Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Order of radioactive disintegration reaction is…… |
|
Answer» zero |
|
| 2. |
Order of paramagnetic character among following elements is/are: |
|
Answer» `MngtFegtCr` |
|
| 3. |
Order of nucleophilicity of oxygen containing nucleophiles |
|
Answer» `RO^(-) GT OH^(-) gt ROH gt H_2O` |
|
| 4. |
Order of hydrolysis for the following ltBrgt (I) RCOCl (II) RCOOR (III) RCONH_(2) ltBrgt (IV) (RCO)_(2)O |
|
Answer» IgtIVgtIIgtIII the order of REACTIVITY of acid derivatives DECREASES in the following order. `RCOCl gt (RCO)_(2)O gt RCOOR gt RCONH_(2)` |
|
| 5. |
Order of nucleophilicity of halide ions in aprotic solvent |
|
Answer» `I^(-) gt BR^(-)gt Cl^(-) gt F^-` |
|
| 6. |
Order of esterification of alcohols is |
|
Answer» `3^(@) gt 2^(@) gt 1^(@)` |
|
| 7. |
Order of base strength of the compounds :- |
|
Answer» IV GT II gt I gt III 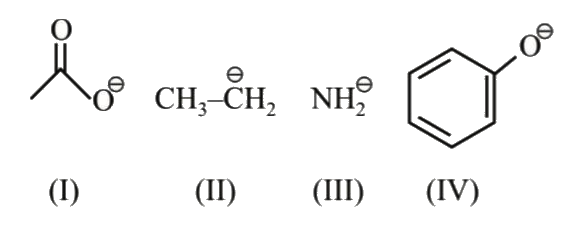 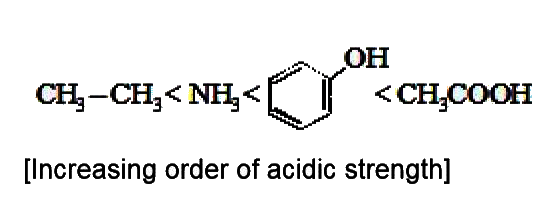 . .
|
|
| 8. |
Order of a reaction is the power of its concentration which appears in the ________ equation. |
| Answer» SOLUTION :RATE LAW | |
| 9. |
Order of acidity of alcohol is |
|
Answer» `1^@` GT `2^@` gt `3^@` |
|
| 10. |
Order of a reaction is decided by |
|
Answer» PRESSURE |
|
| 11. |
Order of a reaction is decided by……. |
|
Answer» PRESSURE |
|
| 12. |
Order of a reaction can not be obtained |
|
Answer» EXPERIMENTALLY |
|
| 13. |
Order of a reaction can be zero. |
| Answer» | |
| 14. |
Order of a reaction can be |
|
Answer» fractional |
|
| 15. |
Order H_(3)PO_(4) gt H_(2)SO_(4) gt HNO_(3) gt HCl is correct for :- |
|
Answer» ACIDIC nature |
|
| 17. |
Order of a chemical reaction. |
|
Answer» Solution :The order of a chemical reaction is defined as the number of molecules (or atoms) WHOSE CONCENTRATIONS influence the rate of the chemical reaction. OR The order of a chemical reaction is defined as the sum of the POWERS (or exponents) to which the concentration terms of the reactants are raised in the rate LAW expression for the GIVEN reaction. |
|
| 18. |
Order and Molecularity of the reaction. |
| Answer» Solution :`{:("Order","Molecularity"),("1. Ot is the sum of the exponents to which the concentration TERMS in rate law EXPRESSION are raised.","1. It is the number of molecules (or ATOMS or ions) of the reactants taking parts in the elementary REACTION."),("2. It is experimentally determined and indicates the dependance of concentration of particular reactants.","2. It is theoretical property and indicates the number of molecules of reactant in each step of the reaction."),("3. It may have values which are integer, freactional or zero.","3. It is always an integer."),("4. Its value depends upon experimental conditions.","4. Its value does depends upon experimental conditions."),("5. It is the property of elementary and complex reactions.","5. It is the property of elementary reactions only."):}` | |
| 19. |
Orbital interaction between the sigma bonds of a substituent group and neighbouringp-orbital is known as |
|
Answer» HYPERCONJUGATION |
|
| 20. |
Orbital angular momentum of p-subshell is:- |
| Answer» Answer :C | |
| 22. |
Orange colouredsodiumcobaltinitrite Na_(3)[Co(NO_(3))_(6)] is used for thedetectionof K^(Theta) whichgives …… pptdue to theformation of pot sod cobaltinitrite K_(2)Na[Co(NO_(2))_(6)] |
|
Answer» White |
|
| 23. |
"Orange solid"overset(Delta)rarrunderset(gas)"diamagnetic"+underset("residue")"green" underset("solid"(X_(2)))"Orange"overset(Delta)rarrunderset("magnetic(gas)")"para"+underset(("residue"))"green"+underset("(solid)")"yellow" X_(1) and X_(2) are respectively: |
|
Answer» `Na_(2)Cr_(2)O_(7),K_(2)Cr_(2)O_(7)` |
|
| 24. |
Orangecolourof Cr_(2)O_(7)^(2-)ioncharges toyellowin alkaliand yellowsolutionturnsout orangeon adding H^(+) ions.Explainwhy ? |
|
Answer» Solution :(i)Whenorangesolutioncontaining`Cr_(2) O_(7)^(2-)` ION is treatedwith analkali a YELLOWSOLUTION of `Cr_(2)O_(4)^(2-)` is obtained . (ii)Similarly when `H^(+) ` ionsare addedto yellowsolutionan orangesolutionof `Cr_(2)O_(7)^(2-) ` isobtaineddue tointerrconversion. |
|
| 26. |
Oral contraceptive drugs contain |
|
Answer» Mestanol |
|
| 27. |
Option with correctly matched reactant and product is/are : |
|
Answer» `"C"Cl_(3)-underset(O)underset(||)C-H underset(Delta)OVERSET(Ooverset(ϴ)H)rarr "C"Cl_(3)-COO^(ϴ)+"C"Cl_(3)-CH_(2)-OH` |
|
| 28. |
Optically pure (S)-(+)-2bromo octane [alpha]_(D)^(25^(@)C)=+36^(@), react with aqueous NaOH in acetone to give optically pure (R)-(-)-2-octanol [alpha]_(D)^(25^(@)C) =-10.3^(@). With partially racemized bromo compound whose [alpha]_(D)^(25^(@)C)=30^(@), the [alpha]_(D)^(25^(@) of the alcohol product is - 6.0^(@). Calculate the percentage of frontside attack : |
| Answer» SOLUTION :N//A | |
| 29. |
Optically pure (S)-(+)-2bromo octane [alpha]_(D)^(25^(@)C)=+36^(@), react with aqueous NaOH in acetone to give optically pure (R)-(-)-2-octanol [alpha]_(D)^(25^(@)C) =-10.3^(@). With partially racemized bromo compound whose [alpha]_(D)^(25^(@)C)=30^(@), the [alpha]_(D)^(25^(@) of the alcohol product is - 6.0^(@). Calculate the per cent optical purity of partially racemized bromo compound : |
| Answer» SOLUTION :N//A | |
| 30. |
Optically active tert alkyl halide given below on hydrolysis gives recemic mixture. Soparation of (d)and (l) form from the recemic mixture is known as resultuion. C_(6)H_(5)-overset(C_(6)H_(13)) overset(|)underset(CH_(3))underset(|)(Br)- overset(HOH)toC_(6)H_(5)C-overset(C_(6)H_(13)) overset(|)underset((X))underset(CH_(3))underset(|)C-OH+HO-overset(C_(6)H_(13)) overset(|) underset((Y))underset(CH_(3)) underset(|)C-C_(6)H_(5) Which of these statements are correct for the given reaction ? 1 Reaction intermediate is carboncation (2) Product formation takes place by formation of TS. (4) There is inversion as well as retention of configuration (4) Reaction is known as hydrolysis, i.e, solvolysis. Of these statements |
|
Answer» 1, 3 and 4 are correct |
|
| 31. |
Optically active compound is , |
|
Answer» 2-chloropropane |
|
| 32. |
Optically pure (S)-(+)-2bromo octane [alpha]_(D)^(25^(@)C)=+36^(@), react with aqueous NaOH in acetone to give optically pure (R)-(-)-2-octanol [alpha]_(D)^(25^(@)C) =-10.3^(@). With partially racemized bromo compound whose [alpha]_(D)^(25^(@)C)=30^(@), the [alpha]_(D)^(25^(@) of the alcohol product is - 6.0^(@). Calculate the precentage of racemization : |
| Answer» SOLUTION :N//A | |
| 33. |
Optically pure 2(S)-butanol is subjected to the following reaction 2 (S) - Butanol underset(Na)(rarr)X overset(CH_1I)(rarr) , which of the following statements is correct for stereo cemical outcome of the reaction |
|
Answer» The reaction proceeds with INVERSION |
|
| 34. |
Optically active tert alkyl halide given below on hydrolysis gives recemic mixture. Soparation of (d)and (l) form from the recemic mixture is known as resultuion. C_(6)H_(5)-overset(C_(6)H_(13)) overset(|)underset(CH_(3))underset(|)(Br)- overset(HOH)toC_(6)H_(5)C-overset(C_(6)H_(13)) overset(|)underset((X))underset(CH_(3))underset(|)C-OH+HO-overset(C_(6)H_(13)) overset(|) underset((Y))underset(CH_(3)) underset(|)C-C_(6)H_(5) Which one of the following statements is NOT true for (A) and (B) the above reaction ? |
|
Answer» (X) and (Y) are enatiomers |
|
| 35. |
Optically active 'P' has the molecular formula C_(6) H_(12) and catalytic hydrogenation converts into achiral C_(6) H_(14) . The structure of 'P' is - |
|
Answer»

|
|
| 36. |
Optically active (S)-alpha-methoxyacetaldehyde on reaction with MeMgX gave a mixture of alcohols. The major diastereomer 'P' on treatment with MeI//K_(2)CO_(3) gave an optically inactive compound. P is - |
|
Answer» `i` The STATEMENT OPTICALLY active `(s) - ALPHA -` methoxy ACETALDEHYDE is incorrect. |
|
| 37. |
Optically active polyhydroxy aldehydes or ketones are known as |
|
Answer» AMINO acids |
|
| 38. |
Optically active isomers but not mirror images are called |
|
Answer» ENANTIOMERS |
|
| 39. |
Optically active isomers are also called: |
|
Answer» Epimers |
|
| 40. |
Optically active 2-iodobutane on treatment with sodium iodide in acetone gives a product which does not show optical activity. Explain. |
|
Answer» Solution :Optically ACTIVE 2-iodobutane DISSOCIATED to form iodide ion and the carbocation. The iodide ion then attacks the flat carbocation from either side resulting in a mixture of two ENANTIOMERIC products in equal amounts, which CONSTITUTES a RACEMIC mixture which does not show optical activity. |
|
| 41. |
Optically active 2-iodobutane on treatment wit NaI in acetone gives a product which does not show optical activity. Expalain. |
|
Answer» Solution :OPTICALLY active 2-iodobutane on treatment with NaI in acetone undergoes racemization and HENCE the product does not show optical activity as explained below: When optically active 21-iodobutane (I) is treated with `I^(-)` ion, it undergoes WALDEN inversion (`S_(N)2` raction) to give 2-iodobutane (II) which is the enantiomer of (I). Now 2-iodobutane (II) undergoes Walden inversion to give enantiomer (I). As a RESULT of repetition of these two Walden inversion, ultimately a 50:50 mixture of the two enantiomers is obtained 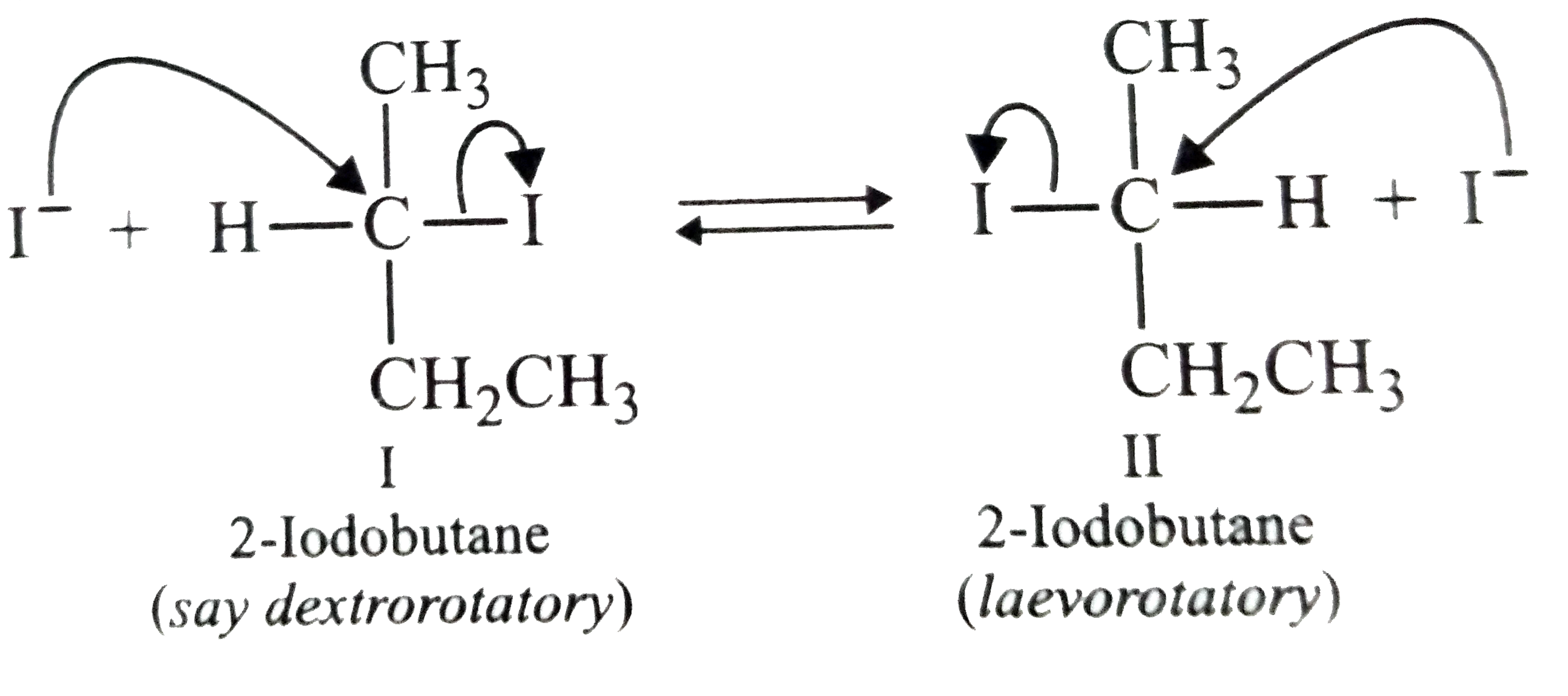
|
|
| 42. |
Optically active 2-iodobutane on treatment with sodium iodide in acetone gives a product which does not show optical activity. Explain |
| Answer» SOLUTION :OPTICALLY ACTIVE 2-iodobutane dissociated to FORM iodide ion and the carbocation. The iodide ion then attacks the flat carbocation from either side resulting in a mixture of two enantiomeric products in equal amounts, which constitutes a racemic mixture which does not show optical activity. | |
| 43. |
Optically active 2-iodobutane on treatment with Nal in acetone gives a product which does not show optical activity. Explain briefly. |
|
Answer» Solution :There occurs breaking and reforming of C-1 bond. This resutls into two enantiomers which FORM racemic mixture which does not show optical activity due to EXTERNAL compensation. `H-UNDERSET(C_(2)H_(5))underset(|)overset(CH_(3))overset(|)(C)-Ioverset((i)-I^(-))underset((ii)+I^(-))rarrH-underset(C_(2)H_(5))underset(|)overset(CH_(3))overset(|)(C)-I+I-underset(C_(2)H_(5))underset(|)overset(CH_(3))overset(|)(C)-H` |
|
| 44. |
Optically active 2-bromobutane on treatment with NaI in acetone gives a product which does not show optical activity. Explain briefly. |
Answer» SOLUTION :The product OBTAINED after the reaction contain equal amount of both d and l-isomers of 2-iodobutane and this RACEMIC mixture does not show OPTICAL activity due to EXTERNAL compensation. 
|
|
| 45. |
Optical isomers which are non-superimpo sable mirror images of each other are called |
|
Answer» ENANTIOMERS |
|
| 46. |
Optical isomers essentially have |
|
Answer» SUPER IMPOSABLE MIRROR image |
|
| 47. |
Optical isomers have A) property of chirality B) almost identical chemical properties C) almost identical physical properties D) similar rotation of plane polarised light |
|
Answer» A,B,C are CORRECT |
|
| 48. |
Optical isomerism not show by :- |
|
Answer» `Cl-CH=C=CH-CH_(3)` |
|
| 49. |
Optical isomers differ in |
|
Answer» CHEMICAL properties |
|