Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Osmotic pressure of a solution containing 2 g dissolved protain per 300cm^(3)id solution is 20 mm of Hg at 27^(@)C. Calculate the molecular mass of protein. (R=0.082 L atm K^(-1)mol^(-1)) |
|
Answer» `pi=20" mm of HG"=20/760=0.02632" atm, "R=0.0821" L atm K"^(-1)mol^(-1)` `T=27^(@)C=27+273=300K, M_(B)=?` `M_(B)=(W_(B)xxRxxT)/(pixxV)=((2.0g)xx(0.0821"L atm K"^(-1)mol^(-1))xx(300K))/((0.02632 atm)xx(0.3 L))=6238.6"g mol"^(-1)`. |
|
| 2. |
Osmotic pressure of a 0.0103 molar solution of an electrolyte was found to be 0.70 atm at 27^(@)C. Calculate Van't Hoff factor. |
|
Answer» `pi=0.70" atm",C=0.0103" mol L"^(-1), R=0.0821" L atm K"^(-1)mol^(-1)` `T=27+273=300 K` `i=pi/(CRT)=((0.70"atm"))/((0.0103" mol L"^(-1))XX(0.0821"L atm K"^(-1)mol^(-1))xx(300K))=2.76~~3`. |
|
| 3. |
Osmotic pressure of 4% (weight/vol) urea solution is 1.64 atm and that of 3.42% (weight/volume) cane sugar is 2.46 atm when equal volumes of the above two solutions are mixed, the osmotic pressure of the resulting solution is: |
|
Answer» 1.64 ATM |
|
| 4. |
Osmotic pressure of 0.1 M solution of NaCl and Na_(2)SO_(4) will be |
|
Answer» Same |
|
| 5. |
Osmotic pressure is the minimum pressure that has to be applied on the …………..to prevent that entry of………..from …………………….to……………… . |
| Answer» SOLUTION :solution SOLVENT, solvent, solution | |
| 6. |
Osmotic pressure observed when benzoic acid is dissolved in benbzene is less than that expected from throretical considerations. This is because |
|
Answer» BENZOIC acid is an ORGANIC solute The number of particles decreases and hence osmotic pressure decreases. |
|
| 7. |
Osmotic pressue of a solution is 0.0821 atm at a temperature of 400 K. Calculate concerntration of solution in moles /litre. |
|
Answer» `"Concentration of solution "( C)=pi/(RT)` `=((0.0821" ATM"))/((0.0821"litre-atm K"^(-1)mol^(-1))xx400K)` `=0.0025 mol^(-1) L^(-1)`. |
|
| 8. |
Osmotic pressure is measured by |
|
Answer» OSTWALD's method |
|
| 9. |
Osmotic pressure is a colligative property and it is proportional to the molarity of solution. What is osmotic pressure ? |
| Answer» Solution :It is the MINIMUM excess pressure to be applied on the solution side to prevent osmosis, when the solution and the SOLVENT are SEPARATED by a SEMIPERMEABLE membrane. | |
| 10. |
Osmostic pressure of a solution (density is 1 g/ml) containing 3 g of glucose (molecular weight =180) in 6o g of water at 15^@C is: |
|
Answer» 0.34 atm |
|
| 11. |
Osmium tetra oxide is a reagent used for |
|
Answer» hydroxylation of acetylenes |
|
| 12. |
Osmosis is the spontaneous flow through a semipermeable membrane of : |
|
Answer» A LESS concentrated SOLUTION into more in to more concentrated solution |
|
| 13. |
Osmotic pressure of 40% (wt./vol.) urea solution is 1.64 atm and that of 3.42% (wt./vol.) cane sugar is 2.46 atm. When equal volumes of the above two solutions are mixed, the osmotic pressure of the resultng solution is: |
|
Answer» 1.64 atm |
|
| 14. |
Osazone formation involves only 2-carbon atoms of glucose because of |
|
Answer» Oxidation |
|
| 15. |
Osazone formation involves only 2 carbon atoms of glucose because of |
|
Answer» chelation |
|
| 16. |
Osazone formation involves only 2 carbon atoms of glucose because of : |
|
Answer» CHELATION |
|
| 17. |
Osazone fomation is sued to characterize |
|
Answer» polymers |
|
| 18. |
Orthophosphoric acid on strong heating forms |
|
Answer» `P_(4)O_(10)` |
|
| 19. |
Orthophosphoric acid represents the molaysis condition due to |
|
Answer» HYDROGEN bonding |
|
| 20. |
Orthophosphoric acid on heating gives |
|
Answer» phosphine |
|
| 21. |
Orthophosphoric acid on heating at 600^∘C gives: |
|
Answer» Phosphine |
|
| 22. |
Orthophosphoric acid is ionised in _______ steps: |
|
Answer» 1 |
|
| 23. |
Orthoboric acid when heated to red hot gives : |
|
Answer» METABORIC acid |
|
| 24. |
Orthoboric acid (H_(3)BO_(3)) and metaboric acid (HBO_(2)) differ in respect off |
|
Answer» No. of `SIGMA`- bond |
|
| 25. |
Orthoboric acid in aqueous medium is |
|
Answer» MONOBASIC `H_(3)BO_(3)` is a weak monobasic acid it does not act as a `H^(+)` donor but behaves as a LEWIS acid. |
|
| 26. |
Orthoboric acid is heated to red hot gives : |
|
Answer» METABORIC acid |
|
| 27. |
Orthoboric acid contains discrete units of : |
|
Answer» `BO_(2)^(-)` |
|
| 28. |
Ortho silicates are also called as …………. |
|
Answer» Into silicates |
|
| 29. |
Ortho-nitrophenol is less soluble in water than p- and m-nitrophenols because |
|
Answer» o-nitrophenol SHOWS INTRAMOLECULAR H-bonding |
|
| 30. |
Ortho -nitrophenol is less soluble in water than p-and m-nitrophenols because |
|
Answer» o-nitrophenol is more volatile STEAM than those of m-and p-isomers 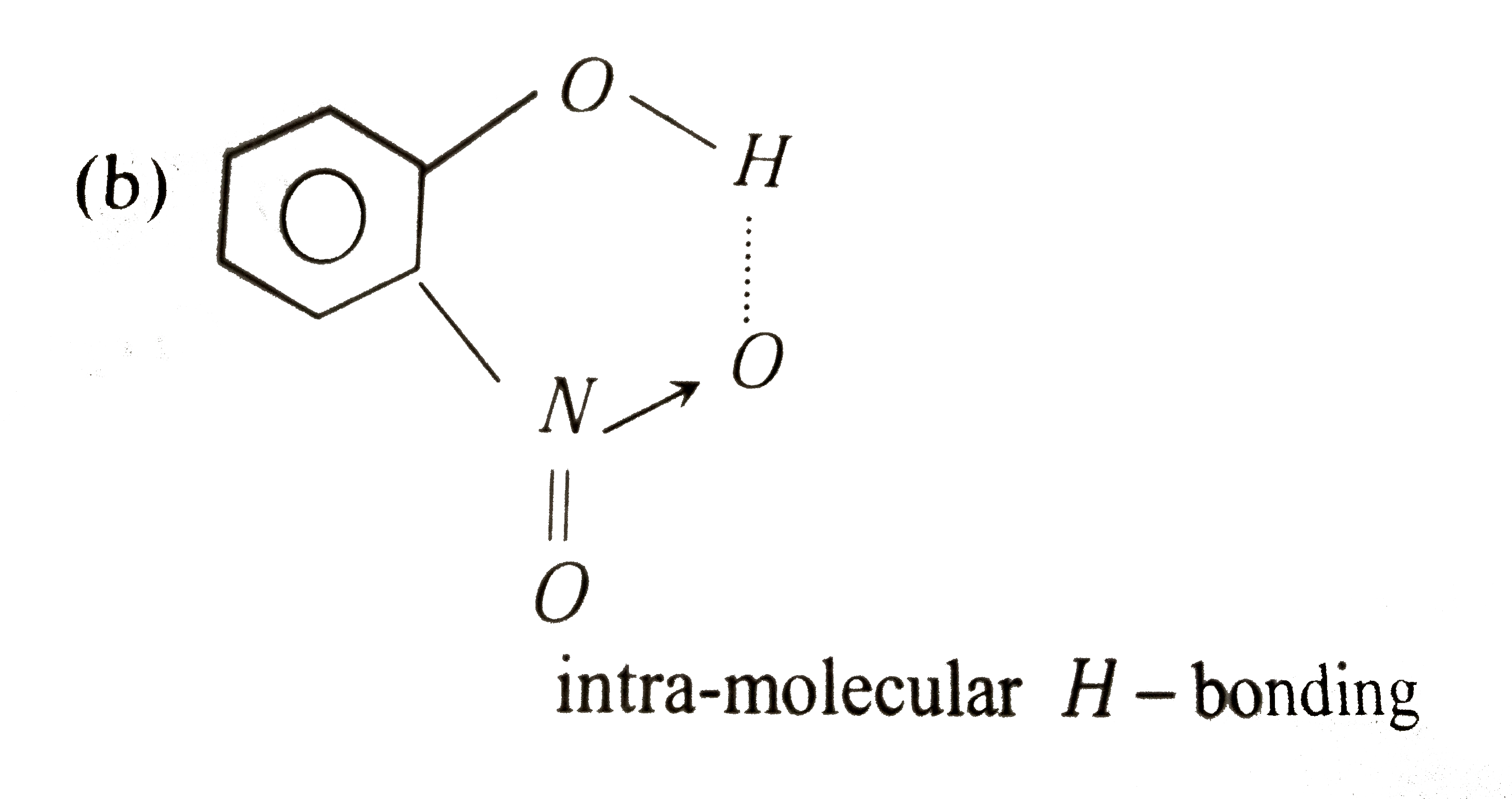
|
|
| 31. |
Ortho-Nitrophenol is less soluble in water than p- and m- Nitrophenols because : |
|
Answer» o-Nitrophenol is more volatile than those of m- and p-ISOMERS. 
|
|
| 32. |
Ortho- nitrophenol is less soluble in water than p-and m-nitrophenols because : |
|
Answer» o-nitrophenol showns intramolecular H bonding |
|
| 33. |
Ortho -Nitrophenol is less soluble in water than p-and m- Nitrophenol because |
|
Answer» <P>o-Nitrophenol is more volatile is steam that those of m- and p- isomers |
|
| 34. |
Ortho-nitro acetanilide is removed from p-nitro acetanilide by crystallisation from: |
|
Answer» HOT water |
|
| 35. |
Ortho isomer of nitrophenol is steam volatiledue to _________. |
|
Answer» INTER molecularhydrogen BONDING |
|
| 36. |
Ortho effect is a special type of effect that is shown by d-substituents, but it is not necessarily just a steric effect.This ortho-effect opertes with the benzoic acid.Irrespective of the polar types nearly all o-substitued benzoic acid are stronger than unsubstituted beznoic acid.Benzoic acid is a resonance hybrid and so the carboxyl group is coplanar with the ring.An o-substituent tends to prevent this coplanarity . Ortho effect also opertes in substituted anilines where as ortho substituent has base weakening effect. What is the order of acidity of following compounds : |
|
Answer» IgtIVgtIIIgtII (iv)Comes NEXT due to intramolecular H bonding with OH GROUP. (iii)Comes next due to ortho EFFECT. |
|
| 37. |
Ortho effect is a special type of effect that is shown by d-substituents, but it is not necessarily just a steric effect.This ortho-effect opertes with the benzoic acid.Irrespective of the polar types nearly all o-substitued benzoic acid are stronger than unsubstituted beznoic acid.Benzoic acid is a resonance hybrid and so the carboxyl group is coplanar with the ring.An o-substituent tends to prevent this coplanarity . Ortho effect also opertes in substituted anilines where as ortho substituent has base weakening effect. What is the order of basicily of following compounds ? |
|
Answer» IltIIltIIIltIV |
|
| 38. |
Ortho-dihydroxy toluenes |
|
Answer» Carvacrol 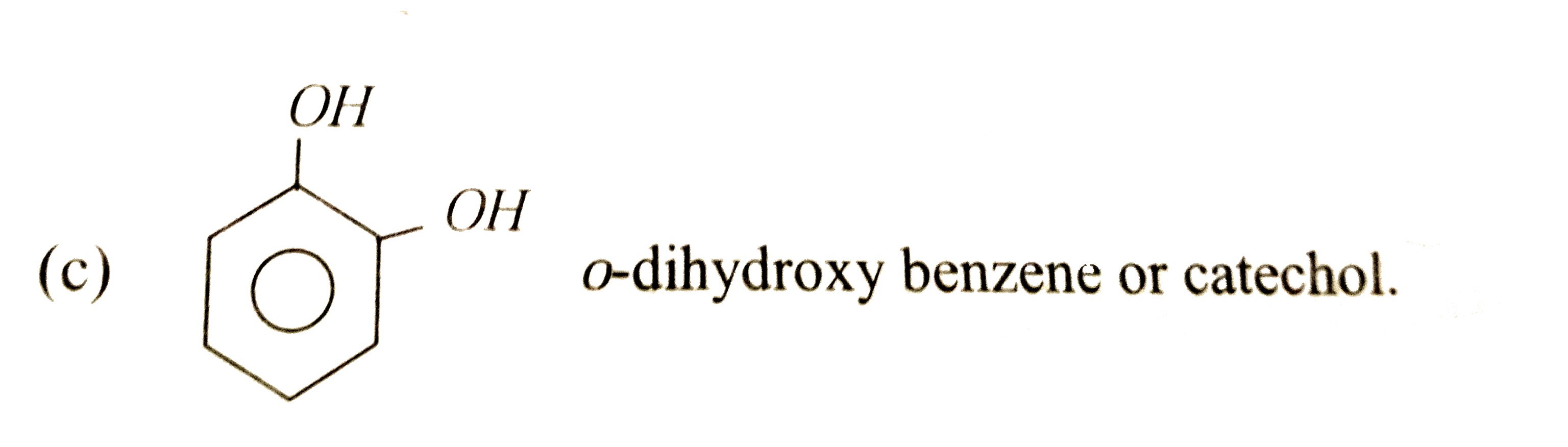 o-dihydroxy BENZENE or catechol. |
|
| 39. |
Ortho cresol and benzyl alcohol are |
|
Answer» Chain ISOMERS |
|
| 40. |
Ortho chlorotoluene on oxidation with KMnO_(4) gives : |
|
Answer» BENZOIC ACID 
|
|
| 41. |
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions |
Answer» SOLUTION : SIMILARLY DRAW the STRUCTURES of p-nitrophenoxide ION. |
|
| 42. |
Ortho and para - nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoide ions. |
Answer» Solution :The RESONANCE structures of o - and p- nitrophenoxide ions and phenoxide ion are SHOWN below :  Due to `-R` EFFECT of the `-NO_(2)` group, o- and p - nitrophenoxide ions are more STABLE than phenodie ion. Also there are additional resonance structures, III and IX, which add to the stability of the phenoxide ion. As a result, o - and p - nitrophenols are more ACIDIC than phenol. |
|
| 43. |
Ortho and para-nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions. |
Answer» Solution :The resonance STRUCTURE of o- and p nitrophenoxide IONS and phenoxide ion are given below: 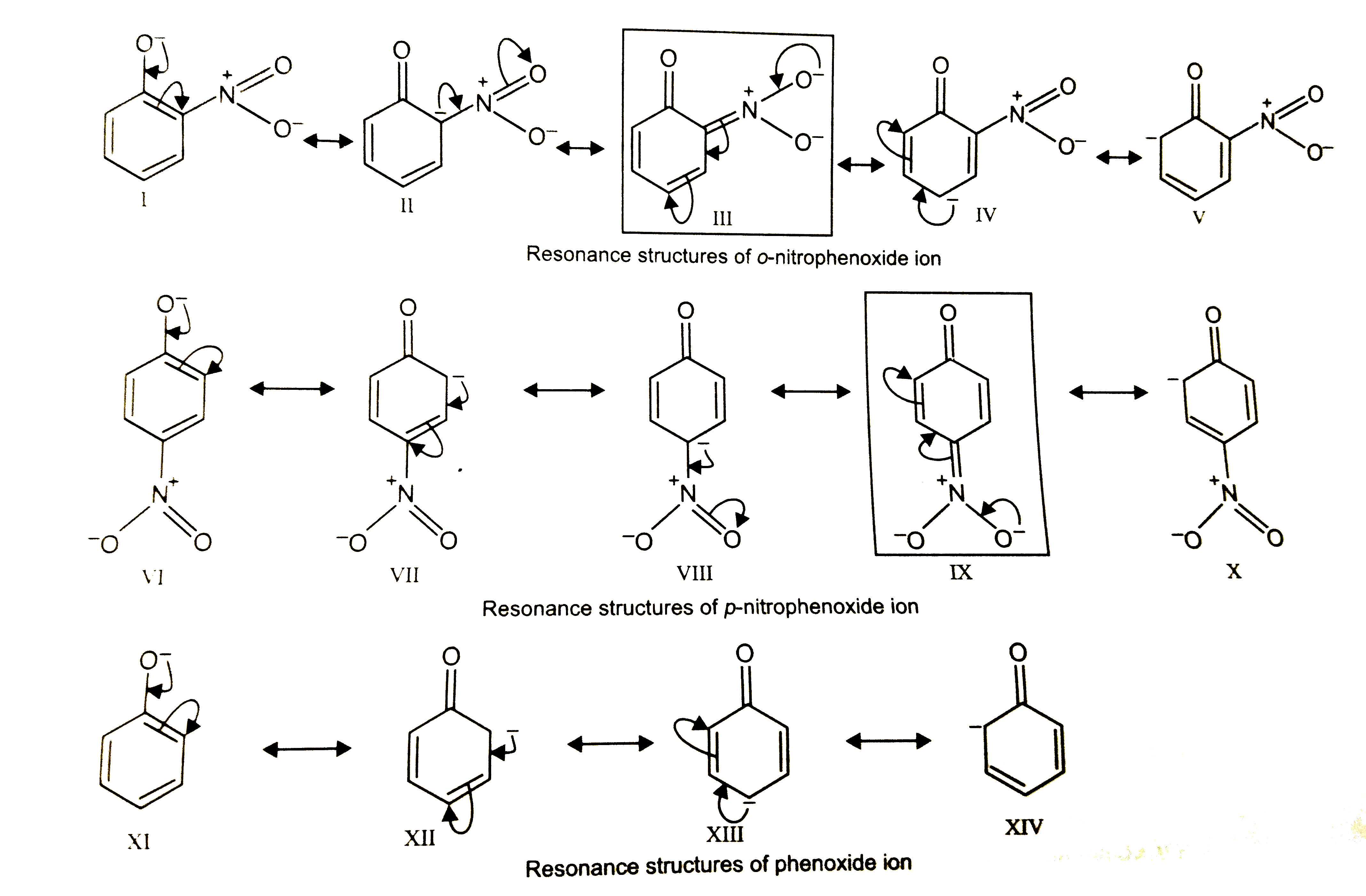 It is evident from the above structures that due to -R-effect of the `-NO_(2)` group, o- and p-nitrophenoxide ions are more stable (because of ADDITIONAL resonance structures, III and IX ENCLOSED in boxes) than phenoxide ion. as a result, o- and p-nitrophenols are more acidic than phenol. |
|
| 44. |
Ortho and para nitrophenols are more acidic than phenol. Draw the reasonationg structures of the corresponding phenoxide ions. |
|
Answer» Solution :`to ` The electron pair of O -H bond is pulled towards the oxygen ATOM because of -M effect of `-NO_2` that decreases the electron density on the oxygen atom. This FACILITATES the RELEASE of protron and stabilizes the phenoxide ion. `to` RESONATING forms of o-nitrophenoxide ion : 
|
|
| 45. |
Ortho and para hydrogen have |
|
Answer» identical CHEMICAL properitesbut differentphysical PROPERTIES |
|
| 46. |
Ortho and para hydrogen differ in the: |
|
Answer» NUMBER of protons |
|
| 47. |
Orlon is prepared frompolymerization of |
|
Answer» `CH_3-CH=CH_2` |
|
| 48. |
Ornamental gold containing copper is an example for what type of solution? |
| Answer» SOLUTION :SOLID is solid TYPE of solution (COLLOIDAL Solution) | |
| 49. |
Orlon or acrilan is |
|
Answer» `(-CH_(2)-UNDERSET(CN)underset("|")"CN"-)_(N)` |
|
| 50. |
Orlon is a polymer of: |
|
Answer» Styrene |
|