Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Number of p-disubstituted benzene compounds possible with molecular formula C_(8)H_(11)N is |
Answer» 
|
|
| 2. |
Number of oxygen atos between two chromium atoms in Cr_(2)O_(7)^(2-) ion is ..... |
|
Answer» |
|
| 3. |
Number of oxygen atoms in an acetamide molecule is |
|
Answer» 1 |
|
| 4. |
Number of oxide orwesd among , (i) Cassiterite (ii) pyrolusite (iii) Kieserite (iv) Azurite (V) chromite (vi) siderite are. |
|
Answer» |
|
| 5. |
Number of optically inactive stereoisomers of the following molecule is . CH_3-CH=CH-undersetunderset(OH)(|)CH-CH=CH-undersetunderset(OH)(|)CH-CH=CH-CH_3 |
|
Answer» |
|
| 6. |
(Number of organic product formed.) |
|
Answer» |
|
| 7. |
Number of optically active tartaric acid is/are possible : |
|
Answer» 1 |
|
| 8. |
Number of optically active isomers of compound [M(AB)b_(2)c_(2)]^(+-n) |
Answer» 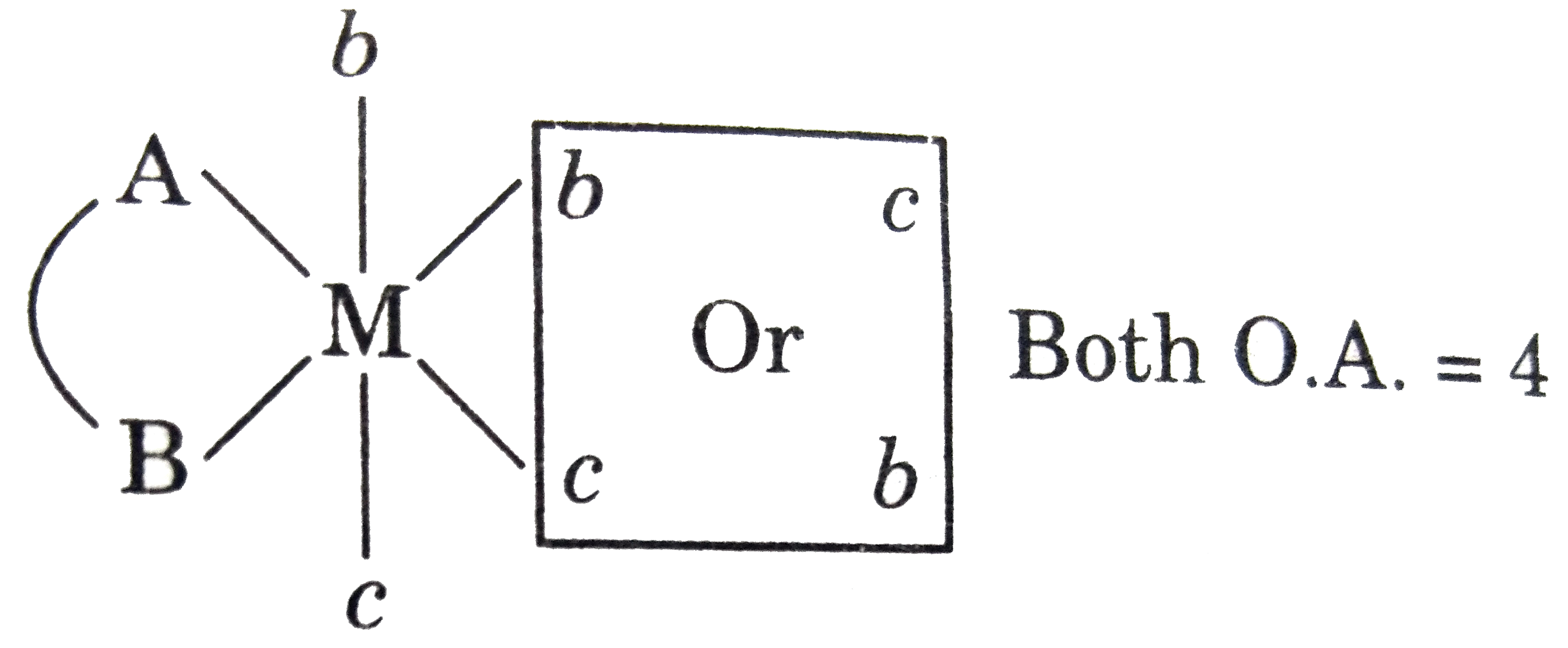
|
|
| 9. |
Number of optically active isomer is/are : |
|
Answer» 0 |
|
| 10. |
Number of optically active isomer is/are : |
|
Answer» 0 |
|
| 11. |
Number of nodal planes for f-orbital are |
|
Answer» 3 |
|
| 12. |
Number of nitrogen atoms present in melamine isx.The number of-imine and - amine groups is y and z,x,y and z are respectively |
|
Answer» 3,1,2 No. of IMINE groups three No. of amine groups three |
|
| 13. |
Number of nitrogen atoms present in melamine is x. The number of -imine and - amine groups is y and z. x,y and z are respectively. |
|
Answer» 3,1,2 |
|
| 15. |
Number of neutrons in a parent nucleus X, which given ._(7)N^(14) nucleus after two successive beta emission would be |
|
Answer» 9 |
|
| 16. |
Number of natural life times (t_(av)) required for a first order reaction to complete 99.9% is |
|
Answer» `6.93` `=10 xx (0*693)/(k) ""(because t_(1//2) = (0*693)/(k))` `=10 xx 0*693 xx t_(AV)"" (because t_(av) = (1)/(k))` `= 6*93 xx t_(av), i.e., 6*93` natural life TIMES. |
|
| 17. |
Number of monocloro structural isomers of : (i) , overset(Cl_(2)//h v) rarr"" (ii) ,overset(Cl_(2)//h v) rarr |
Answer» 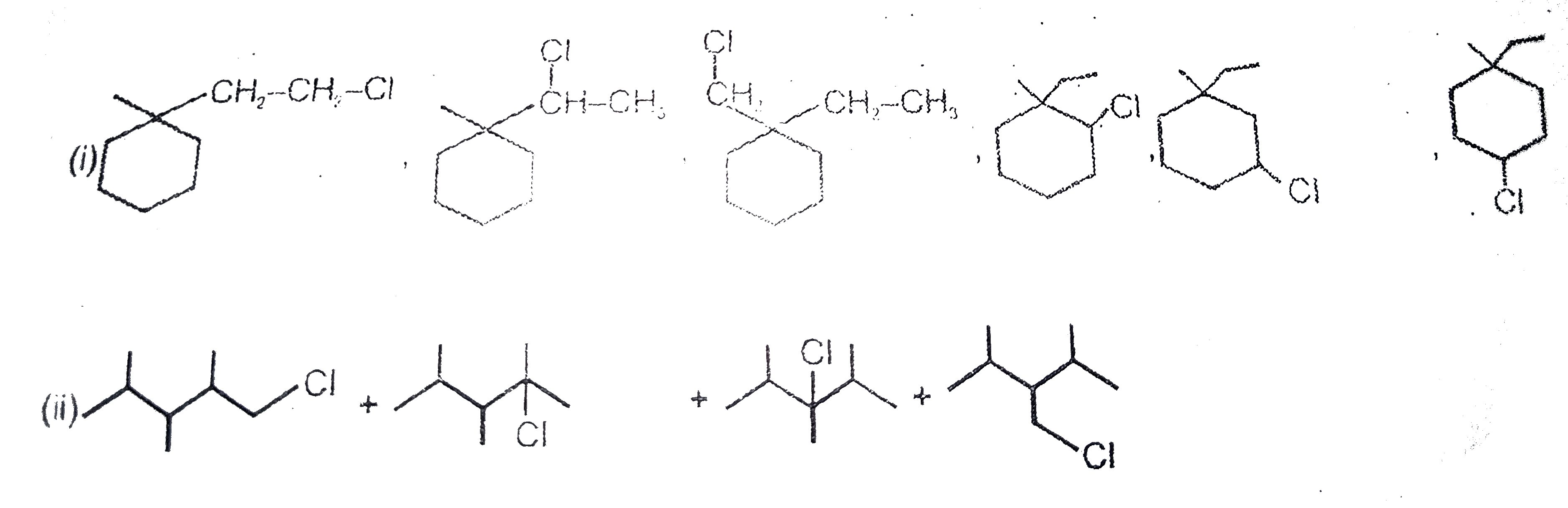
|
|
| 18. |
Number of natural life times (T_(av)) required for a first-order reaction to achieve 99.9% level of completion is : |
|
Answer» `2.3` |
|
| 19. |
Number of moles (P) of HIO_(4) used per moles of compound X is : |
|
Answer» 2 (B) has one amide linkage . (C) Oxidation products of aldohexose. (d) is REDUCTION productsof 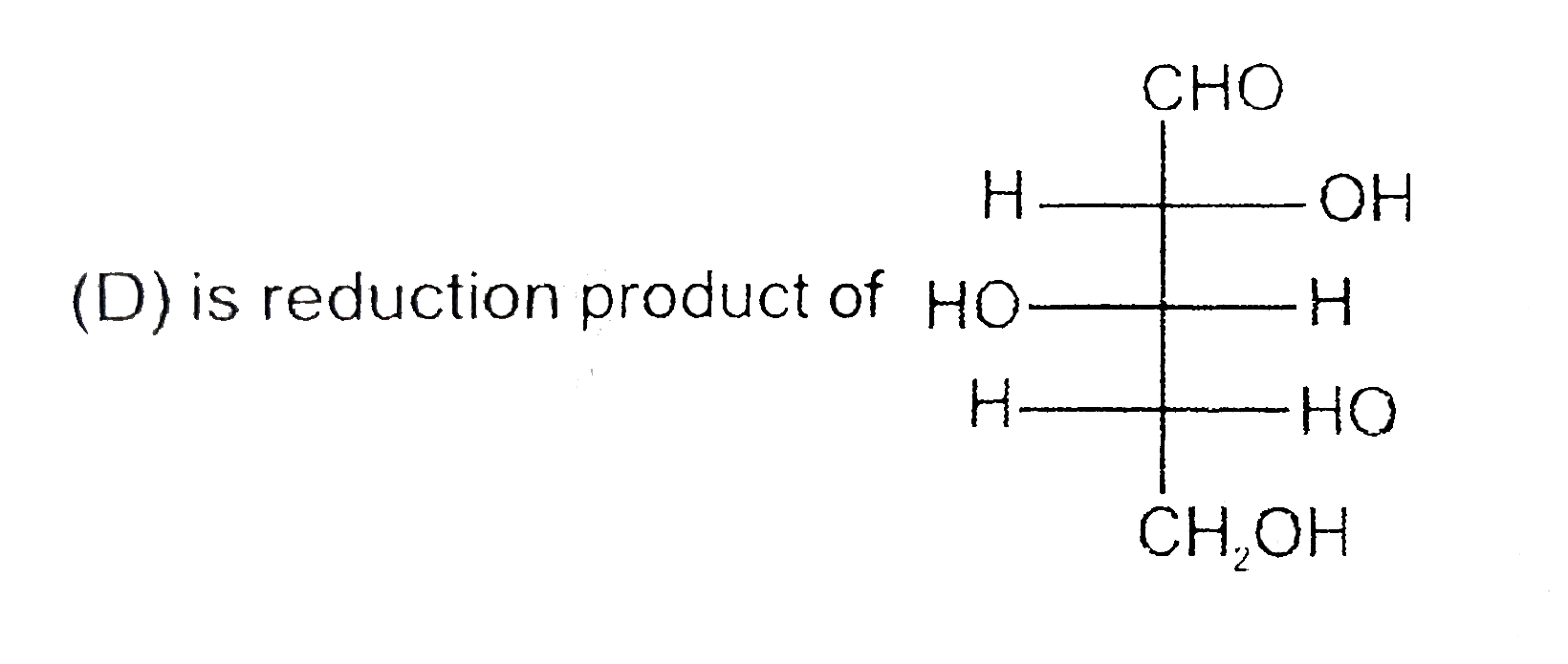
|
|
| 20. |
Number of moles of the solute per kilogram of the solvent is |
|
Answer» MOLE FRACTION |
|
| 21. |
Number of moles of RMgX consumed during reaction are? , underset(2.H_(3)O)overset(1.RMgX("excess"))rarr |
|
Answer» 2 |
|
| 22. |
Number of moles of oxygen required for the complete combustion of butane are |
|
Answer» 6 |
|
| 23. |
Number of moles of Na_(2)CO_(3) that should be dissolved in 4 litres of the solution to obtain 1 N Na_(2)CO_(3) solution is |
|
Answer» Solution :No. of moles of `Na_(2)CO_(3)` REQUIRED for 1 L of 1 N solution = 0.5 `THEREFORE` No. of moles of `Na_(2)CO_(3)` required for 4 L of 1 N solution `=4xx0.5=2.` |
|
| 24. |
Number of moles of [NH_4OH] required to coagulate 1mole of [Fe(OH)_3]Fe^(+3) are |
|
Answer» 3 MOLES of `NH_4OH` required to COAGULATE `[Fe(OH)_] Fe^(+3)` ion = 3 |
|
| 25. |
Number of moles of K_(2)Cr_(2)O_(7) reduced by one mole of Sn^(2+) ions is |
|
Answer» `1/3` |
|
| 26. |
Number of moles of K_(2)Cr_(2)O_(7) in acidic medium required to oxidize one mole of Cu_(3)P" to " CuSO_(4) " and " H_(3)PO_(4) is : |
|
Answer» <P>`11/6` |
|
| 27. |
Number of moles of hydrogen will required for complete hydrogenation of one mole of following compounds : |
|
Answer» `6` |
|
| 28. |
Number of moles of K_(2)Cr_(2)O_(7) can be reduced by 1 mole of Sn^(2+) ions is: |
|
Answer» `1//3` |
|
| 29. |
Number of moles of electrons in 4.2g of N^(3-) ion (nitride ion) is: |
|
Answer» 3 |
|
| 30. |
Number of moles of CH_3-OH in acidic medium react with one mole of glucose . |
|
Answer» 1 |
|
| 31. |
Number of moles of CaO required to remove hardness from 1000 litre water having 324 ppm of calcuim bicarbonate and 74.5 ppm of potassium chloride is : |
|
Answer» 8 |
|
| 32. |
Number of molecules or ions which has/have any bond angle of 120^(@)is CH_(3)^(-), CH_(3)^(o+), NH_(3), CO_(3)^(2-), BCl_(3), PCl_(5) |
|
Answer» 3 |
|
| 33. |
Number of moles, mass, volume, enthalpy, entropy and free energy are examples of _____ properties. |
|
Answer» |
|
| 34. |
Number of moles in 1 m^(3) gas at NTP are |
|
Answer» `4.46` `"1 MOLE = 22.4 L"` `therefore" MOLES present in 1000 L"=(1000)/(22.4)=44.6` |
|
| 35. |
Number of molecules in 1 litre of oxygen at NTP is: |
|
Answer» `(6.02xx10^(23))/(32)` |
|
| 36. |
Number of mole of Grignard comsumed per mol in given molecule.(When grignard reagent is in excess) |
Answer» 
|
|
| 37. |
Number of mole of HIO_(4) required per mol for complatr reaction of alpha-D-methylglycopyranoside. |
|
Answer» |
|
| 38. |
Number of mole of a substance present in 1 litre volume is known as: |
|
Answer» Activity |
|
| 39. |
Number of mole of a substance present in 1 litre volume is known as |
|
Answer» activity |
|
| 41. |
Number of metamers represent by molecular formula C_(4)H_(10)O is |
|
Answer» 4 `CH_(3)CH_(2)OCH_(2)CH_(3)(I),CH_(3)OCH_(2)CH_(2)CH_(3)(II) and CH_(3)OCH(CH_(3))_(2)(III) ` . Here I and II , I and III are PAIRS of METAMERS. |
|
| 42. |
Number of mole of 1 m^3 gas at NTP are : |
|
Answer» 44.6 |
|
| 43. |
Number ofmetalsamongfollowingwhich are obatainedbyelectromentallurgy in molten state are Li, Ba, Na,Al, Fe, Cu, Pb, Sn, Ag, Zn, Ca, Mg. |
|
Answer» |
|
| 44. |
Number of meso compounds are possible with molecular formula C_6H_12Cl_2? |
Answer» 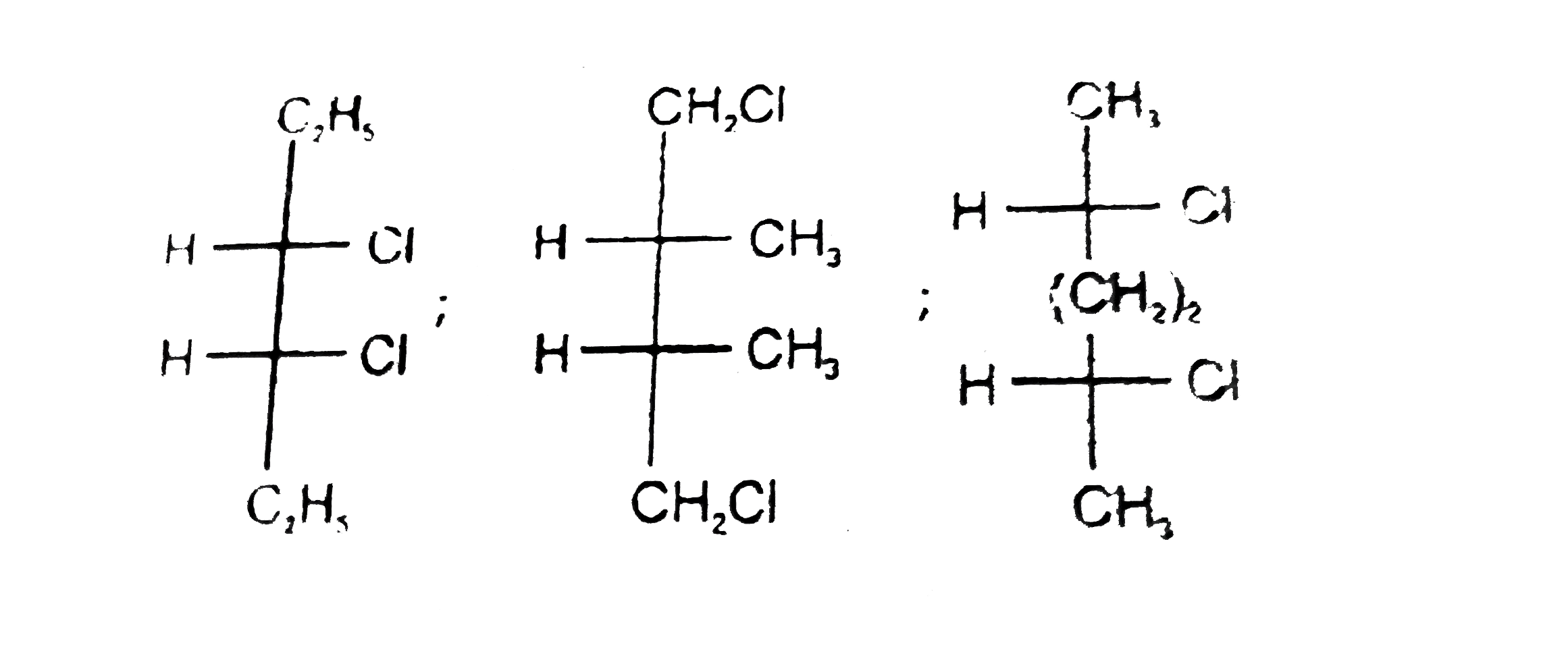
|
|
| 45. |
Number oflone pairs of electrons on Xe atoms XeF_(2),XeF_(4) and XeF_(6) molecules are respectively |
|
Answer» 3,2 and 1 
|
|
| 46. |
Number of lone pairs electrons on Xe atoms in XeF_(2), XeF_(4) and XeF_(6) molecules are respectively. |
|
Answer» 3,2 and 1 |
|
| 47. |
Number of lone pairs, sigma bonds are pi bonds in A are respectively: |
|
Answer» 1,3,1 |
|
| 48. |
Number of lone pairs of electrons on Xe atoms in XeF_(2), XeF_(4), XeF_(6) and XeO_(4) molecules are respectively |
| Answer» Answer :A | |
| 49. |
Number of lone pair and bond pairs present on Xe of XeOF_4molecule is |
|
Answer» 1,2 |
|