Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Orlon is a polymer of : |
|
Answer» VINYL CHLORIDE |
|
| 2. |
Orlon has monometric unit |
|
Answer» acrolein `underset("Acrylonitrile or vinyl cyanide ")(nH_(2)C=overset(CN)overset(|)(C)H) underset("Peroxide catalyst")overset("Polymerisation")to underset("Polyacrylonitrile(PAN)(orlon)")([--CH_(2)-overset(CN)(C)H--]_(n)` |
|
| 3. |
Orlon has a unit of: |
|
Answer» VINYL cyanide |
|
| 7. |
Organometallic compound used in the purifi-cation of its metals is |
|
Answer» `NI(CO)_4` |
|
| 8. |
Organometallic compound used in purification of its metals is |
|
Answer» `NI(CO)_4` |
|
| 9. |
Organometallic compound used in the purification of its metals is: |
|
Answer» `NI(CO)_4` |
|
| 10. |
Organic halogen compound used in industry as solvents are chlorides rather than bromides and iodides. Explain. |
| Answer» Solution :Organic ALKYL chlorides are WIDELY USED as solvents in INDUSTRY because chlorides are not only more stable but are also more volatile than bromide and IODIDES. | |
| 12. |
Organic compounds used in industry as solvents are chlorides rather than bromides and iodides. Why? |
| Answer» SOLUTION :ORGANIC ethyl chlorides are more volatile than bromides and IODIDES. | |
| 13. |
Organic compounds sometimes adjust their electronic as well as steric structure to attain stability. Among the following, the compound having highest dipole moment is : |
|
Answer»
|
|
| 14. |
Organic compounds required in the diet in small amounts to perform specific biological functions for normal maintenance of optimum growth and health of the organism are called |
|
Answer» NUCLEIC ACIDS. |
|
| 15. |
Organic compounds containing amine as functional group are present in a vivid variety of compounds, namely amino acids, hormones, neurotransmitters, DNA, alkaloids, dyes, stc. Drugs including nicotine group in one form or another. Amines are basic because of the presence of lone pair of electrons on nitrogen. Addition of nitrogen into an organic framework leads to the formation of two families of molecules, namely amines and amides. As chemistry students, we must appreciate the versatility of nitrogen. Name the linkage formed when carboxyI end of one amino acid condenses with amino end of other amino acid. |
| Answer» SOLUTION :It is CALLED PEPTIDE LINKAGE. | |
| 16. |
Organic compounds containing amine as functional group are present in a vivid variety of compounds, namely amino acids, hormones, neurotransmitters, DNA, alkaloids, dyes, stc. Drugs including nicotine group in one form or another. Amines are basic because of the presence of lone pair of electrons on nitrogen. Addition of nitrogen into an organic framework leads to the formation of two families of molecules, namely amines and amides. As chemistry students, we must appreciate the versatility of nitrogen. What are essential amino acids ? |
| Answer» Solution :AMINO ACIDS which cannot be synthesised by our body and MUST be PROVIDED through diet are CALLED essential amino acids. | |
| 17. |
Organic compounds containing amine as functional group are present in a vivid variety of compounds, namely amino acids, hormones, neurotransmitters, DNA, alkaloids, dyes, stc. Drugs including nicotine group in one form or another. Amines are basic because of the presence of lone pair of electrons on nitrogen. Addition of nitrogen into an organic framework leads to the formation of two families of molecules, namely amines and amides. As chemistry students, we must appreciate the versatility of nitrogen. Why are amino acids amphoteric ? |
| Answer» Solution :These COMPOUNDS CONTAIN a basic (AMINO group) and an ACIDIC group (carboxylic group). Therefore, these compounds are amphoteric. | |
| 18. |
Organic compounds containing amine as functional group are present in a vivid variety of compounds, namely amino acids, hormones, neurotransmitters, DNA, alkaloids, dyes, stc. Drugs including nicotine group in one form or another. Amines are basic because of the presence of lone pair of electrons on nitrogen. Addition of nitrogen into an organic framework leads to the formation of two families of molecules, namely amines and amides. As chemistry students, we must appreciate the versatility of nitrogen. Give one point of difference between acidic and basic amino acid. |
| Answer» Solution :If the NUMBER of carboxyl groups is more than the number of AMINO groups, it is an ACIDIC amino ACID. If the number of amino groups is more than of carboxyl groups, it is basic amino acid. | |
| 19. |
Organic compounds containing amine as functional group are present in a vivid variety of compounds, namely amino acids, hormones, neurotransmitters, DNA, alkaloids, dyes, stc. Drugs including nicotine group in one form or another. Amines are basic because of the presence of lone pair of electrons on nitrogen. Addition of nitrogen into an organic framework leads to the formation of two families of molecules, namely amines and amides. As chemistry students, we must appreciate the versatility of nitrogen. What are amino acids ? |
| Answer» Solution :COMPOUNDS containing an AMINO group as well as a CARBOXYLIC group are called amino ACIDS. | |
| 20. |
Organic compounds A, B and C are aliphatic saturated hydroxyl compounds when they react with Lucas reagent (anhyd.ZnCl_(2)+ conc.HCl ), the following observations are made(i) Compound A gave turbidity immediately.(ii) Compound B gave turbidity after five minutes(iii) Compound C gave turbidity only on heating. Identify the type of compound A, B and C.(b) Complete the equation:C_(6)H_(5)COOH + HOC_(6)H_(5) overset(H+)(to) (c) Name the main organic product formed when anisole is reacted with Hl. |
|
Answer» Solution :A = Tertiary alcoholB = SECONDARY alcoholC = Primary alcohol (b) `C_(6)H_(5)COOH + HOC_(6)H_(5) overset(H+)(to) underset("Phenyl Benzoate")(C_(6)H_(5)-COO- C_(6)H_(5)COO) + H_(2)O` (c) Phenol |
|
| 21. |
Organic compound with molecular formula C_(3)H_(6)O has two isomers (A) and (B). (A) on heating with NaOH in I_(2) forms a yellow precipitate while (B) does not. Identify the isomers A and B and explain the reactions. |
|
Answer» Solution :TWO POSSIBLE isomers of `C_(3)H_(6)O` are `underset("acetone")underset((A))(CH_(3)COCH_(3))andunderset("PROPANAL")underset((B))(CH_(3)CH_(2)CHO)` `underset("Acetone")underset((A))(CH_(3)COCH_(3))underset("Iodo form reaction")overset(I_(2)//NaOHDelta)tounderset("acetate")underset("Sodium")(CH_(3)COONa)+underset("Yellow ppt")underset("Iodoform")(CHI_(3))` `underset("Proponal")underset((B))(CH_(3)CH_(2)CHO)underset(Delta)overset(I_(2)//NaOH)to"No ppt of "CHI_(3)` Acetone answer Iodo form test whereas proponal does not. |
|
| 22. |
Organic catalysts differ from inorganic catalysts in |
|
Answer» acting at a very high temperature |
|
| 23. |
Orford process is used in extraction of |
|
Answer» Fe |
|
| 24. |
Ore pitch blende is main source of |
|
Answer» Ra |
|
| 25. |
"Ore" overset("Calcination")to "Residue" overset("dil.HNO"_(3))tounderset("Metal"(M)+Zn^(2+)(aq.))underset(darr"Zn dust")("aq. solution")above metallurgy is possible when ore is :- |
|
Answer» `ZnCO_(3)` |
|
| 26. |
Orford process is used in extraction of : |
|
Answer» Fe |
|
| 27. |
Ore of a metal M(X) to overset("roasting")to "Roasted ore" underset("absence of"O_(2))overset(Delta with (X)) to "Metal M" Ore of metal M(Y) to overset("calcination")to "Roasted ore" underset("absence of"O_(2))overset(Delta with (X)) to "Metal M" |
|
Answer» X=chalcocite Y=malachite |
|
| 28. |
Ores. |
| Answer» Solution :The minerals CONTAINING a high percentage of metals from which metals can be profitably EXTRACTED are called ORES. | |
| 29. |
Ore like magnetite or tungstates in tin ores are concentrated by |
|
Answer» FROTHFLOATATION |
|
| 30. |
Ore dressing is the process, in which __________. |
|
Answer» ore SIZE is REDUCED to required size |
|
| 32. |
Oredressingof ironore is done by |
|
Answer» FROTHFLOATATIONPROCESS |
|
| 33. |
Ordinary strong solution of HCl, HNO_3 and H_2 SO_4 contains roughly. |
|
Answer» `1//5, 2//3` and `3//3` FRACTIONS of pure acid and water respectively Therefore, `HCl` "contains" `~~ 20//100 = 1//5` `HNO_3 "contains" ~~ 68 //100 ~~ 2//3` `H_2 SO_4 "contains" ~~ 98//100 ~~ 2//3` Thus, ordinary strong solution of `HCl, HNO_3` and `H_2 SO_4` contains roughly `1//5, 2//3` and `3//3` fractions of pure acid and water respectively. |
|
| 34. |
Ordinary spirit is …… |
|
Answer» Methanol |
|
| 35. |
Ordinary oxygen contain how many isotopes of oxygen ? |
|
Answer» |
|
| 36. |
Ordinary light oscillates in_____plane passing through the line of propagation. |
|
Answer» infinite |
|
| 37. |
Ordinary soaps are defined as |
|
Answer» Al SALTS of HIGHER FATTY acids |
|
| 38. |
Ordinary oxygen contains: |
|
Answer» Only `O^16` isotope |
|
| 39. |
Ordinary hydrogen has preponderance of: |
|
Answer» Hydrogen |
|
| 40. |
Ordinary hydrogen at room temperature is a mixture of |
|
Answer» 25% or tho+75 % para |
|
| 42. |
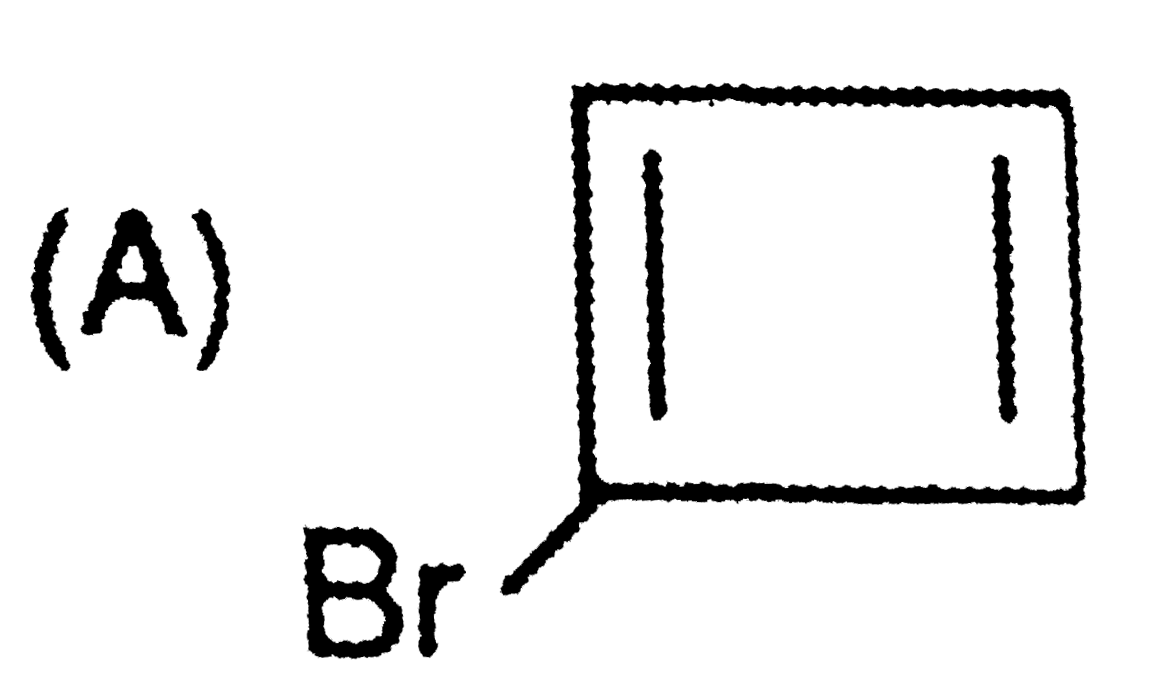
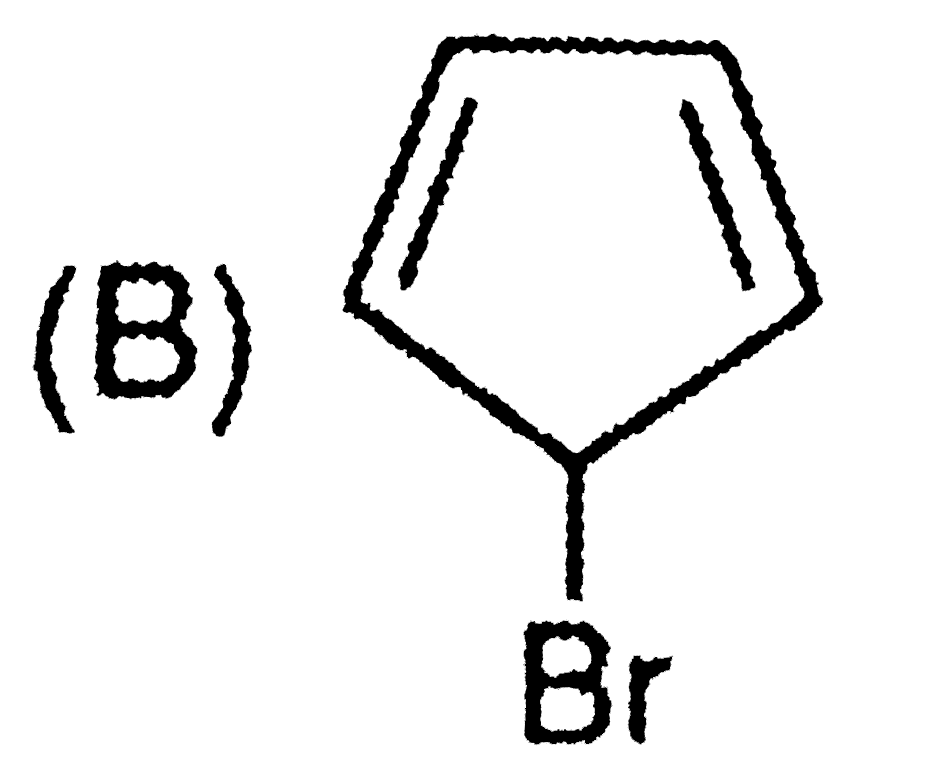
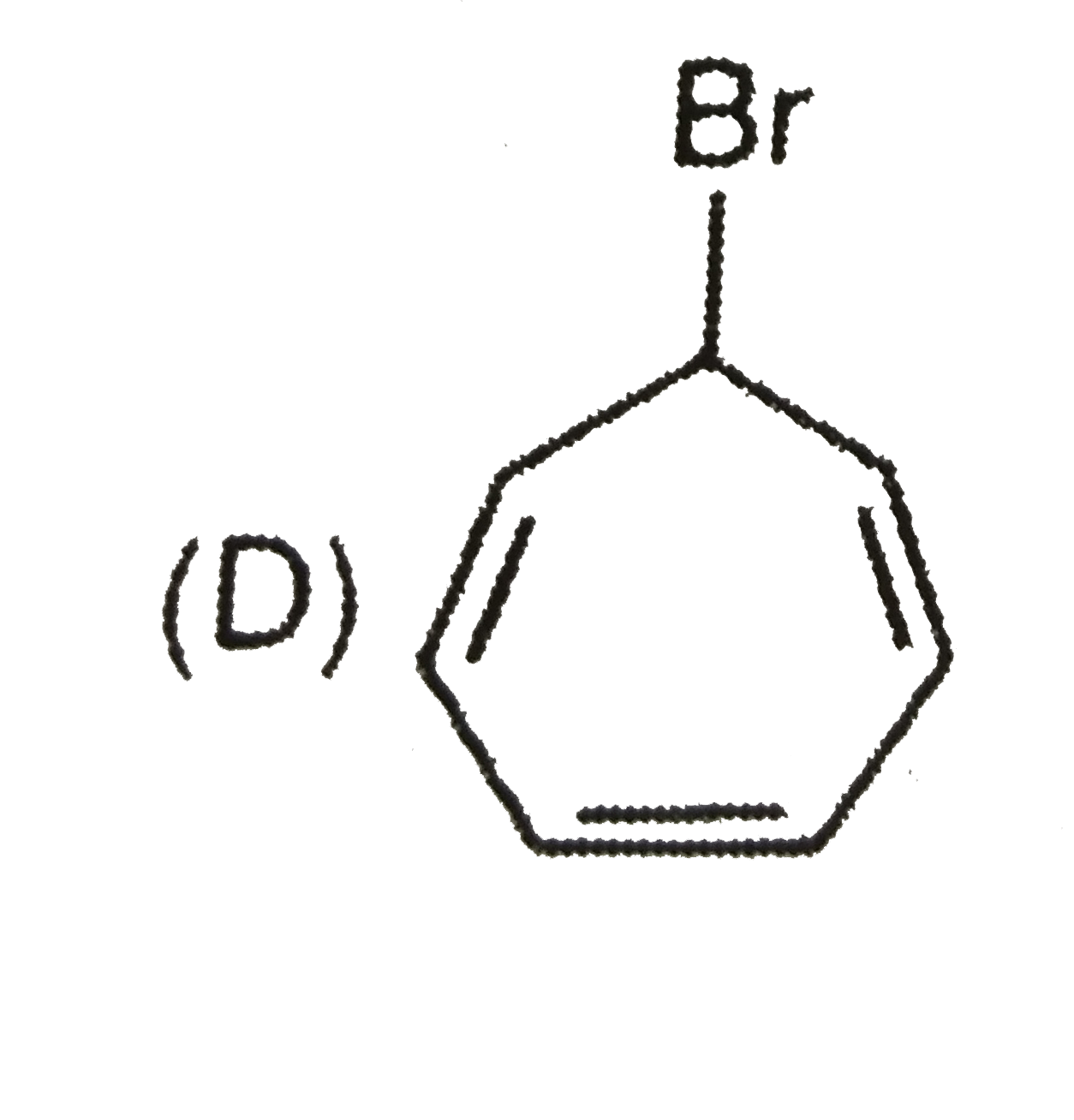
Order of reactivity towards nucleophili substitution reaction of the compounds : |
|
Answer» `(i) GT (II) gt (III) gt (IV)` |
|
| 43. |
Order of relativity of alkanes in substitution reaction is _____ |
|
Answer» |
|
| 44. |
Order of stability of halogen oxides :- |
|
Answer» `I LT Cl lt BR` |
|
| 45. |
order of reactivity of C_2H_6, C_2H_4 and C_2H_2 is |
|
Answer» `C_2H_6 GT C_2H_4 gt C_2H_2` |
|
| 46. |
Order of reactivity of hydrogen halides towards ethers is |
|
Answer» `HCl GT HBR gt HI` |
|
| 47. |
Orderof rateof electrophilicadditionreaction with HBrwill be : |
|
Answer» `IV GT I gt III gt II` |
|
| 48. |
Order of reactivity is |
|
Answer» `R-OVERSET(O)overset(||)(C)-X gt RCONH_(2) gt RCOOCOR gt RCOOR` |
|
| 49. |
Order of reaction is decided by |
|
Answer» molecularity |
|
| 50. |
orderof reactionis |
|
Answer» neverzero |
|