Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Out of coke and CO_(3) which is better reducing agent for the reduction of ZnO ? Why ? |
| Answer» Solution :Ellingham diagram clearly shows that free energy FORMATION of CO or `CO_(2)` from C is LOWER than that of formation of `CO_(2)` from CO than ZNO. Thus coke is a BETTER reducing agent. | |
| 2. |
Out of coke and CO, which is better reducing agent for the reduction of ZnO? Why? |
| Answer» Solution :COKE (C ) is a better REDUCING AGENT for the reduction of ZnO.Because,when we use coke,the reduction can be easily out at 673 K.Thus carbon (Coke) reduce zinc OXIDE more esily than carbon monoxide (CO).From the Ellingham diagrams ,it is quite clear that the reduction of zinc oxide is more favourble USING coke (`DeltaG` for the formation of carbon monoxide from is more negative). | |
| 3. |
Out of [CoF_(6)]^(3-) and [Co(C_(2)O_(4))_(3)]^(3-), which one complex is: (i) diamagnetic (ii) more stable (iii) outer orbital complex and (iv) low spin complex? "" (Atomic no. Of CO = 27) |
|
Answer» Solution :(i) `[Co(C_(2)O_(4))_(3)]^(3-)` (II) `[Co(C_(2)O_(4))_(3)]^(3-)` (iii) `[CoF_(6)]^(3-)` (iv) `[Co(C_(2)O_(4))_(3)]^(3-)` |
|
| 4. |
Out of CO_2, SiO_2, GeO_2, SnO_2 and PbO_2 |
|
Answer» solid CARBON dioxide and silicon dioxide are hard solid `CO_2 + H_2O rarr H_2CO_3` `SnO_2 + H^(+) rarr Sn^(2+) + H_2O` `SnO_2 + OH^(-) rarr Sn(OH)_(6)^(4-)` `2PbO_2 + 2H_2SO_4 rarr 2PbSO_4 + 2H_2O + O_2`. |
|
| 5. |
Out of coke and carbonmonoxide, which is a better reducing agent for iron oxide? |
| Answer» Solution :Both coke and CARBONMONOXIDE are REDUCTANTS for iron OXIDE. At lowtemperature coke and at high temperature carbonmonoxide are GOOD reducing AGENTS. | |
| 6. |
Out of C,N,O,F and Si, the lowest electronegativity is of: |
|
Answer» C |
|
| 7. |
Out of chlorobenzene and benzyl chloride , which one gets easily hydrolysed by aqueous NaOH and why ? |
Answer» Solution :Benzyl chloride gets EASILY hydrolysed with aqueous NaOH because in CHLOROBENZENE C-Cl bond has partial bond character due to RESONANCE due to that `C-Cl ` bond is DIFFICULT to break to carry out hydrolysis reaction with aqueous NaOH.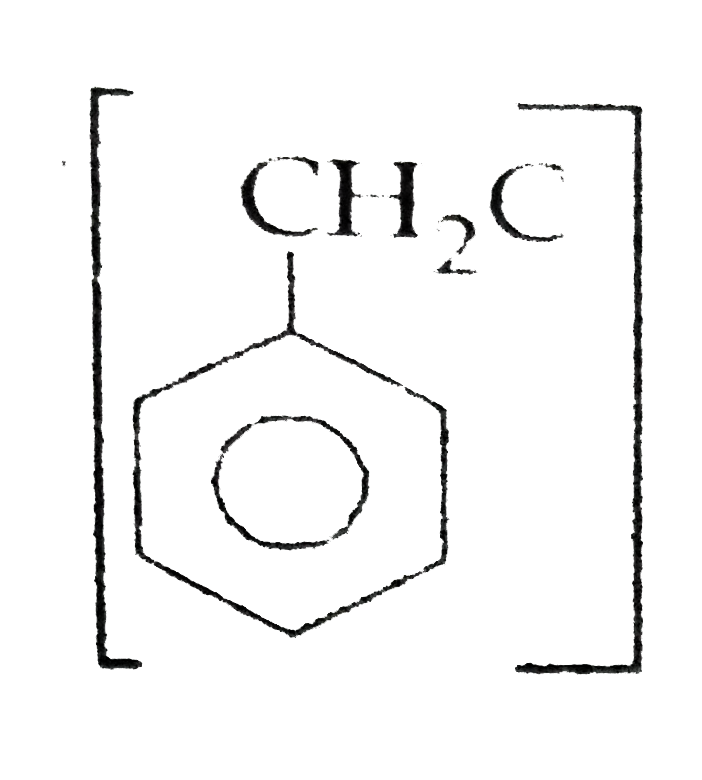 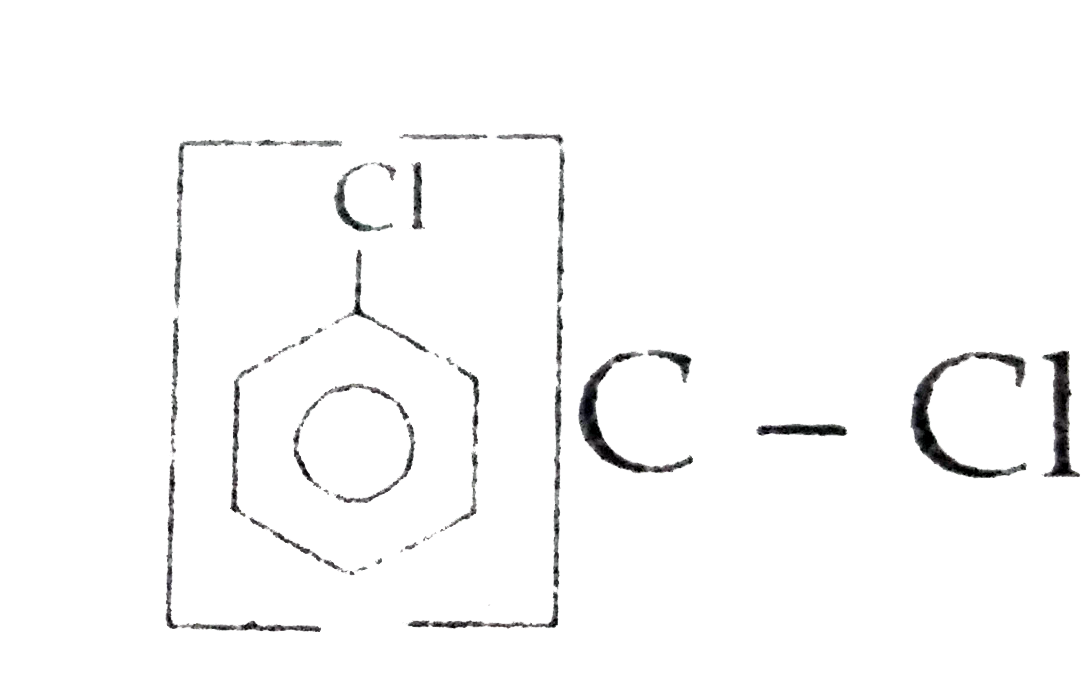 |
|
| 8. |
Out of chlorobenzene and chloromethane which is more reactive towards nucleophilic substitution reactions. |
| Answer» Solution :CHLOROBENZENE can be regarded as a RESONANCE hybrid of five structure. As a result, C-Cl bond in chlorobenzene has some DOUBLE bond CHARACTER and hence more difficult to break than C-Cl bond in chloromethane `(CH_(3)-Cl)` which has pure SINGLE bond character. | |
| 9. |
Out of CH_(3)_underset(C H_(3))underset(|)(C)H-CH_(2)-Cl and CH_(3)-CH_(2)-underset(CH_(3))underset(|)(C)H-Cl which in more reactive towards S_(N^(1))reaction and why? |
|
Answer» SOLUTION :`CH_(3)-CH_(2)-UNDERSET(CH_(3))underset(|)(C)H-Cl` is more reactive. Because SECONDARY CARBOCATION formed in the slow step is more stable as compared to the primary carbocation in the other reaction. |
|
| 10. |
Out of CHCl_(3) , CH_(4) and SF_(4) , the molecule are not having regular geometry are :- |
|
Answer» `CHCl_(3)` only |
|
| 11. |
Out of chlorobenzene and benzyl chloride, which one gets easily hydrolysed by aqueous NaOH and why? |
| Answer» Solution : Benzyl CHLORIDE, DUE to resonance, stable benzyl CARBOCATION is formed. | |
| 12. |
Out of CH_(3)OH, RCH_(2)OH,R_(2)-CHOH and R_(3)COH, which is the least acidic ? |
|
Answer» `CH_(3)OH` has higher boiling POINT than `CH_(3)NH_(2)`. This is because there is strong intermolecular hydrogen bonding in `CH_(3)OH`. Because of lower ELECTRONEGATIVITY of N than O, hydrogen bonding does not take PLACE in `CH_(3)NH_(2)`. |
|
| 13. |
Out of CH_(3)OH and(##SB_CHM_XII_OD_I_2016_E01_004_Q01.png" width="80%">, which one is more acidic ? |
| Answer» Solution :Phenol is more acidic that ETHANOL because of RESONANCE oxygen has POSITIVE charge and tends to RELEASE HYDROGEN radily. Hence phenol is more acidic. | |
| 14. |
Out of (CH_(3))_(3)C-Br and (CH_(3))_(3)C-I, which one is more reactive towards S_(N)l and why? |
| Answer» Solution :`(CH_(3))_(3)Cl_(2)` DUE to large SIZE of IODINE / BETTER leaving group / Due to lower electronegativity. | |
| 15. |
Out of CH_(3)-underset(CH_(3))underset(|)(C)H-CH_(2)-Cl and CH_(3)-CH_(2)-underset(CH_(3))underset(|)(C)H-Cl which is more reactive towards S_(N)1 reaction and why? |
Answer» Solution :Reactivity in `S_(N)1` reactions DEPENDS upon the stability of the carbocation it gives on ionization. Now, the first COMPOUND, i.e., 1-chloro-2-methylpropane gives a `1^(@)` carbocation and the second compound, i.e., 2-chlorobutane gives a `2^(@)` carbocation. 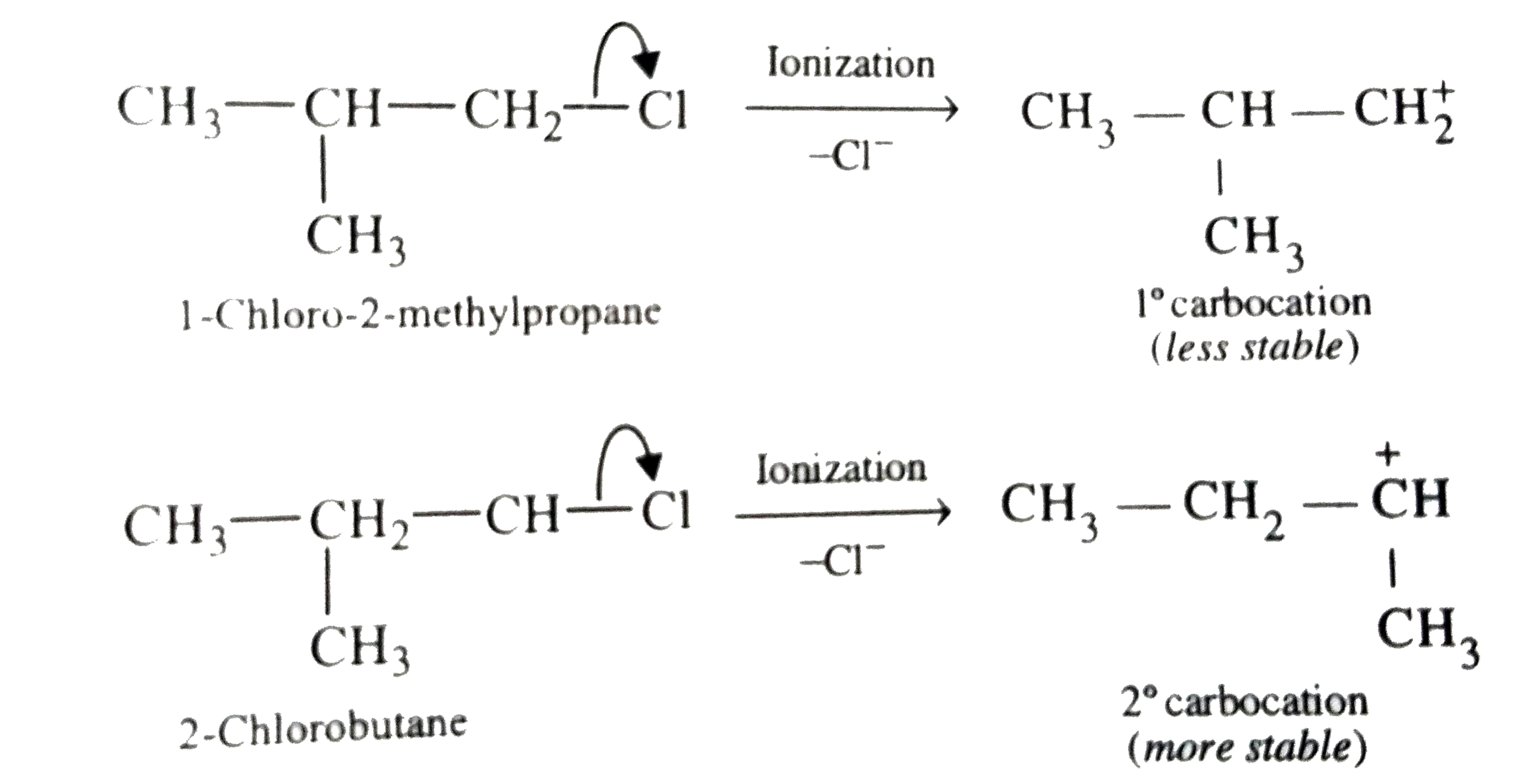 SINCE `2^(@)` carbocations are more stable than `1^(@)` carbocations, therefore, second compound, i.e., 2-chlorobutane reacts faster than the first compound, i.e., 1-chloro-2-methylpropane. Alternatively, first compound, i.e., 1-chloro-2methylbutane is a `1^(@)` alkyl halide while the second compound, i.e., 2-chlorobutane is a `2^(@)` alkyl halide. since in `S_(N)1` reactions, `2^(@)` alkyl halides are more reactive than `1^(@)` alkyl halides, therefore, second compound, i.e., 2-chlorobutane reacts faster than the first compound, i.e., 1-chloro-2-methylpropane. |
|
| 16. |
Out of CH_(3)CH_(2)COCH_(2)CH_(3) and CH_(3)CH_(2)CH_(2)COCH_(3) which gives iodofrom test. |
| Answer» Solution :`CH_(3)CH_(2)CH_(2)COCH_(3)` will give IODOFORM TEST. | |
| 17. |
Out of CH_(3) - underset(CH_(3))underset(|)CH - CH_(2) - Cl " and " CH_(3) - CH_(2) - underset(CH_(3))underset(|)CH - Cl, which is more reactive towards S_(N)1 reaction and why ? |
| Answer» Solution :`CH_(3) - CH_(2) - UNDERSET(CH_(3))underset(|)CH - Cl ` is more reactive TOWARDS `S_(N) 1` because it will FORM the cation easiliy . | |
| 18. |
out of CH_3-F, CH_3Cl, CH_3-BR, and CH_3-I, CH_3-Cl has highest dipole moment. |
| Answer» | |
| 19. |
Out of Cannizzaro's reaction and Aldol condensation which is possible for benzaldehyde ? Give equation . |
Answer» SOLUTION :
|
|
| 20. |
Out of C_(6)H_(5)CH_(2)Cl and C_(6)H_(5)CHClC_(6)H_(5), which is more easily hydrolysed by aqueous KOH. |
|
Answer» Solution :HYDROLYSIS follow `S_(N)1` path. The `S_(N)1` reaction procceds through the formation of more stable intermediate product carbocation. Thus, greater is the stability of carbocation, faster will be the reaction rate. The intermediate product formed during the reaction of given two compounds are : 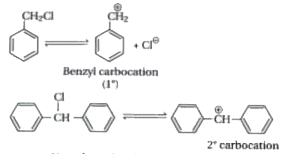 The `2^(@)` carbocation is more STABILIZED the + R effect of two PHENYL groups. Thus, `C_(6)H_(5)underset("Cl ")underset("| ")("C ")H-C_(6)H_(5)` will READILY undergo hydrolysis. |
|
| 21. |
Out of C_(6)H_(5)CH_(2)Cl " and " C_(6)H_(5)CHClC_(6)H_(5) which is more easily hydrolysed by equeous KOH ? |
Answer» Solution :`C_(6)H_(5)CH_(2)Cl " isa " 1^(@) ` aralkyl halide while `C_(6)H_(5)CH(Cl) C_(6)H_(5) " is a " 2^(@)` aralkyl halide.  In `S_(N)1` REACTIONS, reactivity depends upon the stability of CARBOCATIONS. CARBOCATION `C_(6)H_(5)overset (+)CHC_(6)H_(5)` where the `+v` charge is delocalised over two `C_(6)H_(5)` RINGS is more stable than the carbocation , `C_(6)H_(5)overset(+)CH_(2)` where ` + ve` charge is delocalized over one `C_(6)H_(5)` ring. Therefore , `C_(6)H_(5)CHClC_(6)H_(5)` gets hydrolysed more easily than `C_(6)H_(5)CH_(2)Cl " under"S_(N)1` conditions . But under `S_(N)2` conditions, the reactivity is affected by steric hindrance , therefore `C_(6)H_(5)CH_(2)Cl` gets hydrolysed more easily than `C_(6)H_(5)CHCl_(6)H_(5) " under " S_(N)2 ` conditions . |
|
| 22. |
Out of C and CO which is better reducing agentfor ZnO ? |
|
Answer» Solution :C is better REDUCING AGENT for ZNO. `ZnO + Coverset(1673K)to ZN + CO` |
|
| 23. |
Out of C_(6)H_(5)CH_(2)Cl and C_(6)H_(5)CHClC_(6)H_(5) which is more easily hydrolysed by aqueous KOH? |
Answer» Solution :`C_(6)H_(5)CH_(2)Cl` is a `1^(@)` aralkyl HALIDE while `C_(6)H_(5)CH(Cl)C_(6)H_(5)` is a `2^(@)` aralkyl halide. 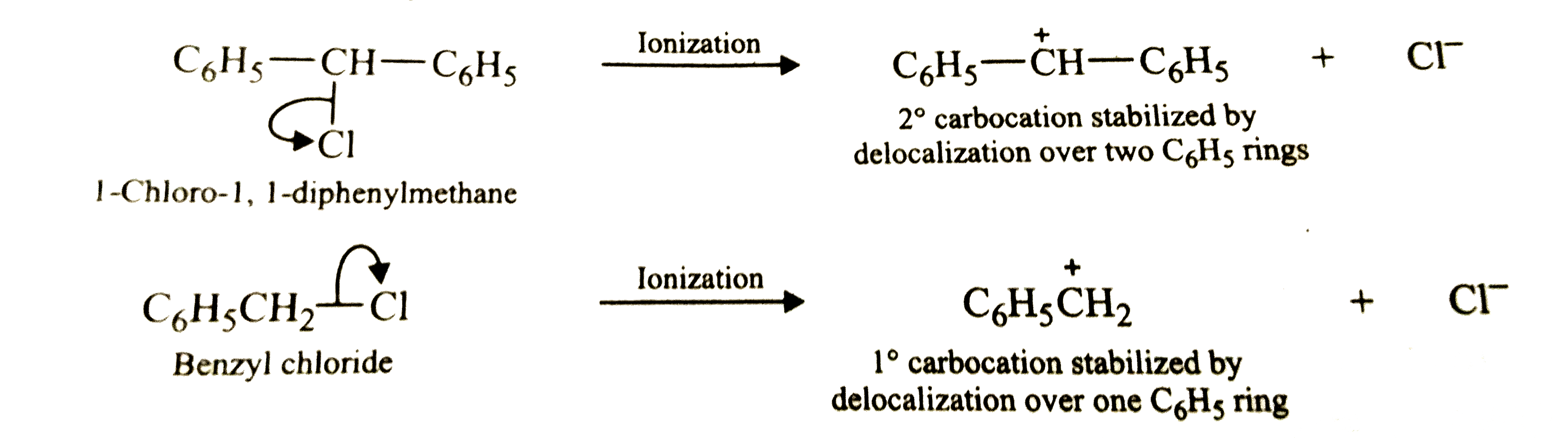 In `S_(N)1` reactions, the reactivity depends upon the stability of carbocations, SINCE the carbocation `C_(6)H_(5)overset(+)(C)HC_(6)H_(5)` (where the +ve charge is delocalised over the `C_(6)H_(5)` RINGS) DERIVED from `C_(6)H_(5)-CHCl-C_(6)H_(5)` is more stable than the carbocation, `C_(6)H_(5)CHClC_(6)H_(5)` gets hydrolysed more easily than `C_(6)H_(5)` ring) derived from `C_(6)H_(5)CH_(2)Cl`, therefore, `C_(6)H_(5)CHClC_(6)H_(5)` gets hydrolysed more easily than `C_(6)H_(5)CH_(2)Cl` under `S_(N)2` conditions. However, under `S_(N)2` conditions, the reactivity depends on STERIC hindrance, therefore, under `S_(N)2` conditions, `C_(6)H_(5)CH_(2)Cl` (with lesser steric hindrance) gets hydrlysed more easily than `C_(6)H_(5)CHClC_(6)H_(5)`. |
|
| 24. |
Out of C_(6)H_(5)CH_(2)Cl and C_(6)H_(5)CHCIC_(6)H_(5), which is more easily hydrolysed by aqueous KOH? Why? |
| Answer» SOLUTION :Beryllium and magnesium atoms in comparison to other ALKALINE earth metals are comparatively smaller and their ionisation enthalpies are very high. Hence,the ENERGY of the FLAME is not sufficient to excite their electrons to higher energy levels. These elements, there- fore, do not GIVE any colour in Bunsen flame. | |
| 25. |
Out ofCandCO, which is betterreducingagentfor ZnO ? |
| Answer» SOLUTION : The` DELTA _f G ^(@) ` of`CO_ 2 `from CO isalways higherthanthatof` Delta_f G ^(@)`of` ZnO `.Therefore, COcannotbe usedforreductionofZnOtoZn. In contrast,` Delta _f G ^(@) `ofCOfrom Cis lowerattemperaturesabove1180 Kwhilethatof` CO_ 2`from C islowerintemperatureabove1270Kthan`Delta_ FG ^(@) `ofZnO.Thus,above1270 K, ZnO canbereducedto Zn byC.Inactualpractice , reductionisusuallycarriedoutaround1673 K so that thereductionprocess essentilly goestocompletion.Thus,outofCandCO, C isabetter reducingagentthanCOforreductionofZnOtoZn. | |
| 26. |
Out of C and CO, which is better reducing agent at 673 K ? |
| Answer» Solution :At 673 K, the `DELTAG^@` VS T LINE for CO, `CO_2` is lower than that of C, CO line. Therefore, CO can be used as better reducing AGENT at 673 K. | |
| 27. |
Out of C and CO which is a better reducing agent for ZnO ? |
|
Answer» Solution :From Ellingham DIAGRAM, At TEMPERATURE above 1120 K, `Delta_f G_((C","CO))^(@) < Delta_fG_((Zn"," ZnO))^@`and at temperature above 1323 K `Delta_fG_((C"," CO_2)) < Delta_fG_((Zn"," ZnO))^(@)` However, `Delta_fG_((CO"," CO_2)) < Delta_fG_((Zn"," ZnO))^(@)` Hence, CARBON is BETTER reducing agent than CO. The reduction of zinc is CARRIED out at 1673 K. `ZnO + C overset(1673 K)(rarr) Zn + CO`. |
|
| 28. |
Out of C and CO, which is a better reducing agent for ZnO? |
| Answer» SOLUTION :Refer to Ellingham diagram FIG. 6.1. The standard free energy of FORMATION `(triangle_(f)G^(0))` of CO from C becomes lower above 1223 K and that of `CO_(2)` from C becomes lower above 1323 K than `triangle_(f)G^(0)" of "ZnO`. Therefore, above these temperatures, C can reduce ZnO to Zn. However, `triangle_(f)G^(0)" of "CO_(2)` from CO is always higher than that of ZnO. Therefore, CO cannot reduce ZnO to Zn even at higher temperature (AROUND 1673 K). Thus, out of C and CO, C is a better reducing agent. | |
| 29. |
Out of C and CO, which is a better reducing agent for ZnO ? |
| Answer» SOLUTION :Carbon is the better reducing agent for REDUCTION of `ZnO`. This is because the temperature REQUIRED for reduction of ZnO with CO is very high when compared with the temperature required for reduction of ZnO with carbon. Reduction temperatures can be SUPPORTED from Ellingham diagram. | |
| 30. |
Out of C and CO which is a better reducing agent for FeO : In the lower part of blast furnace (Higher temperature). |
| Answer» SOLUTION :In the lower part of blast furnace i.e., at higher temperature, C is a better reducing AGENT because the CURVE of `C to CO_(2)` lies below the curve for `2Fe+O_(2) to 2FeO`. | |
| 31. |
Out of C and CO which is a better reducing agent for FeO : In the upper part of blast furnace (Lower temperature). |
| Answer» Solution :In the upper part of blast furnace i.e., at lower TEMPERATURE, CO is a better REDUCING agent because the curve for `CO to CO_(2)` lies below the curve for `Fe to FeO`. | |
| 32. |
Out of C and CO which is a better reducing agent for Feo. (i) In the lower part of blast furnace (Higher temperature) (ii) In the upper part of blast furnace (Lower temperature) |
|
Answer» Solution :C is BETTER reducing AGENT at higher TEMPERATURE (lower part of blast FURNACE). (ii) CO is better reducing agent at lower temperature (higher part of blast furnace). |
|
| 33. |
OutofC and COwhichisabetterreducingagentfor FeO (i) In thelower partofblast furnace (higher temperature) (ii)Intheupper part of blastfunace (lowertemperature ) |
|
Answer» Solution :Althoughboth C and COcan reduce FeOtoFe, but C is abetterreducingatentthan CO in thelower partoftheblastfrunacewhere thetemperatureis high `(gt 1073 K) ` while CO isbetterreducingthat C in theupper partof theblastfurnace where thetemperatureis low`(lt 1073 K ` actually between500 -900 K ) as explainedbelow : (i)In thelowerpart of theblast FURNACE, temperatureishigh ( `gt 1073 K`).Above1073 K, the` Delta _fG^(@) `for COfromC ISMORE negativethan thatofFeO from Fe. Therefore, at high temperatures`(gt 1073 K)`, C caneasilyreduce FeO to Fe. `"" FeO + C overset (gt 1073 K) toFe + CO` (ii)However,in theupperpart ofblastfunace,temperatureis low ( ` lt 1073K` , actually between500 -900 K). Below 1073 K `Delta_f G^(@) ` of` CO_ 2 `from COIS more negativethat that ofFeO from Fe. Therefore, at lower temperatures `( lt1073K) ` , CO isabetterreducing agentthan C. ` ""FeO+CO overset (500 -900 K) to Fe+CO_ 2` |
|
| 34. |
Out of C and CO, which is a better reducing agent at lower temperature range in the blast furnace to extract iron from the oxide ore? |
|
Answer» Solution :CO is a better reducing agent at the LOWER temperature range in the BLAST FURNACE. `{:(3Fe_(2)O_(3)+CO to 2Fe_(3)O_(4)+CO_(2)),(Fe_(3)O_(4)+4CO to 3Fe+4CO_(2)),(Fe_(2)O_(3)+CO to 2FeO+CO_(2)):}`. |
|
| 35. |
Outof C and CO whichisa betterreducingagentat 983K ? |
|
Answer» Solution :Whencarbon reacts withdioxygen TOW reactions are possible: `C(s) + O_(2)(g) to CO_(2) (g)` `2C(s) + O_(2)(g) to 2CO(g)` The CO thus producedcanalso reactwith`O_(2)`to FORM `CO_(2)` . `2 CO(g) + O_(2) (g) to 2CO_(2)(g)`In the firstreaction, the volumeof `CO_(2)`produced is the sameas that of `O_(2)`pordoued is the sameas that of `O_(2)`used, therefore, `DeltaS` is very small and `Delta_(f)G^(@)` does notchangeswithtempreature. Thus, the graph of `Delta_(f)G^(@)` against T is almosthorizontal .(Fig . 6.19)  Thesecond reactionpreducestwo volumesof COforeveryone volumeof ` O _ 2`used.Thus,` DeltaS`is positiveandhence` Delta _f G ^(@) `becomesincreasinglynegativeas thetemperatureincreases.Consequenty, C, CO lineontheEllinghamdiagramslopesdownwards . Thethirdreactionproducestwo volumesof` CO_ 2`foreverythreevolumesofreactantsused.Thus, ` DeltaS` is-veandhence`Delta_ fG^(@) `becomesincreasingly+veas thetemperatureincreases. Consequentrly,CO, `CO_ 2`lineonthe Ellinghamdiagramslopesupwards. Thethreelinescrossat983 K . Belowthistemperature, FORMATIONOF` CO_ 2`fromCaswellasfromCOisenergeticallymorefavourable.Inotherwords, below 983 K ,both Cand Cocanact asreducingagentsbut sinceCOcanbemore easilyoxidisedto`CO_ 2`thanC to`CO_2`,therefore , below983 K, COisamoreeffectivereducingagentthanC. However, above983 K, COis morestableandhenceitsoxidationto` CO _ 2`isless rapidthanthatofC to` CO _ 2`. Therefore,above983 K, C isabetterreducingagentthan CO. |
|
| 36. |
Out of C and CO, which is a better reducing agent at 673K? |
|
Answer» Solution :Carbon reacts with oxygen in one of the following two ways : `{:(C(s)+O_(2)(g) to CO_(2)(s)"""..........."(i)),(2C(s)+O_(2)(g) to 2CO(g)"""..........."(ii)):}` The CO produced in (ii) can also react with `O_(2)` to form `CO_(2)` `2CO(g)+O_(2)(g) to 2CO_(2)(g)""".........."(iii)` In the first REACTION, the volume of `CO_(2)` produced is almost the same as that of `O_(2)` used, therefore, `triangleS` is very small and `triangleG^(0)` does not change with temperature. Thus, the graph of `triangleG^(0)` against T is almost horizontal (see the figure). The second reaction produces two VOLUMES of CO for EVERY one volume of `O_(2)` used. Thus, `triangleS` is positive and hence `triangleG^(0)` becomes increasingly negative as the temperature increases. `triangleG= triangleH- T triangleS`. Consequently, its line on the Ellingham diagram slopes downwards. The third reaction produces two volumes of `CO_(2)` for every three volumes of reactants used. Thus, `triangleS` is `-ve` and hence `triangleG^(0)` becomes increases. Consequently, its line on the Ellingham diagram slops upwards. The three lines cross at 673K (see Fig. 6.3). Below this temperature, formation of `CO_(2)` is energetically more favourable and above this temperature, formation of CO is energetically more favourable. In other words, below 673 K, both C and CO can act as reducing agents but SINCE CO can be more easily oxidised to `CO_(2)` than C to `CO_(2)`, therefore, below 673 K, CO is a more effective reducing agent than C. Above 673 K, `triangleG` vs T curve for `CO to CO_(2)` lies above the corresponding curves for both `C to CO_(2)" and "C to CO`, therefore C is a better reducing agent than CO. 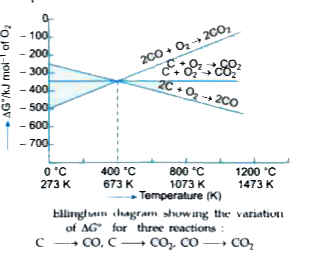
|
|
| 37. |
Out of C and CO, which is a better reducing agent at 673 K? |
|
Answer» Solution :The following reactions TAKE place, when carbon and carbonmonoxide acts as a reducing AGENT. (i) `{:(2C + O_2 , to , 2CO),(C + O_2, to , CO_2):} implies` Carbon as reducing agent. (ii) `2CO + O_2 to 2CO_2 } CO` is reducing agent. From ELLINGHAM diagram, at TEMPERATURE 673 K, the free energy CHANGE of the formation of `CO_2` from `CO` is more negative than the formation of `CO` or `CO_2` from carbon. Hence, at 673 K, CO is better reducing agent than carbon. |
|
| 38. |
Out of butan-1-ol and but-1-amine, which is more soluble ? |
|
Answer» |
|
| 39. |
Out of Br^(-) and I^(-) ions, which is a stronger nucleophile? |
| Answer» Solution :Iodide ION `(I^(-))` is a stronger nucleophile than bromide ion `(Br^(-))` because iodine is LESS electronegative and THEREFORE, electron PAIR can be more readily released by `I^(-)` ion than by `Br^(-)` ion. | |
| 40. |
Out of boilingpont (I), entropy (II), pH (III) and density (IV), Intensiveproperties are : |
|
Answer» I,II |
|
| 41. |
Out of boiling point (I), entropy (II), pH (III) and density (IV), Intensive properties are : |
|
Answer» I, II |
|
| 42. |
Out of benzyl chloride and chlorobenzene, which will give a white precipitate with AgNO_(3) solution? |
| Answer» SOLUTION :BENZYL CHLORIDE. | |
| 43. |
Out of benzene naphtahlene anthracene, and phenantrene, which is more reactive in the Diels - Alder reaction? |
|
Answer» Solution :(i) Antrancene is more REACTIVE in the DIES- Alder reaction (see Illustration `11.10). ii. Benzene does not undergo Diels-Alder reaction because its armonic ring is very stable (high `R.E. = 150.4 kJ mol^(-1)`) (iiI) Naphtalene does not reacts at orinary conditions but can react at very high pressure. It resists losing resonance energy. iv. Phenanthere does not react like benzen. If central ring of phenanthere reacts is LOST . If phenanthrene ACTS as dienophile at `(C^(() = C^(10))` double bonds, then teh aromaticity of the TWO rings is lost `:.` Reactivity order is: Anthrancene gt napthalene gt benzene = phenanthere. |
|
| 44. |
Out of benzene and phenol which is more easily nitrated and why? |
| Answer» SOLUTION :Phenol is NITRATED more easily than BENZENE because `-OH` group is electron releasingand activating group. It increases electron density at o- and p - POSITIONS by means of RESONANCE. | |
| 45. |
Out of below correct matching is |
|
Answer» P-1-i |
|
| 46. |
Out of Be,B, C, N, O, F and Ne the lowest negative electron gain enthalpy is of: |
|
Answer» N |
|
| 47. |
Out of BaCl_2 and KCI, which one is more effective in causing coagulation of a negatively charged colloidal sol ? Give reasons. |
| Answer» SOLUTION :`BaCl_2` PRODUCES dipositive `Ba^(2+)` cations while NaCl produces monopositive `Na^(+)`ions. Therefore `Ba^(2+)` ION are more EFFECTIVE in causing coagulation of a negatively charged colloidal solution. | |
| 48. |
Out of below correct matching is- |
|
Answer» S-4-iv |
|
| 49. |
Out of BaCl_92) and KCl, which one is more effective inj causing coagulation of a negatively charged sollosidal sol ? Give reason. |
| Answer» SOLUTION :`BaCl_(2)` is more effective in causing coagulation of negatively charged colloidal sol because GREATER the valency of the COAGULATING ion (oppoitely charged ion), greater is its POWER to bring about coagulation (`Ba^(2+)` has GRETER velency that `K^(+)`) | |
| 50. |
Out of aniline and N-nitrosoamine........is carcinogenic |
| Answer» SOLUTION : N-nitrosomine | |