Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Of PH_(3) and H_(2)S which is more acidic and why? |
| Answer» Solution :Out of `PH_(3)` and `H_(2)S, H_(2)S` is more acidic because acidic character increases on moving ACROSS the PERIOD. | |
| 2. |
Of NH_3 and N_2 which gas will be adsorbed more readily on the surface of charcoal and why? |
| Answer» Solution :`NH_3` is adsorbed more READILY because it is more easily LIQUEFIABLE as compared to `N_2`. Moreover, `NH_3` molecule has large MOLECULAR size. | |
| 3. |
Of methylamine and aniline, which is a stronger base and why? |
| Answer» SOLUTION :METHYLAMINE is a stronger base because of +I effect of methyl group, while aniline is WEAKER because of -I and -R effects. | |
| 4. |
Of H_(2)S and PH_(3), whch is more acidic. |
|
Answer» Solution :The strength of an acid depends upon the stability of the anion (i.e., conjugate base) it GIVES after release of a proton. Now the disssociation of `PH_(3), H_(2)S` and HCl in AQUEOUS solution occurs as follows : `{:(PH_(3)(g) + aq rarr H^(+) (aq) + H_(2)P^(-)(aq)" ,"" "K_(a) = 1.6 xx 10^(-29)),(H_(2)S (g) + aq rarr H^(+) (aq) + HS^(-) (aq)" ,"" "K_(a) = 1.3 xx 10^(-7)):}` Since S (EN = 2.5) is more electronegative than P (EN = 2.1), therefore, S in `HS^(-)` can accommodate the negative charge more easily than P in `H_(2)P^(-)`. In other words, `HS^(-)` is more stable than `H_(2)P^(-)`, i.e., `H_(2)S` can release a proton more easily than `PH_(3)` and HENCE `H_(2)S` is a stronger acid than `PH_(3)`. |
|
| 5. |
Of methylamine and aniline which is a stronger base and why ? |
| Answer» SOLUTION :METHYLAMINE is a stronger base because of `+I` effect of methyl group, while aniline is WEAKER because of `-I" and "-R` EFFECTS of phenyl group. | |
| 6. |
OF^(-) does not exist , though HOF is known (R ) HOF molecule does not ionise easily |
|
Answer» Both (A) and (R ) are TRUE and (R ) is the CORRECT explanation of (A) |
|
| 7. |
Of all the metals in the periodic table, only………………….. crystallizes in simple cubic pattern. |
| Answer» SOLUTION :POLONIUM | |
| 8. |
Of 0.1 molal solution of glucose and sodium chloride, respectively which one will have ahigher boiling point ? |
| Answer» SOLUTION : 0.1 molal solution of SODIUM CHLORIDE will have a HIGHER boiling point. This is due to dissociationof NaCl into `Na^+` and `CL^-`ions. | |
| 10. |
Odd one out |
|
Answer» Each sphere is in CONTACT with 6 of its neighbouring spheres in ABAB types |
|
| 11. |
Octet rule is not satisfied for which of the following molecules? |
|
Answer» `Cl_2` |
|
| 12. |
Octet rule is not valid for the molecule : |
|
Answer» `CO_2 ` |
|
| 13. |
Octane number is zero for |
|
Answer» n-OCTANE |
|
| 14. |
Octane number of petrol (gasoline) can be increased by |
|
Answer» Aromatisation |
|
| 16. |
Octane number has 0 value for |
|
Answer» Iso-octane |
|
| 17. |
Octanenumberfo gasolinecan be increased by addition ofBTX.BTX stands for: |
|
Answer» BUTANE , TETRAETHYL LEAD and xylene |
|
| 18. |
Octane number can be changed by |
|
Answer» `C_(8)-C_(12)` |
|
| 19. |
Octanenumbercan bechangedby : |
|
Answer» ISOMERISATION |
|
| 20. |
Octanenumbercanbechangedby : |
|
Answer» ISOMERISATION |
|
| 21. |
Octane number 116 is given for: |
|
Answer» 2,2,2-trinethyl pentane |
|
| 22. |
Octane no of 2,2,4-trimethylpentane has been assumed to be: |
|
Answer» 100 |
|
| 24. |
Octahedral complexes involving d^(2)sp^(3) hybridisation are called …… complexes whereas those involving sp^(3)d^(2) hybridisation are called …… . |
| Answer» SOLUTION :INNER ORBITAL, OUTER orbital | |
| 25. |
Octahedral crystalline variety of sulphur contain ''X'' S-S bonds in which covalency of sulphur =y. It contains ''Z'' lonepair of electrons, then (z - x - y)= |
| Answer» | |
| 26. |
OCH_(3)group is |
|
Answer» Stronger + R GROUP than - OH |
|
| 27. |
OCH-CHO overset(OH^(-))rarr HOH_(2)C-COOH The reaction given is |
|
Answer» ALDOL condensation `{:(CHO),(|),(CHO):}+NaOHrarr{:(CH_(2)OH),(|),(COONa):}` |
|
| 28. |
Ocidation number of Fe in Fe_3O_4 are : |
| Answer» Answer :A | |
| 29. |
Occlusion of Hydrogen on Palladium is an example for ………type solution |
|
Answer» GAS in solid |
|
| 30. |
Obtain ethylamine (ethanamine) from acetonitrile (methyl cyanide). |
| Answer» Solution :`underset("acetonitrile")(CH_(3)-CN)+4[H]OVERSET(Na//C_(2)H_(5)OH)rarrunderset("ethylamine")(CH_(3)-CH_(2))-NH_(2)` | |
| 31. |
Obtain a relation log_(10).(k_(2))/(k_(1)) = (E_(a) (T_(2) -T_(1)))/(2.303 R xx T_(1) xx T_(2)) obtaina relationshowingvariationin rateconstant withtemeprature . |
|
Answer» SOLUTION :ByArrhenius equaiton , the rateconstantk of thereactionat a temperatureT is repesented as . `k = A xx e^(-E_(a)//RT)` whereAis a frequncyfactore, R is a gas constantand `E_(a)`is theenergy of activation. By takinglogarithmto the base we, get In = In `(A xx^(-Ea//RT))` = In `A +lne^(-E_(a)//RT) = In A -(E_(a))/(RT)` In e `therefore In k = In A - (E_(a))/(RT)` `therefore2.303 log_(10) k = 2.303 log.(A)/(10) -(E_(a))/(RT)` `therefore log_(10)K = log_(10)A - (E_(a))/(2.303RT)` If `k_(1)` and `k_(2)`are therateconstsntsat temperatures `T_(1)`and `T_(2)`respectively, then `log_(10)k_(1) = log_(10)A -(E_(a))/(2303RT_(1))` `log_(10) k_(2) = log_(10)A-(E)/(2.303RT_(2))` `thereforelog_(10)k_(2) - log_(10)k_(1) = [log_(10)A - (E_(a))/(2.303 RT_(2))] - [log_(10)A-(E_(a))/(2.303RT_(1))]` `log_(10).(k_(2))/(k_(1)) = (E_(a))/(2.303RT) - (E_(a))/(2.303RT)` `= (E_(a))/(2.303R)((1)/(T_(1)) -(1)/(T_(2))) "" log_(10).(k_(2))/(k_(1))=(E_(a)(T_(2) -T_(1)))/(2.303RT_(1) xx T_(2))` Bymeasuringthe rateconstant`k_(1)`and `k_(2)` at TWO DIFFERENTTEMPERATURES `T_(1)` and `T_(2)`the energyof activation `E_(a)`of the reaction can be obtained . |
|
| 32. |
Obtain an expression for half-life period of zero order reaction. |
|
Answer» Solution :The rate LAW expression for zero order reactio'n is,`[A]_(t) = -kt [A]_(0)` Where `[A]_(0)`and `[A]_(0)`are theconcentrationof thereactantat time t - 0 andaftertime t respectively. Half -lifeperiod`t_(1//2)`is the time WHENTHE CONCETRATION reduce form `[A]_(0)`to `[A]_(0)` /2. .E., at `t = t_(1//2)` `[A]_(t) = [A]_(0)`/2. `therefore([A]_(0))/(2) = - kt_(1//2) +[A]_(0)` ` therefore kt_(1//2) = ([A]_(0))/(2) = ([A]_(0))/(2)` `therefore t_(1//2) = ([A]_(0))/(2k)` Hence for a zero order reaction, half-life PERIOD 1s directlyproportional to the initial concentration of the reactant. |
|
| 33. |
Obtain Arrhenius equation from collision theory of bimolecular reactions. |
|
Answer» Solution :Consider following a bimolecular REACTION, `A - B+ C to A + B -C` (i) Collisions of reactant molecules: The rate of reaction depends on the rate and frequency of collisions between reacting species. But the observed rate of reaction is low as compared to the rate of collisions. (ii) Energy of activation : For fruitful collisions, the colliding molecules must possess a certain amount of energy called activation energy `E_(a)`. Due to collisions of three nuclei namely A, B and C the old A-B bond is weakened while new bond is partially formed between B and C, and results in the formation of an activated complex or a transition state.`underset("Reactants")(A-B +C) to underset("Complex")underset("Activated")(A....B....C) to underset("PRODUCT")(A + B -C)` The transition state always has higher energy than reactants or products. Due to high energy, activated complex is unstable, short lived and decomposes into the products. Thefraction (f) of molesules at temperature T having activation energy `E_(a)` is given by`f = e^(-E_(a)//RT)` . If Z is the TOTAL COLLISION frequency then successful collision frequency will be `Z xx e^(-E_(a)//RT)` , and the rateof collisions will be. Total collision rate `= Z xx [AB] xx [C]` Successful collision rate active masses of AB and C respectively. Orientation of reactantmolecules: The collidingmolecules musthaveor Reaction rate =Px successful collision rate `=P xx Z xx e^(-E_(a)//RT) xx [AB] xx c` By rate law, for bimolecular SECOND order, the rate of reaction is, Reaction rate = [AB][C], where k is a rate constant. By comparing above two equations, we can write, `k = P xx Z xx e^(-E_(a)//RT)` Since P and Z are constant at constant temperature for the given reactants, we can write `P xx Z=A` where A is called frequency factor or `k = P xx Z xx e^(-E_(a)//RT)`. Thisis calledArrhenius equation. 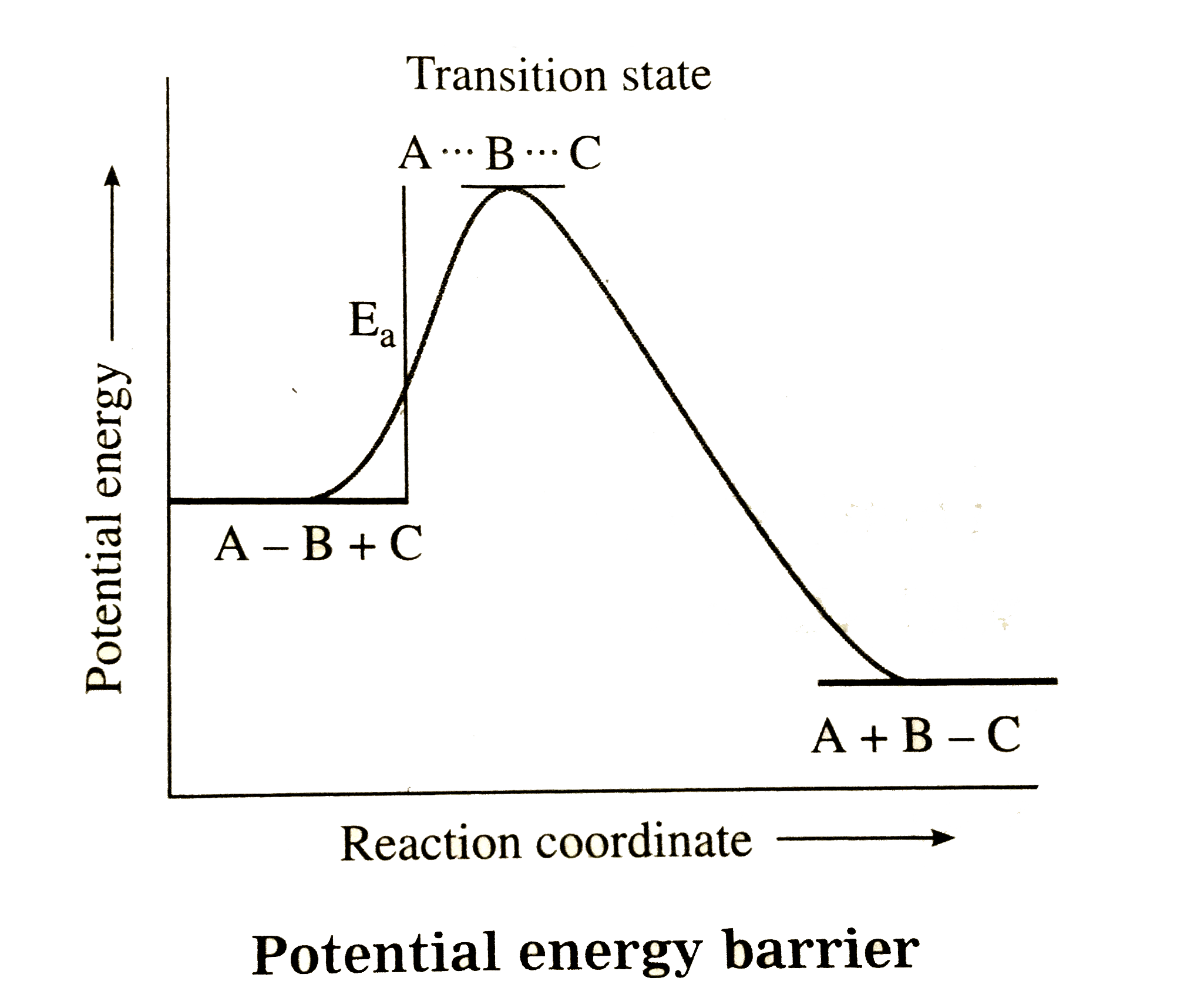
|
|
| 34. |
Obtain ethylamine (Ethanamine) from acetaldoxime. |
| Answer» Solution :`underset("ACETALDOXIME")(CH_(3)-CH)=N-OH+4[H]OVERSET(Na//C_(2)H_(5)OH)rarrunderset("ethylamine")(CH_(3)-CH_(2))-NH_(2)+H_(2)O` | |
| 35. |
Obtain a relation between standard cell potential ( or standard emf) and equilibrium constant of the cell reaction. |
|
Answer» Solution :For any galvanic cell, the overall cell reaction at equilibrium can be represented as, Reactants `HARR`Products. [For example for Daniell cell,`Zn_((S) +Cu_((aq))^(2)) hArr Zn_((aq))^(2+) +Cu_((s))`] The equilibrium constant is related to the standard free energychange `DeltaG^(@)` as , follows. `DeltaG^(@) = - RT` in K If `E_("cell")^(0)` is thestandard cell potential (or emf) of the galvanic cell, then `DeltaG^(@) = - nFE_("cell)^(@)` By comparing above equations, `DeltaG^(@) = - nFE_("cell")^(@) = - RT In k` `therefore E_("cell")^(@) = (RT)/(nF)` In K OR `E_("cell")^(0) = (2.303 RT)/(nF) log_(10)` K At `25^(@)C` `(2.303 xx RT)/(F) = (2.303 xx 8314 xx 298)/(96500) = 0.0592` `therefore E_("cell")^(0) = (0.0592)/(n) log_(10)K` This is a relation between equilibrium constant and `E_("cell")^(0)` |
|
| 36. |
Obtain a relation between radis of anatom and edgelength in the followingcrystalline solids. (1)Body - centred cubic crystal. (2) Face - centred cubiccrystal. |
|
Answer» Solution : (1) Body-centred cubic (BCC) structure : In this unit cell, 8 atomsare presentat 8 corners and ONE additional atom is presentat the body center. The atmosare in contactalongthebodydiagonal BF. Let a be theedgelengthand rthe radiusof an atoms. Consider a triangleBCE. `BE^(2) = BC^(2) + CE^(2) = a^(2) +a^(2) =2a^(2)` Consider triangleBEF. `BF^(2)=BE^(2) + EF^(2) = 2a^(2) + a^(2) =3a^(2)` `thereforeBF = sqrt(3)a` From figure , BF = 4r . `therefore 4r = sqrt(3)a` `thereforer = (sqrt(3))/(4)a` 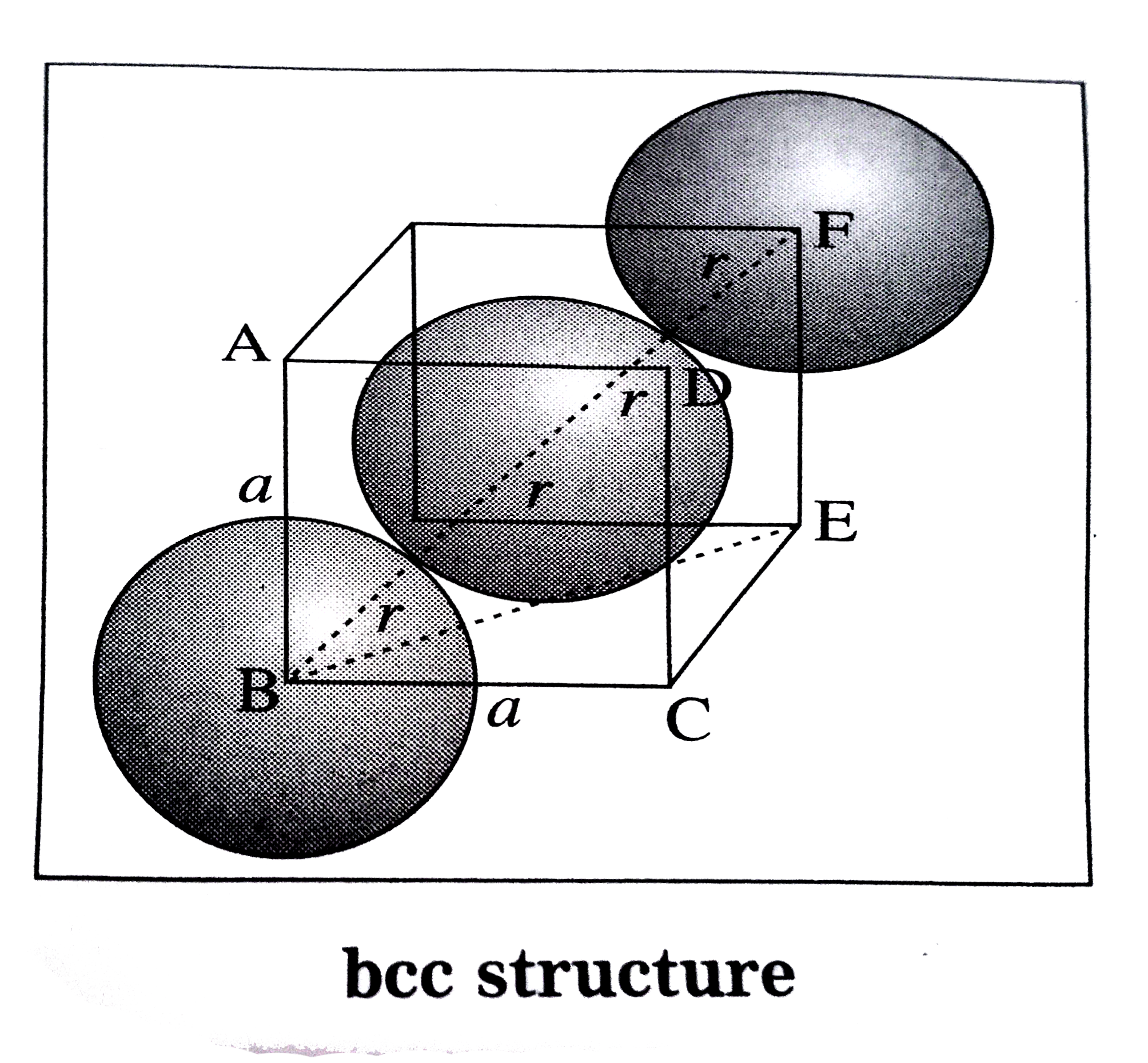 (2) Face-centred cubic (FCC) structure : In the unit cell,there are 8 atomsat 8 coners and 6 atoms at 6 face centre . Theatoms are in contantalongthe facedigaonal BD. Let a be theedgelength and r, theradiusof an atoms. Considera triangleBCD. `BD^(2) = BC^(2) +CD^(2)` `=a^(2) +a^(2) = 2a^(2)` `therefore""BD = sqrt(2a)` From figureBD = 4r `therefore""4r = sqrt(2)a` `therefore ""r=(sqrt(2))/(4)a = (a)/(2sqrt(2))` `r = (a)/(2sqrt(2))` 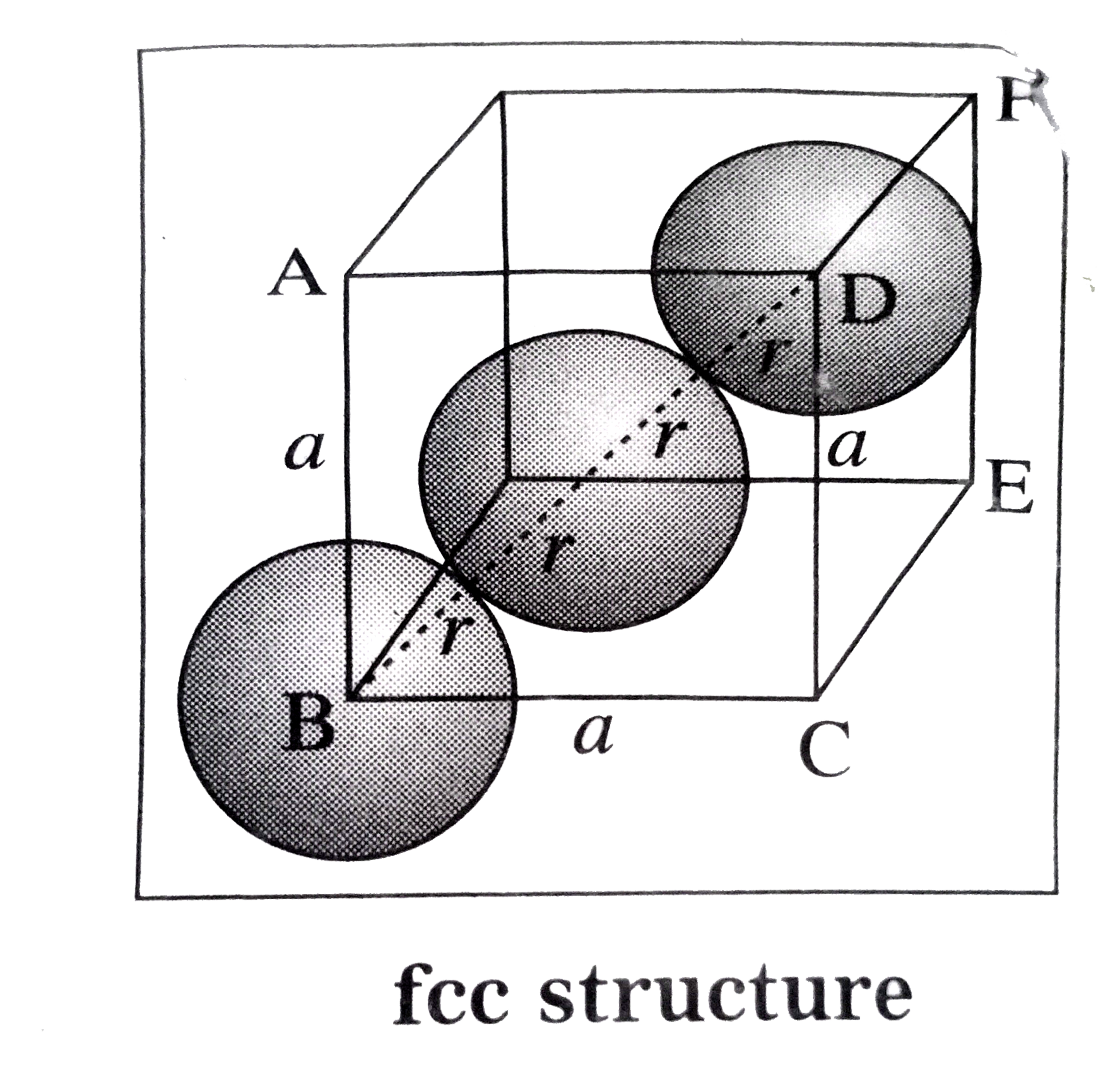
|
|
| 37. |
Obtain a relation between conductivity (K) and molarconductivity (^^_(m)). |
|
Answer» Solution : CONDUCTIVITY or specific conductance (K) is the conductanceof 1 `cm^(3)`of the solution in C.G.S. units, while molar conductivity is theconductance of a solution containing one MOLE of an electrolyte. Consider C molar solution i.,E. C moles of an electrolytepresentin 1 literor `1000 cm^(3)`ofthe solution . `because`C moles of an electrolyte are present in `1000cm^(3)` solution. `therefore` 1 MOLEOF an electrolyteis presentin`(1000)/(c) cm^(3)`solution . Now, `because`Conductance of `1 cm^(3)`of thissolution is k . `therefore` Conductanceof `(1000)/(c) cm^(3)` of thesesolutionis `(k XX 1000)/(C)` Thisrepresentmolarconductivty `^^_(m)` `therefore ^^_(m) = (k xx 100)/(C) Omega^(-1) cm^(2)mol^(-1)` ( in C.G.Sunits). |
|
| 38. |
Observer the following road map of reactions and give answer. The number of structural isomers of (S) formed is: |
|
Answer» 1 |
|
| 39. |
Observer the following road map of reactions and give answer. The number of geometrical isomers of (R) formed is : |
|
Answer» 1 |
|
| 40. |
Observer the following road map of reactions and give answer. The IUPAC name of (Q) is : |
|
Answer» cyclopropanetriketone 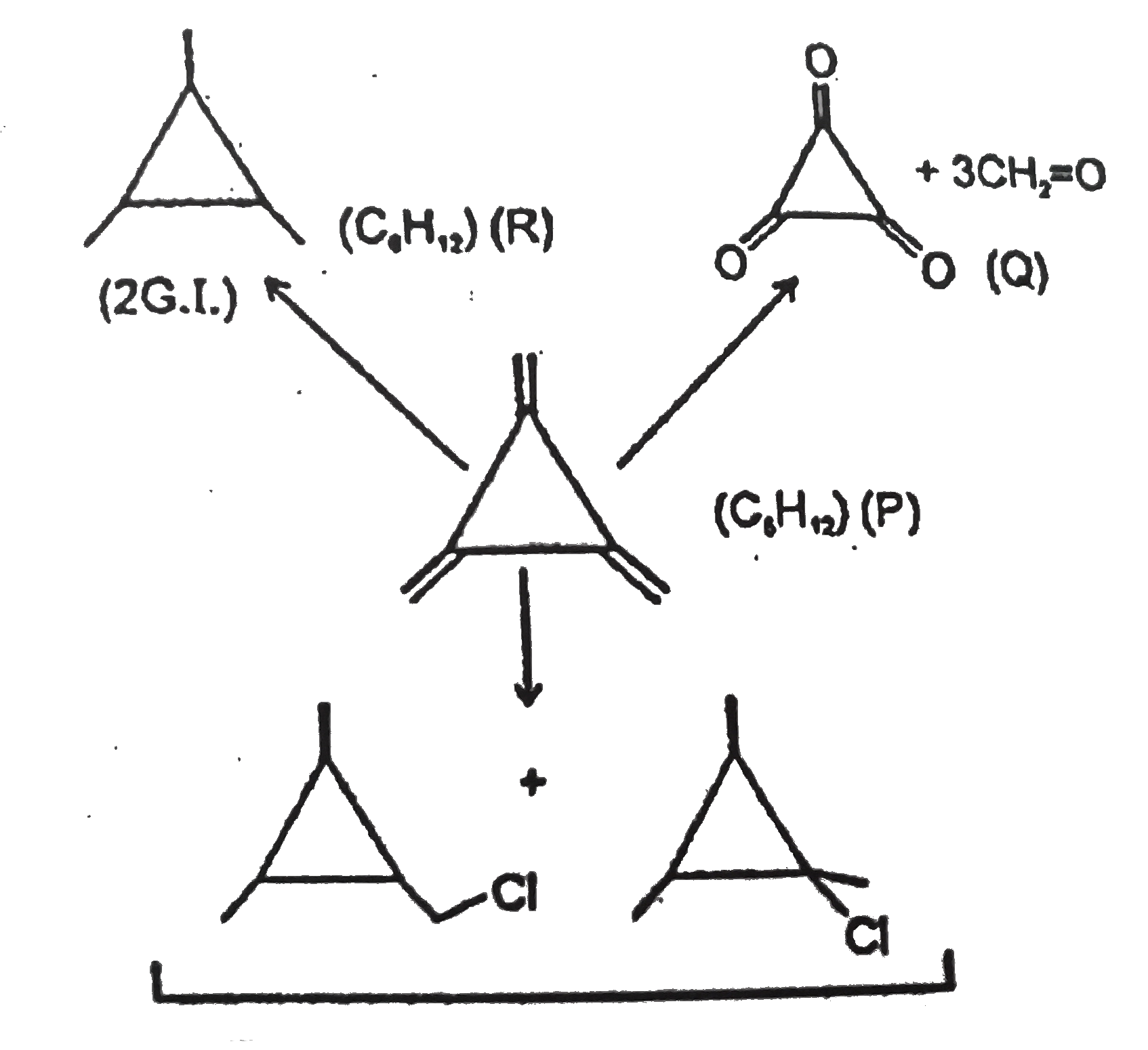
|
|
| 41. |
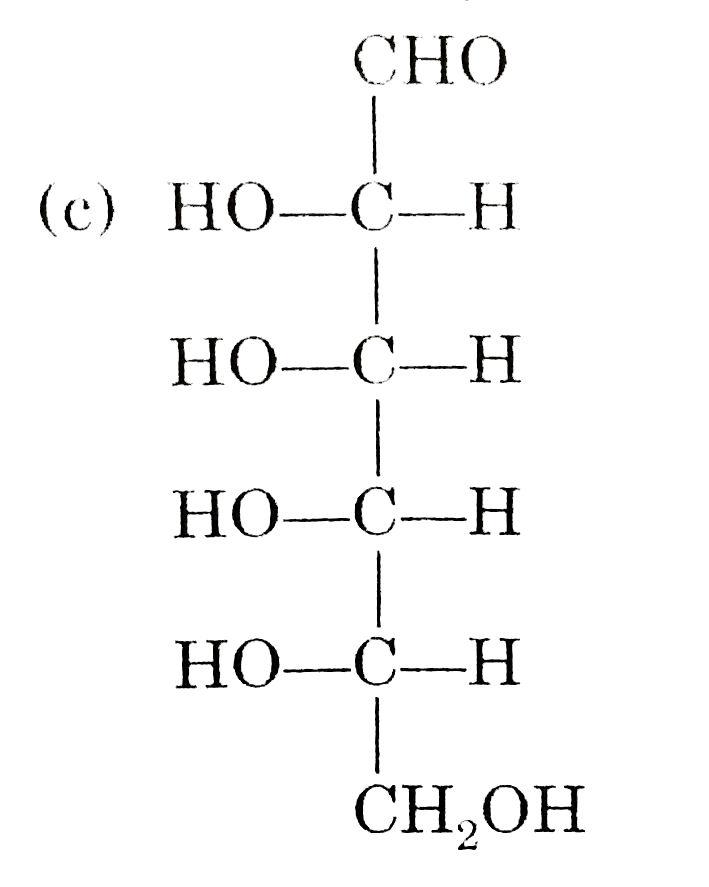
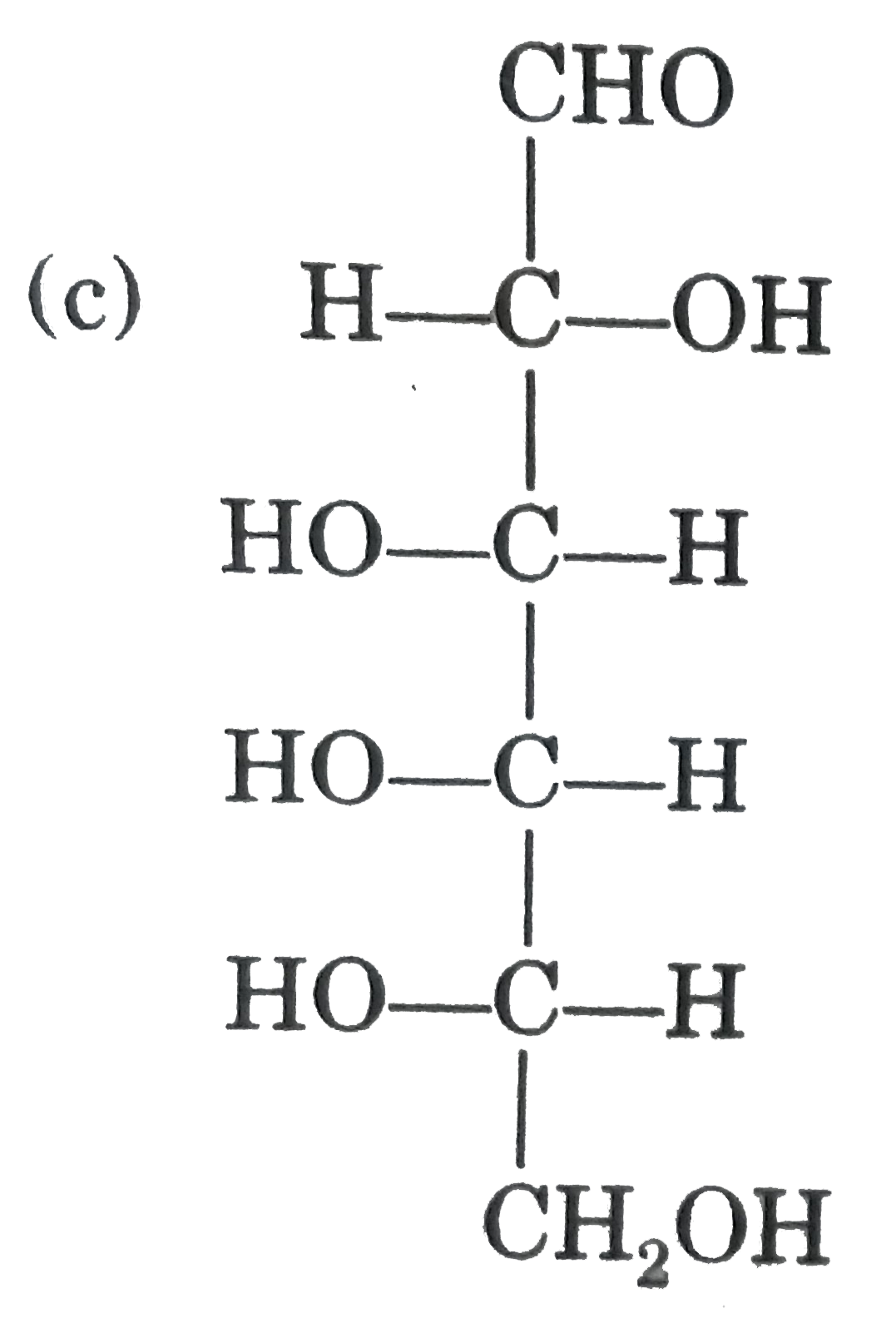
Observe these compounds and give answer of following question : Which of the following pair of compound gives same osazone product ? |
|
Answer» D(+) ALLOSE and D(-) gulose |
|
| 42. |
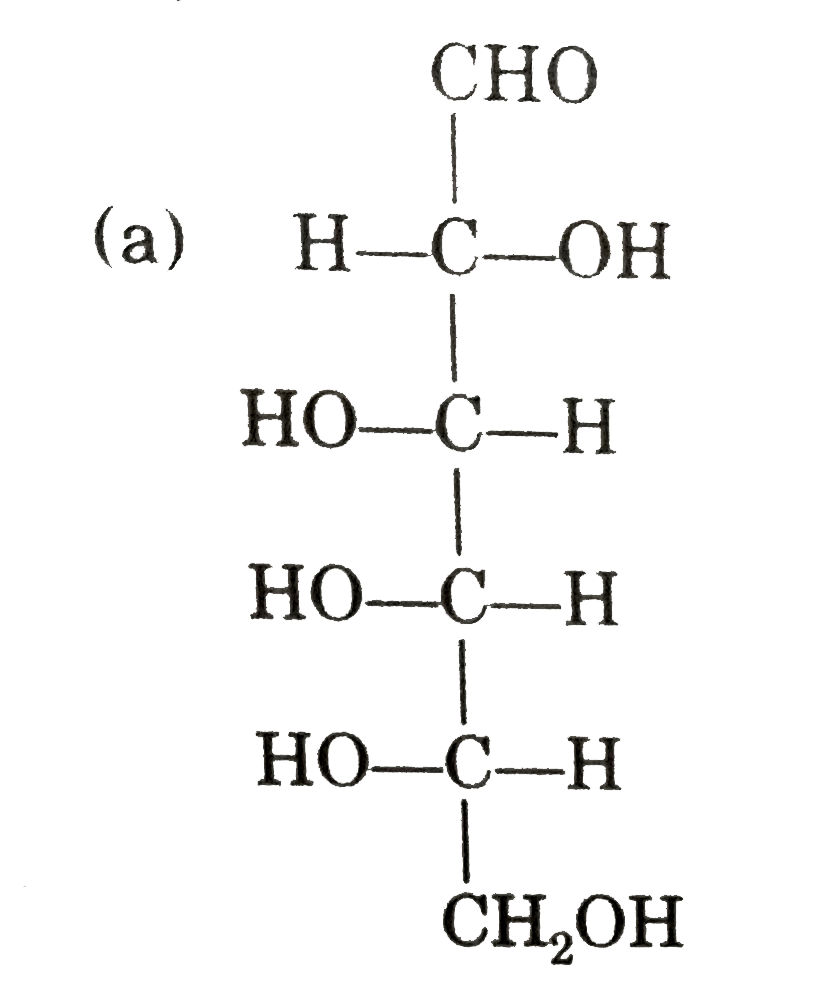
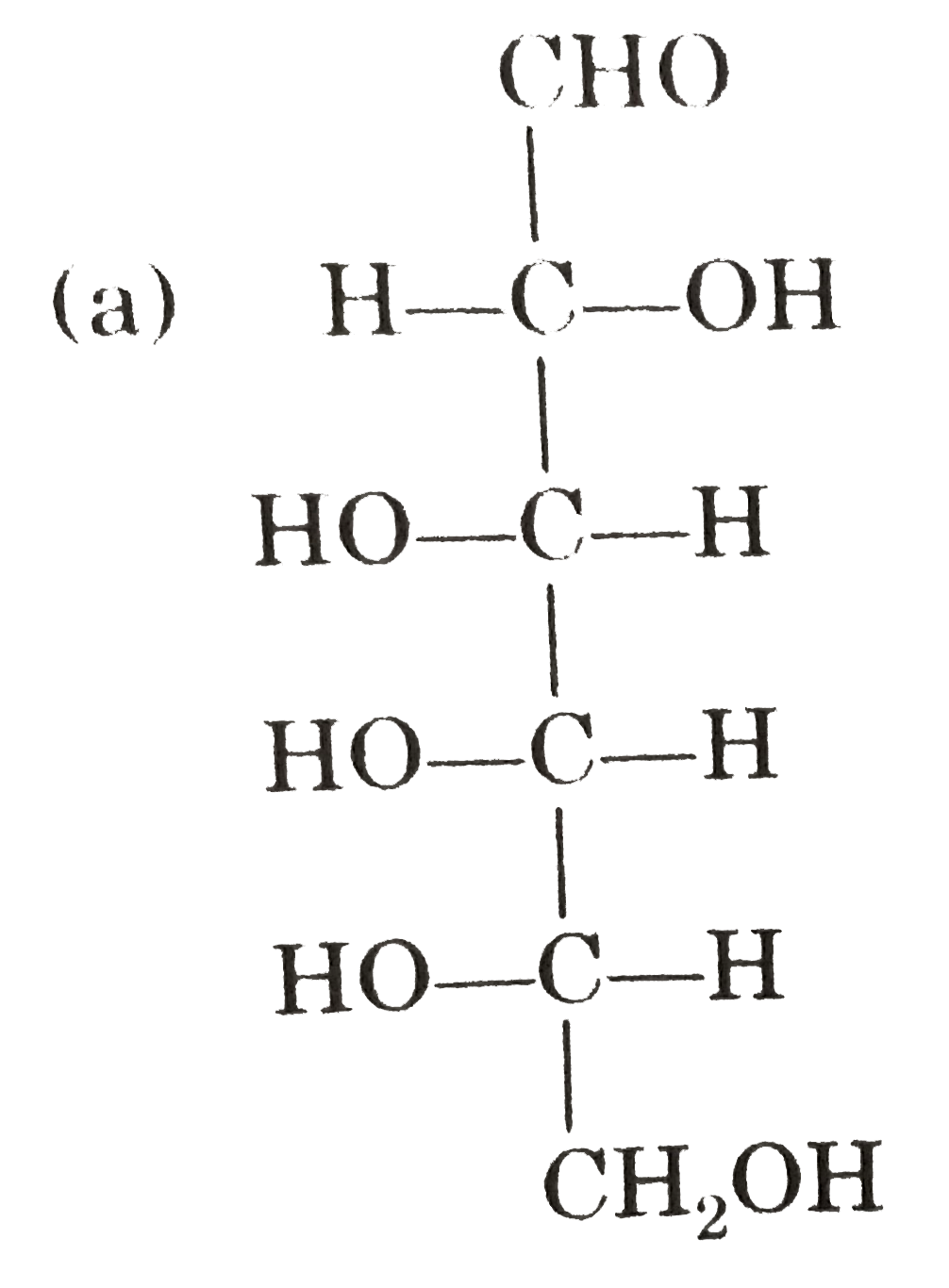
Observe these compounds and give answer of following question : Which of the following is correct presentation of L-galactose ? |
|
Answer»
|
|
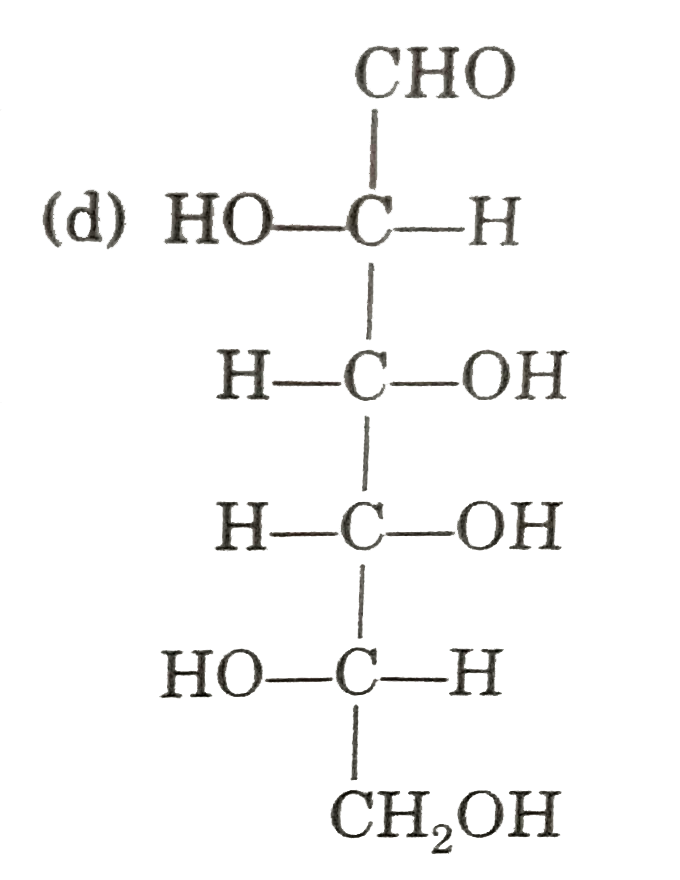
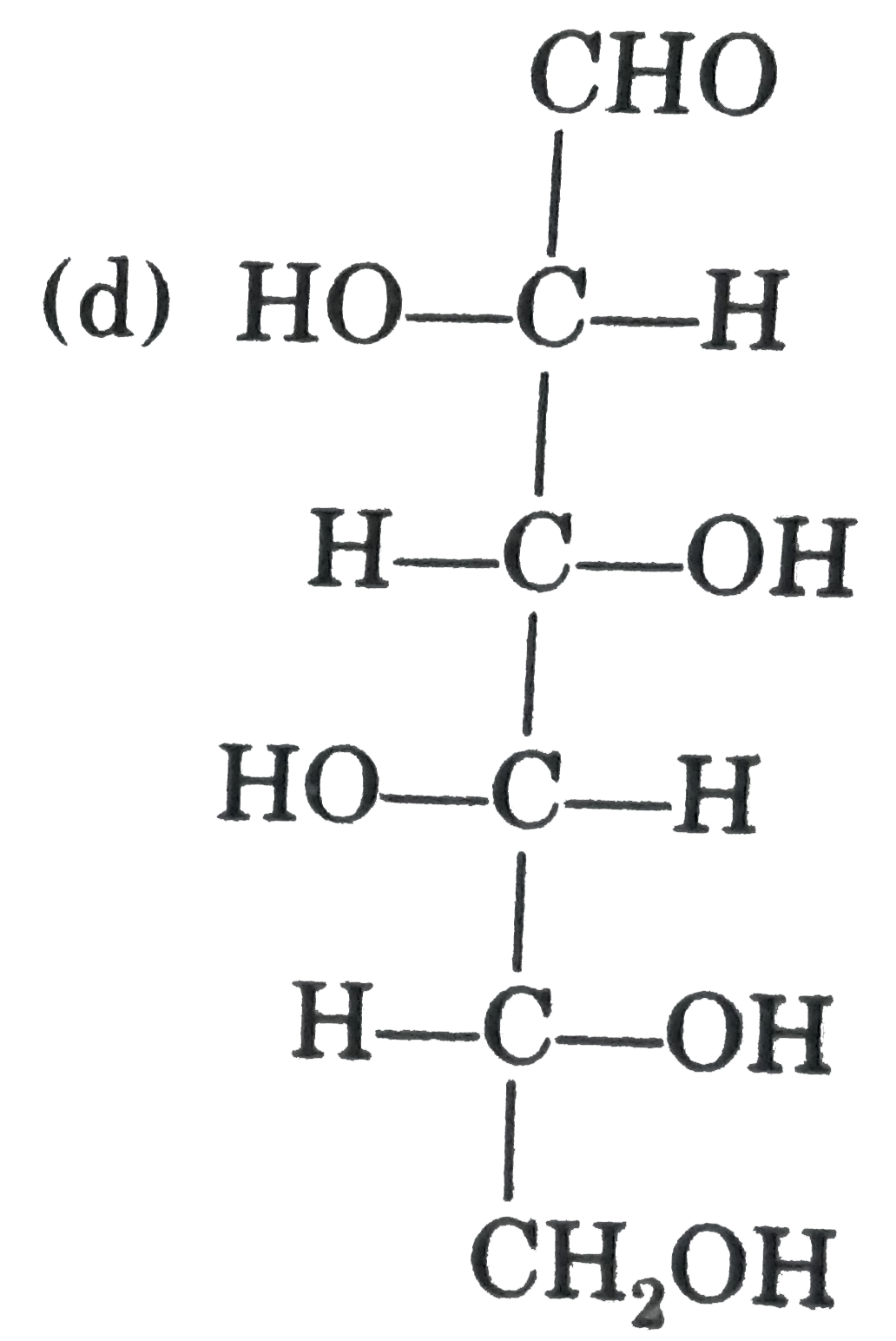
| 43. |
Observe these compounds and give answer of following question : In the given aldise which can form same osazone ? |
|
Answer» D-ribose |
|
| 44. |
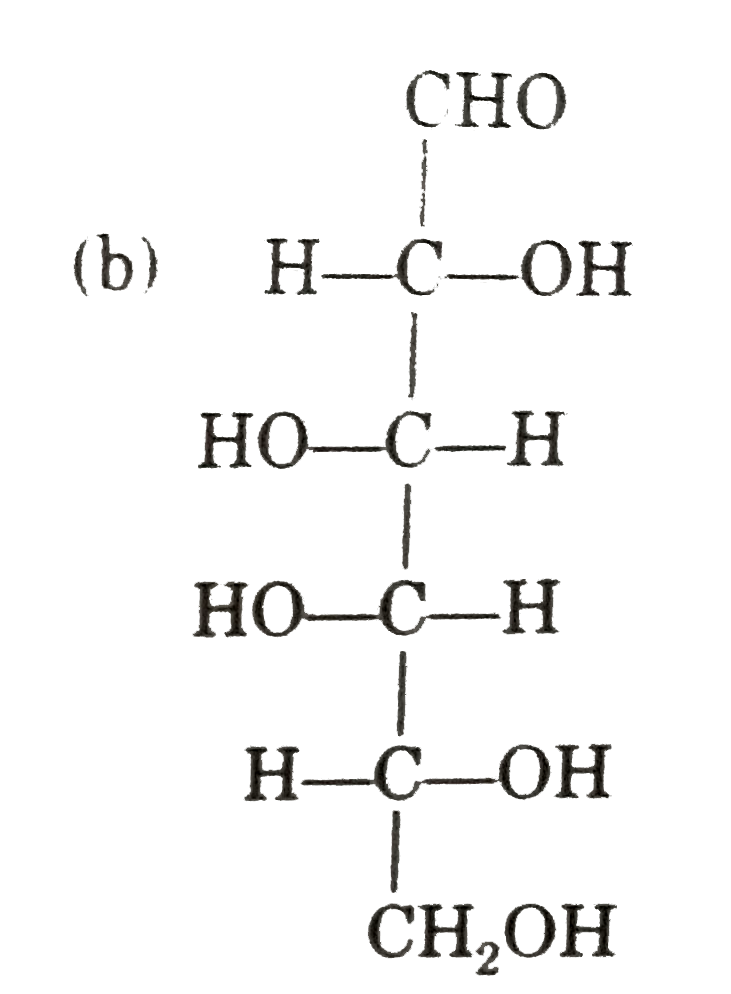
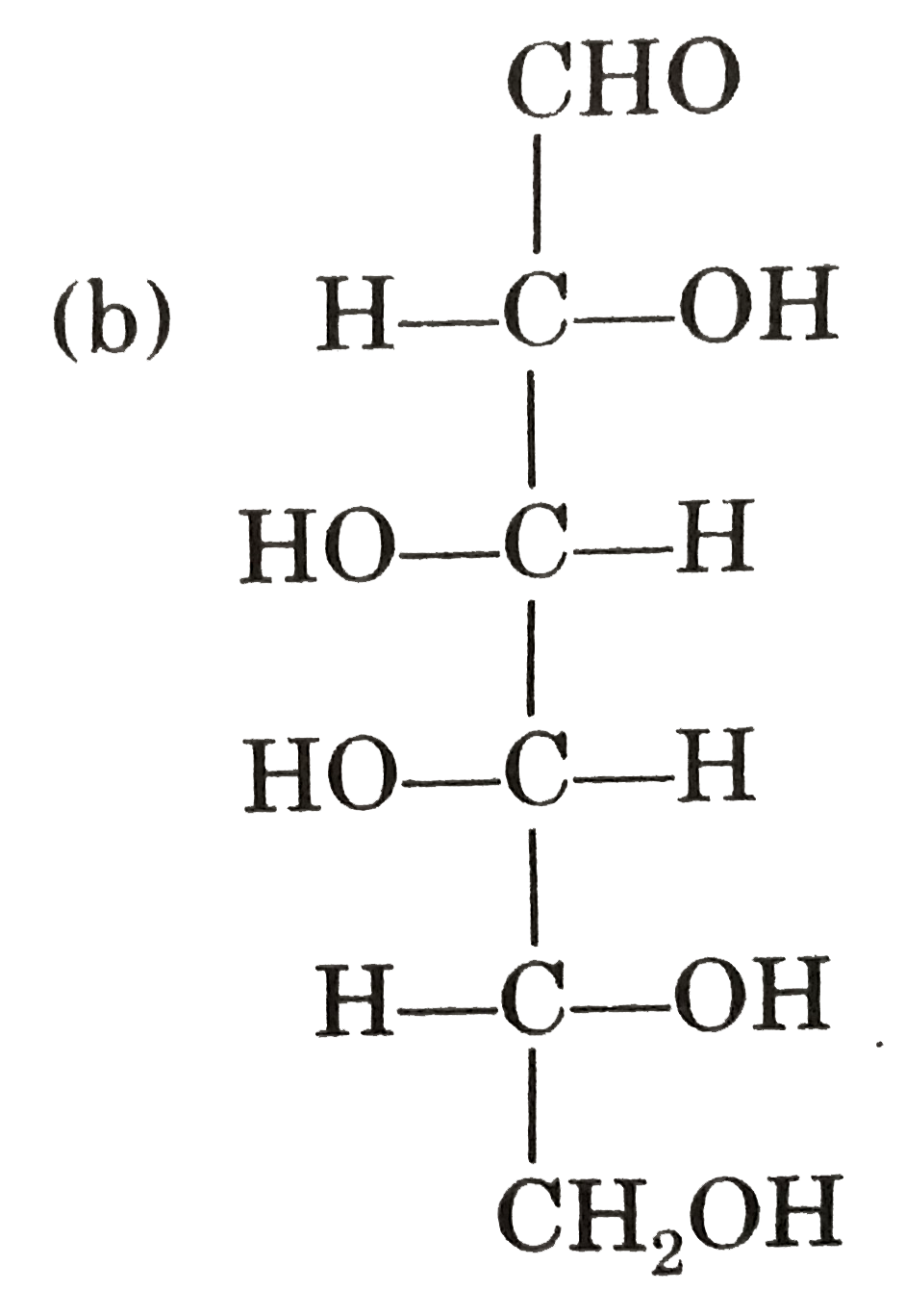
Observe these compounds and give answer of following question : Which of the following is correct presentation of L-glucose ? |
|
Answer»
|
|
| 45. |
Observe these compounds and give answer of following question : What is the relationship betweenC-5-epimer of D-tolose and C-2-epimer of D-taloseand C-2-epimer ofC-3-epimer of F-mannose |
|
Answer» enantiomer |
|
| 46. |
Observe these compounds and give answer of following question : In the given aldise which can form same osazone ? |
|
Answer» D-glucose |
|
| 47. |
Observe these compounds and give answer of following question : Identify the epimers from the following : |
|
Answer» D(+) ALLOSE and D(+) mannose |
|
| 48. |
Observe these compounds and give answer of following question : A carbohydrate undersetgoes the following conversion. D-arabinose underset(HCl)overset(NaCN)rarr'A'overset(H_(2)//Pd-BaSO_(4))rarr'B' overset(H_(3)O^(o+))rarr'C' 'C' can be : |
|
Answer» D-glucose 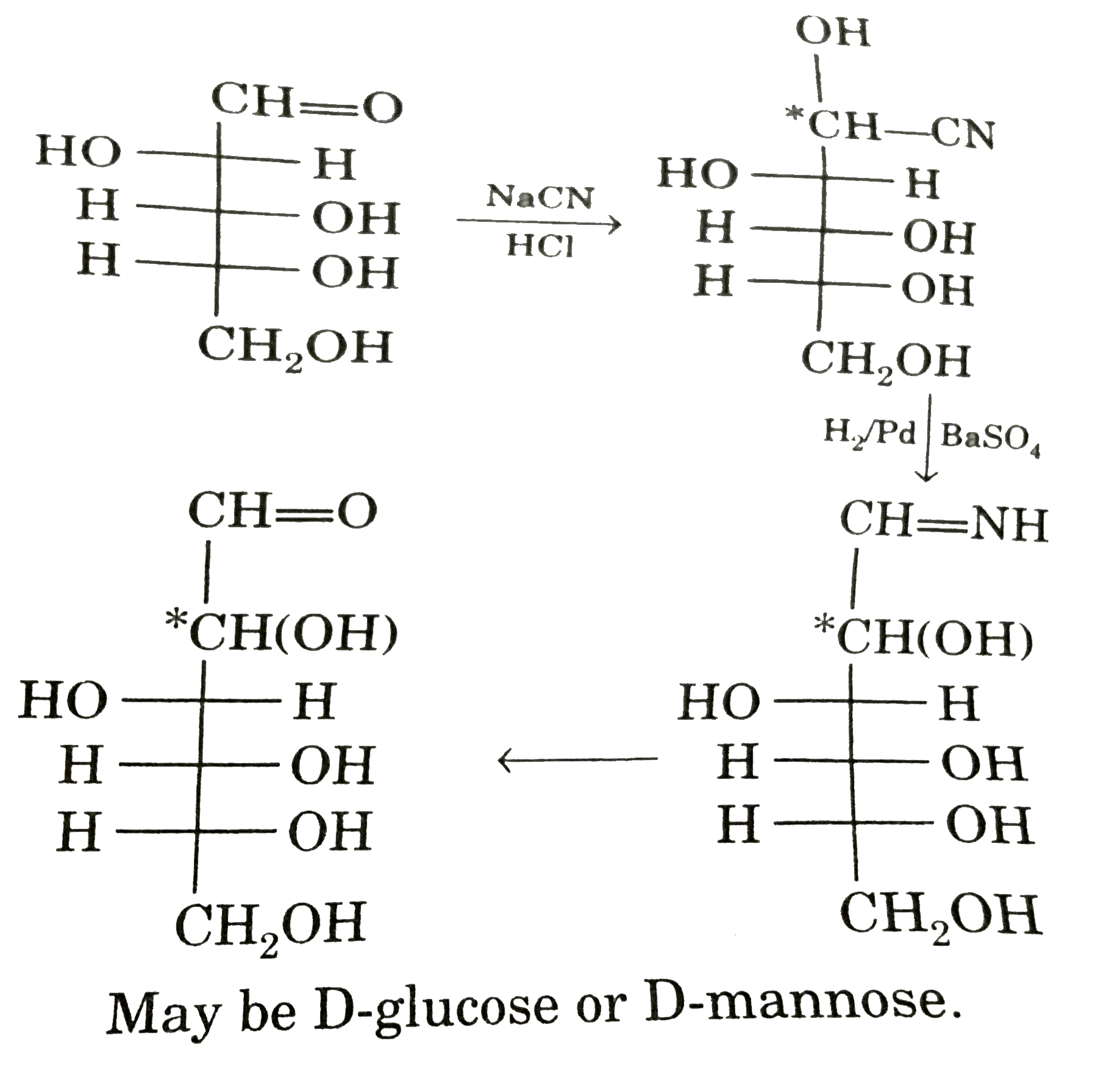
|
|
| 49. |
Observe these compounds and give answer of following question : A overset(Br_(2)//H_(2)O)rarrB(H_(2)O(2)//FeSO_(4)rarrD-Ribose |
|
Answer» D(+) allose |
|
| 50. |
Observe these compounds and give answer of following question : A carbohydrate undersetgoes the following conversion. D-Lyxoseunderset(HCl)overset(NaCN)rarr'A'overset(H_(2)//Pd-BaSO_(4))rarr'B' overset(H_(3)O(o+))rarr'C' 'C' can by : |
| Answer» Solution :D-glucose and L-gulcose areenantiomers. | |