Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
NO is purified by |
|
Answer» ABSORPTION in `(NH_4)_2 SO_4` solution |
|
| 2. |
No gas is liberated when the following HX is treated with MnO_(2)and con .H_(2)SO_(4) |
|
Answer» HCl |
|
| 3. |
No correlation exists between the (R) and (S) designation and the direction of rotation of planepolarised light. Justify the statement. |
Answer» Solution :The rotation of a compound, `[(+) or (-)]` is something that we measure in the laboratory with a polarimeter and it depends on how the molecule interacts with light. On the other hand, the (R) and (S) designation is our own artifical way of describing how the atoms or groups are ARRANGED in space around a chiral CENTRE, i.e., the configuration of a particular chirality centre. Therefore, no necessary correlation exsists between the (R) and (S) designation and the direction of rotation of plane-polarised light. DEXTROROTATORY or (+)- compounds may have R or S configuration. Similarly, LEVOROTATORY or (-)-compounds may have R or S configuration.  . .
|
|
| 4. |
No chemical compound of helium is known Or Helium forms no real chemical compound. |
| Answer» Solution :Firstly, He does not contain d-orbitals in the valence shell and HENCE electrons cannot be promoted to higher energy levels like that in Xe to form bonds. Therefore, helium does not form chemical compounds. SECONDLY, IONIZATION ENTHALPY of He is sufficiently higher (2372 kJ `mol^(-1)`) than those of Xe (1170 kJ `mol^(-1)`) and `O_(2)` (1175 kJ `mol^(-1)`). | |
| 6. |
NO and NO_2 are: |
|
Answer» EVEN ELECTRON molecules |
|
| 8. |
N,N-dimethyl butanamine-2 contains |
|
Answer» six `SP^(3)` hybridised carbon atoms |
|
| 9. |
N,N - dimethylanilinium acetate obtainedfrom |
|
Answer»

|
|
| 10. |
N,N dimethyl acetamide is |
|
Answer» `(CH_(3))_(2)NCOCH_(3)` |
|
| 11. |
N,N- dimethyl ethanamide on reduction with LiAIH_4 gives |
|
Answer» N- methylethanamine |
|
| 12. |
N,N-dimethyl aniline on coupling with C_(6)H_(5)N_(2)Cl yields |
|
Answer» 4-(N,N-dimethyl) amino azo benzene |
|
| 13. |
N,N-Diethyl-3-methylbenzamide (also called N,N-diethyl-m-toluamide. Or DEET) is used many insect repellants. Write its structure. |
Answer» SOLUTION :
|
|
| 14. |
Nitrous acid reacts with all classes of amines. The product obtained from these reactions depend on whether primary, secondary or tetriary and ,wheather the amine or aliphatic or aromatic. Aliphalic primary amines react with acid (NaNO_(2)+HCl) to form alcohol as major product. In , addition to alcohol, alkene and alkyl halides are also formed as minor product. Certain cyclic primaryamines can undergo either ring expansion or ring contraction reactions on treatment with acid. This reaction is called Demajanov ring expansion or contraction. Two compounds (A) and (B) are treated with nitrous acid. |
|
Answer» |
|
| 15. |
Nitrous oxide may easily be distinguished from nitric oxide by: |
|
Answer» adding water and shaking |
|
| 16. |
Nitrous acid reacts with H_2SO_4 to give: |
|
Answer» `NO_2 + SO_2` |
|
| 17. |
Nitrous acid reacts with H_(2)SO_(4) give |
|
Answer» `NO + SO_(2)` |
|
| 18. |
Nitrous acid reacts with all classes of amines. The nature of products obtained from these reactions depends on whether the amine is primary, secondary or tertiary. It also depends on whether the amine is aliphatic or aromatic. Aliphatic primary amines react with nitrous acid (NaNO_(2)+HCl) to form alcohol as the major product. In addition to alcohol, alkene and alkyl halide are also formed as minor products. Certain cyclic primary amines undergo either ring expansion or ring contraction on treatment with nitrous acid. this reaction is called Demjanov ring expansion or contraction. nitrous acid also reacts with nitroalkanes having acidic hydrogen. Q. Which of the following product(s) will be obtained when n-propyl amine is treated with NaNO_(2) and HCl ? |
|
Answer» `CH_(3)-underset(OH)underset(|)(C)H-CH_(3)` |
|
| 19. |
Nitrosoamines (R_2N-N=O) are insoluble in water. On heating with conc. H_2SO_4 , they give secondary amines. The reaction is called |
|
Answer» Liebermann NITROSO reaction |
|
| 20. |
Nitrosonitroalkaneis obtainedby 2^(0) - nitroalkane with |
|
Answer» `HNO_(3)` |
|
| 21. |
Nitrous acid has no action on: |
|
Answer» `CH_(3)CH_(2)NO_(2)` |
|
| 23. |
Nitrosoamines (R_2 N-N=O) are insoluble in water. On heating them with conc. H_2 SO_4, they give sec. amines. The reaction is called |
|
Answer» Liberman's NITROSO REACTION |
|
| 24. |
Nitroso amines (R_2H-N=O) are water soluble .On heating with conc. HCl, they give secondary amines. The reaction is called : |
|
Answer» Perkin REACTION |
|
| 25. |
Nitrosoamines are formed byunderline(" primary amines"). |
| Answer» SOLUTION :NITROSOAMINES are FORMED by SECONDARY AMINES. | |
| 26. |
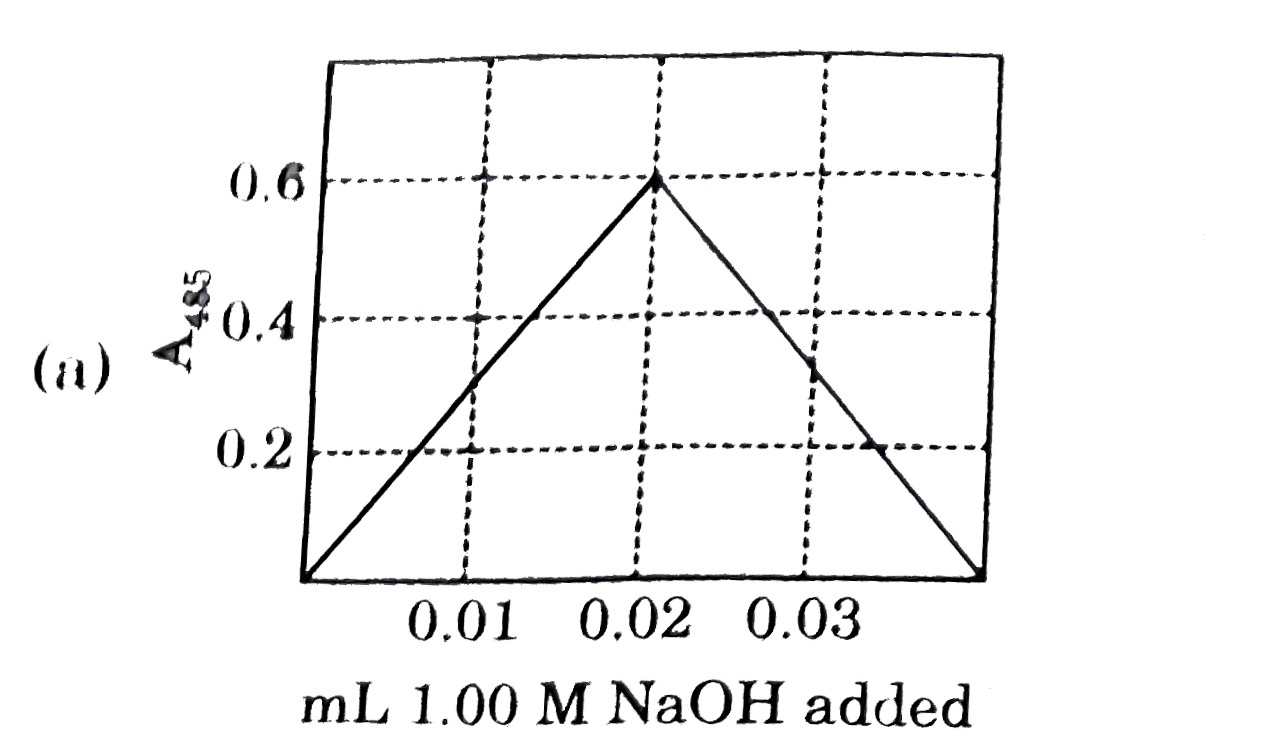
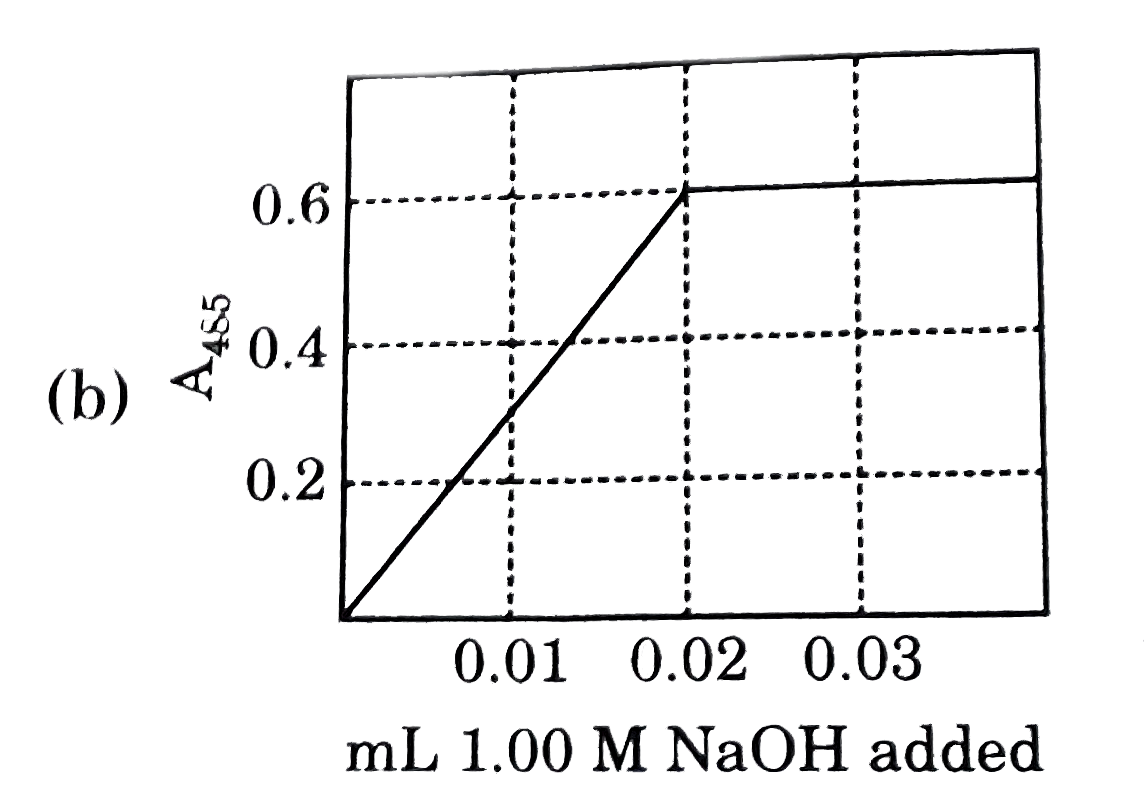
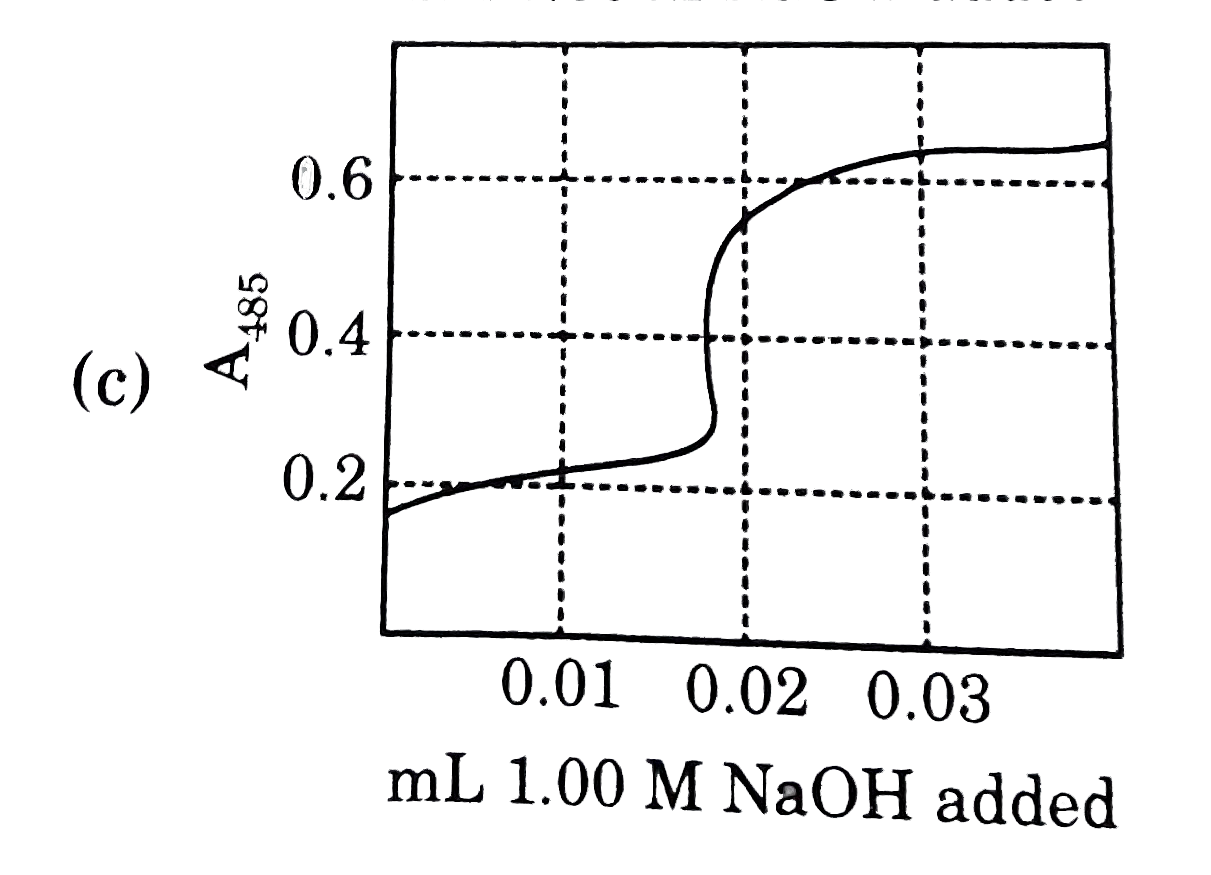
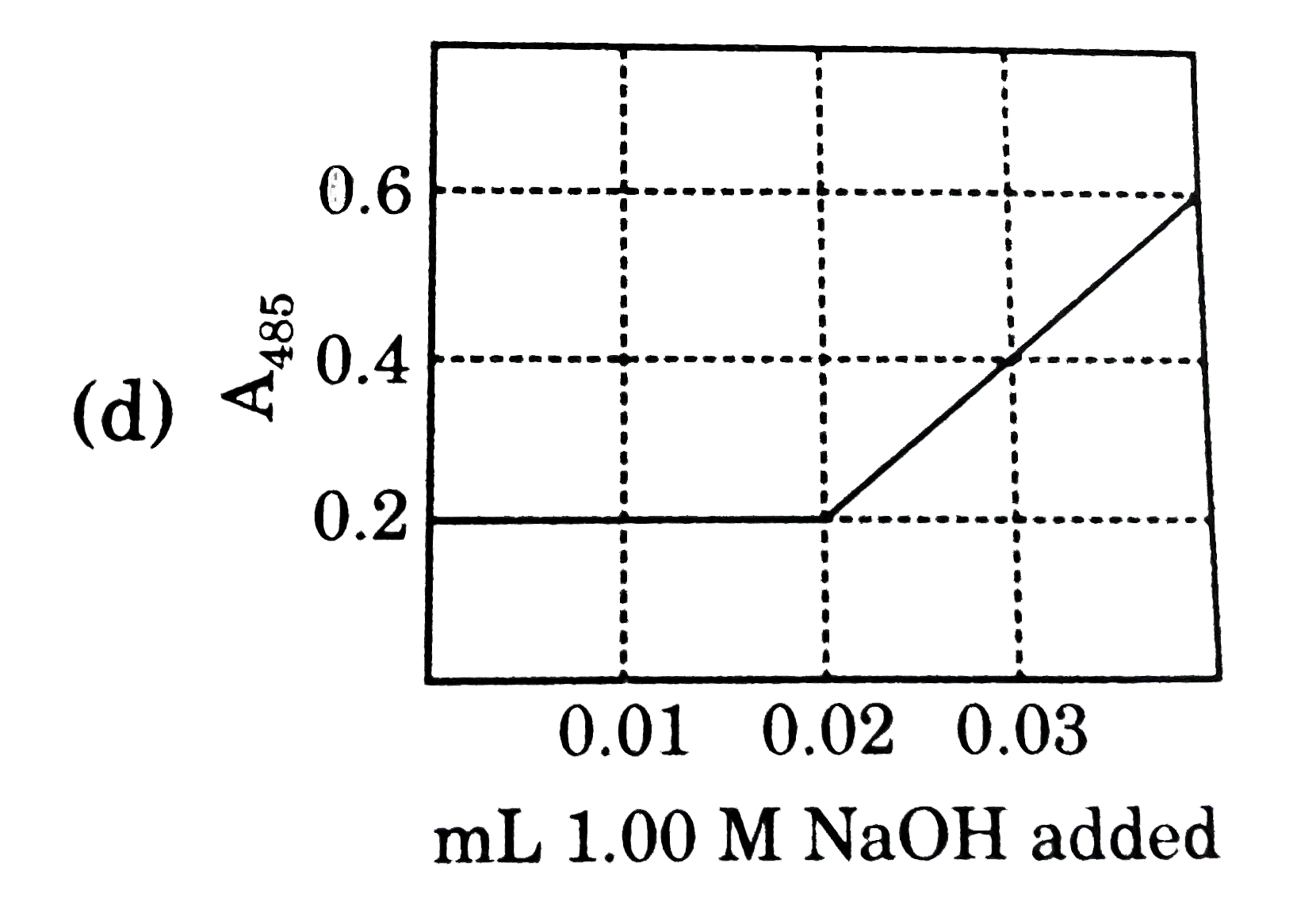
Nitrophenol is a colorless weak monoprotic acid (pK_(a) = 7.2) whose conjugate base is bright yellow. To 2.00 mL of a solution of 0.0100M nitrophenol is added 1.00 M NaOH in 0.01mL portions, and the adsorbance of the solution at 485 nm is monitored. what does the graph of A_(485) as a function of added volume of NaOH look like ? |
|
Answer»
|
|
| 30. |
Nitroparaffins are obtained by oxidation of |
|
Answer» `3^(@)` AMINES |
|
| 31. |
nitromethane will exhibit which type of isomerism? |
|
Answer» METAMERISM |
|
| 32. |
Nitromethane to N-methylmethanamine |
| Answer» Solution :`underset("(Nitromethane)")(CH_(3)NO_(2))overset(Sn//HCl)underset("REDUCTION")RARR CH_(3)NH_(2) overset(CH_(3)CL)rarr underset("Dimethyl a MINE")(CH_(3)-NH-CH_(3))` | |
| 33. |
Nitromethane to methylamine |
|
Answer» Solution :Methylamine : `UNDERSET("NITROMETHANE")(CH_(3)-NO_(2))+6[H]underset(Delta)overset("Sn/conc. HCl")rarrunderset("methylamine")(CH_(3)-NH_(2))+2H_(2)O` |
|
| 34. |
Nitromethane reacts with chlorine in presence of alkali to yield |
|
Answer» Chloromethane |
|
| 35. |
Nitromethane is subjected to the treatment with chlorine in the presence of sodium hydroxide,the main product is : |
|
Answer» Monochloronitromethane |
|
| 36. |
Nitroglycerine is |
|
Answer» An ester |
|
| 40. |
Nitrogethane is acidic only towards |
|
Answer» `Na_(2)CO_(3)` |
|
| 41. |
Nitrogen(1) oxide is produced by: |
|
Answer» THERMAL DECOMPOSITION of ammonium NITRATE |
|
| 42. |
Nitrogen was discovered by: |
|
Answer» Cavendish |
|
| 43. |
Nitrogenforms_______oxides. |
|
Answer» 3 |
|
| 44. |
Nitrogen tetraoxide (N_(2)O_(4)) decomposes as : N_(2) O_(4)(g) rarr 2NO_(2)(g) If the pressure of N_(2)O_(4) falls from 0.50 atm to 0.32 atm is 30 minutes the rate of appearance of NO_(2)(g) is : |
|
Answer» 0.006 ATM `"min"^(-1)` `= (Delta[N_(2)O_(4)])/(Deltat)= (0.50-0.32)/(30)` `= 0.006 atm min^(-1)` Rate of APPEARANCE of `NO_(2)` = Twice rate of disappearance of `N_(2)O_(4)` `2xx0.006 = 0.012 atm "min"^(-1)` |
|
| 45. |
Nitrogen shows oxidation state +5, but it does not form pentahalide as |
|
Answer» it is the first NUMBER ofgroup 15 |
|
| 46. |
Nitrogen sequioxide is. |
|
Answer» `N_2 O_3` |
|
| 47. |
In which of the following phosphorus is in +1 oxidation state? |
|
Answer» `H_3PO_2` |
|
| 48. |
Nitrogen shows different oxidation states in different oxides. In which of the following oxides, nitrogen is in +4 oxidation state? NO ,N_2 O ,N_2 O_3 ,NO_2 |
|
Answer» NO |
|
| 49. |
Nitrogen sesquoxide colour is ……………….. . |
|
Answer» colourless |
|
| 50. |
Nitrogen pentoxide decomposes according to equation 2N_(2)O_(5)(g) to 4NO_(2)(g) + O_(2)(g) The first order reaction was allowed to proceed at 140^(@)C and the data below were collected. a) Calculate rate constant in all the cases. b) What will be the concentration of N_(2)O_(5) after 100 minutes? c) Calculate initial rate of reaction. |
|
Answer» SOLUTION :a) For FIRST order reaction , k`=(2.303)/tloga/(a-x)` In first CASE `k=2.303/(20 min)log(0.400)/0.289 = 2.303/(20 min)log400/289 = 2.303/(20 min)[log 400 - log 289]` `=2.303/(20 min) [2.6201 - 2.4609]=(2.303 xx 0.1412)/(20 mm) = 1.63 xx 10^(-2) min^(-1)` In second case `k=2.303/(40 min) log 0.400/0.209 = 2.303/(40 min) log 400/209 = 2.303/(40 min)[log 400- log 209]` `=2.303/(40 min) [2.6021 - 2.3201] = (2.303 xx 0.2820)/(40 min)= 1.62 xx 10^(-1) min^(-1)` In third case |
|