment of Cr |
| Answer» Solution :CHROMIUM `(3d^(5) 4s^(1))` has six unpaired ELECTRONS. THEREFORE, its MAGNETIC moment is `1.1 B. M xx 6 = 6.6 B. M ` | |
| 49. |
One unit cell of NaCl contains |
|
Answer» `1Na^(+)` and `1CL^(-)` |
|