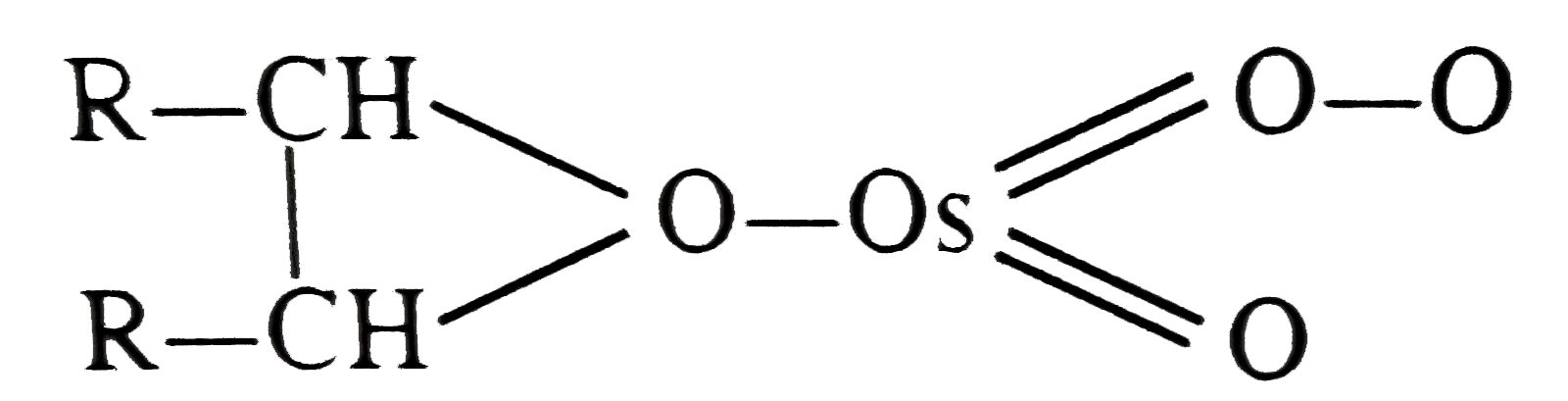Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Out of AlCl_(3) and NaCl which is more effective in causing coagulation of a negative sol and why ? |
| Answer» | |
| 2. |
Out of AgF, CaF_(2), BeF_(2), MgF_(2) |
|
Answer» AgF and `BeF_(2)` are soluble, and `CaF_(2) and MgF_(2)` are INSOLUBLE in water |
|
| 3. |
Out of alanine and phenyalanine, ..........is an essential amino acid. |
| Answer» SOLUTION :PHENYLALANIE | |
| 4. |
Out of acetophenone and benzophenone, which one will give iodoform test? Write the reaction involved. (The compound should have CH_3CO group to show the iodoform test). |
| Answer» SOLUTION :ACETOPHENONE `(C_6H_5 COCH_3)`CONTAINS the group `(CH_3CO)`attached to the carbon atom and hence give iodoform TEST while benzophenone does not CONTAIN this group and hence does not give iodoform test. | |
| 5. |
Out of acetophenone and benzophenone, which gives iodoform test? Write the reaction involved. |
|
Answer» Solution :Acetophenone `(C_(6)H_(5)COCH_(3))` contains the GROUP `CH_(3)CO` attached to carbon and hence gives iodoform test while benzophenone does not contain this group and hence does not give idoform test. `underset("Acetopheone")(C_(6)H_(5)COCH_(3))+3I_(2)+4NaOH underset("Iodoform")(CHI_(3)) + underset("Sod. benzoate")(C_(6)H_(5)COONa)+3NaI+3H_(2)O` `underset("Benzophenone")(C_(6)H_(5)COC_(6)H_(5)) overset(I_(2)//NAOH)to ` No reaction. |
|
| 6. |
Out og 300 g substance [decomposes as per 1st order], hou much will remains after 18 hr? (t_(0.5)=3hr) |
|
Answer» 4.6 gm |
|
| 7. |
Out of 2- chloroethanol and ethanol, which is more acidic and why? |
| Answer» Solution :2 - chloroethanol, DUE to `-I` effect of CHLORINE ATOMS. | |
| 8. |
Out of 2-chloroethanol and ethanol which is more acidic and why? |
Answer» Solution :Due to -I-effect of the CL atom, ELECTRON density in the O-H bond decreases. As a result, O-H bond in 2-chloroethanol BECOMES WEAKER than in ethanol and hence 2-chloroethanol is a stronger ACID than ethanol. 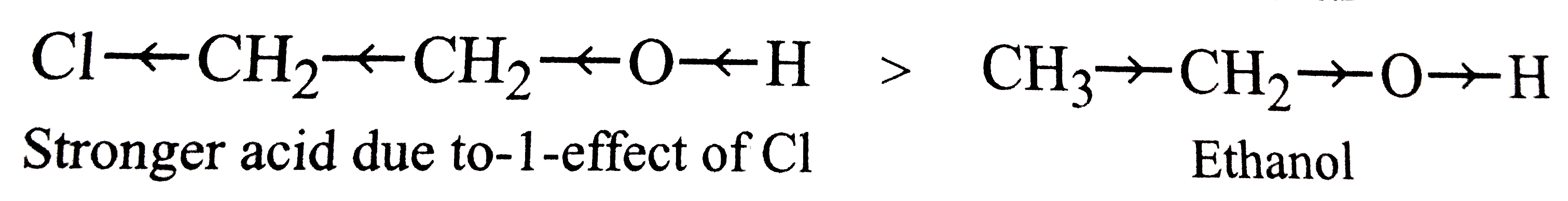
|
|
| 9. |
Out of 1*0 g dioxygen, 1*0 g (atomic) oxygen and 1*0 g ozone, the maximum number of oxygen atoms are contained in |
|
Answer» `1*0` g of atomic oxygen i.e., Atoms of oxygen `= (1xx2)/(32)` in `O_(2)` `=(1)/(16)` in atomic oxygen `=(1xx3)/(48)` in `O_(3)` |
|
| 10. |
Out of 2-chloroethanol and ethanol, which is more acidic and why ? |
| Answer» Solution :`to` 2-chloroethanol is more ACIDIC than ETHANOL. This is because of the presence of chlorine atom that exerts the STRONG -I effect that facilitates the release the PROTON making the ethoxide ion more STABLE. | |
| 11. |
Out of 1M glucose solution and 2M glucose solution which has higher boiling point and why ? |
| Answer» SOLUTION :2M GLUCOSE solution | |
| 12. |
Out of 1M BaCl_2 and 1M Al_2(SO_4)_3, which will have more osmotic pressure ? |
| Answer» Solution :` IM Al_2(SO_4)_3` will have more OSMOTIC pressure but it will produce more IONS in solution per MOLE as compared to 1M `BaCl_2` solution. | |
| 13. |
Out of 1 M glucose and 2 M glucose, which one has a higher boiling point and why? |
| Answer» Solution :The ELEVATION in BOILING of a solution is a colligative property. Hence, it depends upon the number of moles of the solute present in the solution. HIGHER the concentration, greater will be elevation in boiling point. Therefore, 2 M GLUCOSE will have higher boiling point than 1 M glucose solution. | |
| 14. |
Out of (1) butane(2) butan -1- ol (3) butanal(4) butanone . The decreasing order of their B.P is |
|
Answer» `1 GT 2 gt 3 gt 4` |
|
| 15. |
Out of 1-bromobutane, 2-bromobutane and 2-bromo-2 methylpropane, which has the lowest boiling point ? |
| Answer» SOLUTION :2-bromo -2-methylpropane | |
| 16. |
Our of C,N,O and S, the smallest is size of : |
|
Answer» O |
|
| 17. |
Other than the X-ray diffactions, how could be the followingpair of isomers be distinguised from one another by ? [Cr(NH_(3))_(6)][Cr(NO_(2))_(6)] and[Cr(NH_(3))_(4)(NO_(2))_(2)] [Cr(NH_(3))_(2)(NO_(2))_(4)] |
|
Answer» cryoscopic method |
|
| 18. |
Other things being equal , the EMF of a Daniel cell may be increased by |
|
Answer» keeping low temperature `ZN + Cu^(2+) to Zn^(2+) + Cu` , `E_("cell") = E_("cell")^(@) - (RT)/(nF) "ln" ([Zn^(2+)])/([Cu^(2+)])` . Decrease of temperature and increase of `[Cu^(2+)]` will increase the EMF . |
|
| 19. |
Other theories explaining the bonding in coordination compounds are |
|
Answer» Ligand field theory |
|
| 20. |
Other forms of iron can be produced from : |
|
Answer» COST IRON |
|
| 21. |
otal number of pi-electrons in benzene is |
|
Answer» 2 |
|
| 22. |
Ostwald's process for the manufacture of HNO_3 involves the |
|
Answer» OXIDATION of `N_(2)" to "NO` |
|
| 23. |
Ostwald's dilution law is applicable in the case of the solution of: |
|
Answer» `CH_3COOH` |
|
| 24. |
Ostwald's dilution law is applicable in the case of the solution of |
|
Answer» `CH_3 COOH` |
|
| 25. |
Ostwald dilution law is expressed as: |
|
Answer» `K_a = (C CDOT ALPHA^2)/(1- alpha)` |
|
| 27. |
Ostwald dilution law is applicable to the dissociation of |
|
Answer» AMMONIUM HYDROXIDE |
|
| 28. |
"Ostwald dilution law" constituents one of the postulates of the "Arrhenius theory of electrolytic dissociation". It is valid for |
|
Answer» STRONG ELECTROLYTES |
|
| 29. |
Osteomalacia is caused due to deficiency of …………………. |
|
Answer» VITAMIN D |
|
| 30. |
OsO_(4) exists but OsF_(8) does not exist. Why? |
| Answer» Solution :In both CASES O.S. of OS in +8 but in `OsO_(4)`. Os is bonded to OXYGEN by 4 double bonds whereas in `OsF_(8)`, will have to form 8 SINGLE bonds which require much more space around central atom. | |
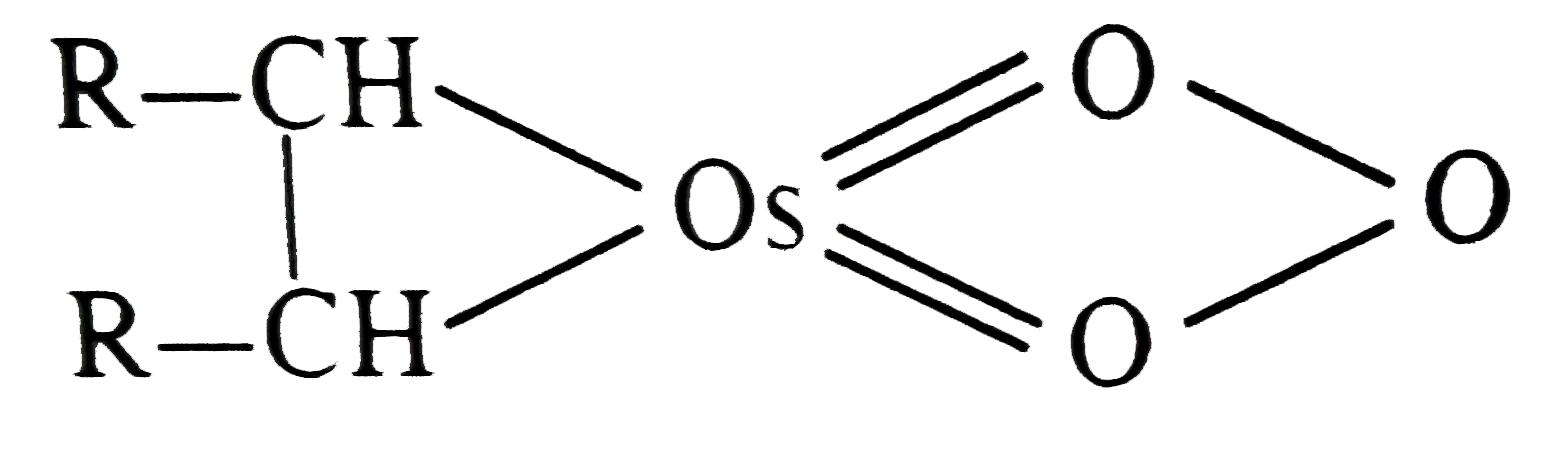
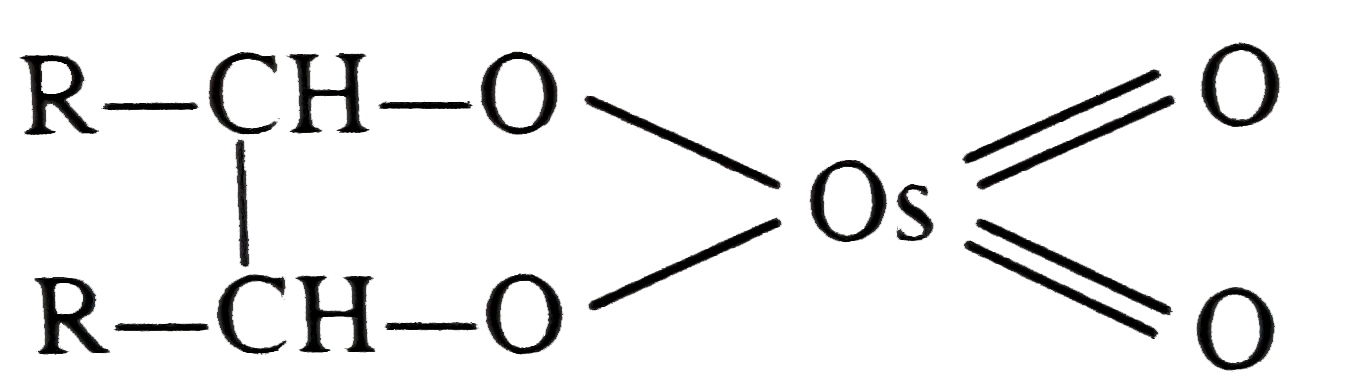
| 31. |
OsO_4 when treated with R-CH=CH-R gives |
|
Answer» 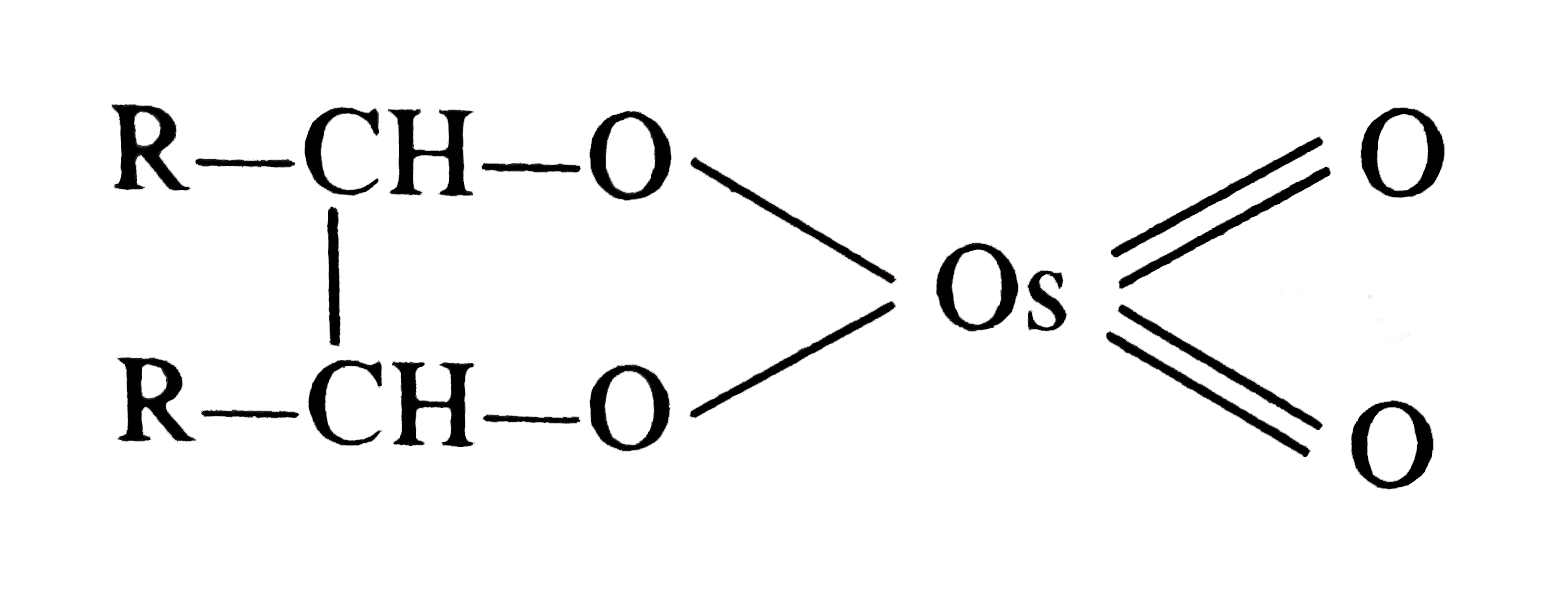
|
|
| 32. |
Osmotic pressure of the solution can be increased by, |
|
Answer» INCREASING the temperature of the solution `pialphaT` |
|
| 33. |
Osmotic pressure of a solution containing 0.1 mole of solute per litre at 273 K is: |
|
Answer» `(0.1)/1 xx0.08205xx273 ` ATM |
|
| 34. |
osmotic presure of a sugar solution at 27^(@)C is 2.46 atmosphere. The concentration of the solution in mol per litre is : |
|
Answer» 1 M `:. C= pi/(RT)= 2.46/(0.082 xx (27 + 273))=0.1 M` |
|
| 35. |
Osmotic pressure of solution (density is 1 g/ml) containing 3g of glucose(molecular weight=180 g) in 60 g of water at 15^(@)C is |
|
Answer» 5.57 ATM |
|
| 36. |
Osmotic pressure of naphthalene solution at 27^@C is 500 mm Hg. When thesolution is diluted and temperature is raised to 47^@C, osmotic pressure of the solutionbecomes 105 mm Hg. The extent of dilution approximately is :- |
|
Answer» 3 TIMES `500/105 = (n_1 /V_1 xx300)/(n_1/V_2 xx 320)` `500/105=V_2/V_1 xx 300/320` `500/105xx320/300 =V_2/V_1` `5 = 320/63=V_2/V_1` |
|
| 37. |
Osmotic pressure of insulin solution at 298K is found to be 0.0072 atm. Hence height of water column due to this osmotic pressure is (density of Hg = 13.6 g cm^(-3)) |
|
Answer» `0.76mm` `P = hdg` (for Hg) 1 atm = 76 MM of Hg `:. 0.0072` atm `-= 0.0072 xx 760mm` of Hg `P = 0.0072 xx 760 xx 13.6 xx 9.8` (for Hg) `P = h xx 1 xx 9.8` (for water) (density of water `= 1g//cm^(3))` `:. 9.8 xx h = 0.0072 xx 60 xx 13.6 xx 9.8` `h = 0.0072 xx 760 xx 13.6mm` `= 74.42 mm` |
|
| 38. |
Osmotic pressure of non-aqueous solution is measured by: |
|
Answer» BERKELEY and HARTLEY METHOD |
|
| 39. |
Osmotic pressure of insulin solution at 298 K is found to be 0.0072 atm. Hence, height of the water column due to this pressure will be (Given density of Hg = "13.6 g mL"^(-1)) |
|
Answer» 7.4 mm But P = 0.0072 ATM. Hence, `h=0.0072xx76cm` of Hg column, d = density of `Hg="13.6 g cm"^(-3), g ="981 cm s"^(_2)` Hence, `P=0.0072xx76xx13.6xx981"(for Hg column)"` For WATER, `d="1 g cm"^(-3)`. Hence, for water column `P=hxx1xx981` Thus, `hxx981=0.0072xx75xx13.6xx981` `"or"h=7.4 cm` |
|
| 40. |
Osmotic pressure of blood is 7.65 atm at 310 K. An aqueous solution of glucose that will be isotonic with blood is …….wt./vol. |
|
Answer» `5.41%` |
|
| 41. |
Osmotic pressure of aqueous solution of urea is 500 mm at 283 K. If its temperature is maintained at 298 K, then on how much dilution its osmotic pressure would be 105.3 mm ? |
|
Answer» 10 times `THEREFORE (500//760)/(105.3//760)=(283xxV_(2))/(V_(1)xx298) therefore (V_(2))/(V_(1))=5` |
|
| 42. |
Osmotic pressure of a urea solution at 10^(@)C is 500mm. Osmotic pressure of the solution become 105.3 mm. When it is diluted and temperature raised to 25^(@)C. The extent of dilution is |
|
Answer» 6 Times `(500 V_(1))/(105.3V_(2))=(nRxx283)/(nR xx298), (V_(1))/(V_(2))=(1)/(5)` so `V_(2)=5V_(1)` |
|
| 43. |
Osmotic pressure of a sugar solution at 24^(@)C is 2.5 atm. The concentration of the solution in gm mole per litre is : |
|
Answer» 10.25 or `C= pi//RT=(2.5)/(0.082xx 297)=0.1025` M |
|
| 44. |
Osmotic pressure of a sugar solution at 24^@C is 2.5 atmosphere. The concentration of the solution in mol per litre is: |
|
Answer» 10.25 |
|
| 45. |
Osmotic pressure of a solution is |
|
Answer» directly PROPORTIONAL to MOLAR concentration of the solution |
|
| 46. |
Osmotic pressure of a solution is 0.0821 atm at temperature of 300 K. The concentration of solution in mol//litre will be |
|
Answer» 0.33 |
|
| 47. |
Osmotic pressure of a solution containing 7 g of a protein per 100 mL of solution is 25 mm Hg at 310 K. Calculate the mass of the protein. (R=0.0821L atm K^(-1)mol^(-1)) |
|
Answer» `T=310 K, R=0.0821" L atm K"^(-1)mol^(-1), M_(B)=?` ` M_(B)=(W_(B)xxRxxT)/(pixxV)=((7g)xx(0.021"L atm K"^(-1)mol^(-1))xx(310 K))/((0.032895" atm")xx(0.1L))=54159.29" g mol"^(-1)` |
|
| 48. |
Osmotic pressure of a solution containing 7 g of a protein present in deciliter of a solutionis 3.3xx10^(-2) bar at 37^(@)C. Calculate the molar mass of protein. |
| Answer» SOLUTION :`54579" G "MOL^(-1)` | |
| 49. |
Osmoticpressureof a solutioncontaining6.8 xx 10^(-3)kg of aproteinper 1xx 10^(-4)m^(3) of solutionis 3.02 xx 10^(3)Pa at37^(@) C Calculatethe molarmassof protein (R= 8.314J K^(-1) mol^(-1)) |
| Answer» Solution :58. 06 kg `mol^(-1)` | |