Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Of the following metuls that cannot be obtained by elecrolysis of the aqueous solution of their satts is are |
|
Answer» Ag |
|
| 2. |
Of the following molecules, the one which haspermanent dipole moment is |
|
Answer» `SiF_4`  `MU NE 0` but `SiF_4 , BF_3` and `PF_5` are symmetrical MOLECULARS |
|
| 3. |
Of the following metals that cannot be obtained by electolysis of the aqueous solultion of their salts is /are |
| Answer» Answer :D | |
| 4. |
Of the following matals that cannot be obtained by electrolysis of the aqueous solutions of their cathode are : |
|
Answer» AG andMg |
|
| 5. |
Of the following hydrides which one has the lowest boiling point ? |
|
Answer» `AsH_(3)` |
|
| 7. |
Of the following, how many can retard the decomposition of concentrated H_(2)O_(2) ? Ethanol, Acetic acid, Ethylene glycol, Benzene, Sulphuric acid, Phosphoric acid? |
|
Answer» |
|
| 9. |
Of the following four reactions, formic and acetic acids differ in which respect |
|
Answer» Replacement of hydrogen by sodium Whereas `CH_(3)COOH underset("solution")overset("Fehling")to`No reaction. |
|
| 10. |
Of the following four groups are m- directing when present on a benzenering. The one which is not meta-directing is: |
|
Answer» `-COOH` |
|
| 11. |
Of the following four reactions, formic and acetic acid differ in which respect ? |
|
Answer» Formation of ester with alcohol |
|
| 12. |
Of the following elements, the one with maximum electropositive character is |
|
Answer» `Cl_(2)`<BR>`Br_(2)` |
|
| 13. |
Of the following compounds which will react with Br_(2) at room temperature in dark. {:((a),"Benzene "(C_(6)H_(6)),,),((b),"Cyclohexene " (C_(6)H_(10)),,),((c),"Cyclohexene " (C_(6)H_(12)),,),((d),"Propanoic Acid" (C_(2)H_(5)CO_(2)H),,),((e),"Phenol " (C_(6)H_(5)OH),,),((f),"Nitrobenzene" (C_(6)H_(5)NO_(2)),,),((g),"Hexyne " (C_(6)H_(10)),,),((h),2"," 2-"dichloropropane" (C_(3)H_(6)Cl_(2)),,):} |
|
Answer» |
|
| 14. |
Of the following element, the one showing only one oxidation state is |
|
Answer» `I_(2)` |
|
| 15. |
Of the following crystalline solids which one is molecular solid, and which one is network solid? (i) P_(4) (ii) Se |
|
Answer» <P> Solution :molecular solid: `P_(4)`, NETWORK solid: SE, |
|
| 16. |
Of the following compounds, which will have zero dipole moment ? |
|
Answer» 1, 1-Dichloroethylene |
|
| 17. |
Of the following compounds ,which will have a zero dipole moment : |
|
Answer» 1, 1-DICHLOROETHYLENE |
|
| 18. |
Of the following compounds which will have a zero dipole moment ? |
|
Answer» 1,1-dichloroethylene |
|
| 19. |
Of the following compounds, the one which is a Lewis acid is: |
|
Answer» `PCl_3` |
|
| 20. |
Of the following compounds, which one is the strongest Bronsted acid in an aqueous solution ? |
|
Answer» `HClO_(3)` |
|
| 21. |
of the following compounds, the one which is a Lewis acid is :^(**"**) |
|
Answer» `PCl_3` |
|
| 22. |
Of the following compounds the most acidic is |
|
Answer» <P>`As_(2)O_(3)` |
|
| 23. |
Of the following complexes, the one with the largest value of the crystal field splitting is: |
|
Answer» `Fe(H_2O)_6^(3+)` |
|
| 24. |
Of the following complex ions. The one that probably has the largest overall formation constant K_(f) is :- |
|
Answer» `[Co(NH_(3))_(6)]^(3+)` `[Co(e n)_(3)]^(3+)` has highest value of `K_(f)` among complex due to chelation |
|
| 25. |
Of the following complex ions, which is diamagnetic in nature ? |
|
Answer» `[CoF_6]^(3-)` |
|
| 26. |
Of the following complex ions, one is a Bronsted-Lowery acid. That one is: |
|
Answer» `[CU(NH_(3))_(4)]^(2+)` |
|
| 27. |
Of the following complex ions, the one that probably has the largest overall formation constant, K_(f) is : |
|
Answer» `[Co(NH_(3))_(6)]^(3+)` |
|
| 28. |
Of the following atoms, which one of the highest n/p ratio |
|
Answer» `NE^(16)` `._(10)Ne^(16) RARR ._(n =6)^(p = 10) (n)/(p) = (6)/(10) = 0.6, ._(8)O^(16) rArr._(n=8)^(p=8) n//p = 1` `._(9)F^(16) rArr._(n =7)^(p =9)(n)/(p) = (7)/(9) = 0.778 ? , N^(16) rArr ._(n =9)^(p = 7)n//p = (9)/(7) = 1.2857` |
|
| 29. |
Of the following complex ions, one exhibits isomerism. That is : |
|
Answer» `[Ag(NH_(3))_(2)]^(+)` |
|
| 30. |
Of the following amines how many can give foul smell with CHCl_(3) and alcoholic KOH : |
|
Answer» |
|
| 31. |
Of the following alcohols, the one that would react fastest with conc. HCl and anhydrous ZnCl_(2) is ...... |
|
Answer» Butan-1-ol |
|
| 32. |
Of the following acids, the one which possesses oxidising and reducing properties and has the capability to form complex compound is |
|
Answer» `HNO_2` |
|
| 33. |
Of the following 0.2 m aqueous solutions, which one will exhibit largest freezing point depression? |
| Answer» ANSWER :D | |
| 34. |
Of the followign substane the one which doesnot contain oxygen is |
|
Answer» Bauxite |
|
| 35. |
Of the following acids, the one that is strongest is |
|
Answer» `HBrO_(4)` |
|
| 36. |
Of the following 0.10 m aqueous solutions, which one will exhibit the largest freezing point depression ? |
|
Answer» KCl i is highest for `Al_(2)(SO_(4))_(3)` |
|
| 37. |
Of the following 0.10 m aqueous solutions, which one will exhibit the largest freezing point depression |
|
Answer» `Al_(2)(SO_(4))_(3)` |
|
| 38. |
Of the following 0.10 m aqueou solution, which one will exhibit the largest freezing point depression ? |
|
Answer» KCl `DeltaT_(f) propiand i " for "KCl=2," for "C_(6)H_(12)O_(6),i=1," for"` `Al_(2)(SO_(4))_(3),i=5" and for "K_(2)SO_(4),i=3`. HENCE, `Al_(2)(SO_(4))_(3)` will show largest freezing point depression. |
|
| 39. |
Of the five isomeric hexanes, the isomers which can give five monochlorinated structural products |
|
Answer» n-Hexane |
|
| 40. |
Of the five isomeric hexanes, the isomer which can give two monochlorination compounds is ? |
|
Answer» `n`-HEXANE |
|
| 41. |
Ofthe five isomeric hexanes, the isomer which can give two monochlorinated compounds is |
|
Answer» n-hexane 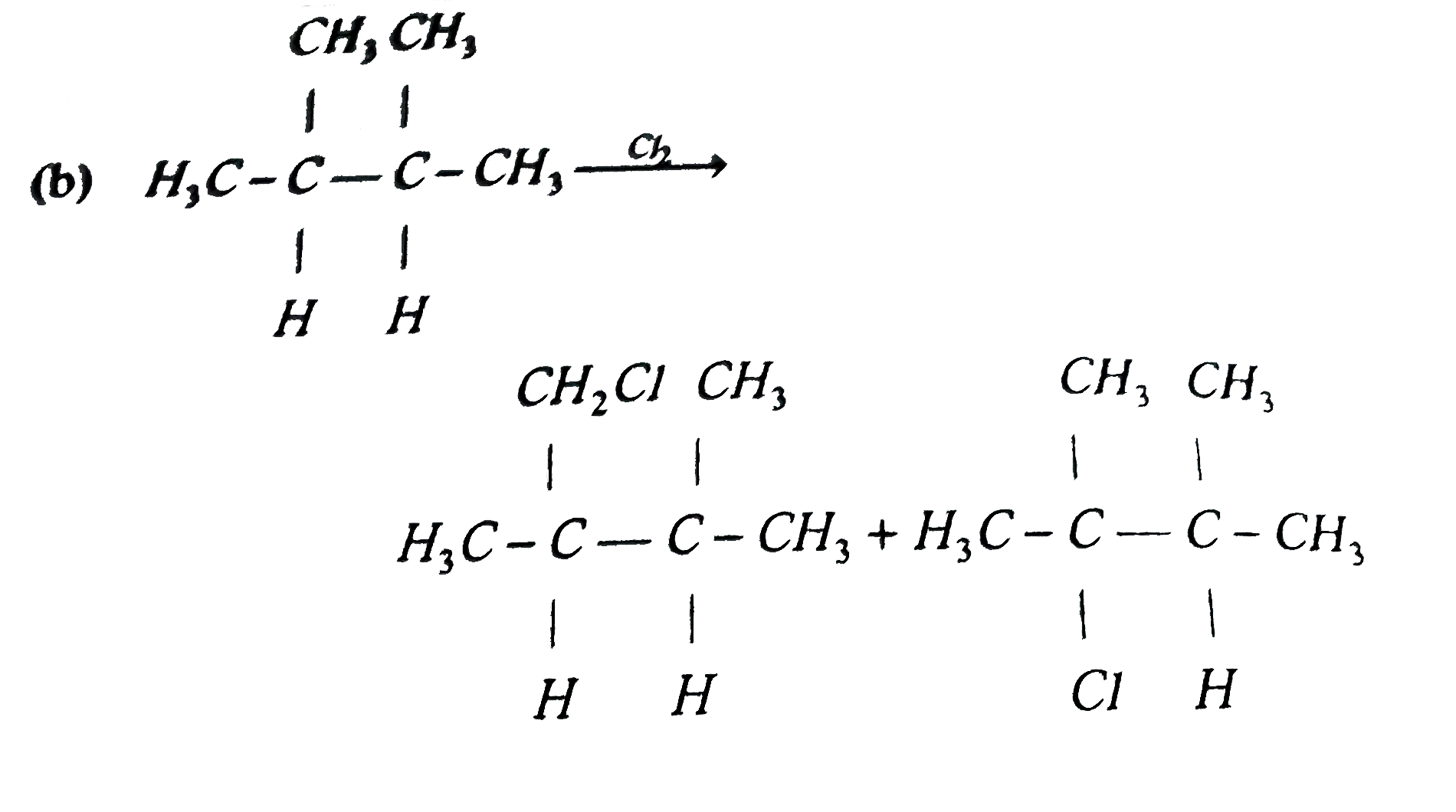
|
|
| 42. |
Of the five isomeric hexanes, the isomer which can give two monochlorinated compound is : |
|
Answer» n-hexane |
|
| 43. |
Ofthefiveisomerichexane, theisomerwhichcangivetwomonochlorinatedcompoundsis : |
|
Answer» N- HEXANE |
|
| 44. |
Of the elements Sr, Zr, Mo, Cd and Sb, all of the which are in the 5th period , the ones that are paramagnetic are |
|
Answer» SR, Cd and Sb |
|
| 45. |
Of the elements Sr,Zr,Mo,Cd and Sb, all of which are in V period, the paramagnetics are: |
| Answer» Answer :D | |
| 46. |
Of the chemical reactions given below which can result in the formation of C_(6)H_(5)CH_(2)NH_(2) ? |
|
Answer»
|
|
| 47. |
Of the different allotropic forms of phosphorus, the one which has a metallic lustre is |
|
Answer» black PHOSPHORUS |
|
| 48. |
Of the different allotropes of phosphorus, the one which is most reactive is |
|
Answer» VIOLET PHOSPHORUS |
|
| 49. |
Of physisorption or chemisorption, which has a higher enthalpy of adsorption ? |
| Answer» SOLUTION :CHEMISORPTION has a HIGHER ENTHALPY of ADSORPTION. | |
| 50. |
Of physisorption and chemisorption, which type of adsorption has a higher enthalpy of adsorption ? |
| Answer» Solution :CHEMISORPTION has higher enthalpy of adsorption on ACCOUNT of formation of CHEMICAL BOND. | |


