Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Nitrogen , oxygen and fluorine are the highly electronegative elements. When they are tied to a hydrogen atom to form covalent bond, the electrons of the covalent bond are shifted towards the more electronegative atom.This partially positively charged hydrogen atom forms a bond with the other electronegative atom. This bond is called as hydrogen bond and is weaker than covalent bond.For example, in HF molecule, thehydrogen bond exists between hydrogen atom of one molecule and fluorine atom of another molecule as depicted ---H^(delta+)-F^(delta-)---H^(delta+)-F^(delta-)---H^(delta+)-F^(delta-) Here, hydrogen bond acts as bridge between atoms which holds one atom by covalent bond and the other by hydrogen bond.Hydrogen bond is represented by a dotted line (---) while a solid line represents the covalent bond. Thus, hydrogen bond can be defined as the attractive force which binds hydrogen atom of one molecule with the electronegative atom (F,O and N) of another molecule. If boiling point of ortho-nitro phenol and para nitro phenol are t_1 and t_2 respectively and mixture of these two are heated at temperature 't' where 't' is between these two temperature.Vapours will significantly contains |
|
Answer» ORTHO NITRO phenol |
|
| 2. |
Nitrogen , oxygen and fluorine are the highly electronegative elements. When they are tied to a hydrogen atom to form covalent bond, the electrons of the covalent bond are shifted towards the more electronegative atom.This partially positively charged hydrogen atom forms a bond with the other electronegative atom. This bond is called as hydrogen bond and is weaker than covalent bond.For example, in HF molecule, thehydrogen bond exists between hydrogen atom of one molecule and fluorine atom of another molecule as depicted ---H^(delta+)-F^(delta-)---H^(delta+)-F^(delta-)---H^(delta+)-F^(delta-) Here, hydrogen bond acts as bridge between atoms which holds one atom by covalent bond and the other by hydrogen bond.Hydrogen bond is represented by a dotted line (---) while a solid line represents the covalent bond. Thus, hydrogen bond can be defined as the attractive force which binds hydrogen atom of one molecule with the electronegative atom (F,O and N) of another molecule. What statement is incorrect about D_2O (solid) |
|
Answer» It will float in `D_2O` liquid |
|
| 3. |
Nitrogen pentoxide decomposes according to equation : 2N_(2)O_(5)(g) rarr 4NO_(2)(g) + O_(2)(g). This first order reaction was allowed to proceed at 40^(@)C and the data below were collected : (a) Calculate the rate constant. Include units with your answer. (b) What will be the concentration of N_(2)O_(5) after 100 minutes ? (c ) Calculate the initial rate of reaction ? |
|
Answer» Solution :(a) `K = (2.303)/(t)log.(A_(0))/([A])` `k = (2.303)/(20 min)log.(0.400)/(0.289)` `k = 0.0163 min^(-1)` (b) `k = (2.303)/(t)log.(A_(0))/([A])` `=0.0163 - (2.303)/(100)log.(0.400)/([A])` `[A] = 0.078 M` (C ) Initial rate R `= k[N_(2)O_(5)]` `= 0.0163 min^(-1) xx (0.400 M)` `=0.00652 M min^(-1)` |
|
| 4. |
Nitrogen , oxygen and fluorine are the highly electronegative elements. When they are tied to a hydrogen atom to form covalent bond, the electrons of the covalent bond are shifted towards the more electronegative atom.This partially positively charged hydrogen atom forms a bond with the other electronegative atom. This bond is called as hydrogen bond and is weaker than covalent bond.For example, in HF molecule, thehydrogen bond exists between hydrogen atom of one molecule and fluorine atom of another molecule as depicted ---H^(delta+)-F^(delta-)---H^(delta+)-F^(delta-)---H^(delta+)-F^(delta-) Here, hydrogen bond acts as bridge between atoms which holds one atom by covalent bond and the other by hydrogen bond.Hydrogen bond is represented by a dotted line (---) while a solid line represents the covalent bond. Thus, hydrogen bond can be defined as the attractive force which binds hydrogen atom of one molecule with the electronegative atom (F,O and N) of another molecule. What would be correct about bonding in chloral hydrate C Cl_3CH(OH)_2? |
|
Answer» Only INTRA molecular H-BONDING 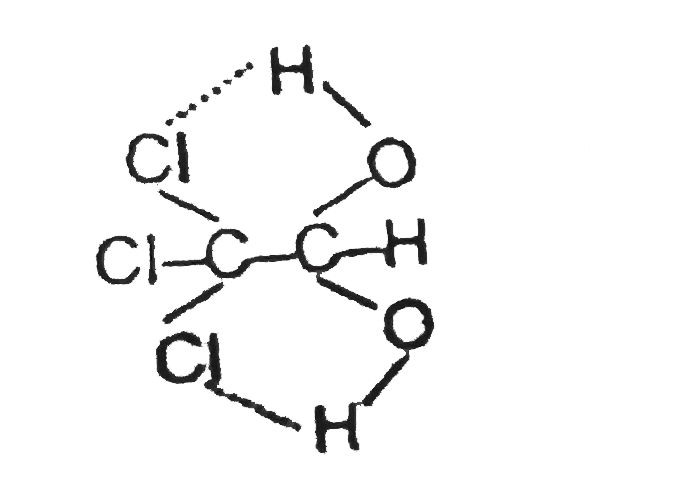
|
|
| 5. |
Nitrogen (N), phosporus(P), and potassium (K) are the main nutrients in the plant fertilizers. According to an industry convention , the numbers on the label refer to the mass % of N ,P_(2)O_(5), and K_(2)O,in that order. Calculate the N : P : K ratio of a 30 :10 : 10 fertilizer in terms of moles of each elements, and express it as x : y :1.0. (P = 31 , K = 39 ) |
|
Answer» |
|
| 6. |
Nitrogen oxides produced from the emission of automobiles and power plants. are the source of line airborne particles which lead to |
|
Answer» PHOTOCHEMICAL SMOG |
|
| 7. |
Nitrogen oxides photochemically react with unsaturated hydrocarbons to from_______. |
|
Answer» |
|
| 8. |
Nitrogen oxides emitted from the exhaust system of supersonic jet aeroplanes slowly deplete the concentration of ozone layer in upper atmosphere. Comment. |
|
Answer» Solution :There is a possibility that nitrogen oxides (particularly nitric oxide), which are emitted from the exhaust SYSTEMS of supersonic jet planes react with ozone. This PROCESS slowly depletes the concentration of ozone layer in the UPPER atmosphere. The REACTION involved is as under : `NO(g) + O_3(g) to NO_2(g) + O_2(g)` |
|
| 9. |
Nitrogen monoxide is |
|
Answer» neutral |
|
| 10. |
Nitrogen molecule is chemically less active because it has a________between two nitrogen atoms: |
|
Answer» SINGLE bond |
|
| 11. |
Nitrogen is relatively inert as compared to phosphorus. Why ? |
| Answer» Solution :P-P Single bonds in phosphorus are MUCH WEAKER than `N-=N` triple bond in `N_2`. THEREFORE, NITROGEN is relatively inert. | |
| 12. |
Nitrogen is relatively inert as compared to phosphorus. Why. |
| Answer» Solution :P-P Single bonds in phosphorus are much WEAKER than `N-=N` triple bond in `N_2`. THEREFORE, NITROGEN is relatively INERT. | |
| 13. |
Nitrogen is obtained when NaNO_2 reacts with |
| Answer» Answer :A | |
| 14. |
Nitrogen is relatively inactive element because is atom has a stable electronic configuration |
|
Answer» Its atom has a stable electronic CONFIGURATION |
|
| 15. |
Give reason: Nitrogen is less reactive at room temperature. |
| Answer» Solution :High BOND DISSOCIATION ENTHALPY makes nitrogen less reactive. | |
| 16. |
Nitrogen is comparatively inactive because : |
|
Answer» Of its stable ELECTRONIC configuration |
|
| 17. |
Nitroeng is chemically inert. Why? |
| Answer» SOLUTION :Although each of the above has same electronic distribution in bonding ORBITALS, NITROGEN is inert because of non-polar nature of bond and high `N-=N` dissociation energy. Rest all `(CO, CN^(-), NO^(+))` are polar. | |
| 18. |
Nitrogen is chemically inert due to |
|
Answer» LOW density |
|
| 20. |
Nitrogen is an essential constituent of all:. |
|
Answer» Proteins |
|
| 21. |
Nitrogen is a relatively inactive element because: |
|
Answer» Its ATOM has STABLE electronic configuration |
|
| 22. |
Nitrogen in the air is removed in the form of ________ is fisher-Ringe method |
|
Answer» `Mg_3N_2` |
|
| 23. |
Nitrogen (I) oxide is produced by: |
|
Answer» THERMAL DECOMPOSITION of AMMONIUM nitrate |
|
| 24. |
Nitrogen has unique ability to form P pi- P pi multiple bonds with itself and with other elements due to |
|
Answer» SMALL size and LOW electronegativity |
|
| 25. |
Nitrogen has several isotopes where atomic masses ranging from 10 to 25. Out of which ._(7)N^(17) is radioactive and is converted into ._(8)O^(17) by emission of |
|
Answer» Alpha particle |
|
| 26. |
Nitrogen gas is, ……………….. . |
|
Answer» INERT |
|
| 27. |
Nitrogen gas is absorbed by |
|
Answer» CALCIUM HYDROXIDE |
|
| 29. |
Nitrogen gas in atmosphere is separated industrially fromliquid air by ………………. . |
|
Answer» SIMPLE DISTILLATION |
|
| 30. |
Nitrogen gas is absorbed by: |
|
Answer» ALUMINIUM carbide |
|
| 31. |
Nitrogen forms the largest number of oxides as it is capable of forming stable multiple bonds with oxygen.They range from N_2O(O.S of nitrogen+1) through NO, N_2O_3,NO_2,N_2O_4 to N_2O_5(O.S of nitrogen +5). Following points are important regarding the study of oxides of nitrogen. (a)All oxides of nitrogen except N_2O_6 and endothermic as a large amount of energy is required to dissociate the stable molecule of oxygen and nitrogen. (b)The small electronegativity difference between oxygen and nitrogen make N-O bond easily breakable to give oxygen and hence oxides of nitrogen are said to be better oxidising agents. (c )Except N_2O_5, all are gases at ordinary temperature. N_2O_3 is stable only at lower temperature (253 K). (d) Except N_2O and NO which are neutral oxides, all are acidic oxides which dissolve in water forming corresponding oxy acids. (e)They are also good example for illustrating the concept the resonance. Identify the incorrect statement. |
|
Answer» In `N_2O_4`the N-N bond length is longer than the usual N-N single bond distance  (C )`:N-=underset(SP)N:tounderset(ddot)overset(ddot)O:` and the structure suggests that it will have some dipole moment. |
|
| 32. |
Nitrogen forms the largest number of oxides as it is capable of forming stable multiple bonds with oxygen. They range of N_(2)O (O.S of nitrogen +1) through NO, N_(2)O_(3),NO_(2),N_(2)O_(4) to N_(2)O_(5) (O.S of nitrogen +5). Following points are improtant regarding the study of oxides of nitrogen. (a) All oxides of nitrogen expect N_(2)O_(5) are endothermic as a large amount of energy is required to dissociate the stable molecule of oxygen and nitrogen. (b) The small electronegativity difference between oxygen and nitrogen make N-O bond easily breakle to give oxygen and hence oxides of nitrogen are said to be better oxidising agents. (c) Expect N_(2)O_(5), all are gases at ordinary temperature. N_(2)O_(3) is stable only at lower temperature (253K). (d) Expect N_(2)O and NO which are neutal oxides, all are acidic oxides which dissolve in water forming coresponding oxy acids. (e) They are also good example for illustrating the concept of resonance. Which of the following statements is corect for the oxides of nitrogen? |
|
Answer» DINITROGEN trioxide dissolves in potassium hydroxide FORMING potassium nitrate. |
|
| 33. |
Nitrogen forms the largest number of oxides as it is capable of forming stable multiple bonds with oxygen. They range from N_(2)O (O.S of nitrogen +1) through NO, N_(2)O_(3), NO_(2), N_(2)O_(4), " to " N_(2)O_(5) (O.S of nitrogen +5). Following points are important regarding the study of oxides of nitrogen (a) All oxides of nitrogen except N_(2)O_(5) are endothermic as a large amount of energy is required to dissociate the stable molecule of oxygen and nitrogen. (b) The small electronegativity difference between oxygen and nitrogen make N - O bond easily breakble to give oxygen and hence oxides of nitrogen are said to be better oxidising agents. (c) Except N_(2)O_(5), all are gases at ordinary temperature N_(2)O_(3) is stable only at lower temperature (253 K). (d) Except N_(2)O and NO which are neutral oxides, all are acidic oxides which dissolve in water forming corresponding oxy acids. (e) They are also good example for illustrating the concept of resonance Which of the following statements is correct for the oxides of nitrogen ? |
|
Answer» Dinitrogen trioxide dissolves in potassium hydroxide FORMING potassium nitrate |
|
| 34. |
Nitrogen forms the largest number of oxides as it is capable of forming stable multiple bonds with oxygen.They range from N_2O(O.S of nitrogen+1) through NO, N_2O_3,NO_2,N_2O_4 to N_2O_5(O.S of nitrogen +5). Following points are important regarding the study of oxides of nitrogen. (a)All oxides of nitrogen except N_2O_6 and endothermic as a large amount of energy is required to dissociate the stable molecule of oxygen and nitrogen. (b)The small electronegativity difference between oxygen and nitrogen make N-O bond easily breakable to give oxygen and hence oxides of nitrogen are said to be better oxidising agents. (c )Except N_2O_5, all are gases at ordinary temperature. N_2O_3 is stable only at lower temperature (253 K). (d) Except N_2O and NO which are neutral oxides, all are acidic oxides which dissolve in water forming corresponding oxy acids. (e)They are also good example for illustrating the concept the resonance. Which of the following statement is correct for the oxides of nitrogen ? |
|
Answer» DINITROGEN trioxide dissolves in potassium hydroxide forming potassium nitrate (B)`H_2O + 2NO_2 to HNO_2` (reducing agent ) +`HNO_3` (oxidising agent) (C )Neutral to litmus as neutral oxide (D)In the liquid state, `N_2O_4` tends to ionise `2NO_2 hArr N_2O_4 hArr NO^(+) + NO_3^-` |
|
| 35. |
Nitrogen forms p pi - p pi multiple bonds. Nitrogen exists as triply bonded diatomic gaseous molecule. Bond strength is very high which is responsible for inertness at ordinary conditions. The other members of nitrogen family form d pi- p pi bonding . Choose the correct answer : Maximum covalency of Sb will be |
|
Answer» 3 |
|
| 36. |
Nitrogen forms pentahalides. |
| Answer» SOLUTION :NITROGEN does not FORM PENTAHALIDES. | |
| 37. |
Nitrogen forms p pi - p pi multiple bonds. Nitrogen exists as triply bonded diatomic gaseous molecule. Bond strength is very high which is responsible for inertness at ordinary conditions. The other members of nitrogen family form d pi- p pi bonding . Choose the correct answer :Which is most acidic in nature ? |
|
Answer» `Bi_(2)O_(3)` |
|
| 38. |
Nitrogen forms p pi - p pi multiple bonds. Nitrogen exists as triply bonded diatomic gaseous molecule. Bond strength is very high which is responsible for inertness at ordinary conditions. The other members of nitrogen family form d pi- p pi bonding . Choose the correct answer : Thermal and electrical conductivity is highest in |
|
Answer» N |
|
| 39. |
Nitrogen forms N_2, but phosphorus is converted into P_4 from P, the reason is |
|
Answer» Triple bond is PRESENT between phosphrous ATOM |
|
| 40. |
Nitrogen forms five stable oxides having formulae N_2O, NO, N_2O_3, N_2O_4 and N_2O_5 . The formation of these oxides explains the |
|
Answer» LAW of definite PROPORTION |
|
| 41. |
Nitrogen forms five compounds with oxygen in which 1.0 g of nitrogen combines with 0.572, 1.14, 1.73, 2.28 and 2.85 g of oxygen respectively. Show that these figures agree with law of multiple proportions. |
| Answer» Solution :Masses of oxygen which combine with 1 G of N are in the ratio of `1:2:3:4:5`. | |
| 42. |
Nitrogen forms a number of oxides and oxoacids. Which of the following is a neutral oxide of Nitrogen?N_2 O, N_2 O_5, NO_2, N_2 O_4 |
|
Answer» `N_2O` |
|
| 43. |
Nitrogen forms a simple diatomic molecule but other elements of same group do not form. Explain. |
| Answer» Solution :Atomic size of nitrogen is very less. Hence .N. atom can approach close to another .N. atom. This FACILITATES the LATERAL overlapof the p-orbitals forming `pi` - BONDS. Due to large size, other atoms of the same GROUP form only single bonds, but not multiple bonds. | |
| 44. |
Nitrogen exists as diatomic molecule and Phosphrus as P_(4). Why? |
|
Answer» <P> Solution :Nitrogen has a TRIPLE bond between its two atoms because of its small size and high ELECTRONEGATIVITY. Phosphorus `P_(4)` has single bond, that is why it is tetra - atomic. |
|
| 45. |
Nitrogen forms a number of oxides and oxoacids. The gas liberated when calcium phosphide is treated with dil. HCI is |
|
Answer» `Cl_2` |
|
| 46. |
Nitrogen exists as diatomic molecule and phosphorus as P_4. Why ? |
|
Answer» Solution :Nitrogen because of its small size and HIGH electronegativity forms `p pi-p pi` multiple bonds. THUS `N_2` exists as gas at ROOM temperature. Phosphorus being larger in size than nitrogen does not form `p pi- p pi`multiple bonds. Hence, phosphorus exists as DISCRETE `P_4` molecule. |
|
| 47. |
Nitrogen exists as diatomic molecule and phosphorus as P_4. |
| Answer» Solution :Because of its SMALL size and high electronegativity nitrogen forms `p pi - p pi `multiple bonds. Therefore, it exists as a diatomic molecule having a triple bond between the two N-atoms. PHOSPHORUS, on the other hand, has larger size and LOWER electronegativity and usually does not form `p pi - p pi` multiple bonds with itself. Instead it PREFERS to form P-P SINGLE bonds and hence it exists as tetrahedral, `P_4` molecules. | |
| 48. |
Nitrogen exist as |
|
Answer» DIATOMIC molecule |
|
| 49. |
Nitrogen exist diatomic molecule and phosphorus as P_(4). Why ? |
| Answer» Solution :DUE to its small size and high ELECTRONEGATIVITY, nitrogen atom forms `ppi-ppi` multiple bonds with itself (triple BOND), so it EXISTS as a discrete diatomic molecule in elemental form, But phosphorus atom has large size and less electronegativity, so it as forms single bond instead of `ppi-ppi` multiple bond. THEREFORE, phosphorus exists as `P_(4)` molecule in its elemental form. | |
| 50. |
Nitrogen exhibits its group valency in |
|
Answer» SILVER nitrate |
|