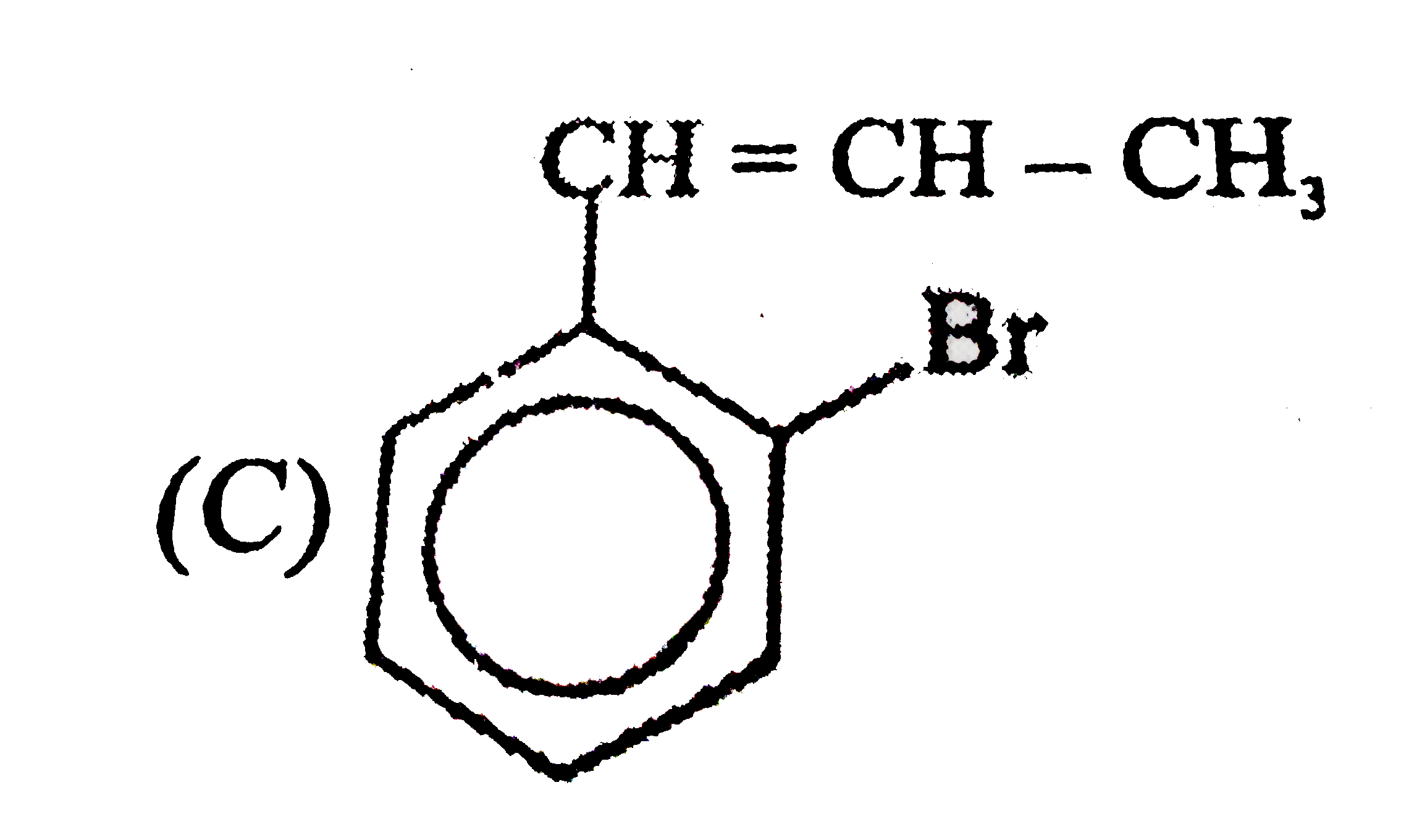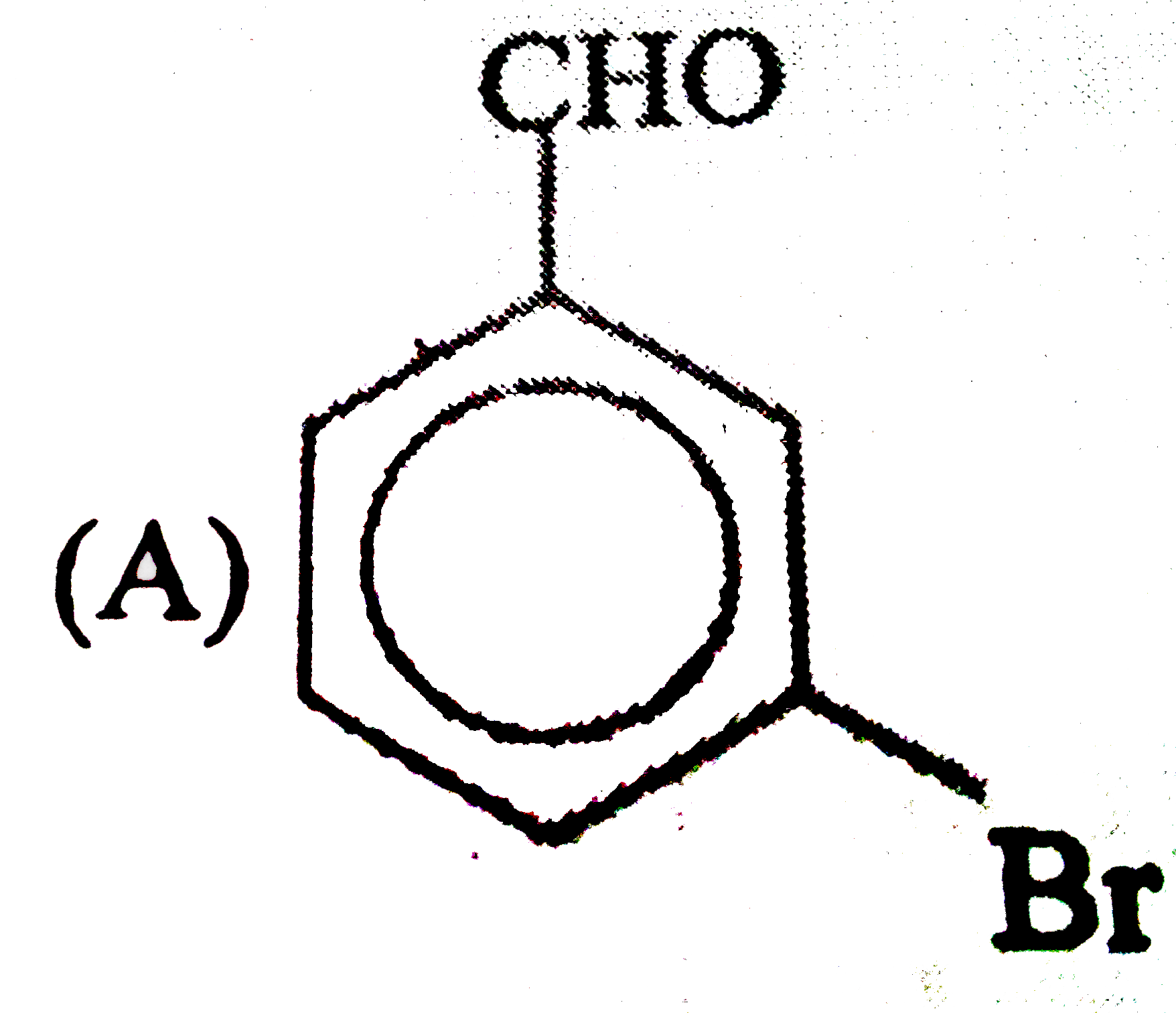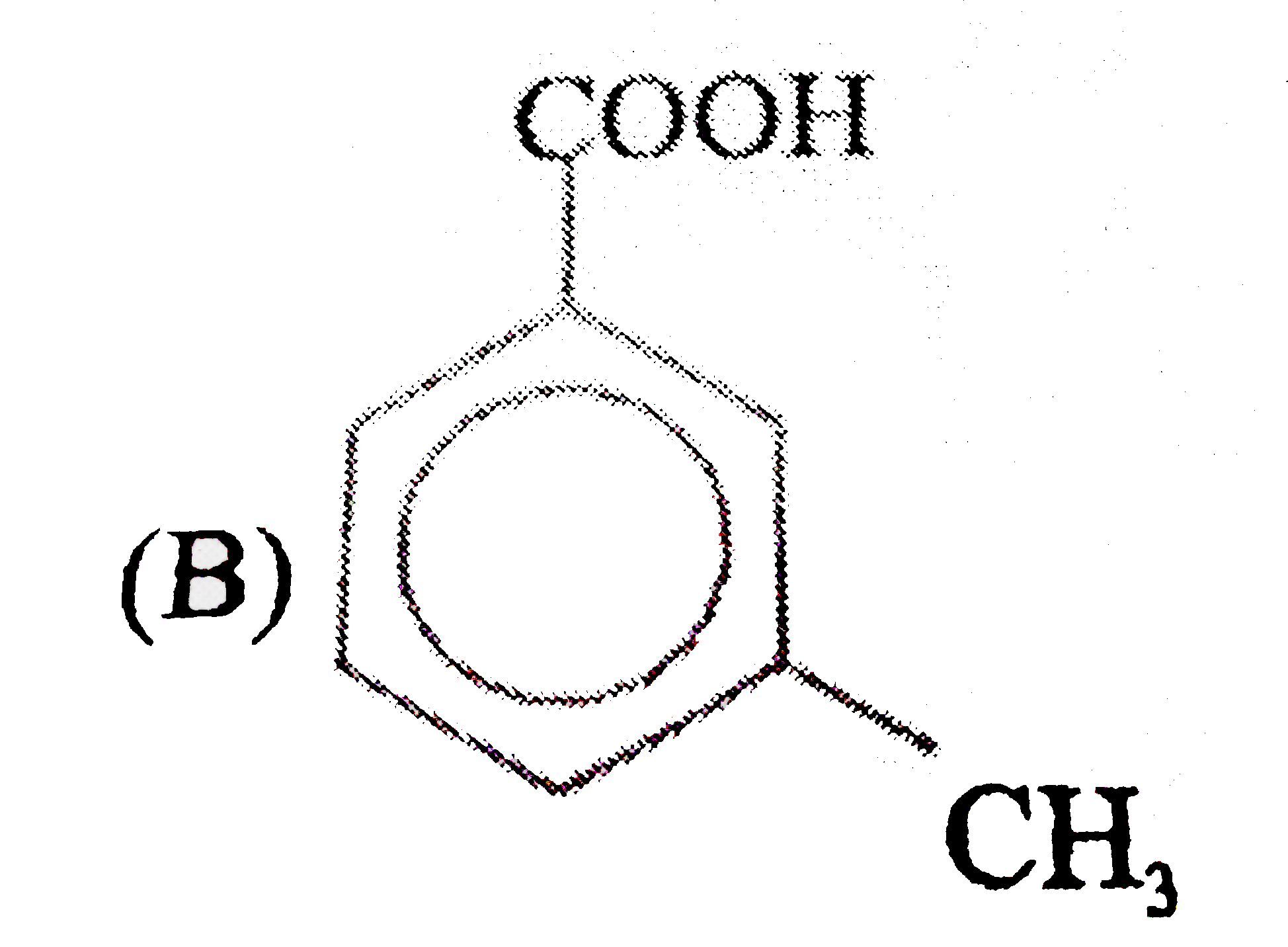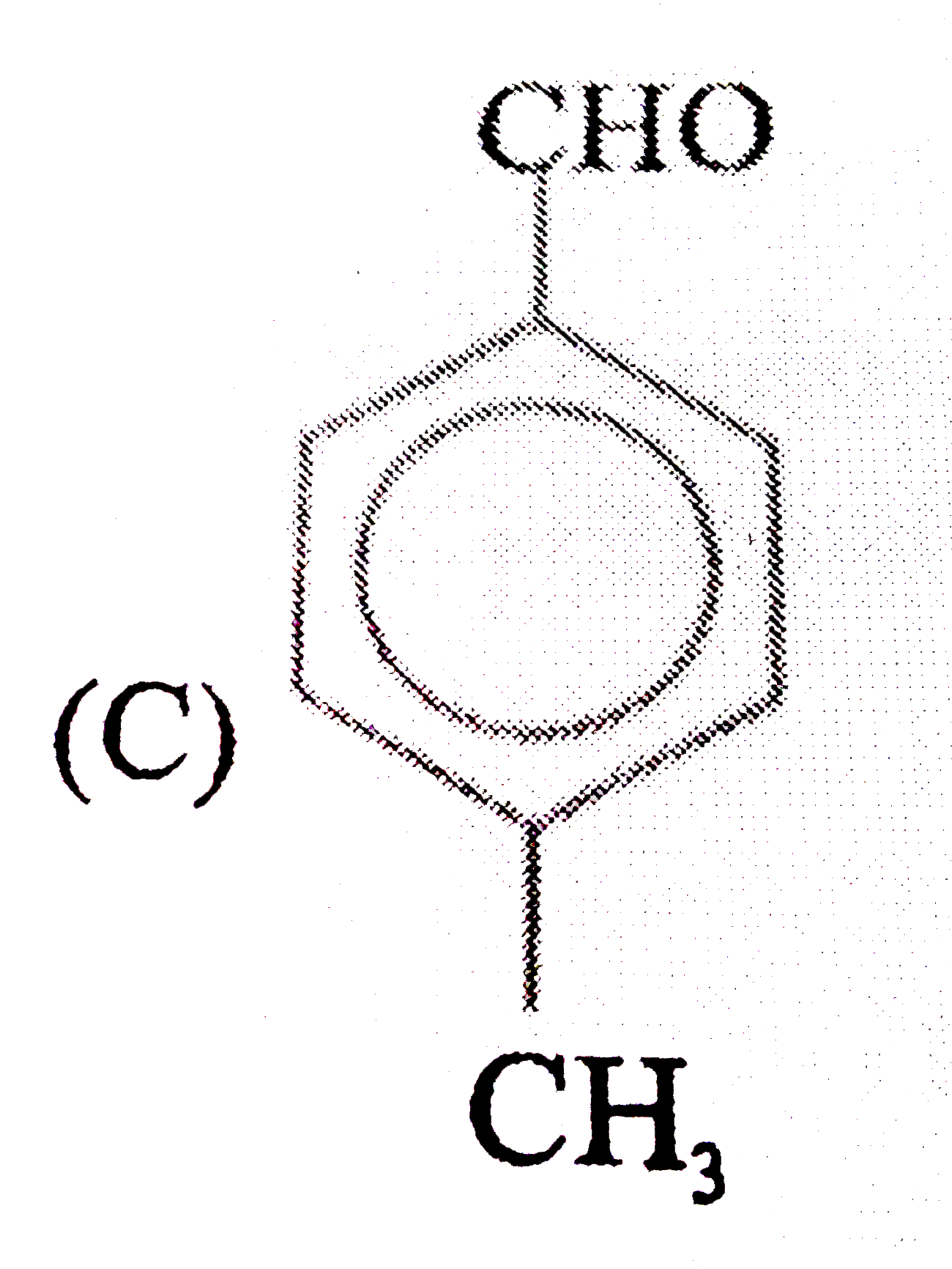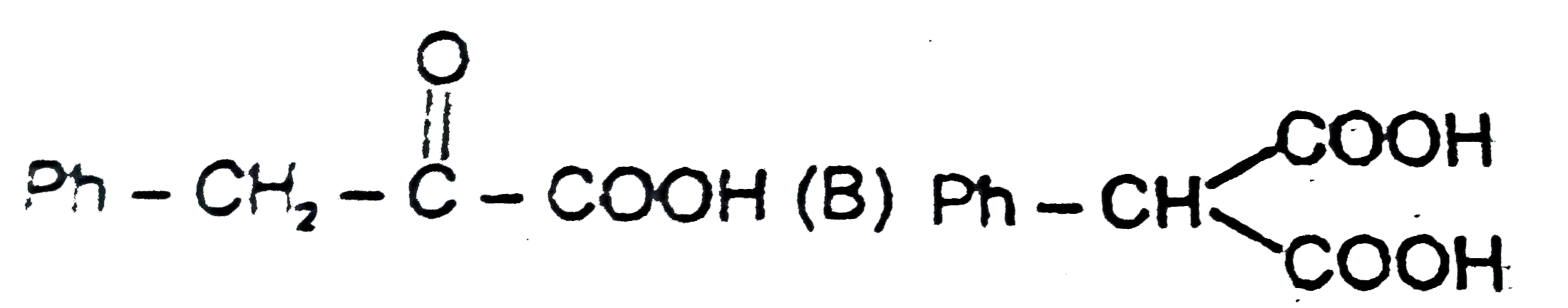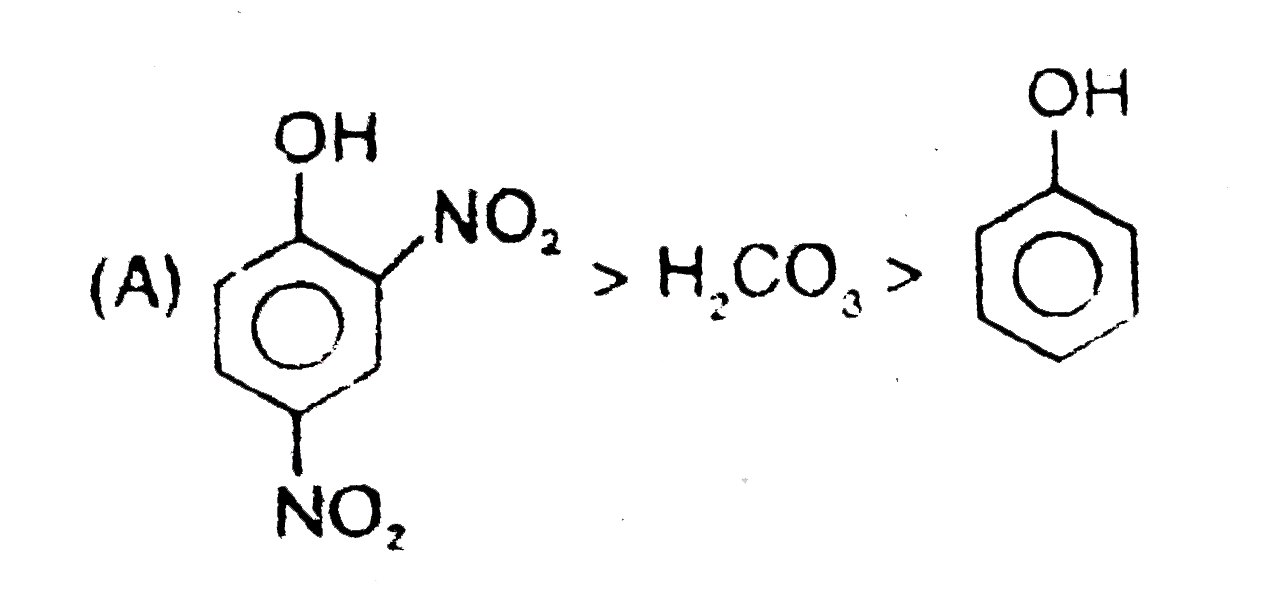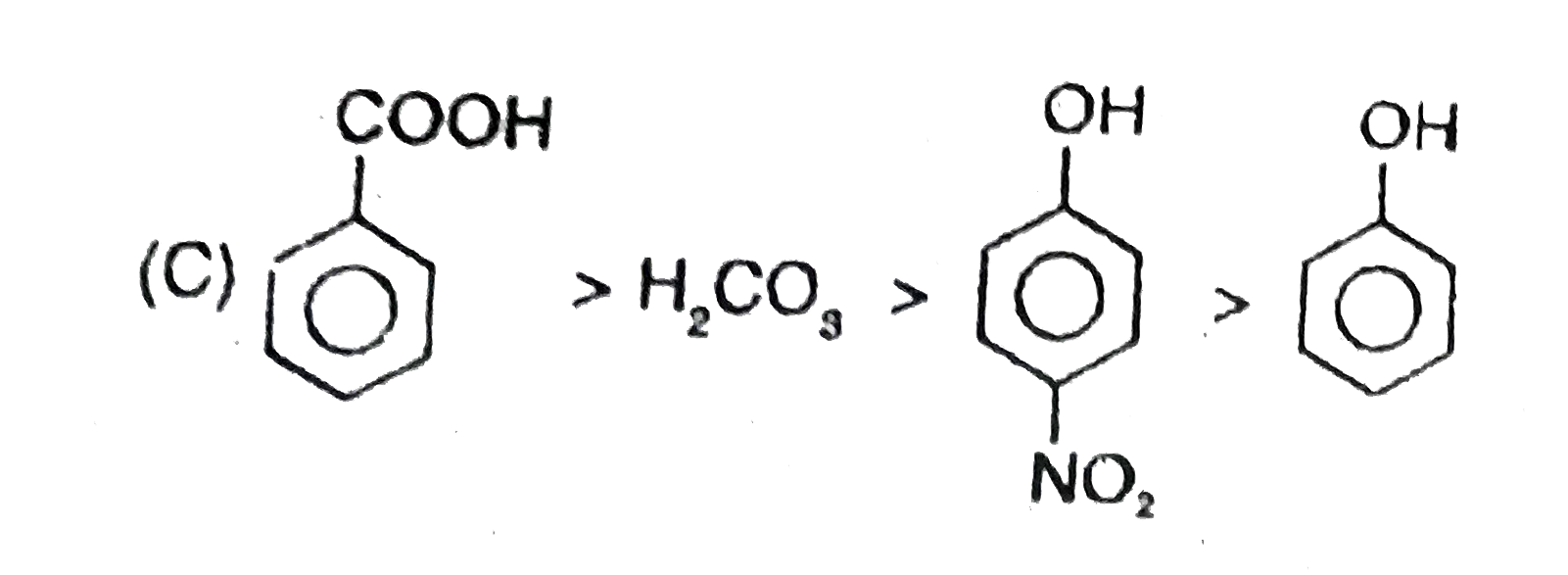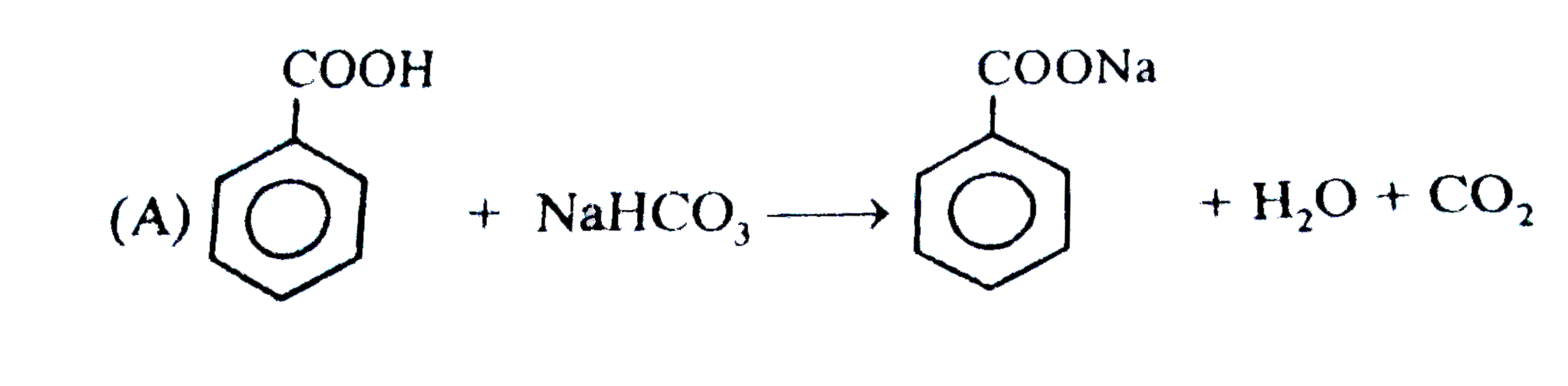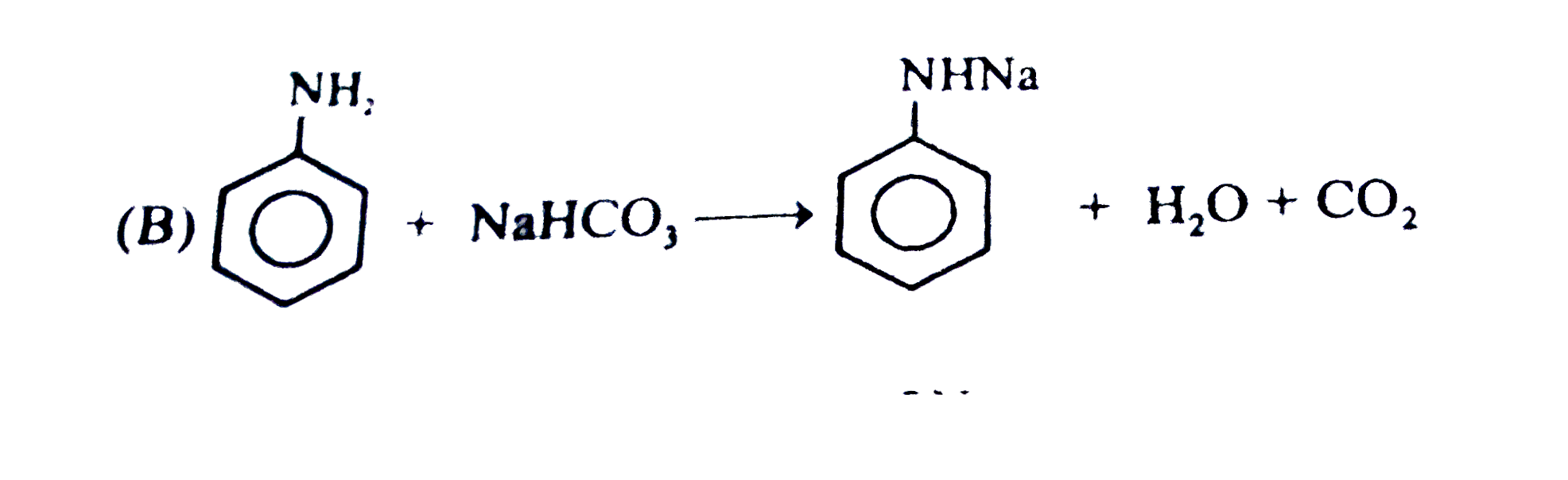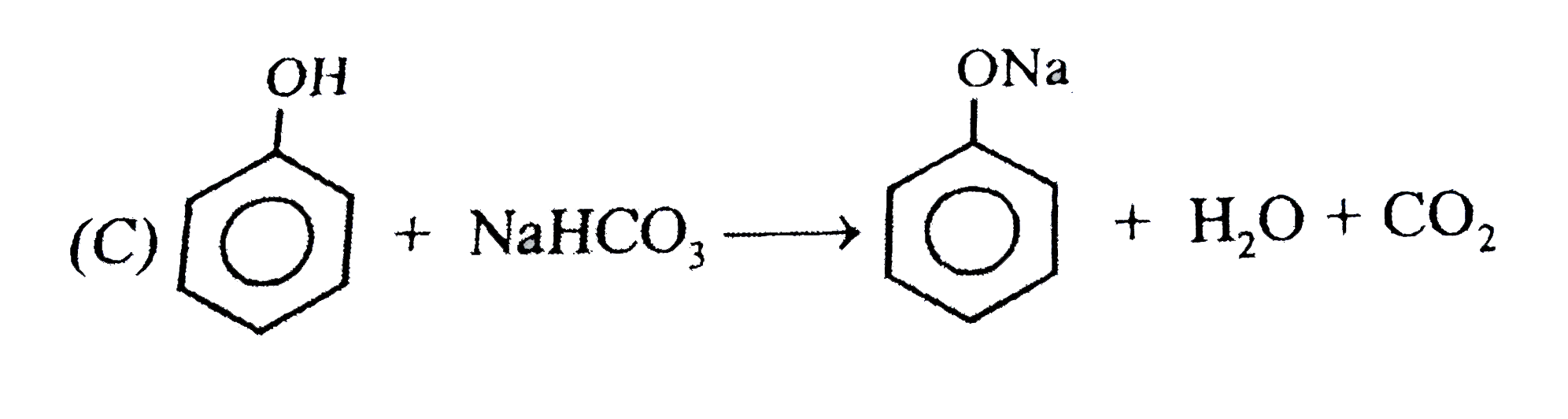Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Observe the following two reactions: (I). CH_(3)-underset(O)underset(||)C-CH+C_(2)H_(5)overset(18)Hoverset(H_(2)SO_(4))to"products"+"water" (II). (X)+"water"overset(("sodium hydroxide")/(Delta))toCH_(3)-underset(O)underset(||)(C)-overset(18)(O)Na+C_(2)H_(5)OH the correct statements are: |
|
Answer» In reaction(I) the PRODUCTS is `CH_(3)-underset(O)underset(||)C-overset(18)O-C_(2)H_(5)`  (ii). `CH_(3)-underset(O)underset(||)C-OC_(2)H_(5)+H_(2)overset(18)(C)` `underset("Basic medium")overset((Naoverset(18)(O)H)/(Delta))toCH_(3)-underset(O)underset(||)C-overset(18)(O)Na` `+C_(2)H_(5)OH` |
|
| 2. |
Observe the following statements? Bleaching powder is used in the preparation of Chloroform II. Bleaching powder decomposes in the presence of CoCl_(2) to liberate O_(2) III. Aqueoue KHF_(2) is used in the preparation of Fluorine. |
|
Answer» I, II and III are correct |
|
| 3. |
Observe the following statements (i) Silicon doped with P is a p-type semiconductor (ii) Presence of Schottky defects decreases the density (iii) Among simple cubic (sc), body centered cubic (bcc) and cubic close packing (ccp) stru ctu res, the p ack in g e fficie n cy is highest for ccp The correct statements are |
|
Answer» I,II and III |
|
| 4. |
Observe the following statements 1) Lanthanides actively participate in chemical reactions 2) The basic nature of hydroxides of lantha nides increases from La(OH)_3to Lu(OH)_3 3) Lanthanides do not form coordinate compounds as readily as d-block metals The correct statements are |
| Answer» Answer :C | |
| 5. |
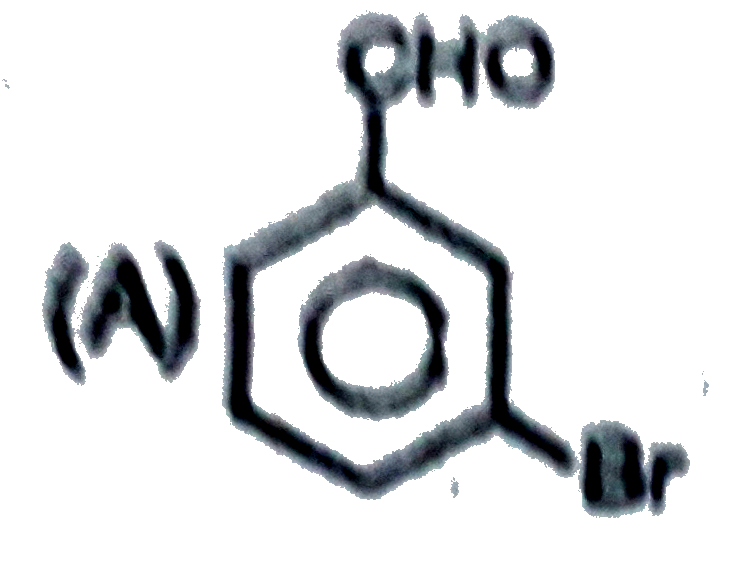
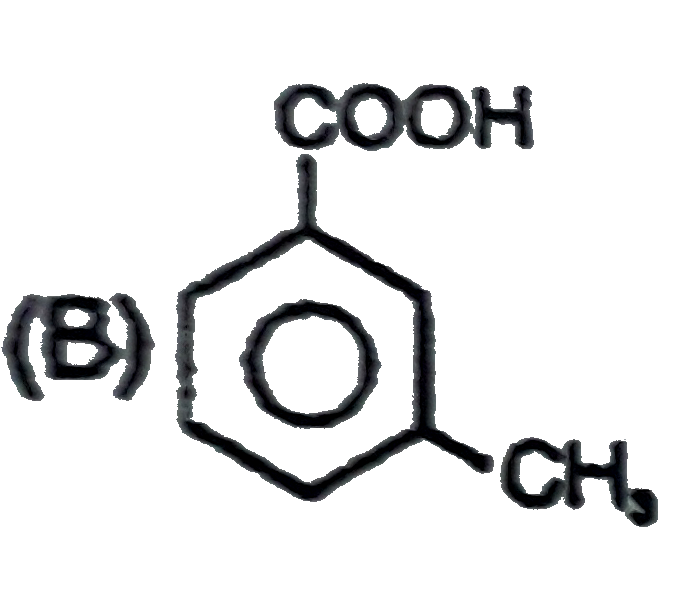
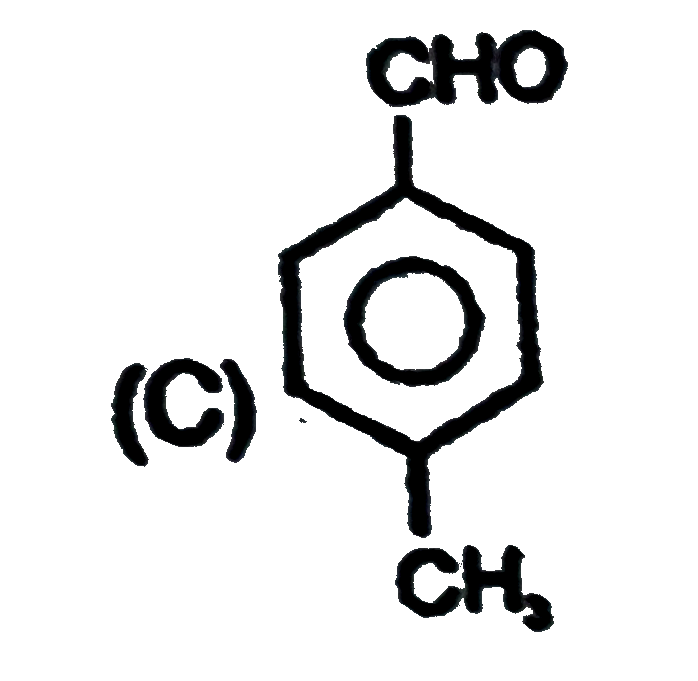
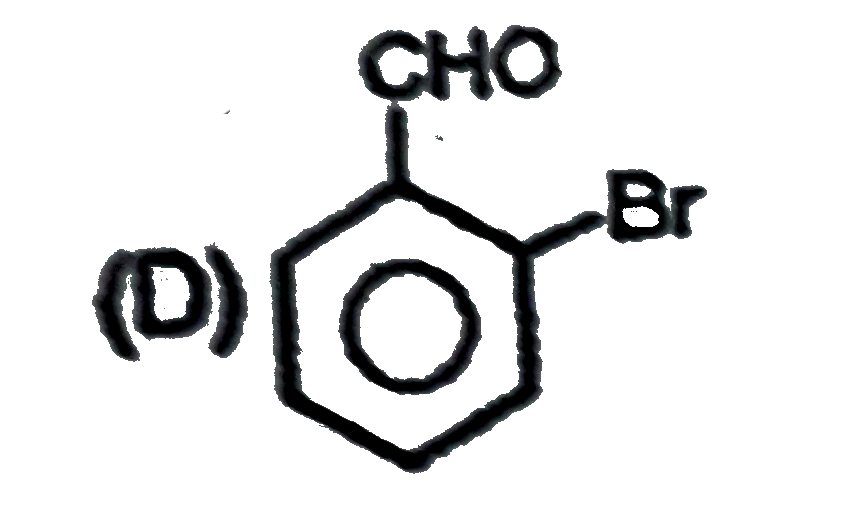
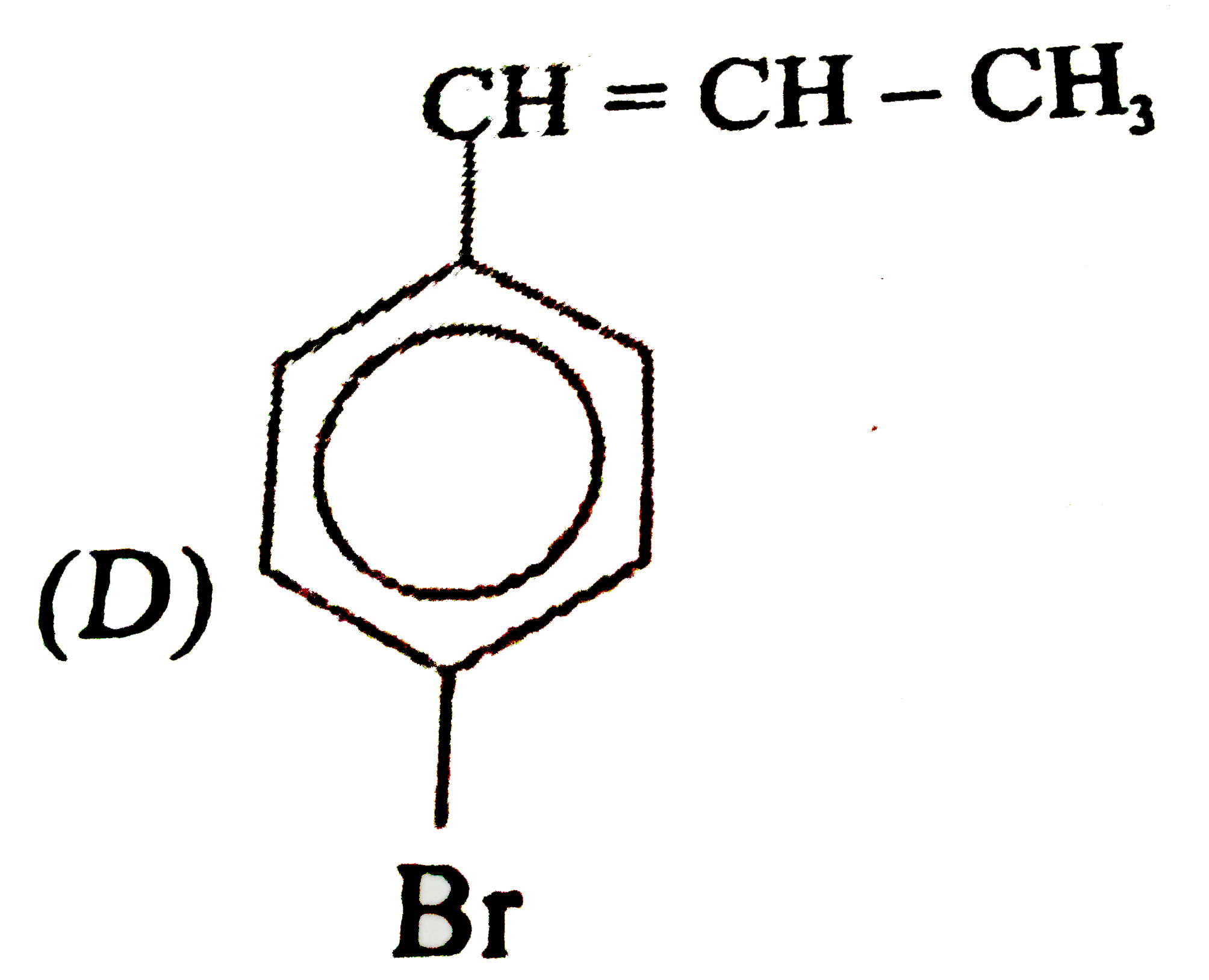
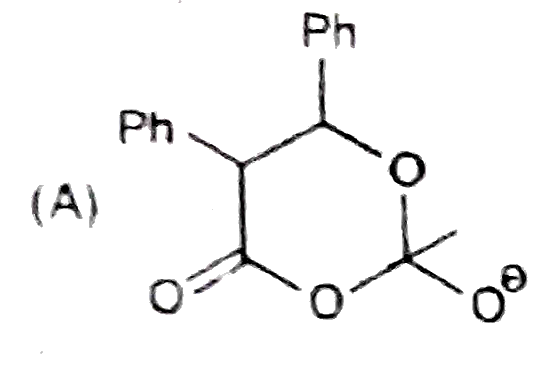
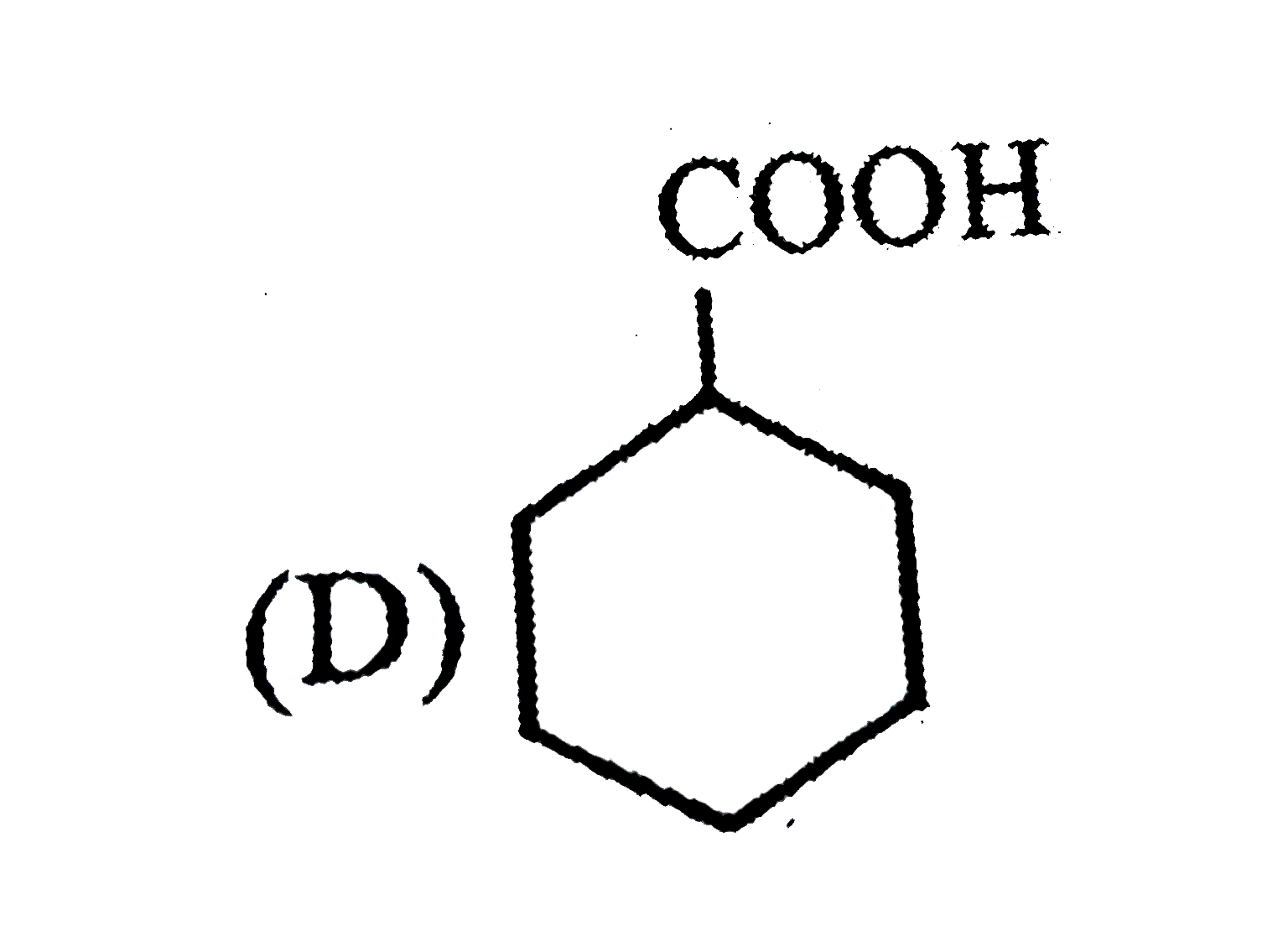
Observe the following sequence of reations P shows geometrical isomersm.Q gives positive Tollen's test and the oxidation product of Tollen's test followed by acidification is the strongest acid among its all position isomers. R gives positive lab tests with 2,4-DNP, Fehling solution and I_2//NaOH reagents. Identify the structure of R. |
|
Answer» HCOOH 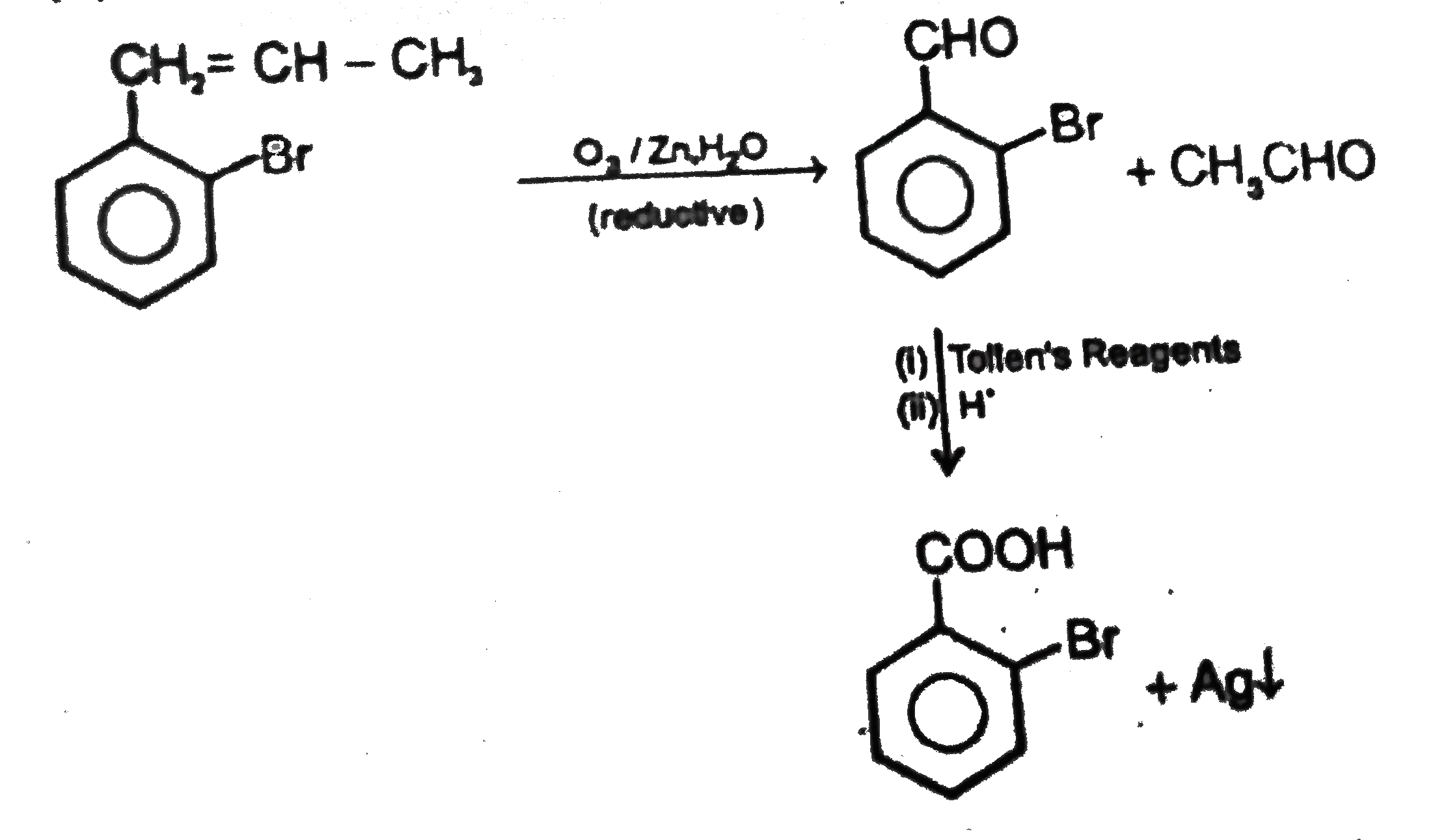
|
|
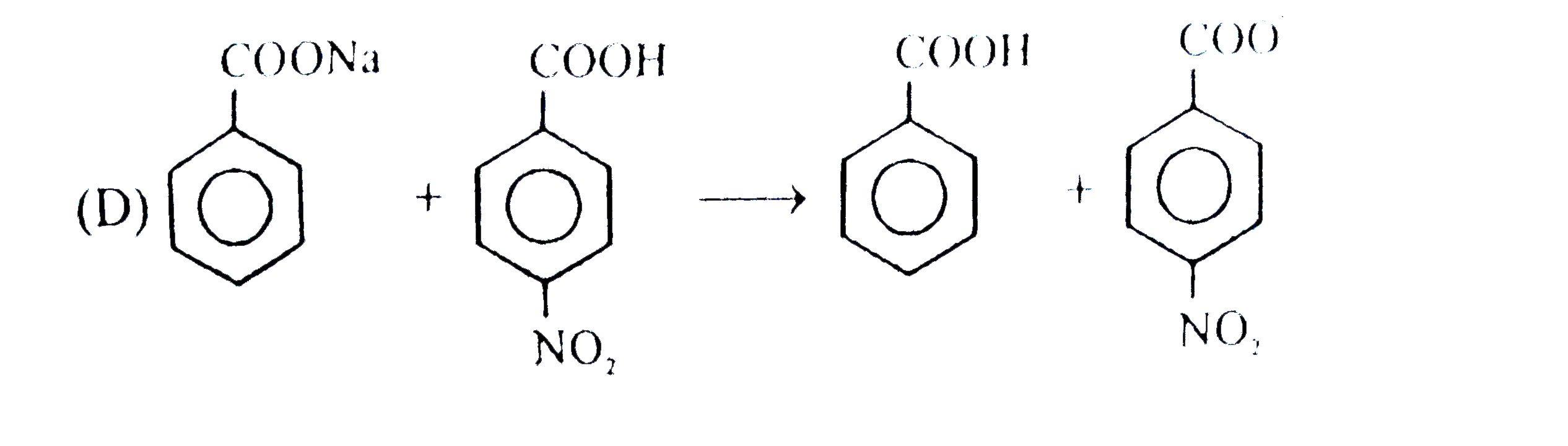
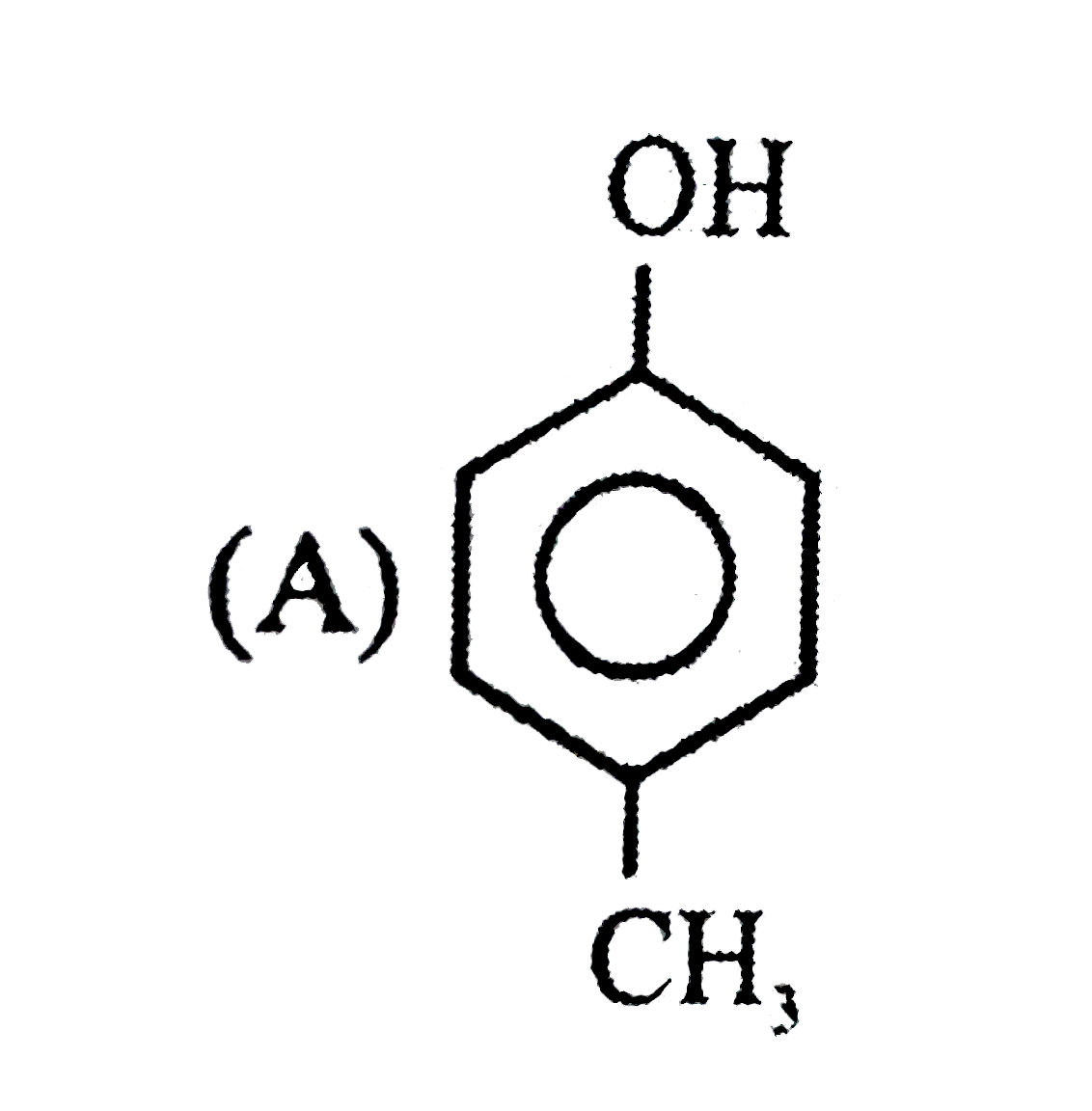
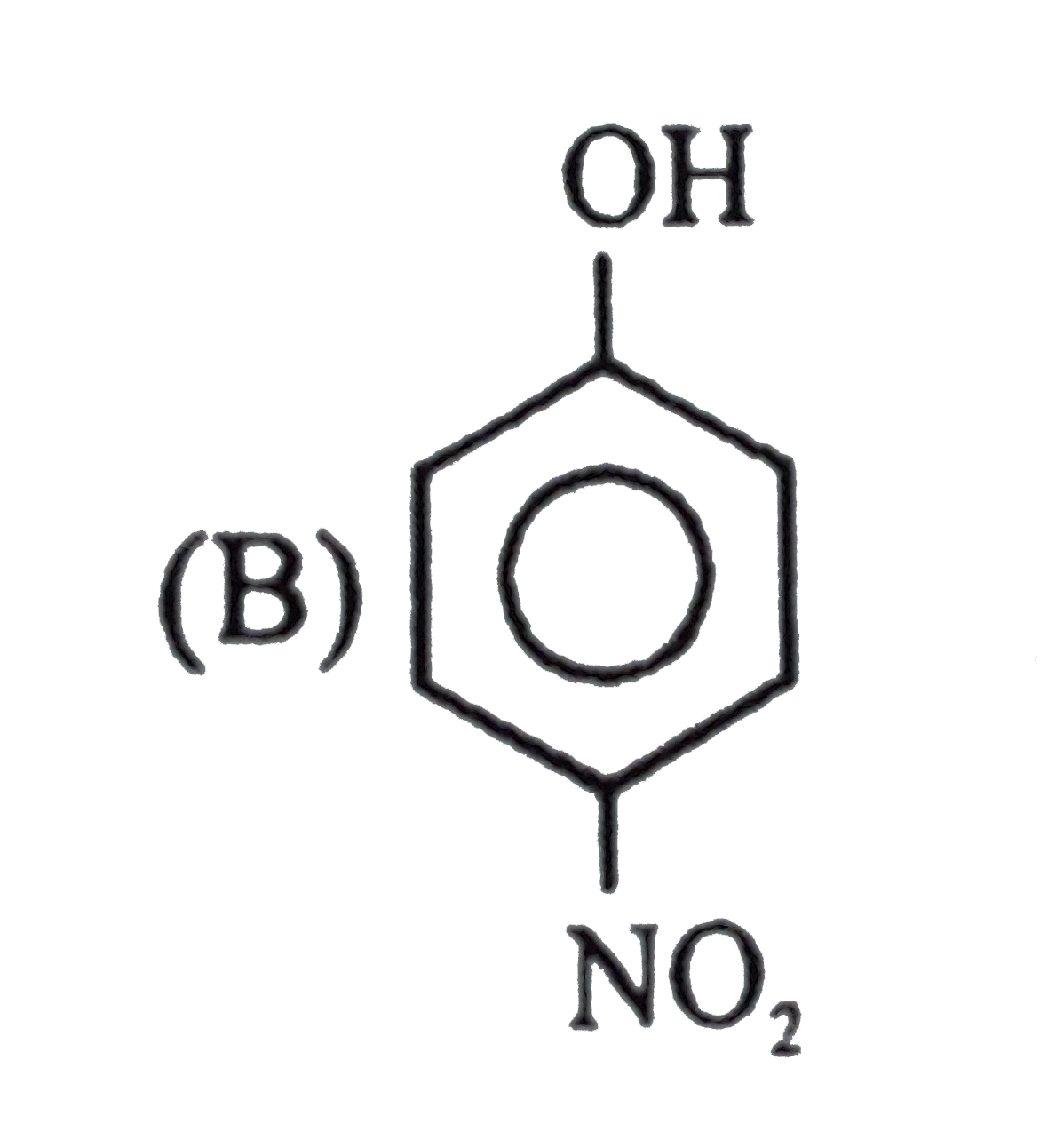
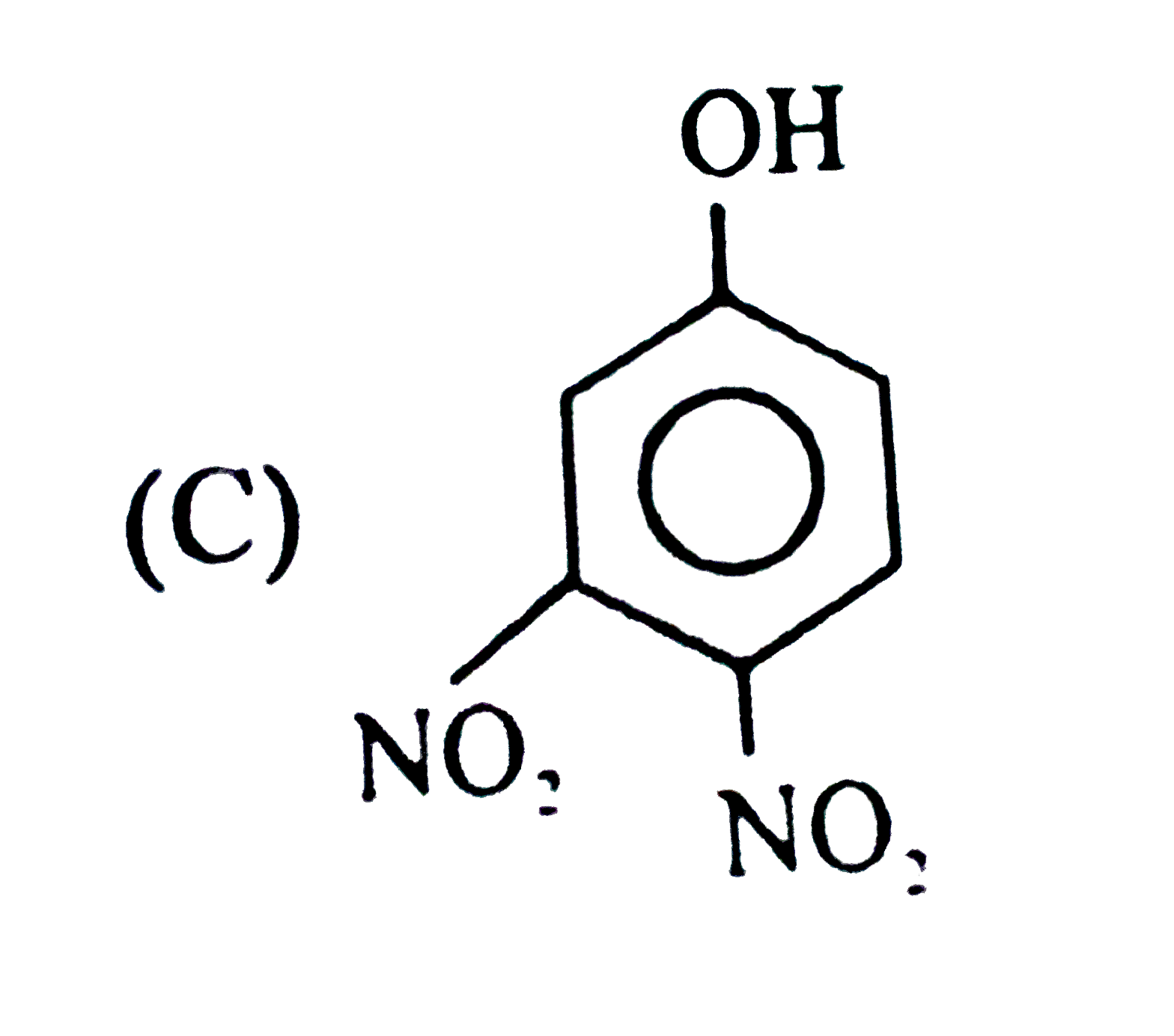
| 6. |
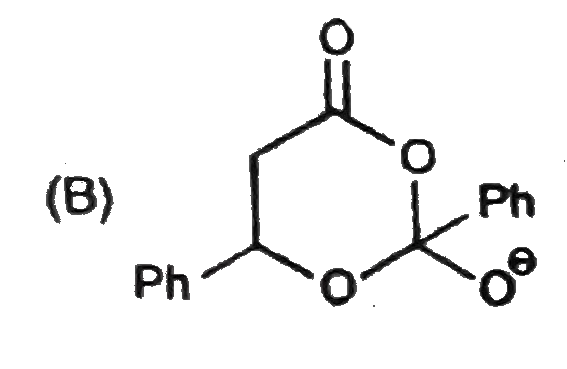
Observe the following sequence of reations P shows geometrical isomersm.Q gives positive Tollen's test and the oxidation product of Tollen's test followed by acidification is the strongest acid among its all position isomers. R gives positive lab tests with 2,4-DNP, Fehling solution and I_2//NaOH reagents. What could be the structure of Q ? |
|
Answer»
|
|
| 7. |
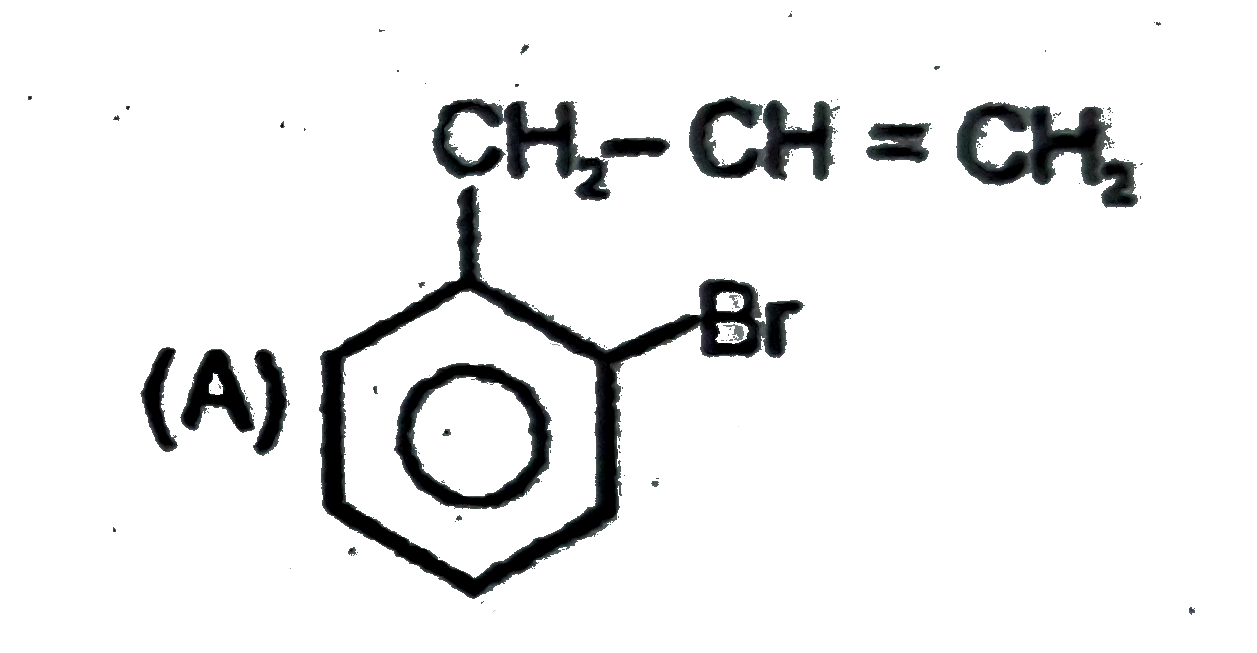
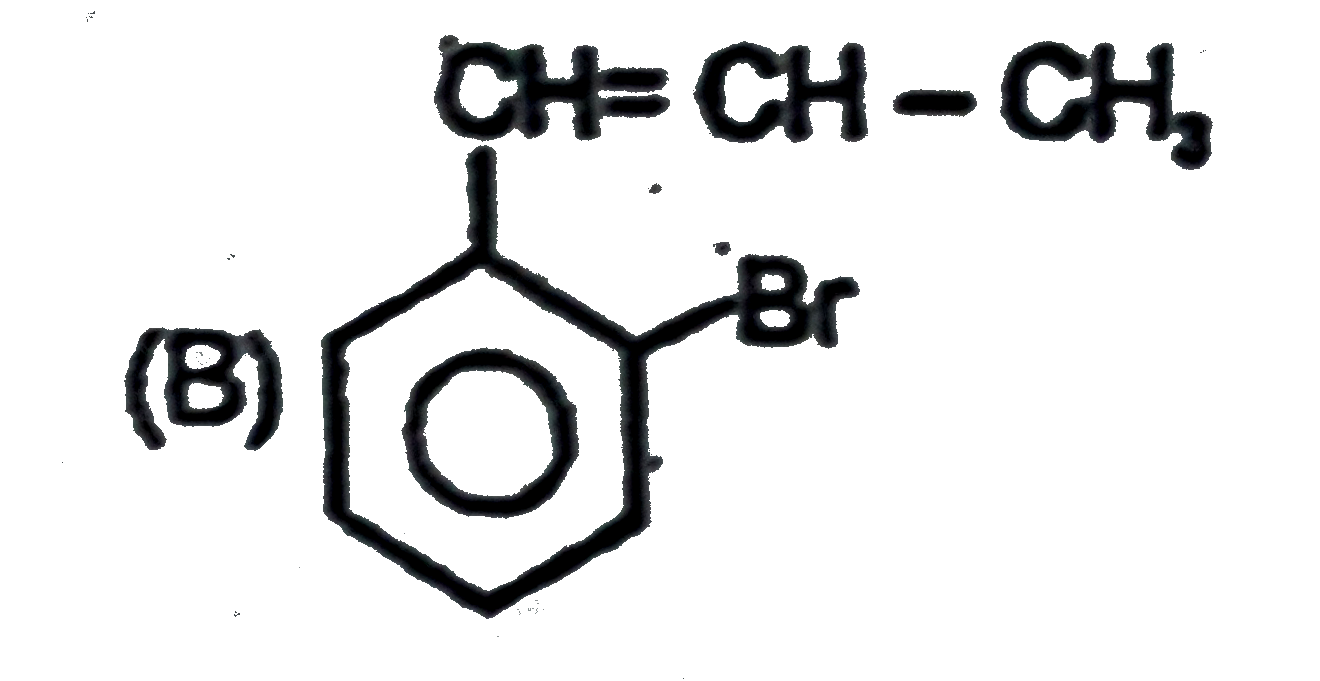
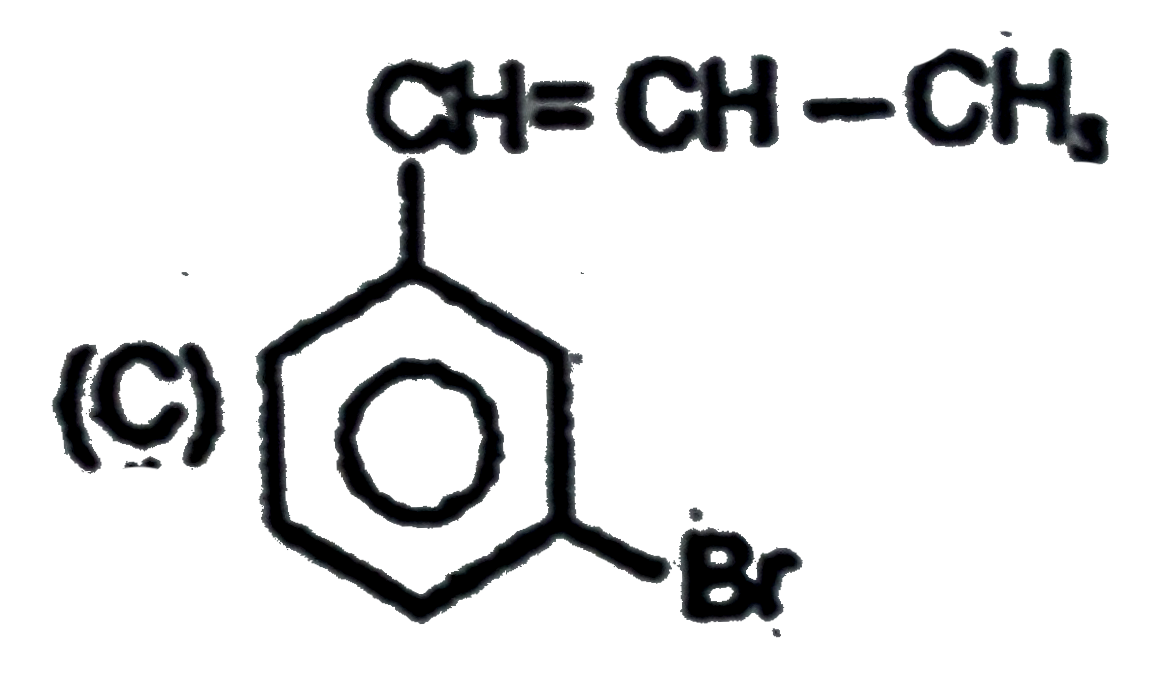
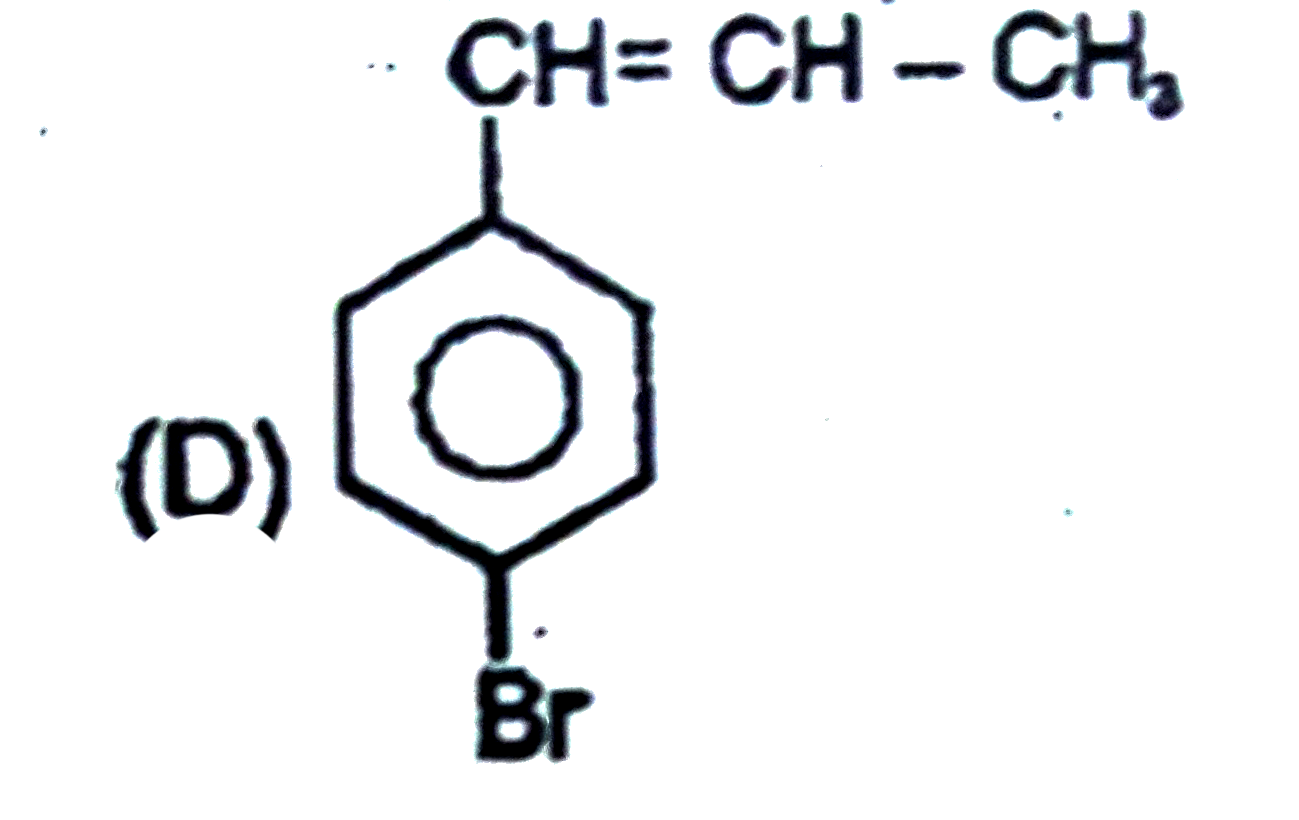
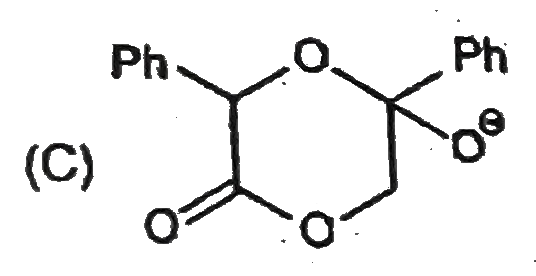
Observe the following sequence of reations P shows geometrical isomersm.Q gives positive Tollen's test and the oxidation product of Tollen's test followed by acidification is the strongest acid among its all position isomers. R gives positive lab tests with 2,4-DNP, Fehling solution and I_2//NaOH reagents. The compound P can be |
|
Answer»
|
|
| 8. |
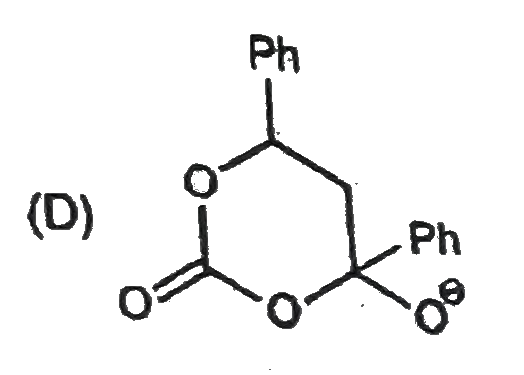
Observe the following sequence of reactions P shows geometrical isomersm. Q gives positive Tollen's test and the oxidation product of Tollen's test followed by acidification is the strongest acis among its all position isomers. R gives positive lab test with 2,4,-DNP, Fehling solutioni and I_(2)//NaOH reagents. Identify the structure of R[GOC-POC] |
|
Answer» `HCOOH`<BR>`CH_(3)CHO` |
|
| 9. |
Observe the following sequence of reactions P shows geometrical isomersm. Q gives positive Tollen's test and the oxidation product of Tollen's test followed by acidification is the strongest acids among its all position isomers. R gives positive lab test with 2,4,-DNP, Fehling solution and I_(2)//NaOH reagents. The compound P can be |
|
Answer» |
|
| 10. |
Observe the following sequence of reactions P shows geometrical isomers. Q gives positive Tollen's test and the oxidation product of Tollen's test followed by acidification is the strongest acis among its all position isomers. R gives positive lab test with 2,4,-DNP, Fehling solutioni and I_(2)//NaOH reagents. What could be the structure of Q? [GOC-POC] |
|
Answer» |
|
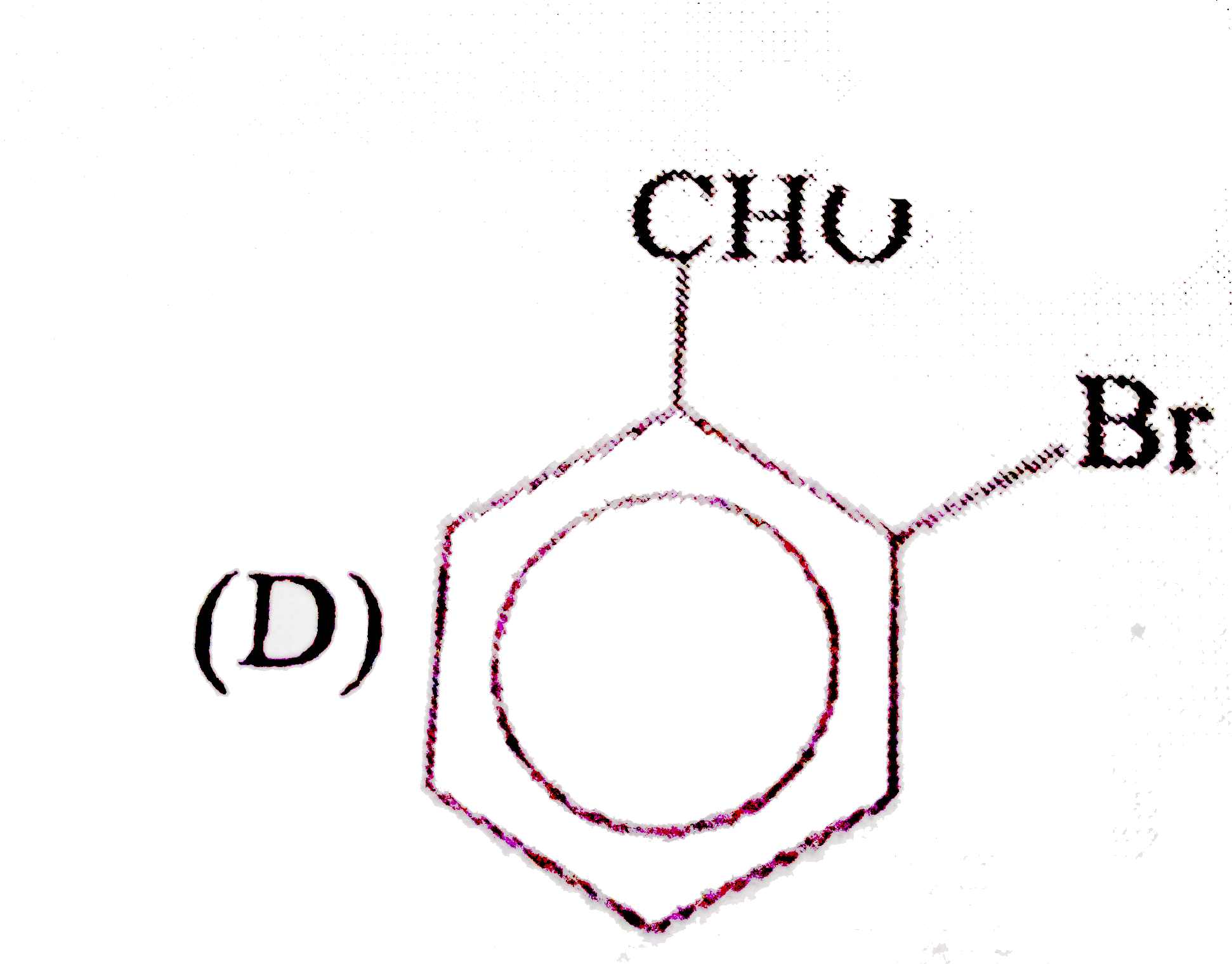
| 11. |
Observe the following sequence of reaction and answer the questions based on it Phenylacetyleneoverset(CH_3MgBr)underset(-CH_4)toxunderset((ii)H^(o+))overset((i)CO_2)toyunderset(HgSO_4)overset(H_2O // H_2SO_4)tozoverset(Delta)tow Which of the following compound give benzoic acid on KMnO_4 oxidation |
|
Answer» w 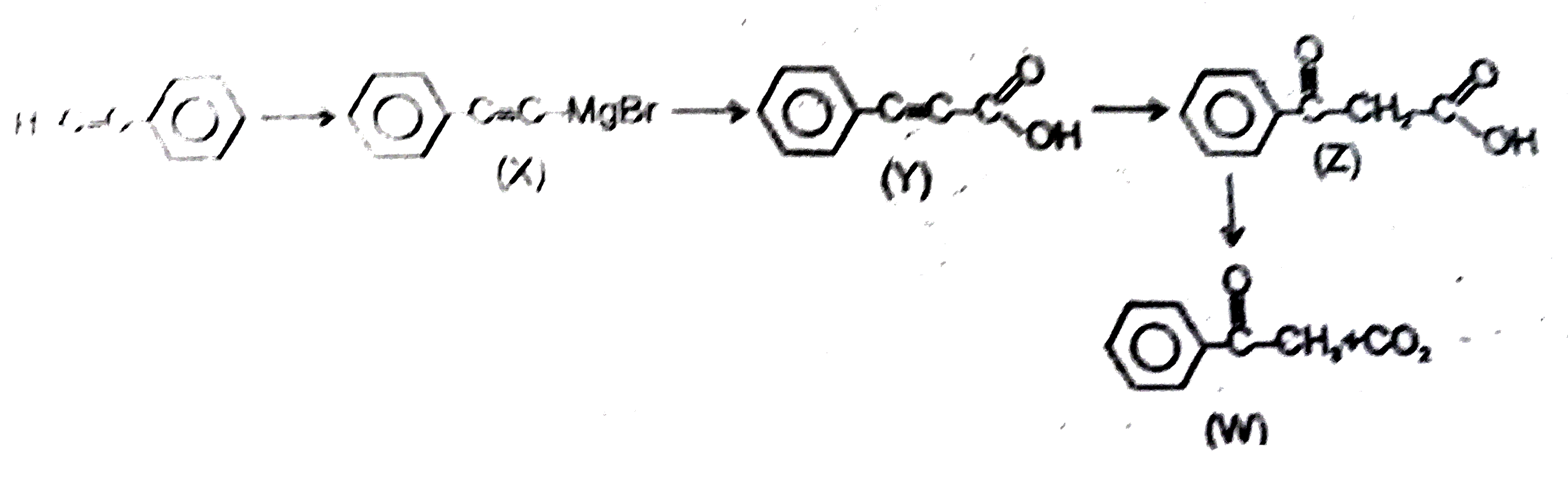
|
|
| 12. |
Observe the following sequence of reaction and answer the questions based on it Phenylacetyleneoverset(CH_3MgBr)underset(-CH_4)toxunderset((ii)H^(o+))overset((i)CO_2)toyunderset(HgSO_4)overset(H_2O // H_2SO_4)tozoverset(Delta)tow Which of the following statement is not correct |
|
Answer» y decolourises `Br_2//H_2O` solution 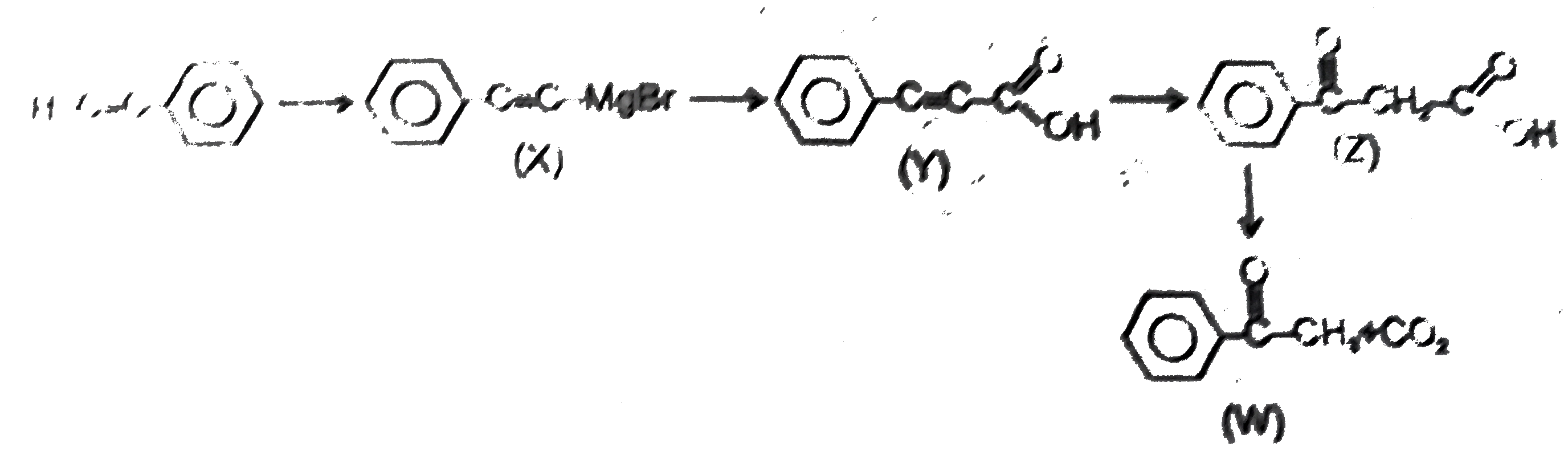
|
|
| 13. |
Observe the following sequence of reaction and answer the questions based on it. Phenylacetylene overset(CH_(3)MgBr)underset(-CH_(4))to x overset((i)CO_(2))underset((ii)H^(o+))to y overset(H_(2)O //H_(2)SO_(4))underset(HgSO_(4))to z overset(Delta)to w Which of the following compound give benzoic acid on KMnO_(4) oxidation |
|
Answer» w |
|
| 14. |
Observe the following sequence of reaction and answer the questions based on it. Phenylacetylene overset(CH_(3)MgBr)underset(-CH_(4))to x overset((i)CO_(2))underset((ii)H^(o+))to y overset(H_(2)O //H_(2)SO_(4))underset(HgSO_(4))to z overset(Delta)to w Which of the following statement is not correct |
|
Answer» y decolourises `Br_(2)//H_(2)O` solution |
|
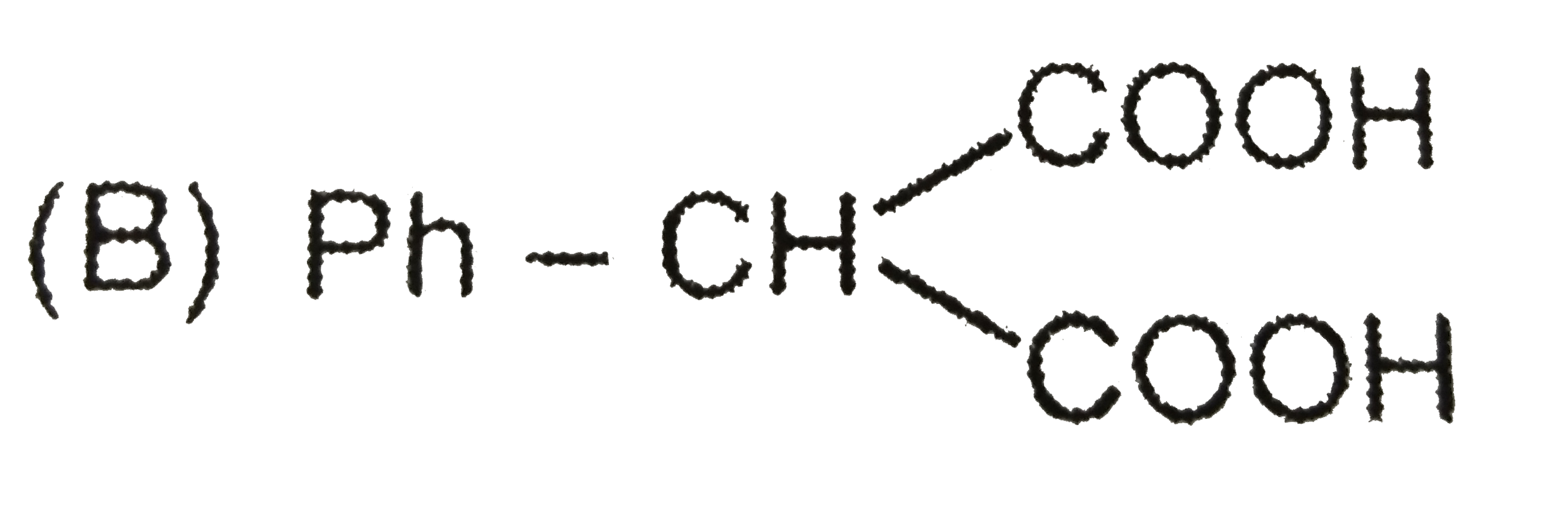
| 15. |
Observe the following sequence of reaction and answer the questions based on it. Phenylacetylene overset(CH_(3)MgBr)underset(-CH_(4))to x overset((i)CO_(2))underset((ii)H^(o+))to y overset(H_(2)O //H_(2)SO_(4))underset(HgSO_(4))to z overset(Delta)to w Compound z is |
|
Answer» `Ph-CH_(2)-OVERSET(O)overset(||)(C)-COOH` |
|
| 16. |
Observe the following reactions and its mechanistic steps and intermediate products. The leaving group of step (V) in reaction -2: |
|
Answer» `CH_3COO^(THETA)` 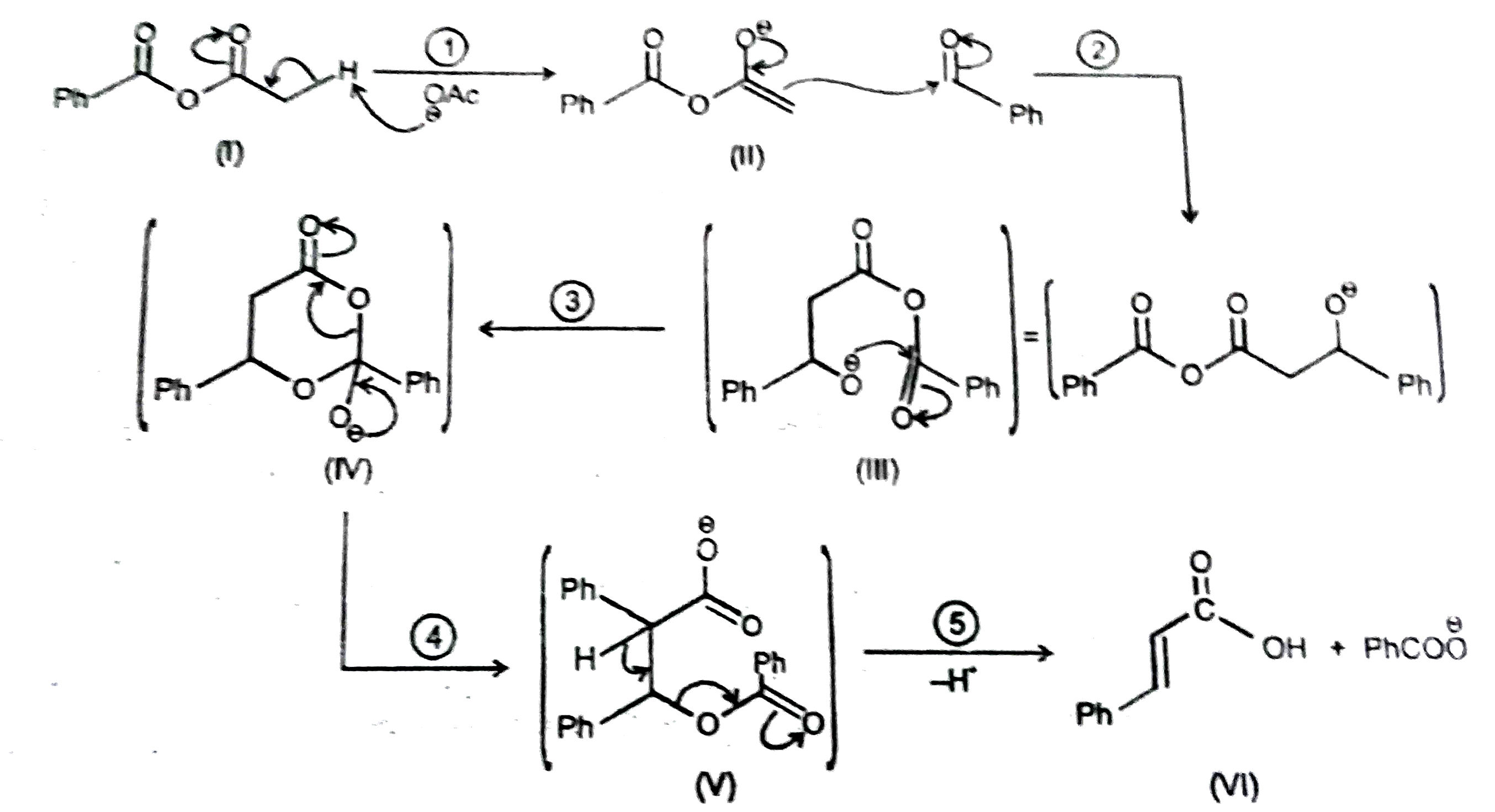
|
|
| 17. |
Observe the following sequence of reaction and answer the questions based on it Phenylacetyleneoverset(CH_3MgBr)underset(-CH_4)toxunderset((ii)H^(o+))overset((i)CO_2)toyunderset(HgSO_4)overset(H_2O // H_2SO_4)tozoverset(Delta)tow Compound z is |
|
Answer» `Ph-CH_2-oversetoverset(O)(||)C-COOH` 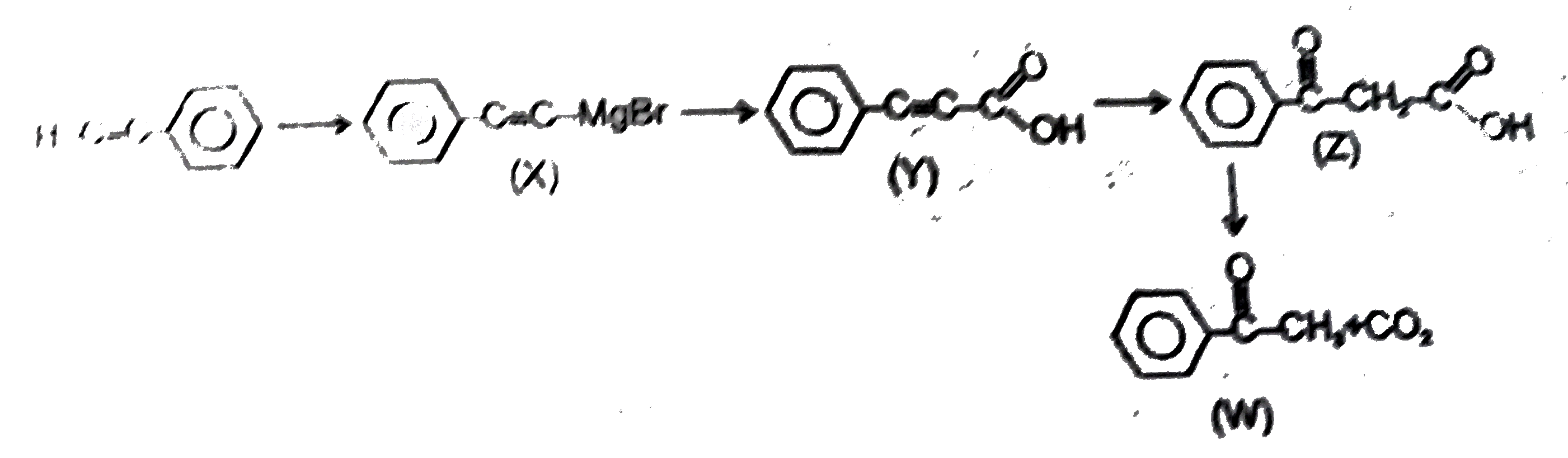
|
|
| 18. |
Observe the following reactions The reagent R_(1) and R_(2) can be respectively. |
|
Answer» NaI/Acetone, aq. `AgNO_(3)` |
|
| 19. |
Observe the following reactions (I)to (IV) and answer questions Z and W are |
|
Answer» IDENTICAL compounds 
|
|
| 20. |
Observe the following reactions (I)to (IV) and answer questions X and Y are |
|
Answer» POSITIONAL isomers 
|
|
| 21. |
Observe the following reactions and its mechanistic steps and intermediate products. If Ph-CH=O^(18) is used then O^18 is traced inreaction-2: |
|
Answer» `Ph-undersetunderset(O)(||)C-O^(18)-H` 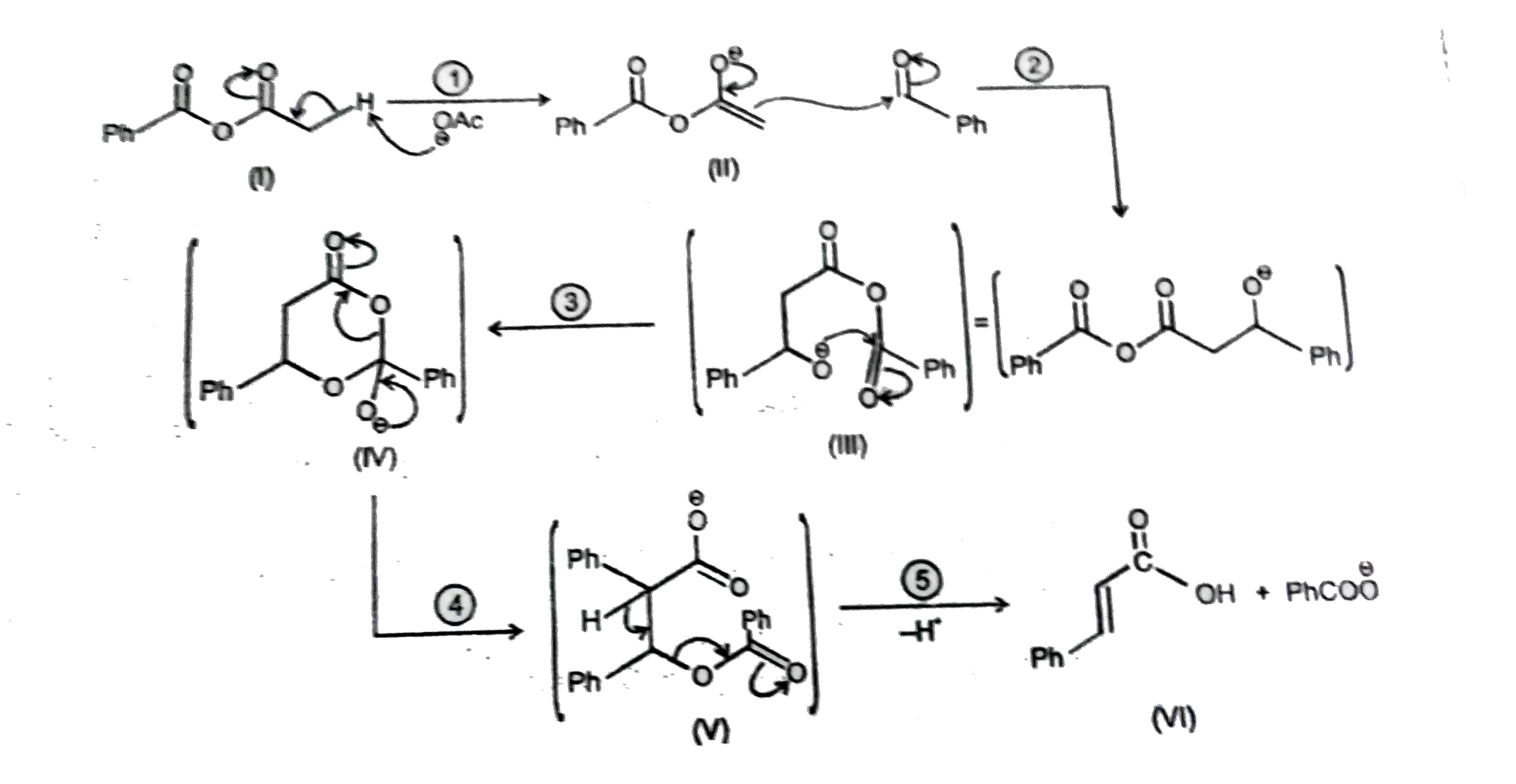
|
|
| 22. |
Observe the following reactions and its mechanistic steps and intermediate products. The intermediate product similar to (IV) following in the reaction-(2): |
|
Answer»
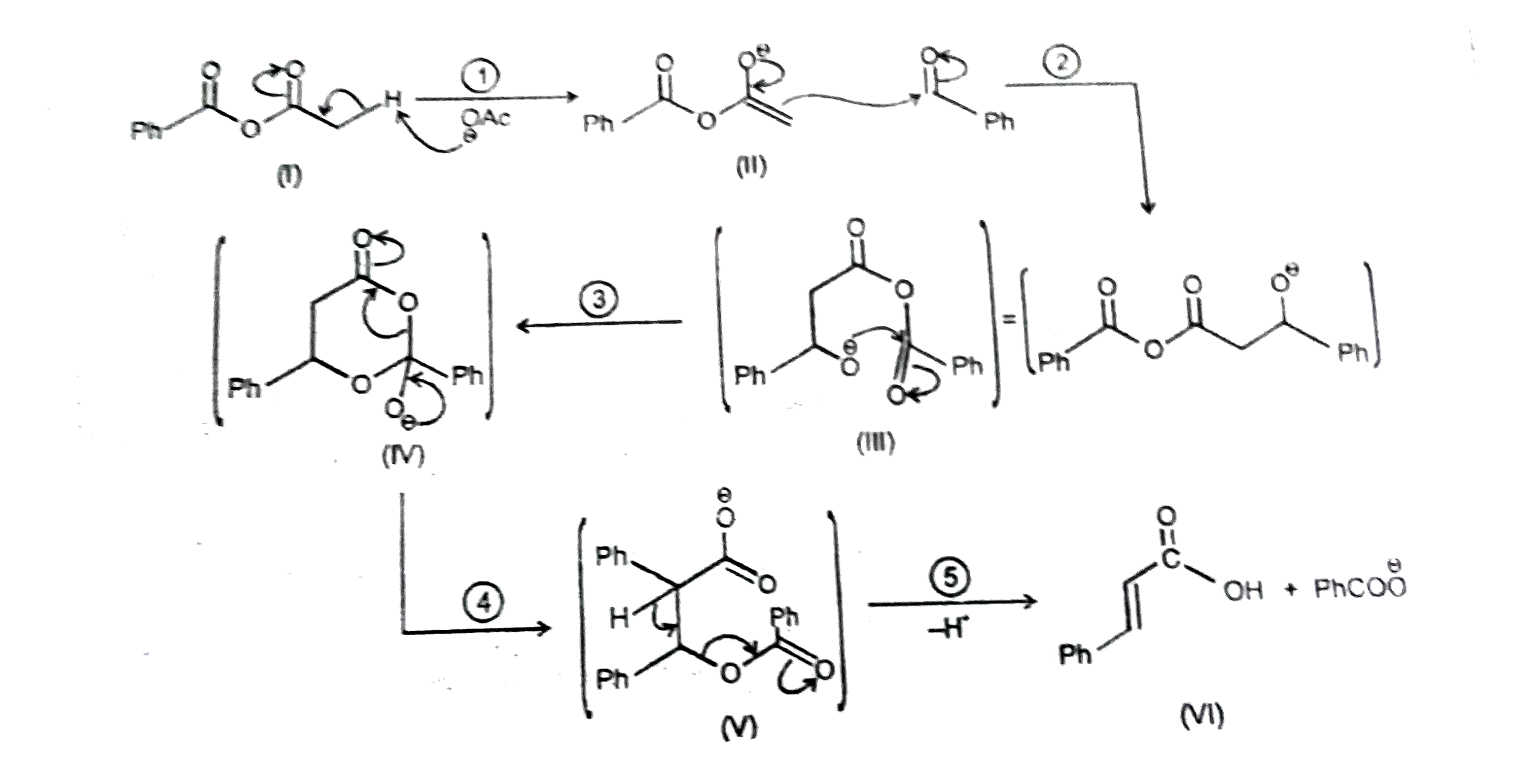
|
|
| 23. |
Observe the following reactions and answer the following questions The product T is |
|
Answer»
|
|
| 24. |
Observe the following reactions and answer the following questions The product mixture 'V' does not have. |
|
Answer»

|
|
| 25. |
Observe the following reaction sequence X can be |
|
Answer»
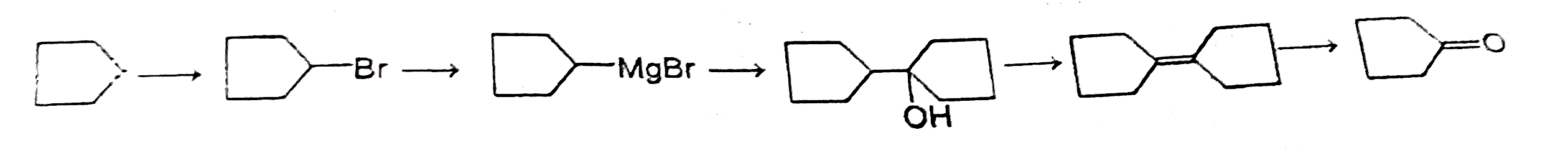
|
|
| 26. |
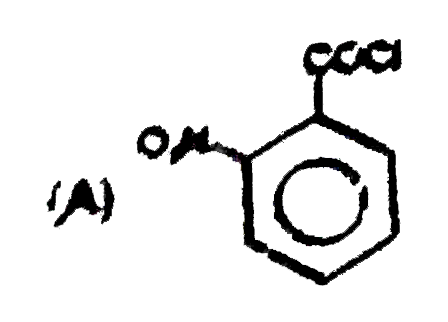
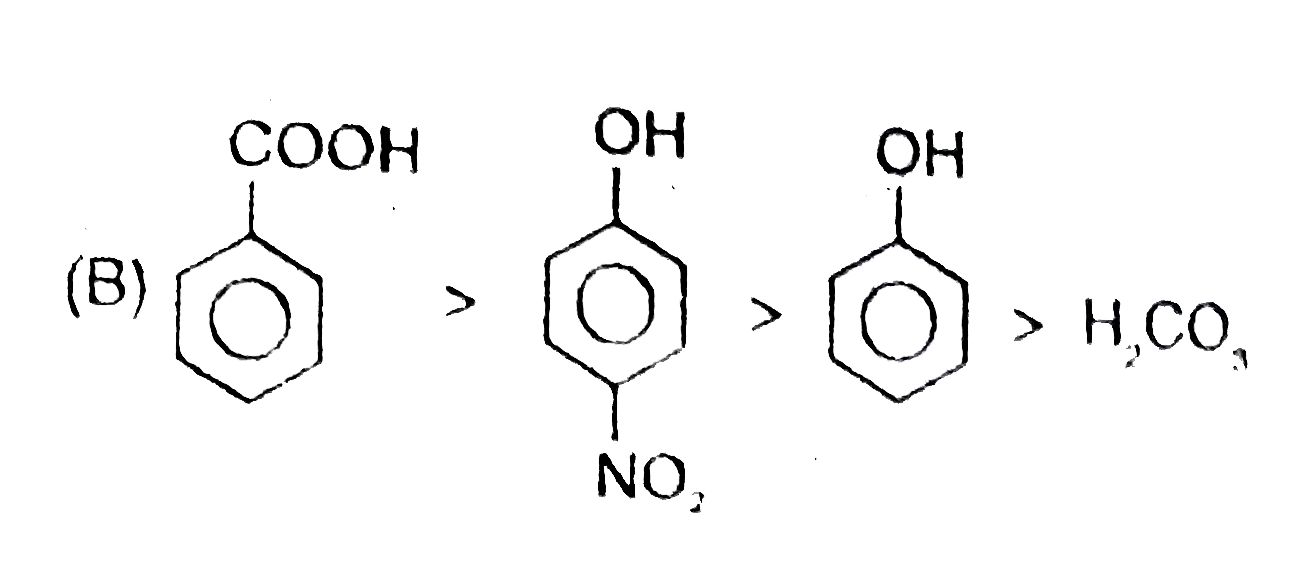
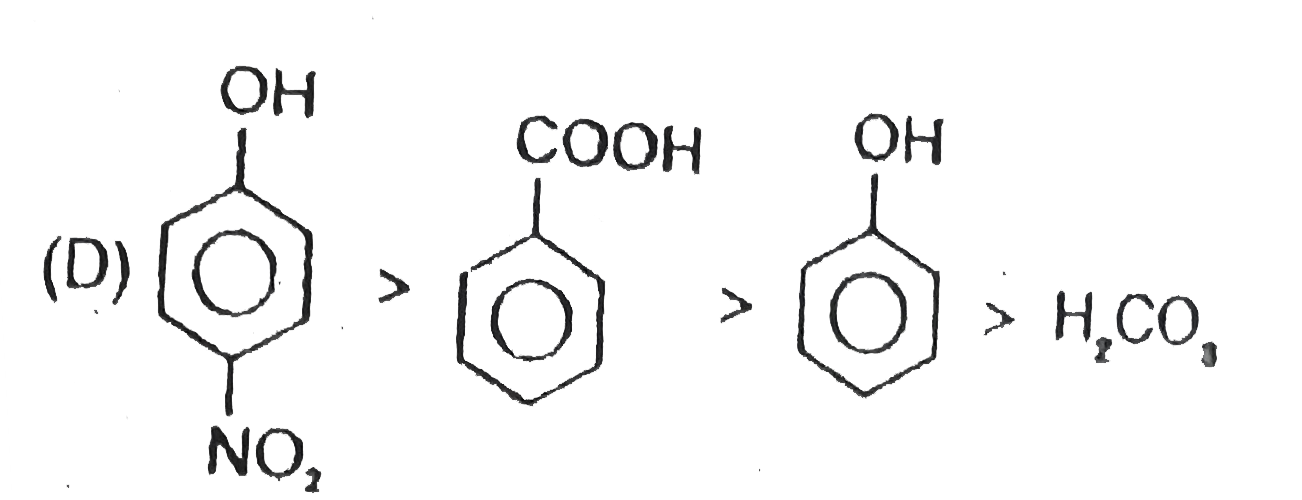
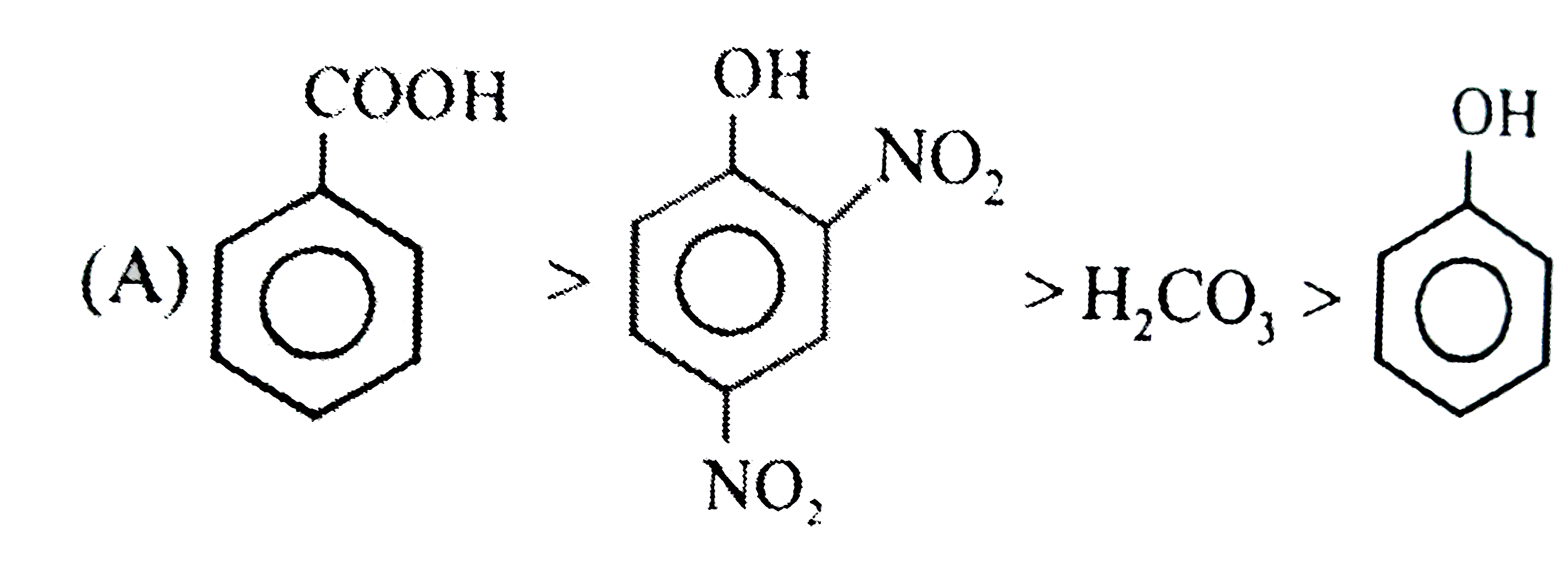
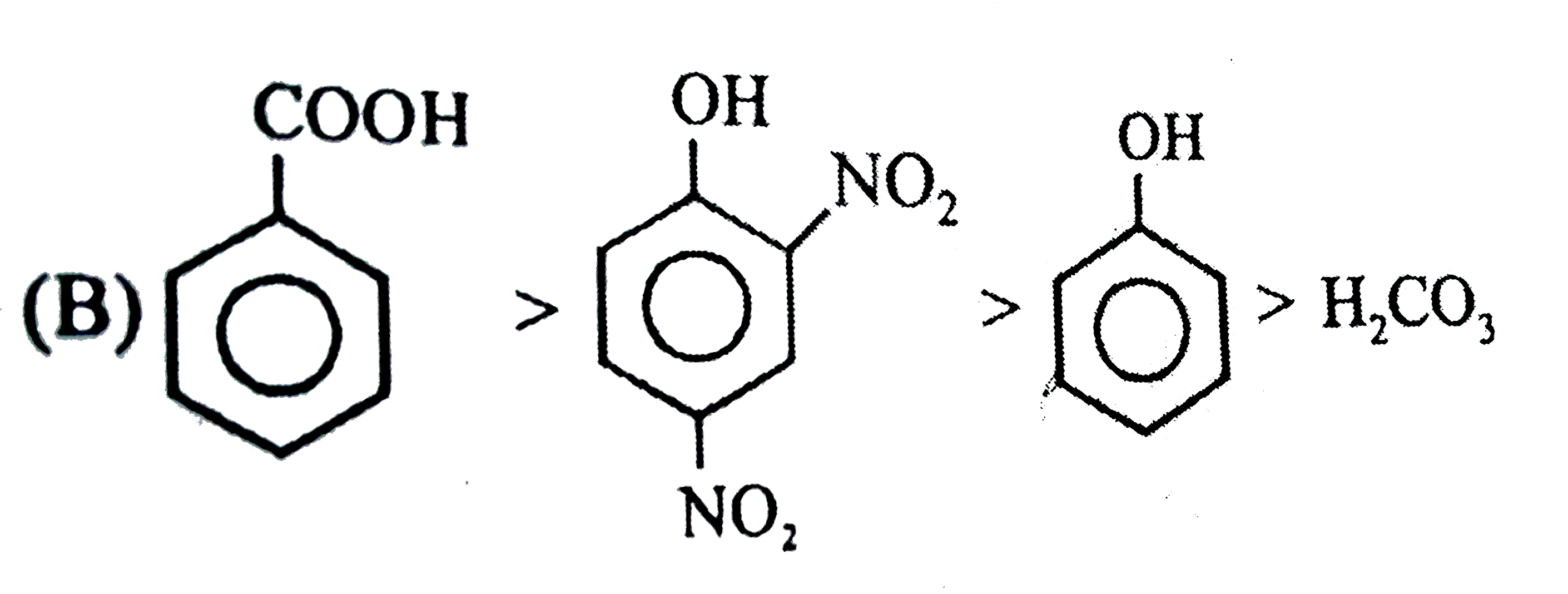
Observe the following reaction : Which of the following is/are incorrect order of acid strength : |
|
Answer»
 Since a strong acid DISPLACES a WEAK acid from its SALT and FORMS its own salt. |
|
| 27. |
Observe the following reaction sequence carefully.Find the total number of stereoisomers formed in step (6) |
Answer» 
|
|
| 28. |
Observe the following reaction sequence Answer the following question in the given format: P=Number of organic products formed in step-1 Q=Number of organic products formed in step-2 (singnificant products) R=Number of organic products formed in step-3 (singnificant products) S=In which step aromatic electrophilic substitution has taken place |
Answer» SOLUTION :
|
|
| 29. |
Observe the following reaction and mention the number which show slowest and rate determine step. |
|
Answer» |
|
| 30. |
Observe the following reaction and mark the correct statement given below- |
|
Answer» METHYL glucosides do not react with Fehling's or Tollen's reagent |
|
| 31. |
Observe the following reaction and find out that how many number of reactant stereoisomers can be reduced to optically inactive meso products. |
Answer» 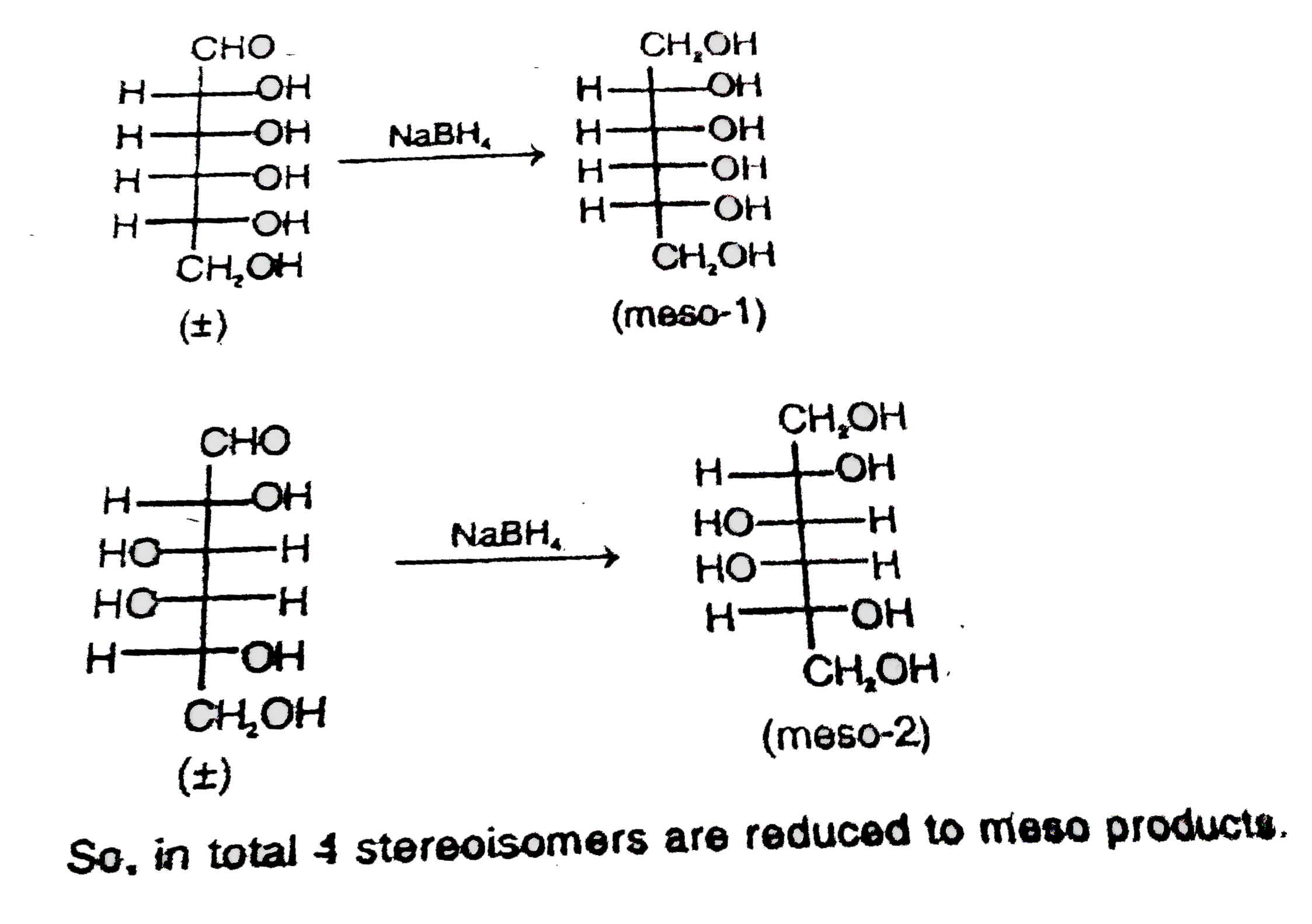
|
|
| 32. |
Observe the following reaction and determine True statement |
|
Answer» If aromatic RING I have `-NO_(2)` group then RATE of REACTION will decrease. |
|
| 33. |
Observe the following reaction, 2A+B rarr C The rate of formation of C is 2.2xx10^(-3)" M m in"^(-1). What is the value of -(d[A])/(dt) ("in moL"^(-1)" m in "^(-1)) ? |
|
Answer» `2.2xx10^(-3)` `(d[C])/(dt)=(1)/(2) ((-d[A])/(dt)) rArr (-d[A])/(dt) =4.4xx10^(-3)` |
|
| 34. |
Observe the following reaction A_((g))+3B_((g))to2C_((g))The rate of this reaction -(d[A])/("d t") is 3xx0^(-3)"mol. lit"^(-1)."min"^(-1). What is the value of -(d[B])/("d t") in "mol. lit"^(-1)."min"^(-1) ? |
|
Answer» `3xx10^(-3)` |
|
| 35. |
Observe the following reaction:2A + B to C The rate of formation of C is 2.2xx10^(3) mol L^(-1) min^(-1).What is the value - (d(A))/(dt) ("in "mol L^(-1) mi n^(-1)) |
|
Answer» `2.2xx10^(-3)` |
|
| 36. |
Observe the following flow chart and write the sum of the number of x,y, z. |
Answer» 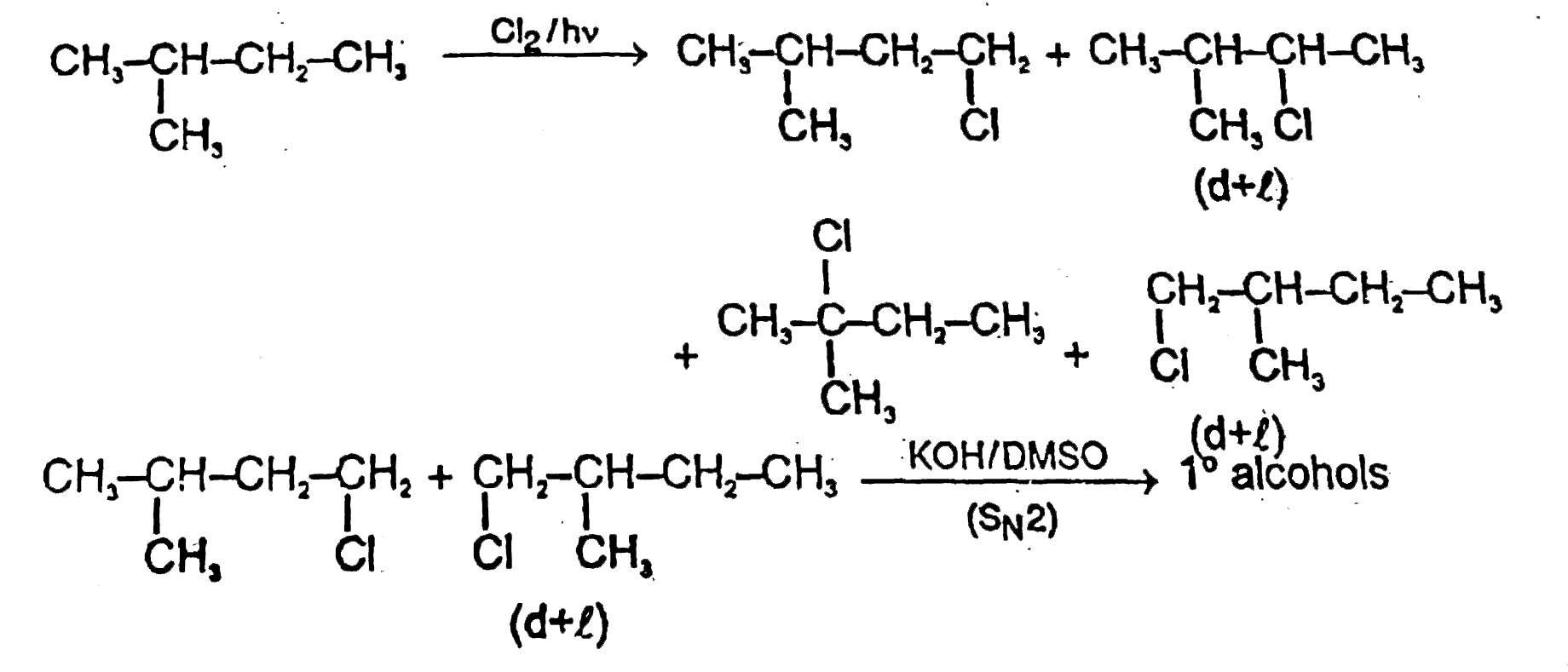
|
|
| 37. |
Observe the following feasible reaction: Q. Which of the following is the correct order of acidic strength: |
|
Answer»
|
|
| 38. |
Observe the following feasible reaction: Q. Identify the feasible reactions |
|
Answer»
|
|
| 39. |
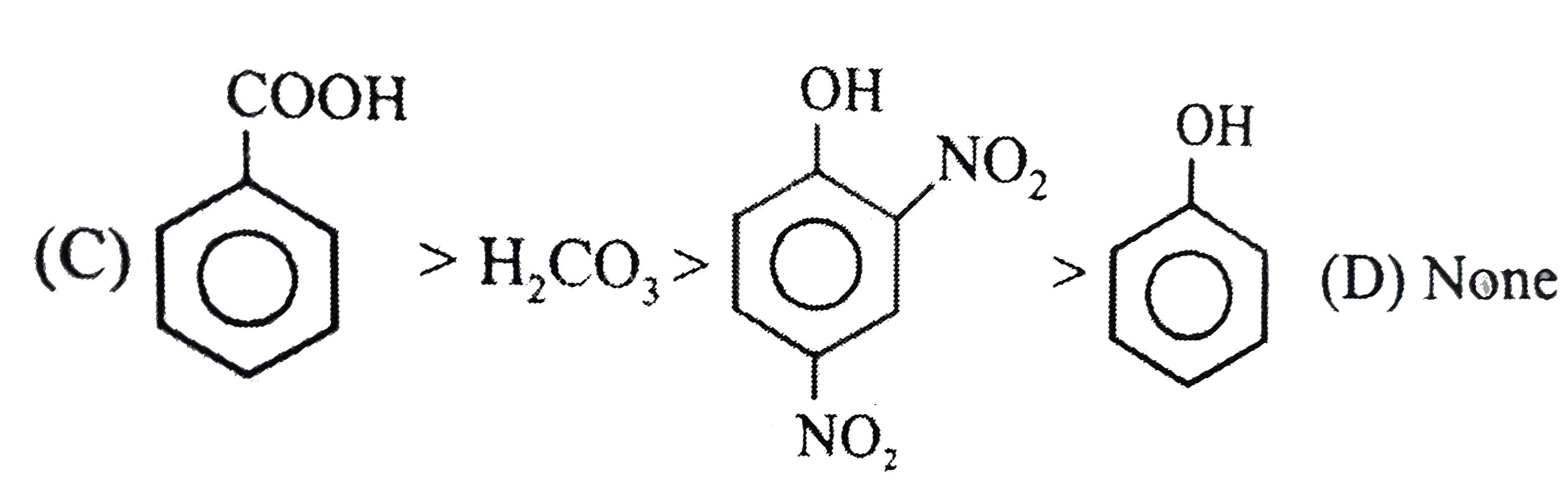
Observe the following feasible reaction: Q. Which of the following compound does not react with NaHCO_(3). |
|
Answer»
|
|
| 40. |
Observe the following experiment |
Answer» 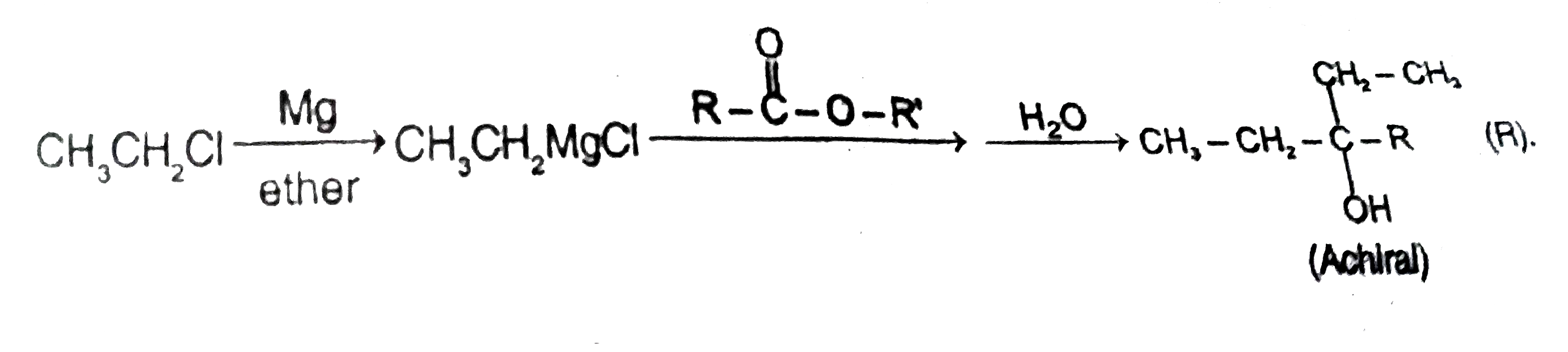
|
|
| 41. |
Observe the following data regarding {:(""2NH_(3)overset(W)(to)N_(2)+3H_(2)),("Pressure (in atm) " :"51020"),("Half life (min)" : "3.61.80.9"):} The unit of K is |
|
Answer» `"min"^(-1)` |
|
| 42. |
Observe the following compounds Number of coumpound which can gives positive Haloform test = (x) Number of compound which can gives positive Lucas reagent test = (y) Report your answer (x+y) |
|
Answer» |
|
| 43. |
Observe the following compound and write the number of hydrogen atom involved in hyper conjugation (with carbocation)? |
Answer» 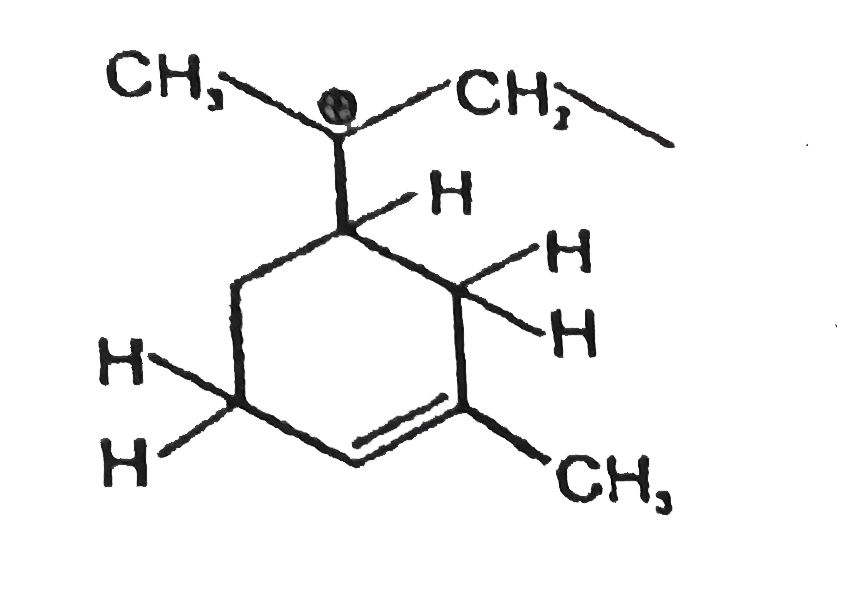 TOTAL NUMBER of HYDROGEN involved in HYPERCONJUGATION with CARBOCATION =6 |
|
| 44. |
Observe the following compound and select +ve & -ve test respectively. |
|
Answer» `+ + + -` 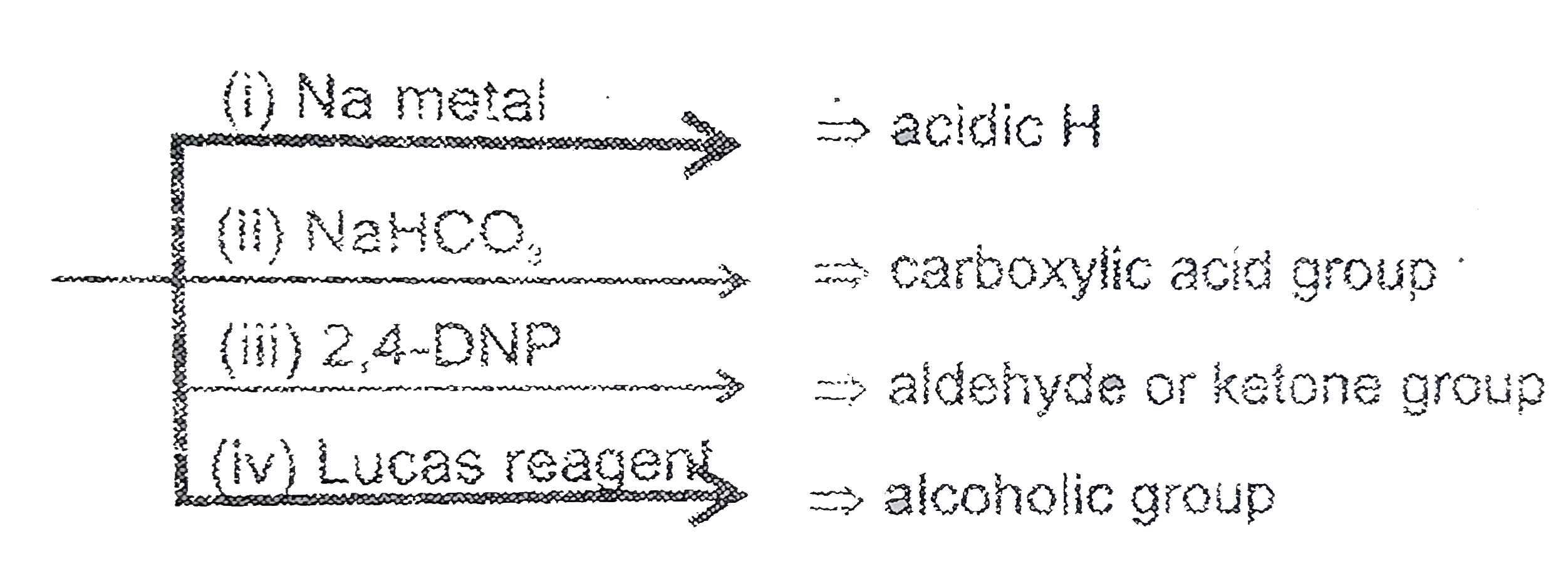
|
|
| 45. |
Observe the following abbrevations pi_(obs) = observed colligative property assuming normal behaviour of solute. Van't Haff factor (i) is given by |
|
Answer» `i=pi_(OBS)XX pi_(CAL)` |
|
| 46. |
Observe the esterification mechanisms for primary and teriary alcohols. CH_(3)-COOH+C_(2)H_(5)overset(18)(O)H overset(Conc. H_(2)SO_(4))to(P) CH_(3)-COOH+(CH_(3))_3C-overset(18)(O)H overset(Conc. H_(2)SO_(4))to (Q) In the above reaction (P) and (Q) are respectively |
|
Answer» `CH_(3)- overset(O)overset(||)(C)-O-C_(2)H_(5),CH_3-overset(O)overset(||)(C)-overset(18)(O)-overset(CH_(3))overset(|)UNDERSET(CH_(3))underset(|)(CH)_(3)-CH_(3)` 
|
|
| 47. |
Observe the esterification mechanisms for primary and teriary alcohols. (+) Octan -2-ol esterifies with Acetic acid to give optically inactive racemised product. It must have gone by |
|
Answer» TYPE I mechanism |
|
| 48. |
Observe the esterfication mechanisms for primary and tertiary alcohols. CH_3-COOH+Ph-undersetunderset(C_2H_5)(|)oversetoverset(CH_3)(|)C-OHoverset(Conc.H_2SO_4)to(Y) |
|
Answer» (X) is optically ACTIVE while (Y) is optically INACTIVE Y is formed by type-2 mechanism hence recemic product is OBTAINED. |
|
| 49. |
Observe the esterfication mechanisms for primary and tertiary alcohols. CH_3-COOH+C_2H_6overset(18)OHoverset(Conc.H_2SO_4)to(P) CH_3-COOH+(CH_3)_3C-overset(18)OHoverset(Conc.H_2SO_4)(Q) In the above reaction (P) and (Q) are respectively : |
|
Answer» <P>`CH_3-oversetoverset(O)(||)C-O-C_2H_6,CH_3-oversetoverset(O)(||)C-overset(18)(O)-undersetunderset(CH_3)(|)oversetoverset(CH_3)(|)C-CH_3` |
|
| 50. |
Observe the esterfication mechanisms for primary and tertiary alcohols. (+) Octan-2-ol esterifies with Acetic acid to give optically inactive recemised product.It must have gone by |
|
Answer» TYPE I MECHANISM |
|