Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Though nitrogen exhibit +5 oxidiation state, it does not form pentahalide. Why? |
| Answer» Solution :NITROGEN dose not haved - orbitals to EXPAND thecovalent beyond four. HENCE it dose not FORM pentahalide . | |
| 2. |
Nitrogen dioxide (NO_(2)) reacts with fluorine (F_(2)) to from nitrul fluoride (NO_(2)F). 2NO_(2)(g)+F_(2)(g)to 2NO_(2)F(g) Write the rate of reaction in terms of (i) rate of formation of NO_(2)F (ii) rate of disappearance of NO_(2) (iii) rate of disappearance of F_(2) |
|
Answer» |
|
| 3. |
Nitrogen does not form pentahalides. Give reason. |
| Answer» Solution :NITROGEN does not FORM pentahalides because it does not possess d-orbitals and hence cannot EXPAND its octet. | |
| 4. |
Nitrogen does not form pentahalides as it does not have ............ . |
| Answer» SOLUTION :d-orbitals | |
| 5. |
Nitrogen does not form any pentahalide like Phosphorus. Why? |
| Answer» SOLUTION :NITROGEN does not FORM pentahalides due to NON - availability of the d - orbitals in its valence SHELL. | |
| 6. |
Nitrogen does not combine directly with: |
|
Answer» Ca |
|
| 7. |
Nitrogen dioxide is released by heating |
|
Answer» `PB(NO_(3))_2` |
|
| 8. |
Nitrogen dioxide is not produced on heation |
|
Answer» `KNO_(3)` `{:(""2KNO_(3)overset(Delta)rarr 2KNO_(2) + O_(2)),(2 Pb(NO_(3))_(2)overset(Delta)rarr 2PbO + 4 NO_(2)+O_(2)),(2Cu(NO_(3))_(2) rarr 2 CuO + 4 NO_(2) + O_(2)),(" "2AgNO_(3) overset(Delta)rarr 2AG + 2NO_(2) + O_(2)):}` |
|
| 9. |
Nitrogen dioxide cannot be obtained by heating : |
| Answer» Answer :A | |
| 10. |
Nitrogen dioxide and sulphur dioxide have some properties in common. Which property is shown by one of these compounds, but not by the other ? |
|
Answer» Is soluble in water |
|
| 11. |
Nitrogen dioxide |
|
Answer» dissolves in water FORMING nitric ACID |
|
| 12. |
Nitrogen dioxide and sulphur dioxide have some properties in common. Which property is shown by one of these compounds, but not by the other? |
|
Answer» Is solubole in WATE |
|
| 13. |
Nitrogen differs from rest of the members on the account of various factors. Which of the following properties can be classified as anomalous properties of nitrogen ? |
|
Answer» BOND enthalpy of `N -= N` is 941.4 kJ/mol |
|
| 14. |
Nitrogen differs from other elements among the VA group, due to |
|
Answer» Small ATOMIC SIZE |
|
| 16. |
Nitrogen containing organic compound when fused with sodium metal forms : |
| Answer» Solution :N//A | |
| 17. |
Nitrogen can be purified from the impurities of oxides of nitrogen and ammonia by passing through: |
|
Answer» CONC. HCl |
|
| 18. |
Nitrogen can form..........type of oxides. |
| Answer» Solution :`NO, N_(2)O, NO_(2), N_(2)O_(3), N_(2)O_(4),N_(2)O_(5)` | |
| 19. |
Nitrogen can form N^(3-) ion because of |
|
Answer» HIGH electronegativity |
|
| 20. |
Nitrogen cannot form NCl_5 due to______. |
| Answer» SOLUTION :The ABSENCE of d-orbitals(2D) in the VALENCY SHELL(L-shell) of N-atom. | |
| 21. |
Nitrogen can be purified from the impurities of oxides of nitrogen and ammonia by passing through : |
|
Answer» |
|
| 22. |
Nitrogen can be obtained from air by removing |
|
Answer» OXYGEN |
|
| 23. |
Nitrogenatomis amines is |
|
Answer» `SP^(2)` - hydridised |
|
| 24. |
Nitrogen atom in ammonia undergoes _______ hybridization and the geometry of the molecule is______ . |
| Answer» SOLUTION :`sp^3` PYRAMIDAL | |
| 25. |
Nitrogen atom in ammonia undergoes .......... hybridization and the geometry of the molecule is ............... |
| Answer» SOLUTION :`sp^3` PYRAMIDAL | |
| 26. |
Nitrogen and phosphorus are elements in the same group but property of catenation is shown only by phosphorus, why? |
| Answer» Solution :NITROGEN has high bond dissociation energy and forms triple bond in `N-= N`. THEREFORE it cannot SHOW the PROPERTY of CATENATION. Phosphorus has low bond dissociation energy and forms P-P bond which can extend further to exhibit catenation. | |
| 27. |
Nitroethane can exhibit one of the following kind of isomerism |
|
Answer» Metamerism 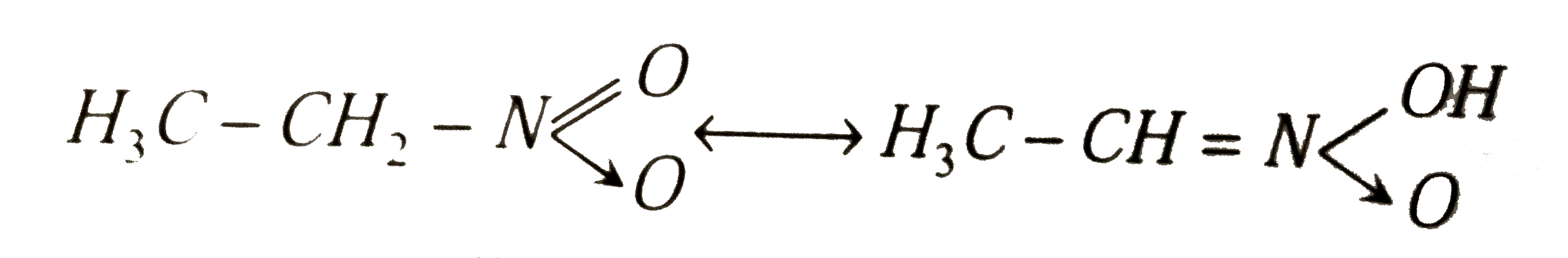
|
|
| 28. |
Nitrocompounds are |
|
Answer» DERIVATIVES of alkane |
|
| 29. |
Nitroethane on reduction by Sn//HCl gives nitrosoethane. |
| Answer» SOLUTION :ETHANAMINE | |
| 30. |
Nitrocompound (A) on reaction with nitrous acid gives a compound (B). B gives a red solution with NaOH. The compound (A) is |
|
Answer» `CH_(3)CH_(2)NO_(2)` |
|
| 32. |
Nitrobezene is formed as the major product along with a minor product in the reaction of benzene with a hot mixture of nitric acid and sulphuric acid. The minor product consists of carbon: 42.86%, hydrogen: 2.40%, nitrogen: 16.67%, and oxygen: 38.07% (i) Calculate the empirical formula of the minor product, (ii) when 5.5 gm of the minor product is dissolved in 45 gm of benzene, the boioling point of the solution is 1.84^(@)C higher than that of pure benzene. Calculate the molar mass of the minor product and determine its molecular its molecular and structural formula.(Molal boiling point elevation constant of benzene is 2.53 K kg mol^(-1).) |
|
Answer» Solution :The rations of atoms in the minor products are : `C:H:N:O``{:( ::(42.86)/(12):(2.40)/(1):(16.67)/(14):(38.07)/(16)),( ::3.57: 2.40 : 1.19 : 2.38), ( :: 3 : 2: 1: 2):}` Empirical formula of the minor PRODUCT: `C_(3)H_(2)NO_(2)` Molar empirical formula mass of the minor product is `(3xx12+2xx1+1xx14+2xx16)` gm `mol6(-1)` `= 84 gm mol^(-1)` Let M be the molar mass of the minor product. For `5.5` gm of the minor product dissloved in 45 gm benzene, the molality of solution is given by `m = (55gm//M)/0.045 kg)` Subsituting this in the expression of ELEVATION of boiling point, we get`DELTA T_(a) = K_(b)m` `1.84 K = (2.53K kg mol^(-1))` `((55gm//M)/(0.045kg))` `M = ((2.53xx55)/(1.84xx0.045))gm mol^(-1)` `=168gm mol^(-1)` Number of unit of empirical formula in the moleculat formula `= (168gm mol^(-1))/(84gm mol^(-1))=2` Hence, the molecular formula of the minor product is `2(C_(3)H_(2)NO_(2))`, i.e., `C_(6)H_(4)(NO_(2))` . The product is m-dinitrobenzene. |
|
| 33. |
Nitrobenzene when reduced with Zn and NH_(4)OH yields a product (A). Identify (A). Will (A) reduce Tollen's reagent? |
Answer» Solution :The product (A) is PHENYL hydroxylamine. It reduces Tollen.s reagent.  This reaction is MULLIKEN Test for DISTINGUISHING `-NO_(2)` group. |
|
| 34. |
Nitrobenzene to Hydrazobenzene. Here the reagent is |
|
Answer» `Sn+HCl` |
|
| 35. |
Nitrobenzene to benzoic acid |
Answer» SOLUTION :
|
|
| 37. |
Nitrobenzene on reduction . With Al - Hg and water gives : |
|
Answer» Azobenzene |
|
| 38. |
Nitrobenzene on reaction with HNO_(3)//H_(2)SO_(4) on 80^(@)-100^(@)C forms which one of the following products |
|
Answer» 1,4-Dinitrobenzene |
|
| 39. |
Nitrobenzene on reduction with Zn//NH_4 Cl gives |
|
Answer» ANILINE |
|
| 40. |
Nitrobenzene on reaction with conc. HNO_(3)//H_(2)SO_(4) at 80-100^(@)C forms which one of the following products? |
|
Answer» 1,4-Dinitrobenzene |
|
| 41. |
Nitrobenzene on reaction with conc. HNO_3//H_2SO_4 at 80-100^@C forms which one of the following products |
|
Answer» TNT 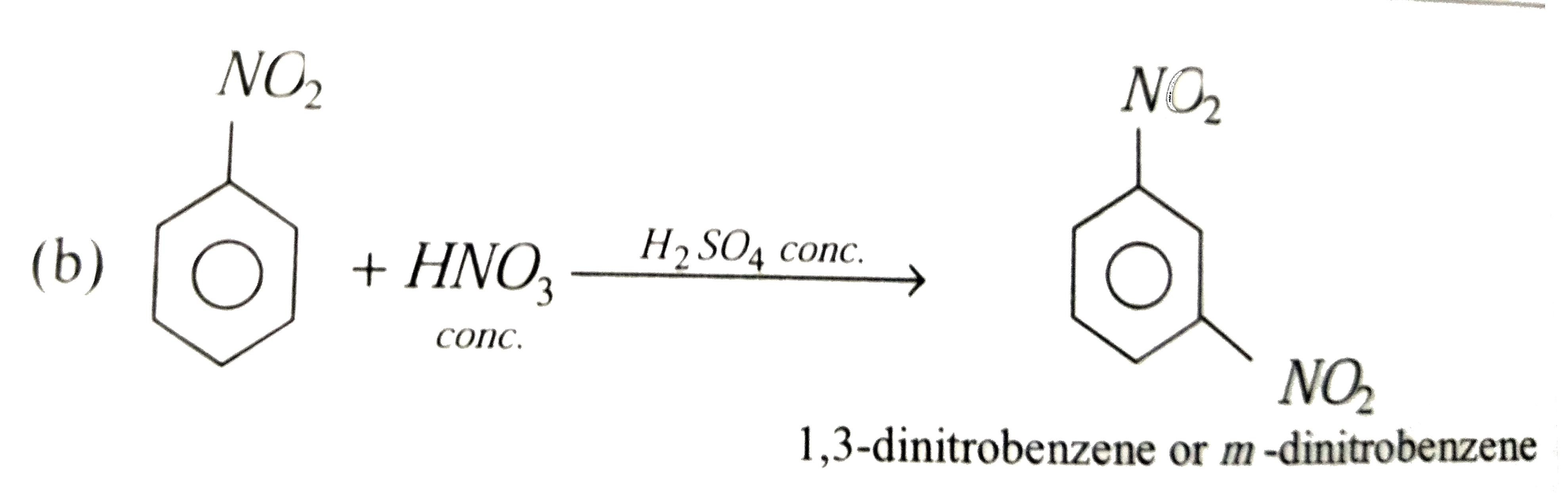
|
|
| 42. |
Nitrobenzene on reaction with at 80-100^(@)C form which one of the following products? |
|
Answer» 1,4-dinitrobenzene 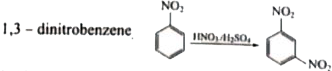
|
|
| 43. |
Nitrobenzene on reaction with conc. HNO_3//H_2SO_4 at 80 - 100^@C forms which one of the following products ? |
|
Answer» 1,4- DINITROBENZENE |
|
| 44. |
Nitrobenzene on further excessive nitration gives |
|
Answer» Trinitrobenzene  Further substitution is checked because of two highly deactivating `- NO_(2)` GROUPS which DECREASE the reactivity of the RING TOWARDS electrophilic subtitution considerably. |
|
| 45. |
Nitrobenzene is subjected to reduction with zinc dust and ammonium chloride. The main product formed will be: |
|
Answer» Benzenamine |
|
| 46. |
Nitrobenzene on electrolytic reduction is strongly acidic medium gives |
|
Answer» aniline |
|
| 47. |
Nitrobenzene on alkaline medium reduction gives………………….. |
|
Answer» |
|
| 48. |
Nitrobenzene is formed as the major product along with a minor product in thereaction of benzene with a hot mixture of HNO_3 and H_2SO_4. The minor product consists of C: 42.86%, H: 2.40%, N: 16.67% and 0:38.07%. (i) Calculate the empirical formula of the minor product, (ii) when 5.5 g of the minor product is dissolved in 45 g of benzene, the b.p. of the solution is 1.84^@Chigher than that of pure benzene. Calculate the molecular weight of the minor product and determine its molecular and structural formula. K_b(C_6H_6) = 2.53 K kg "mol"^(-1) |
| Answer» SOLUTION :`C_3H_2NO_2`, 168, m-dinitrobenzene | |
| 49. |
Nitrobenzene on eelctrolutic reduction gies :. |
|
Answer» Azobebzene |
|