Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Nitrobenzene is a versatile compound that may be converted inot a wide variety of subsituted benzenes. Five such synthesis are shown below. In each reaction box above an arrow write letters designating the reagents and conditions, selected from the list at the bottom of the page, that would effect the transformation. The reagents must be written in the answer box in the correct order of their use. You may assume appropriate heating or cooling takes place, and more than one equivalent of the reagent may be used if needed. {:(,"Reagents",,"Reagents",),(A,H_(2)","Ni "catalyst",F,Cl_(2) " & " FeCl_(3),),(B,KBr " & " Cu_(2) Br_(2),G,NaOH 10 % " solution",),(C,KCN " & " Cu_(2) (CN)_(2),H,(CH_(3)CO)_(2)O", pyridine",),(D,HNO_(2) 0^(@)C,I,HNO_(3)//H_(2)SO_(4),),(E,CH_(3)I " & pyridine",,,):} |
|
Answer» |
|
| 2. |
Nitrobenzene does not undergo Friedel - Crafts reaction - Give reason. |
| Answer» Solution :NITROBENZENE does not UNDERGO Friedel - Crafts REACTIONS due to the strong DEACTIVATING nature of `-NO_2` GROUP. | |
| 3. |
Nitrobenzene combines with hydrogen in the presence of platinum to produce. |
|
Answer» Toluene |
|
| 4. |
Nitrobenzene can be prepared from benzene diazonium chloride by using the reagents |
|
Answer» `HBF_(4)//NaNO_(2)` |
|
| 5. |
Nitrobenzene does not undergo Friedel-Crafts alkylation. Give reasons. |
| Answer» Solution :Due to POWERFUL electron WITHDRAWING effect of the `-NO_(2)` group, the benzene ring in `C_(6)H_(5)NO_(2)` is deactivated towards Friedel CRAFT’s alkylation and hence it is USED as a solvent in MANY Fridel-Craft’s reaction. | |
| 6. |
Nitrobenzene can be prepared from benzene by using a mixture of cone. HNO_3 and conc. H_2SO_4 . In the nitrating mixture HNO_3acts as a |
|
Answer» base |
|
| 7. |
Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO_3 and conc. H_2SO_4. In the nitrating mixture, HNO_3 acts as a |
|
Answer» Base 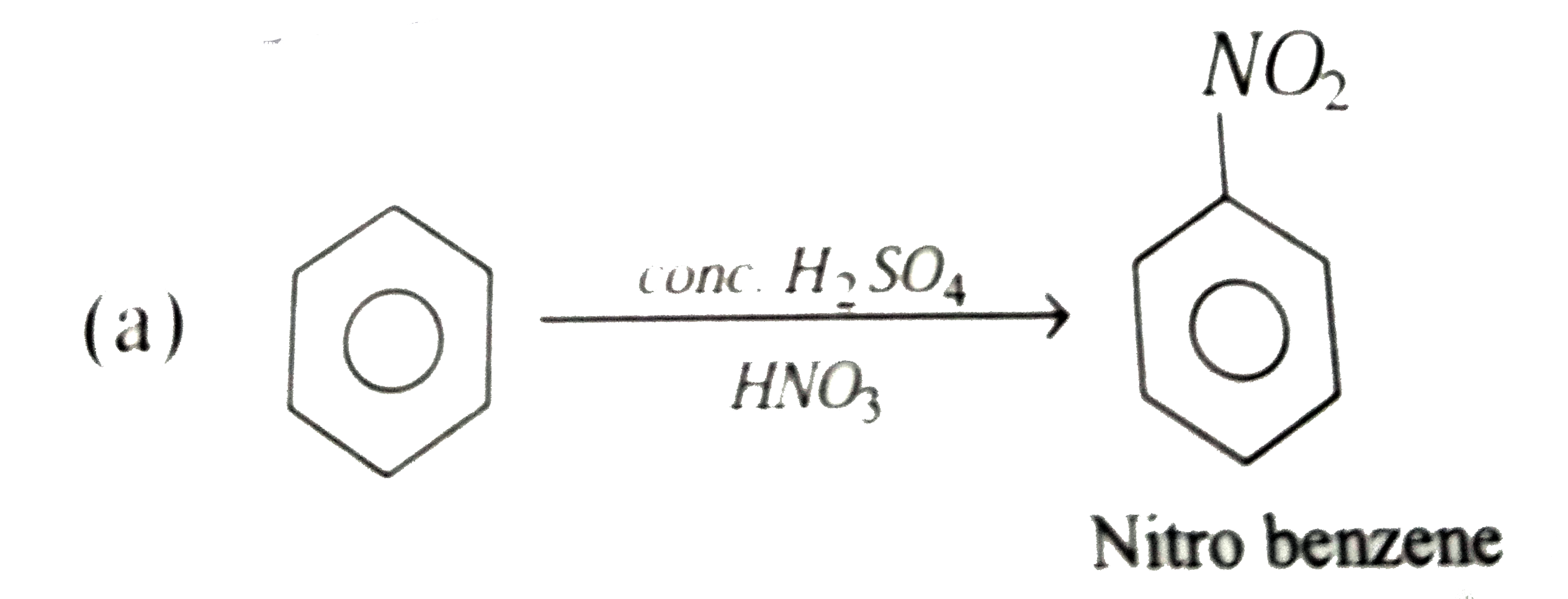
|
|
| 8. |
Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO_(3) and conc. H_(2)SO_(4). In the nitrating mixture HNO_(3) acts as a |
|
Answer» base |
|
| 9. |
Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO_(3) and conc. H_(2)SO(4). In the nitrating mixture, HNO_(3) acts as a |
|
Answer» base |
|
| 10. |
Nitrobenzene can be prepared from benzene by using a mixture of conc. HNO_(3) and conc. H_(2)SO_(4). In the nitrating mixture, nitric acid acts as a |
|
Answer» base In this reaction nitric acid acts like a base. |
|
| 11. |
Nitrobenzenen can be prepared from benzene by using a mixture of conc HNO_(3) and conc. H_(2)SO_(4) in the nitrating mixture. Nitric acid acts as a |
|
Answer» BASE |
|
| 12. |
Nitrobenzene can be converted to aniline by the following reagents : |
| Answer» Answer :D | |
| 13. |
Nitrobenze on electrolytic reduction in strongly acidic medium gives |
|
Answer» aniline 
|
|
| 14. |
Nitroaniline is subjected to the treatement of various reagents in the following sequence (i) NaNO_(2)//HCl (1280 K) (ii) KI (iii) Cu Powder. The product is |
|
Answer» 3, 3' Diaminobiphenyl 
|
|
| 15. |
Nitroation of nitrobenzene results in |
|
Answer» o-dinitrobenzene |
|
| 16. |
Nitroalkanes are reduced by Zn+ NH_(4)Cl gives |
|
Answer» `1^(@)`- amines |
|
| 17. |
Nitroalkanes can be reduced by using __________. |
| Answer» Answer :D | |
| 18. |
Nitroalkane is reduced by stannous chloride and HCl gives |
|
Answer» ` 2^(@)` - amine |
|
| 21. |
Nitroalkane condensedwithaldehydeandketoneto fromnitroalchol. The maincondition ofnitroalkane is |
|
Answer» absenceof `ALPHA` -H atoms |
|
| 22. |
Nitroalkane and alkyl nitrite can be distinguished by the action of: |
|
Answer» alkali |
|
| 24. |
Nitro group in nitrobenzene is : |
|
Answer» ORTHO director |
|
| 25. |
Nitrocompounds are reduced to amines by |
|
Answer» PASSING HYDROGEN GAS in the presence of FINELY divided Ni. |
|
| 26. |
Nitro benzene cannto be prepared from Bromo benzene by direct nitration. Give reason. |
| Answer» SOLUTION :Nitro benzene cannot be PREPARED from Bromo benzene because the BROMINE directly attached to the benzene ring cannot be CLEAVED easily. | |
| 27. |
Nitro benzene on reduction with SnCl_(2)+KOH gives……………… |
|
Answer» |
|
| 28. |
Nitro benzene cannot be prepared from Bromo benzene by direct nitration. Give reason. |
|
Answer» |
|
| 29. |
Nitro-acinitro tautomerism is exhibited by………….. |
|
Answer» NITRO methane |
|
| 30. |
Nitrite (NO_(2)^(Θ)) interfers in the ring -test of nitrate (NO_(3)^(Θ)) some of the following reagent can be used for the removed of nitrate |
|
Answer» `AgF` |
|
| 31. |
Nitrite and mitrate bothrespond toring test Nitrate areremoved bymeating with |
|
Answer» CONC `HNO_(3)` |
|
| 32. |
Nitriles react with SnCl_(2) and HCl followed by hydrolysis give aldeyhydes. What is this reaction known as ? |
| Answer» SOLUTION :Stephen.s REACTION | |
| 33. |
Nitric oxide reacts with hydrogen to give nitrogen and water (2" NO"+2" H"_(2)to"N"_(2)+2" H"_(2)"O"). The kinetics of this reaction is explained by the following steps : (i) 2 "NO"+"H"_(2)to"N"_(2)+"H"_(2)"O"_(2)("slow")"""(ii) ""H"_(2) "O"_(2)+"H"_(2)to2" H"_(2)"O"("fast") What is the predicted rate law ? |
|
Answer» |
|
| 34. |
Nitrile rubber is a copolymer of : |
|
Answer» isobutylene and isoprene |
|
| 36. |
Nitric oxidereacts withH_(2)accordingto thereaction 2NO_((g))+ 2H_(2(g)) to N_(2(g))+ 2H_(2)O_((g)) What isthe relationshipamong (d[NO])/(dt) = (d[H_(2)])/(dt) = (d[N_(2)])/(dt)" and" (d[H_(2)O])/(dt)? |
|
Answer» Solution :GIVEN :`2NO_((G)+ 2H_(2(g)) toN_(2(g))+ 2H_(2)O_((g))` Therelationshipamong therate ofconsumption of thereactants and therate offormationof products isas follows: Rateof reaction`:R= - (1)/(2) (d[NO])/(dt) = (1)/(2) (d[H_(2)])/(dt) = (d[N_(2)])/(dt)= (1)/(2) (d[H_(2)O])/(dt)` |
|
| 37. |
Nitric oxide reacts with bromine as :2NO(g)+Br_(2(g))hArr 2NOBr(g)When 0.087 mol of NO and 0.0437 mol of Br_(2) are mixed in a closed container at constant temperature, 0.0518 mol of NaBr is obtained at equilibrium. The equilibrium constant is : |
|
Answer» `12.86` |
|
| 38. |
Nitric oxide (NO) reacts with oxygen to produce nitrogen dioxide 2NO_((g))+O_(2(g))rarr 2NO_(2(g)) If the mechanism of reaction is NO+O_(2)overset(K)hArr NO_(3) (fast) NO_(3)+NOoverset(K_(1))hArr NO_(2)+NO_(2) (slow) |
|
Answer» Rate `K'[NO][O_(2)]` |
|
| 39. |
Nitricoxide, NO reacts with oxygen to produce nitrogen dioxide : 2" NO"(g)+"O"_(2)(g)to2"NO"_(2)(g) What is the predicted rate law, if the mechanism is : "NO"+"O"_(2)overset(k)iff"NO"_(3)("fast") "NO"_(2)+"NO"overset(k_(1))to"NO"_(2)+"NO"_(2)("slow") |
|
Answer» From fast step, EQM. CONST. `k=([NO_(3)])/([NO][O_(2)])""…(ii)` Substituting the value of `[NO_(3)]" from (ii) in (i), we get: Rate"=k'[NO]^(2)[O_(2)]` |
|
| 40. |
Nitric oxide (NO) reacts with oxygen to produce nitrogen dioxide : 2NO(g)+O_(2)(g)to2NO_(2)(g) What is the predicated rate law if the mechanism is {:(NO(g)+O_(2)(g)hArrNO_(3)(g),,("fast")),(NO_(3)(g)+NO(g)to2NO_(2)(g),,("slow")):} |
|
Answer» Solution :From the slow step, Rate `=k_(1)[NO_(3)][NO]`…………`(i)` From fast step EQULIBRIUM constant `(K)=([NO_(3)])/([NO][O_(2)])`…………`(II)` Substituting the VALUE of `[NO_(3)]` from equation `(ii)` into equation `(i)`, we get Rate `=k.[NO]^(2)[O_(2)]` |
|
| 41. |
Nitric oxide (NO) is paramagnetic in gaseous state |
|
Answer» GASEOUS state |
|
| 42. |
Nitric oxide is prepared by the action of cold dil. HNO_3 on: |
|
Answer» Fe |
|
| 43. |
Nitric oxide is prepared by the action of cold dil. HNO_3 on |
|
Answer» Cu |
|
| 44. |
Nitric oxide becomes brown when released in air. Why ? |
|
Answer» `2NO+O_(2) rarr 2NO_(2)` |
|
| 45. |
Nitric oxide is: |
|
Answer» ACIDIC TOWARDS litmus |
|
| 46. |
Nitric acid whether dilluted or concentrated : |
|
Answer» REACTS with AL to GIVE `H_2` |
|
| 47. |
Nitric acid prepared in large scales using, ……………… . |
|
Answer» OSTWALD's PROCESS |
|
| 48. |
Nitric acid is the most important oxi-acid formed by nitrogen. It is one of the major idustrial chemicals and is widely used. Nitric is manufactured by ostwald process in which catalytic oxidation of ammonia is done in following sequence as shown by reactions 4 NH_(3) (g)+50_(2)(g) overset("Pt/Rh")underset("Catalyst")(rarr) 4 NO(g)+6 H_(2)O(g) ...(i) 2NO(g)+O_(2)(g) overset(1120 K)(rarr) 2NO_(2)(g) ...(ii) 3 NO_(2)(g)+H_(2)O(l) rarr 2 HNO_(3)(aq)+NO(g) ...(iii) In this process the aqueous nitric acid is obtained which can be concentrated by distillation to ~ 68.5% by weight. Then concentration to 98% acid can be achieved by dehydration with concentrated sulfuric acid. If 170 kg of NH_(3) is heated in excess of oxygen, then the volume of H_(2)O(l) produced in 1st reaction at STP is (rho_(H_(2)O)=1 g//mL) |
|
Answer» `33.6xx10^(3) L` `:.` moles of `H_(2)O` formed `=6/4xx10xx10^(3)` `:.` mass of `H_(2)O` formed `=6/4xx10xx10^(3)xx18` `:.` volume of `H_(2)O` formed `=15xx18L =270 L` |
|
| 49. |
Nitric acid is the most important oxi-acid formed by nitrogen. It is one of the major idustrial chemicals and is widely used. Nitric is manufactured by ostwald process in which catalytic oxidation of ammonia is done in following sequence as shown by reactions 4 NH_(3) (g)+50_(2)(g) overset("Pt/Rh")underset("Catalyst")(rarr) 4 NO(g)+6 H_(2)O(g) ...(i) 2NO(g)+O_(2)(g) overset(1120 K)(rarr) 2NO_(2)(g) ...(ii) 3 NO_(2)(g)+H_(2)O(l) rarr 2 HNO_(3)(aq)+NO(g) ...(iii) In this process the aqueous nitric acid is obtained which can be concentrated by distillation to ~ 68.5% by weight. Then concentration to 98% acid can be achieved by dehydration with concentrated sulfuric acid. If 180 L of water completely reacts with NO_(2) produced to form nitric acid according to the above reactions, then the volume of air required at STP containing 20% of NH_(3) is (rho_(H_(2)O)=1 g//mL) |
|
Answer» `1.56xx10^(6) L` `:.` moles of `NO_(2)` required `=30xx10^(3)=` mole of `NH_(3)` required `:.` volume of air at `STP=(30xx10^(3)xx22.4)/0.2=3.36xx10^(6) L`. |
|
| 50. |
Nitric acid oxidises sulphur to: |
|
Answer» `SO_2` |
|