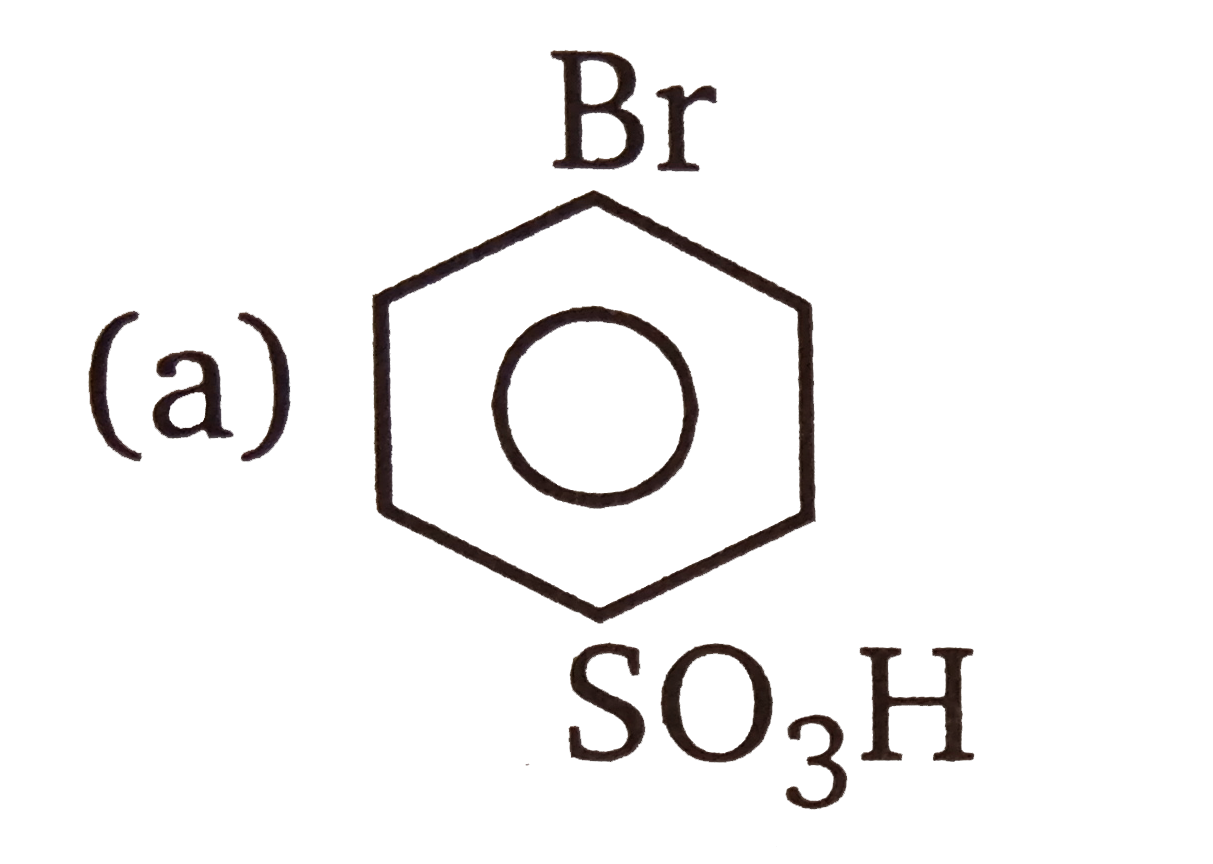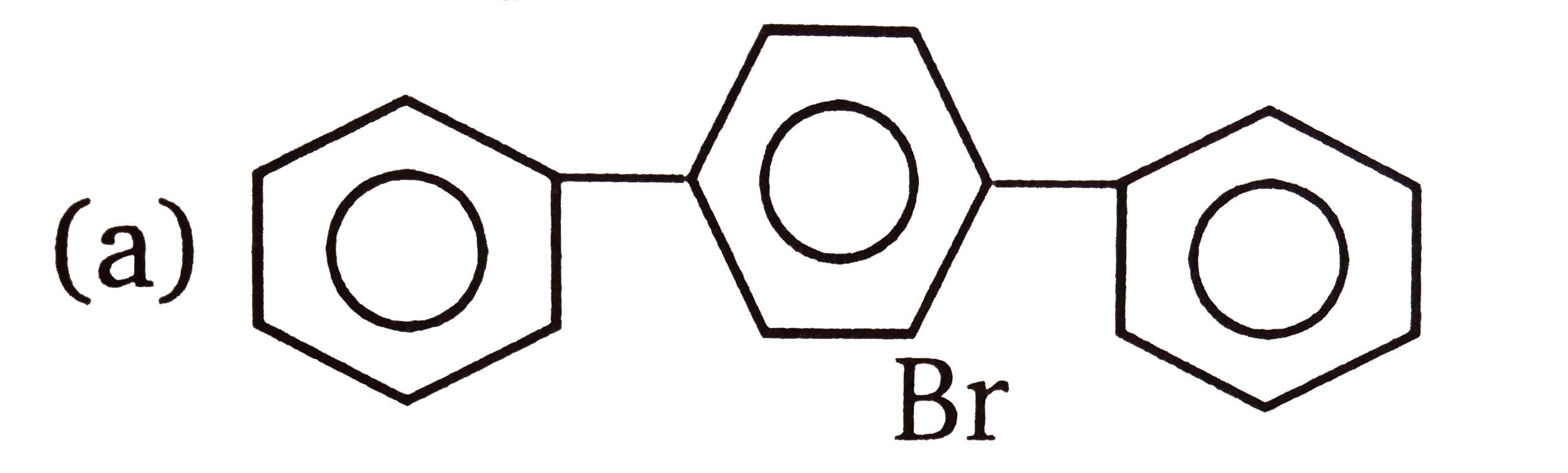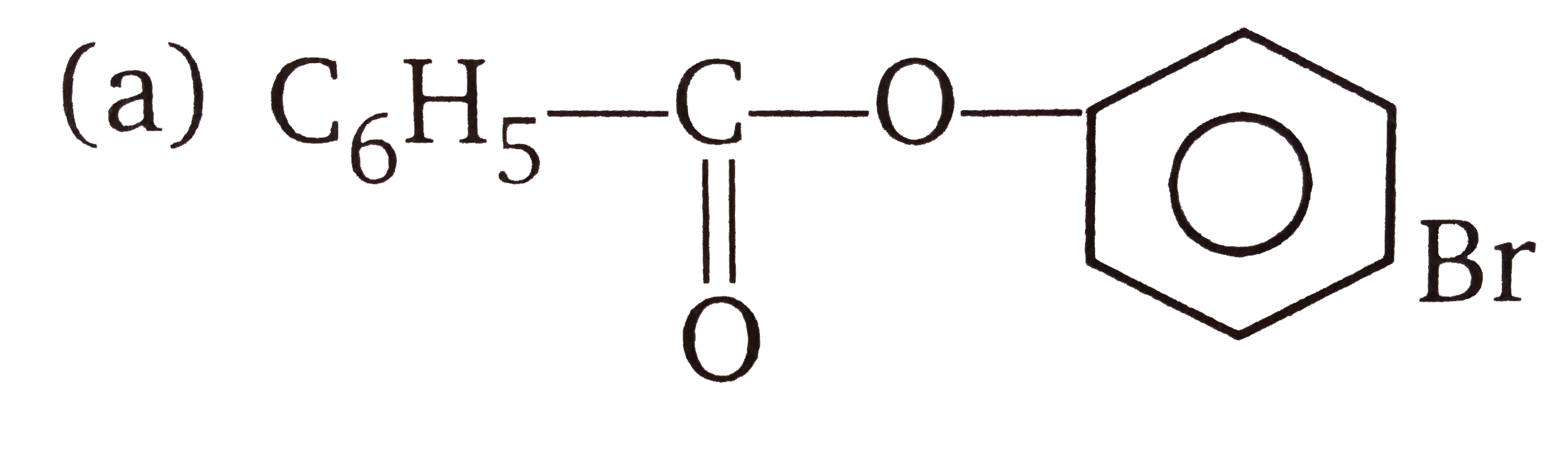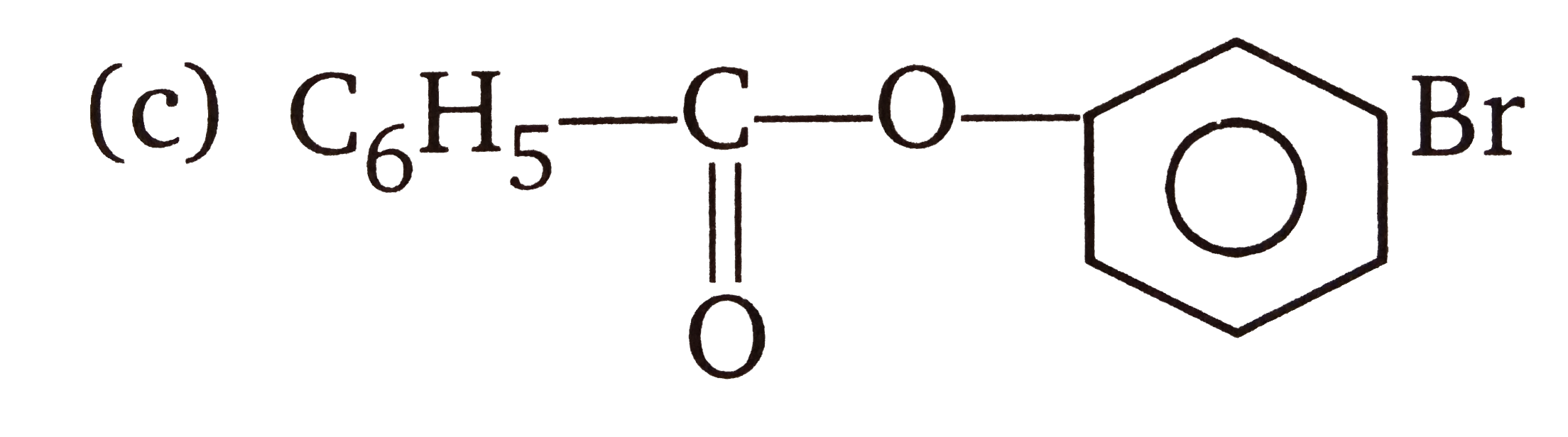Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The compounds A and B in the reaction sequence A overset( " phenol ") larrCH_(3) COCloveserset(CH_(3) COONa) to B |
|
Answer» `CH_(3)CO–O–COCH_(3), C_(6)H_(5)CH_(2)OH` 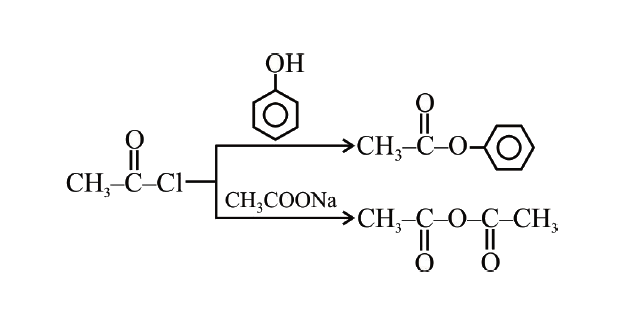
|
|
| 2. |
The compounds A and B are mixed in equimolar proportion to form the products, A+BleftrightarrowC+DAt equilibrium, one third of A and B are consumed. The equilibrium constant for the reaction is |
|
Answer» Solution :`A+BleftrightarrowCV+D` Initial state1100 Atequilibrium`1-(1)/(3)``1-(1)/(3)``(1)/(3)``(1)/(3)` `K_(C)=([C][D])/([A][B])=((1)/(3)XX(1)/(3))/((2)/(3)xx(2)/(3))=(1)/(4)`=0.25 |
|
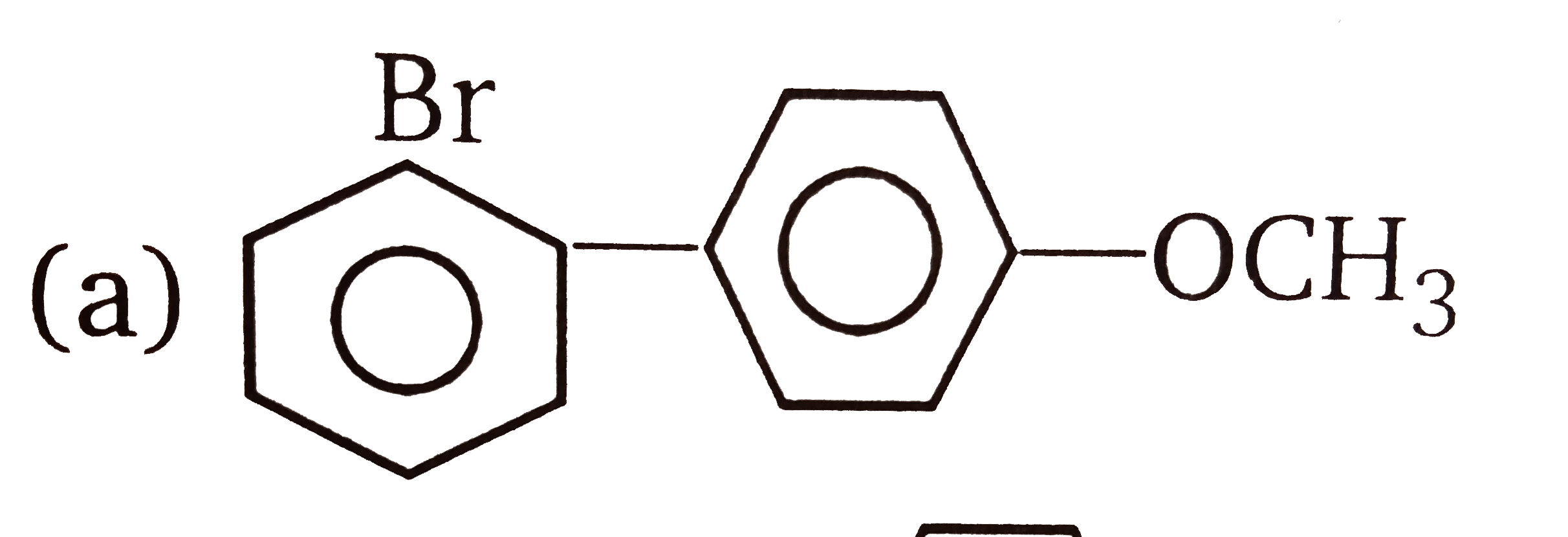
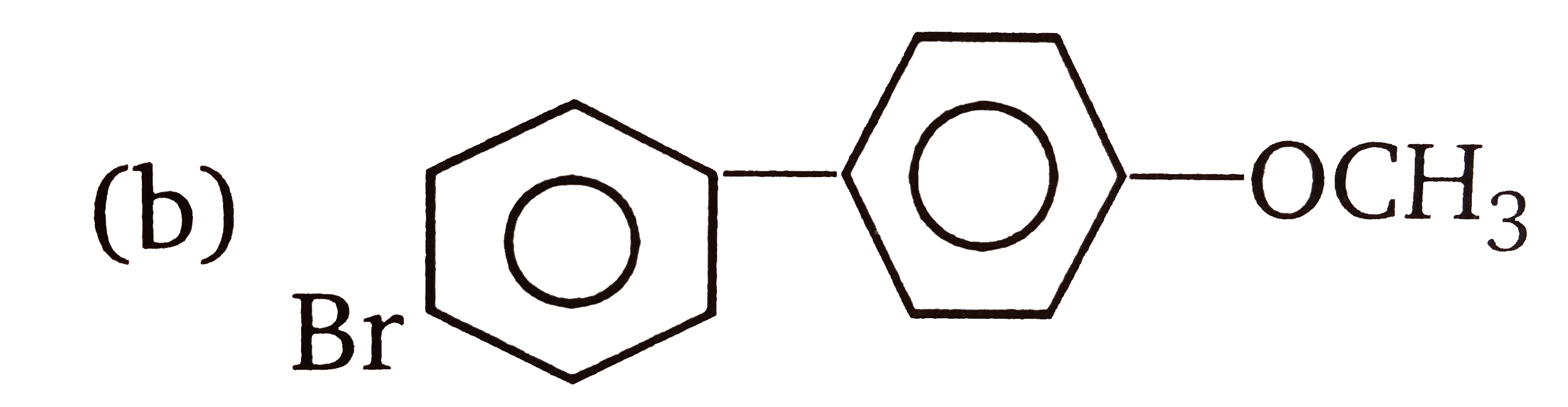
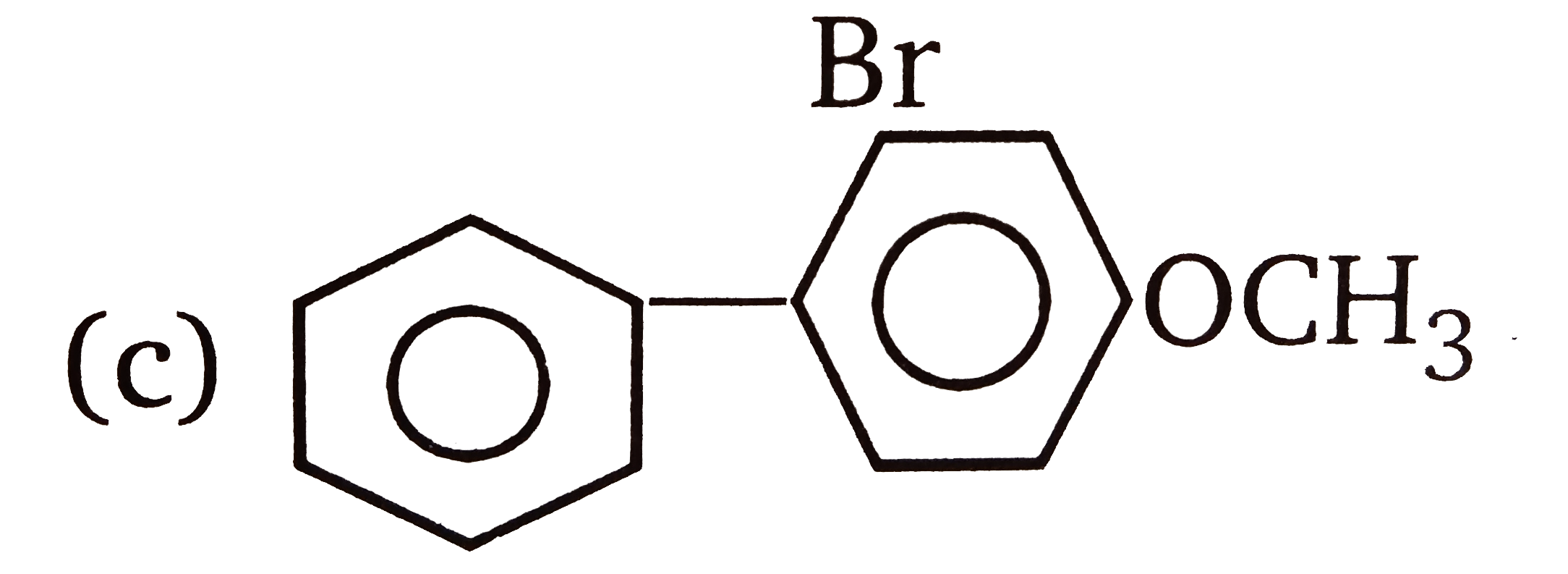
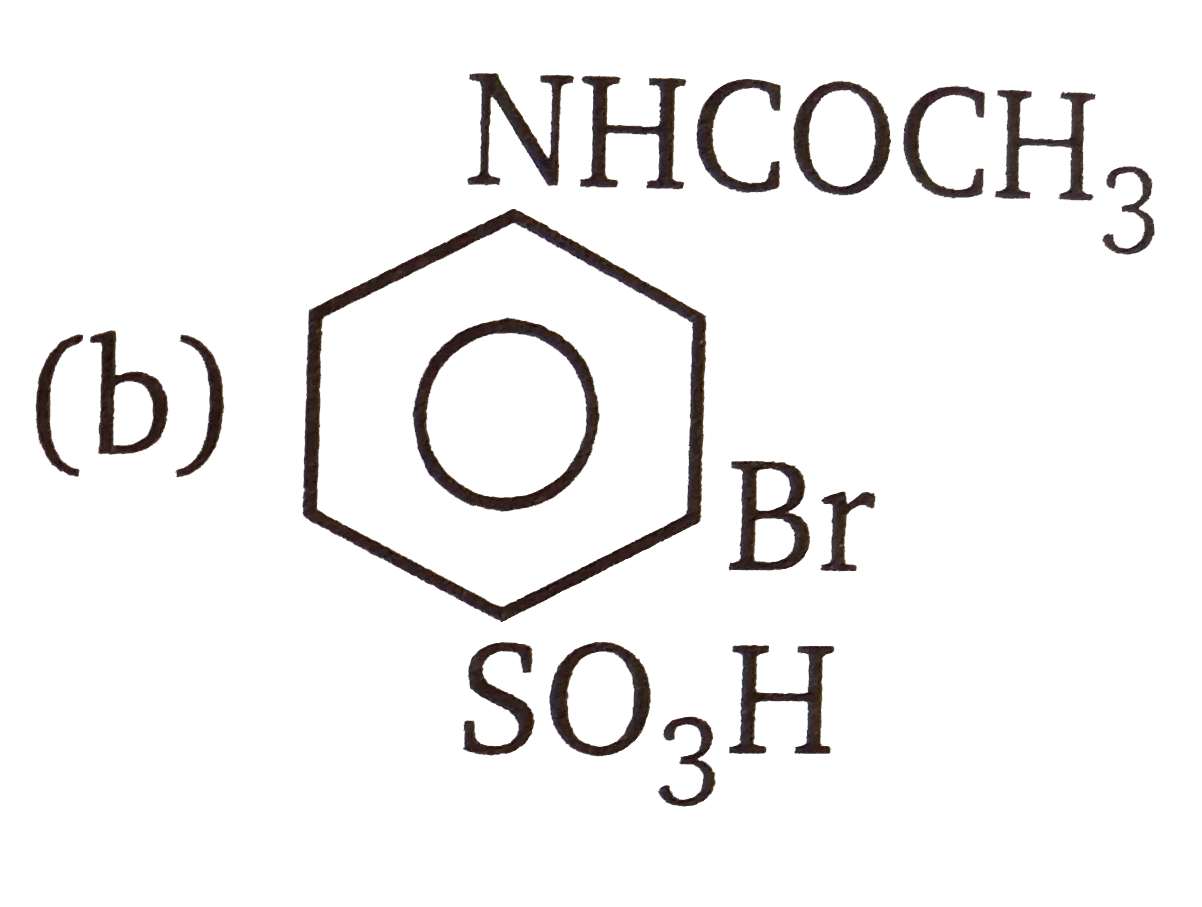
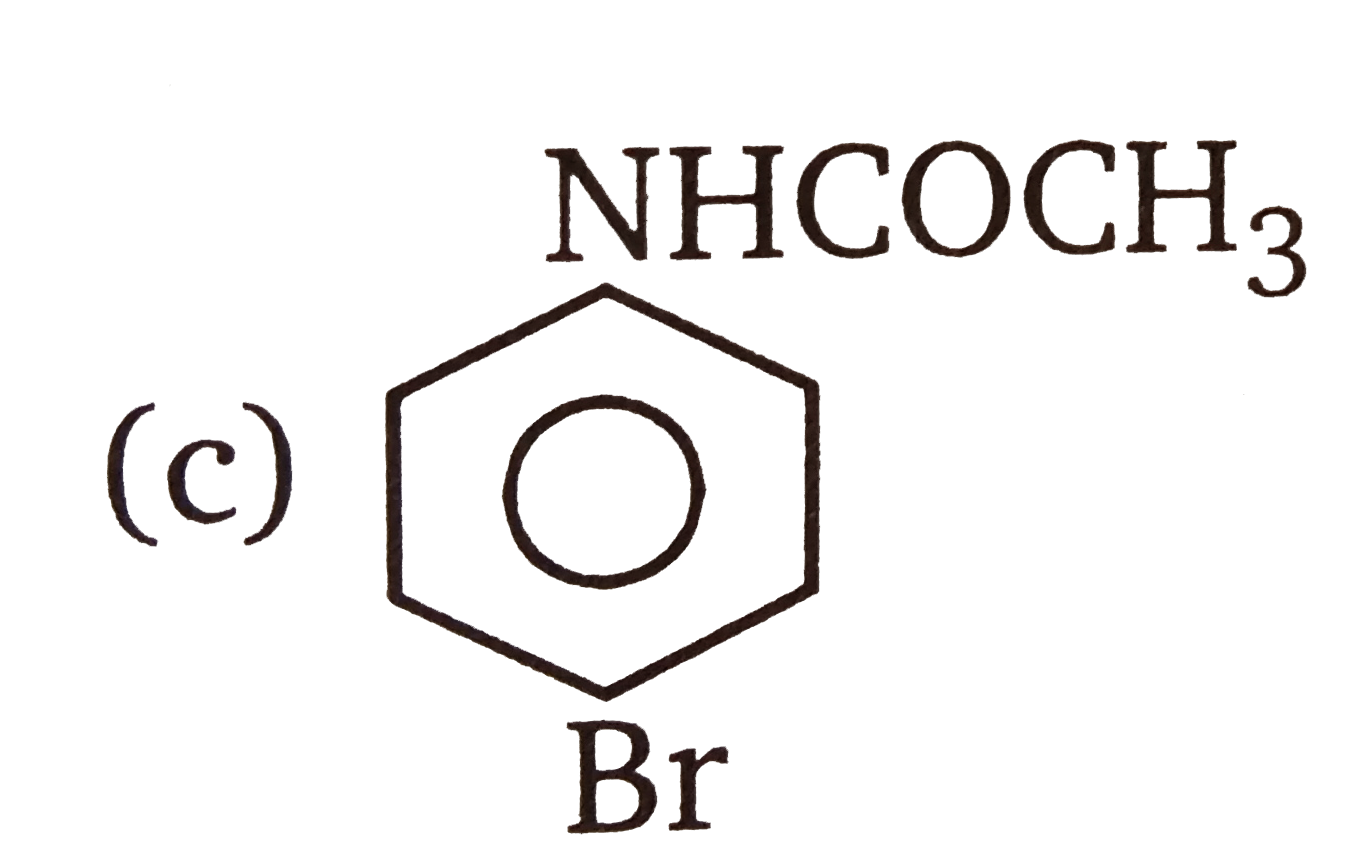
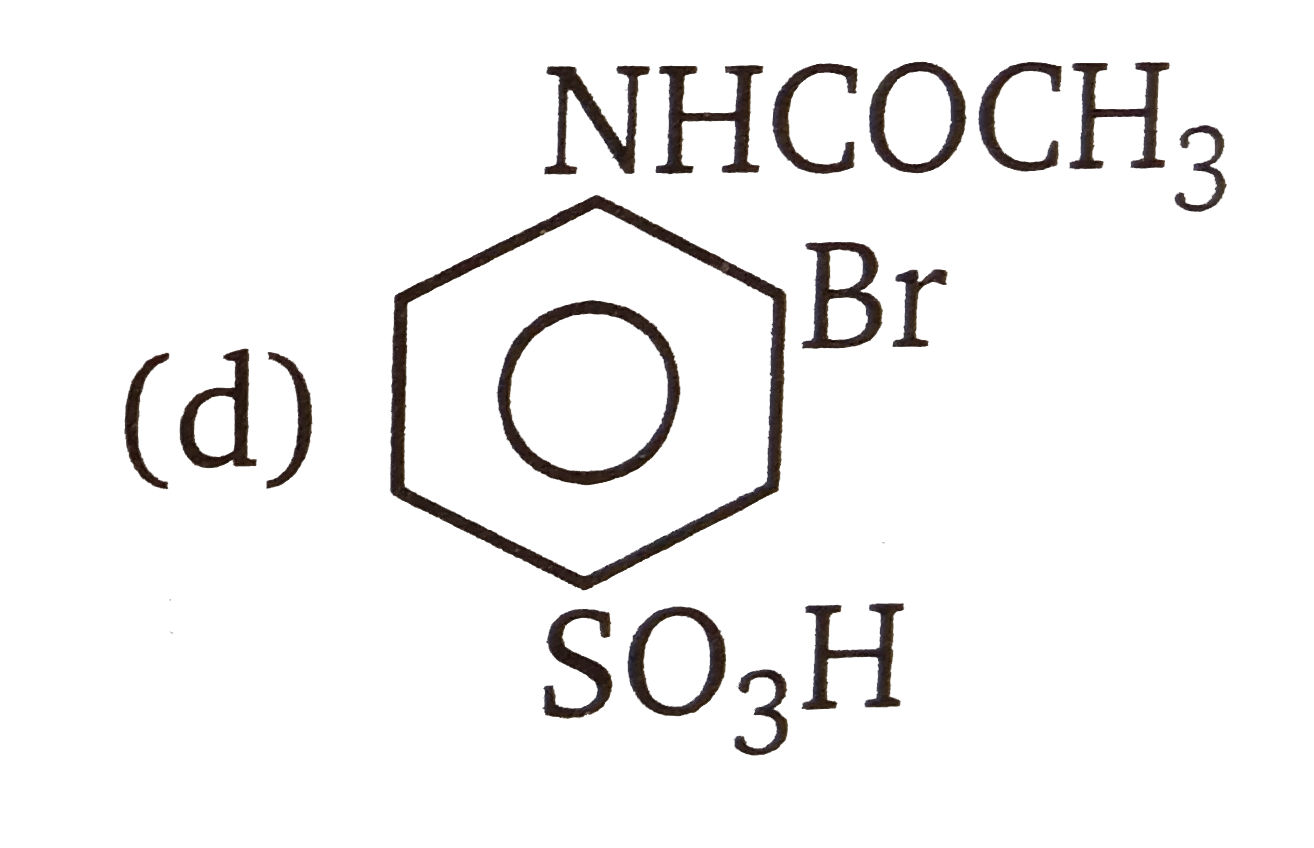
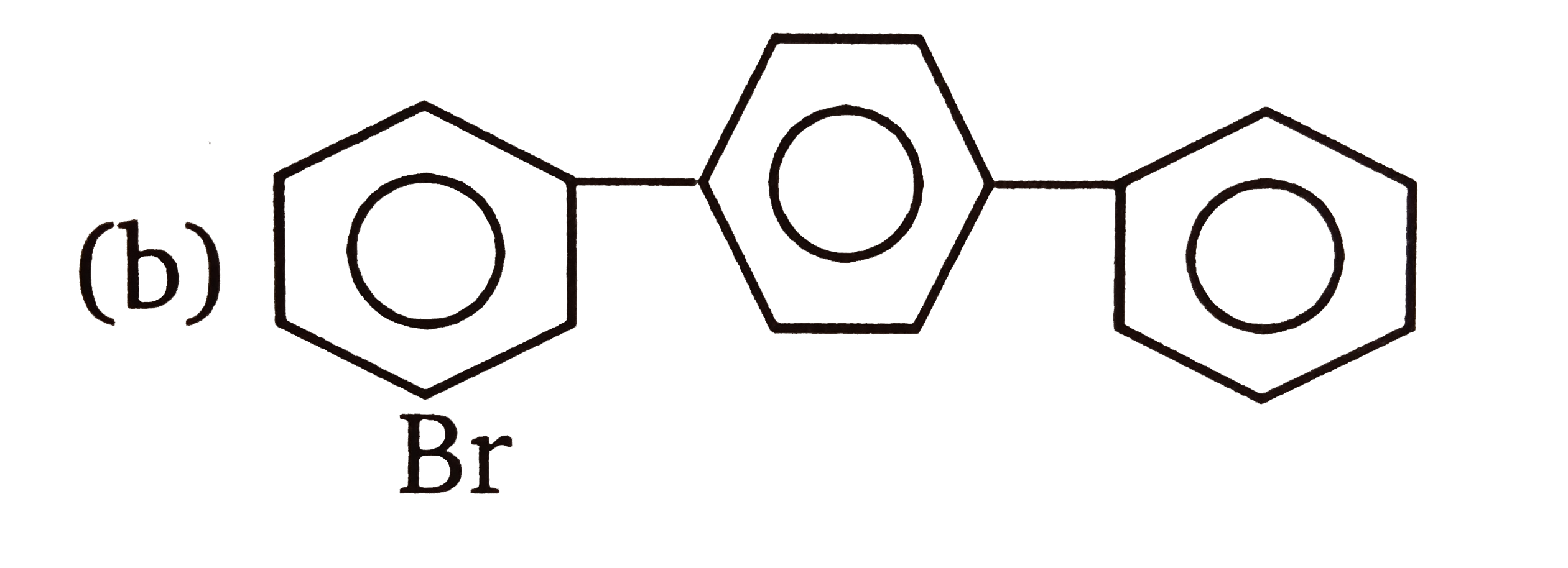
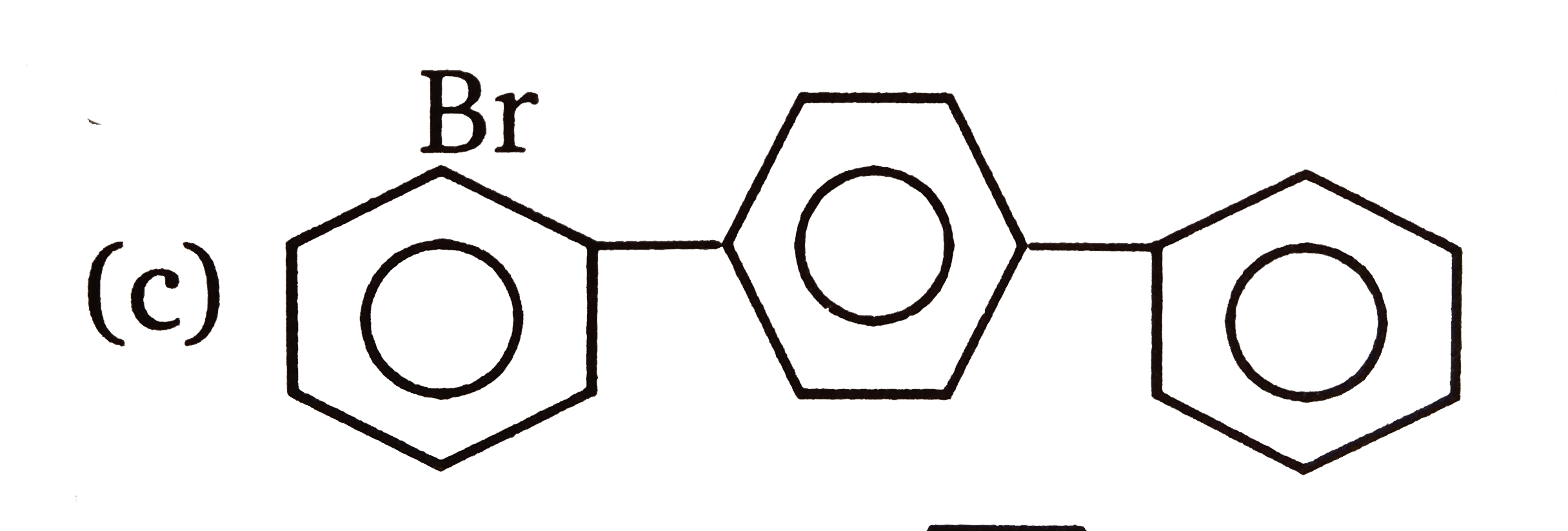
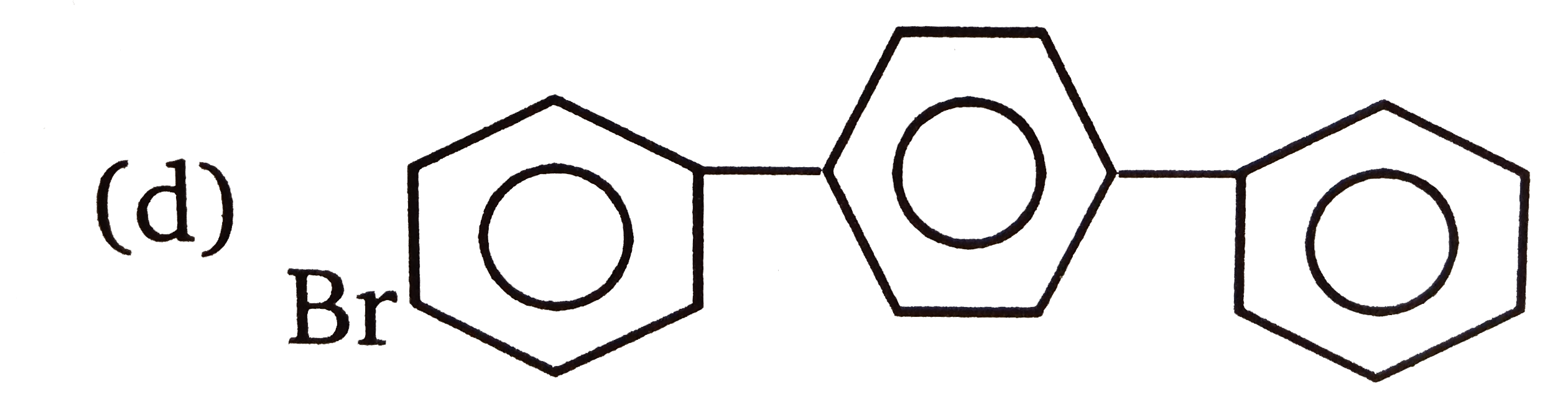
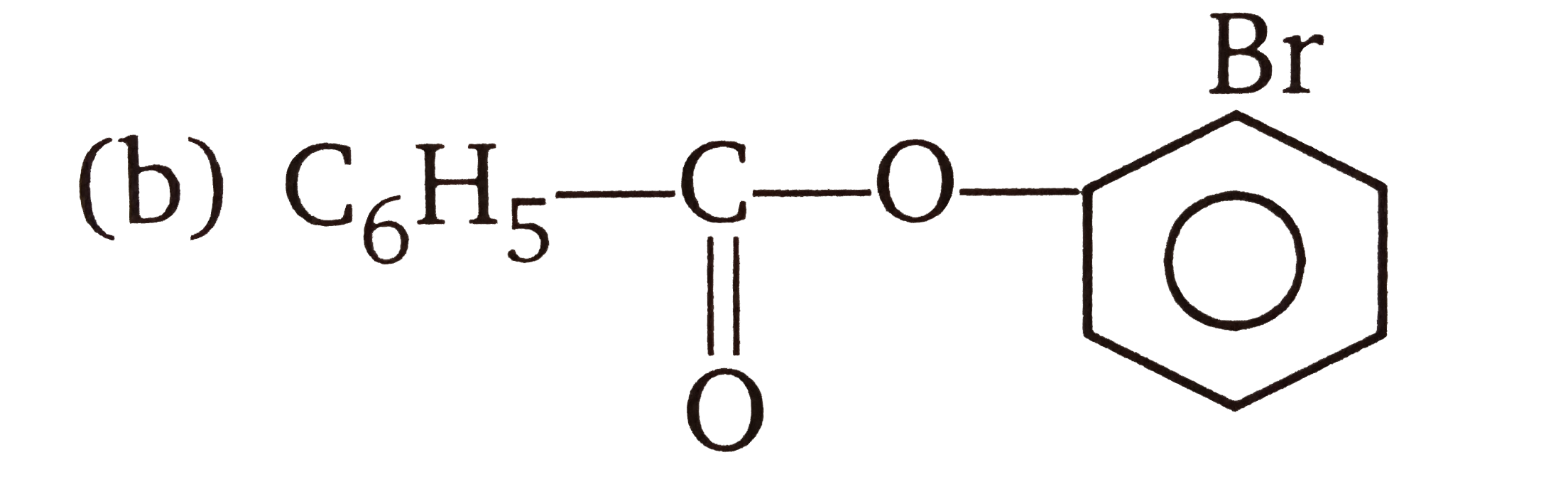
| 3. |
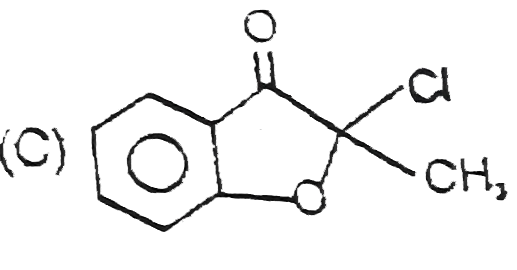
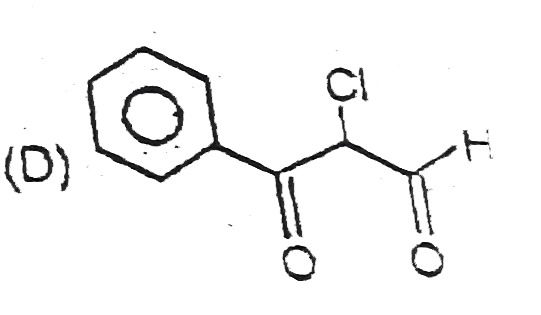
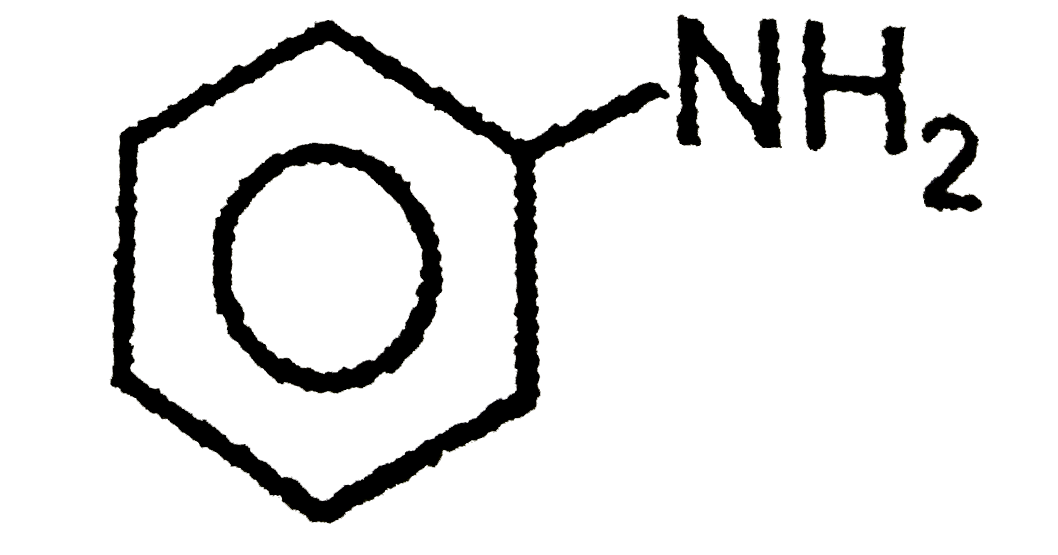
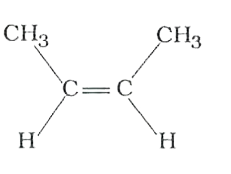
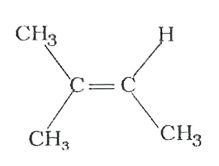
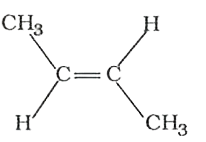
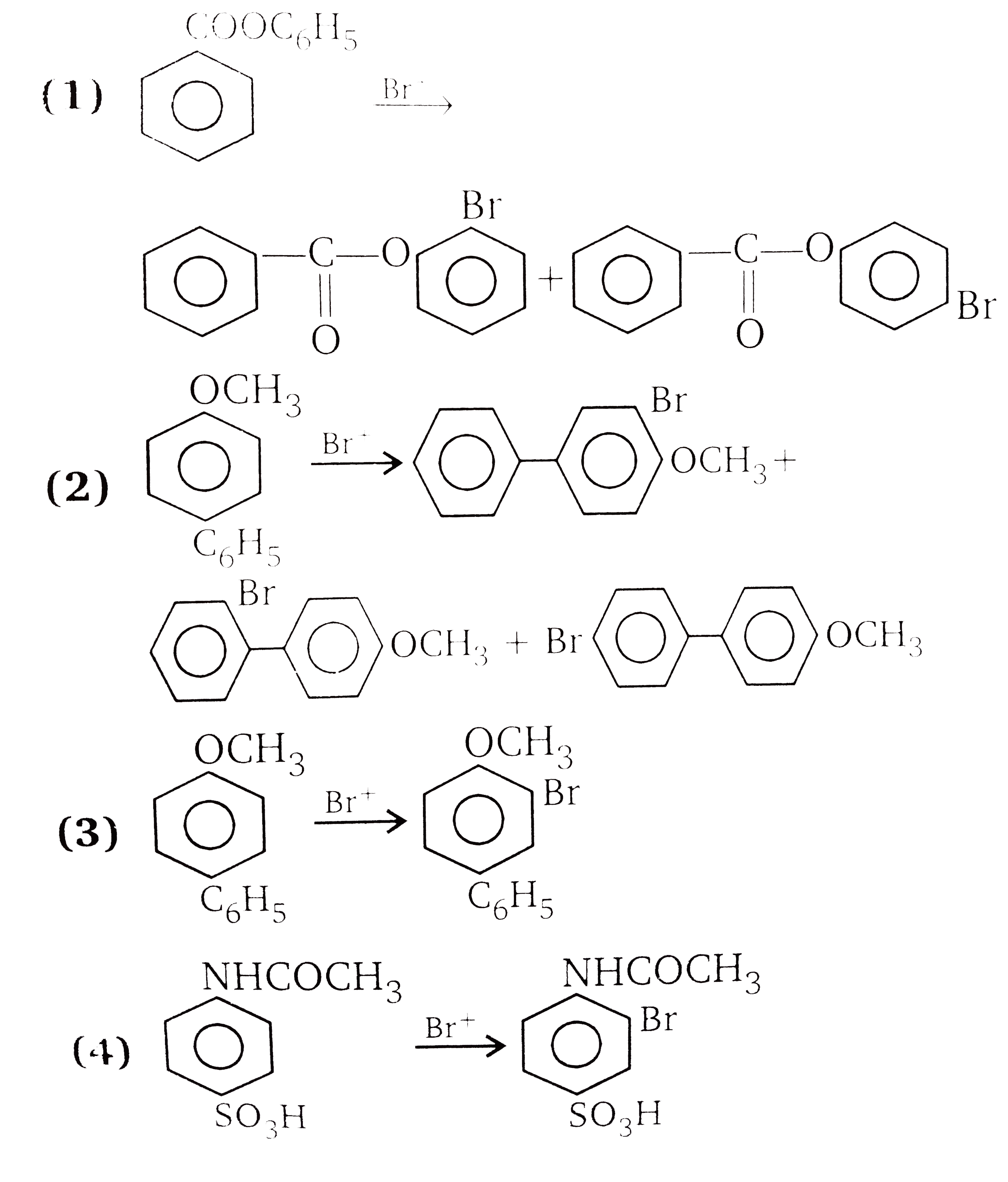
The compounds 1,2,3,4 given below are allowed in undergo electrophilic substitution by bromonium ions assuming only monobromo substitution the substitutes products are A,B,C,D respectively. The compound B is : |
|
Answer»
|
|
| 4. |
The compounds A and B are formed in the sequence of the reaction CH_(3)CH_(2)CH_(2)OH overset(PCl_(3))rarr A underset("KOH")overset("alc.")rarr B In the above reactionB is |
|
Answer» Propylene |
|
| 5. |
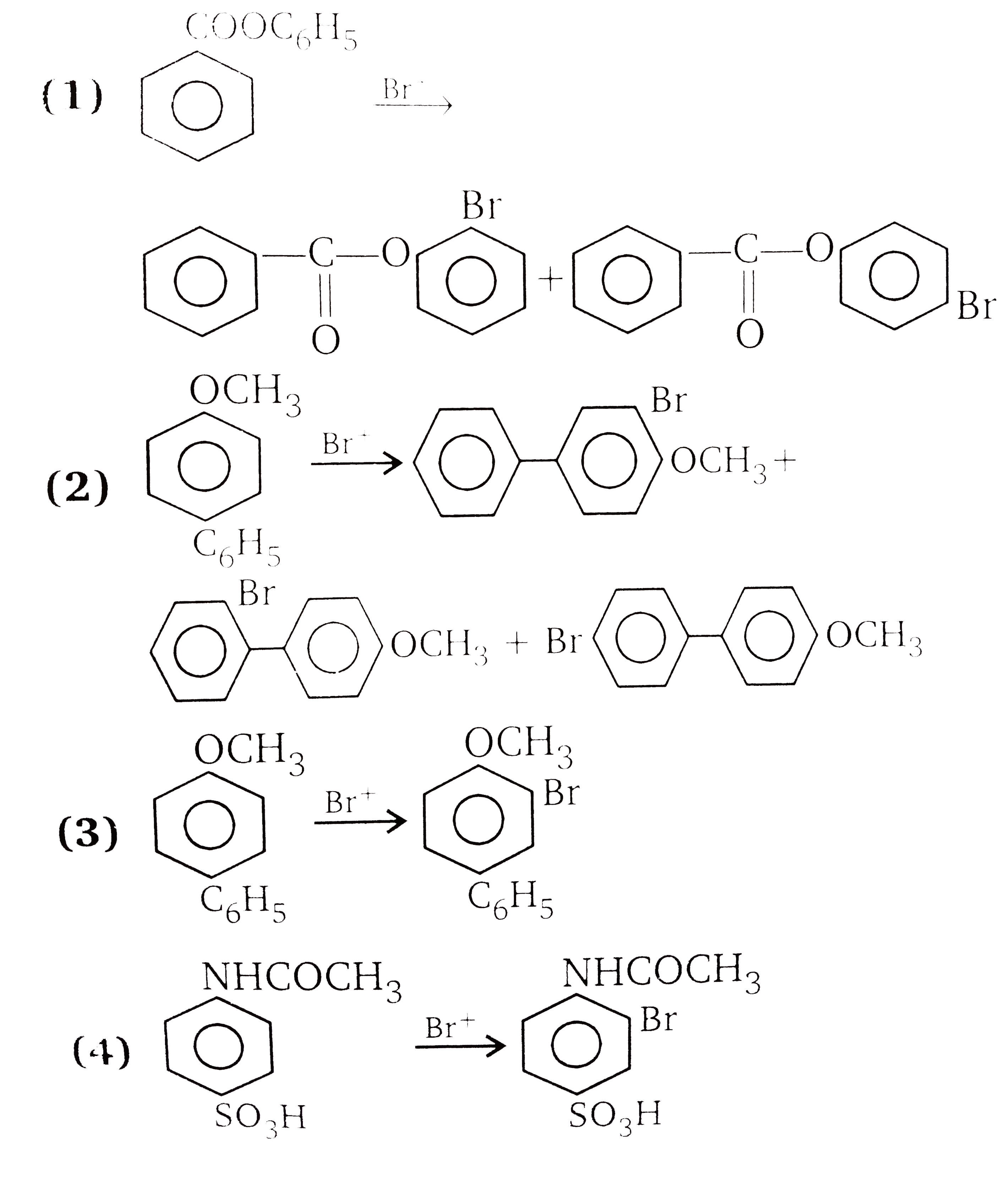
The compounds 1,2,3,4 given below are allowed in undergo electrophilic substitution by bromonium ions assuming only monobromo substitution the substitutes products are A,B,C,D respectively. The compound D is: |
|
Answer»
|
|
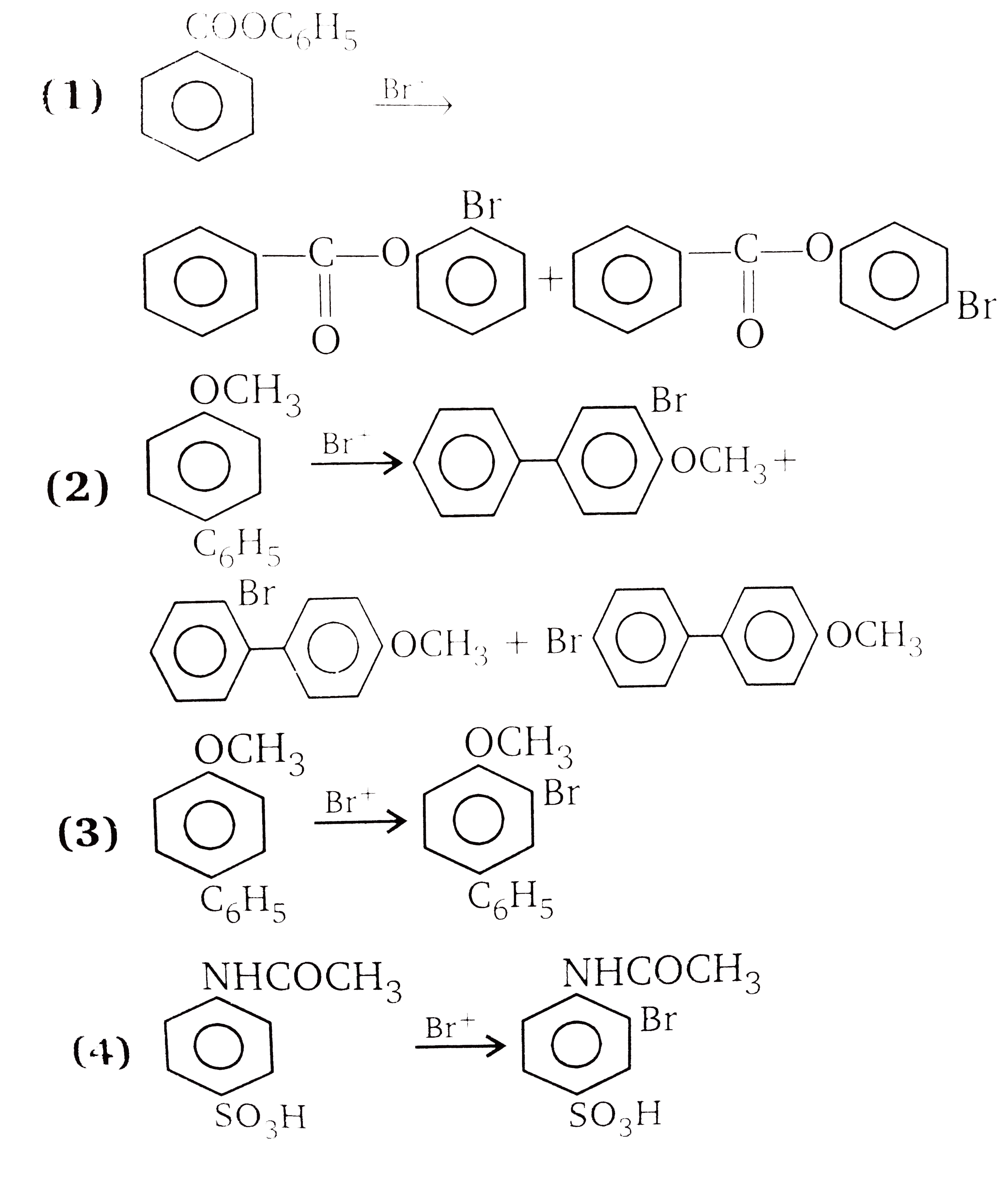
| 6. |
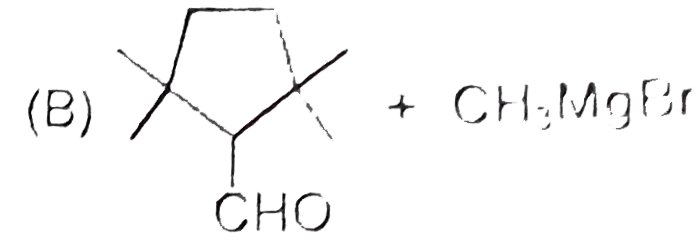
The compounds 1,2,3,4 given below are allowed in undergo electrophilic substitution by bromonium ions assuming only monobromo substitution the substitutes products are A,B,C,D respectively. The compound C is: |
|
Answer»
|
|
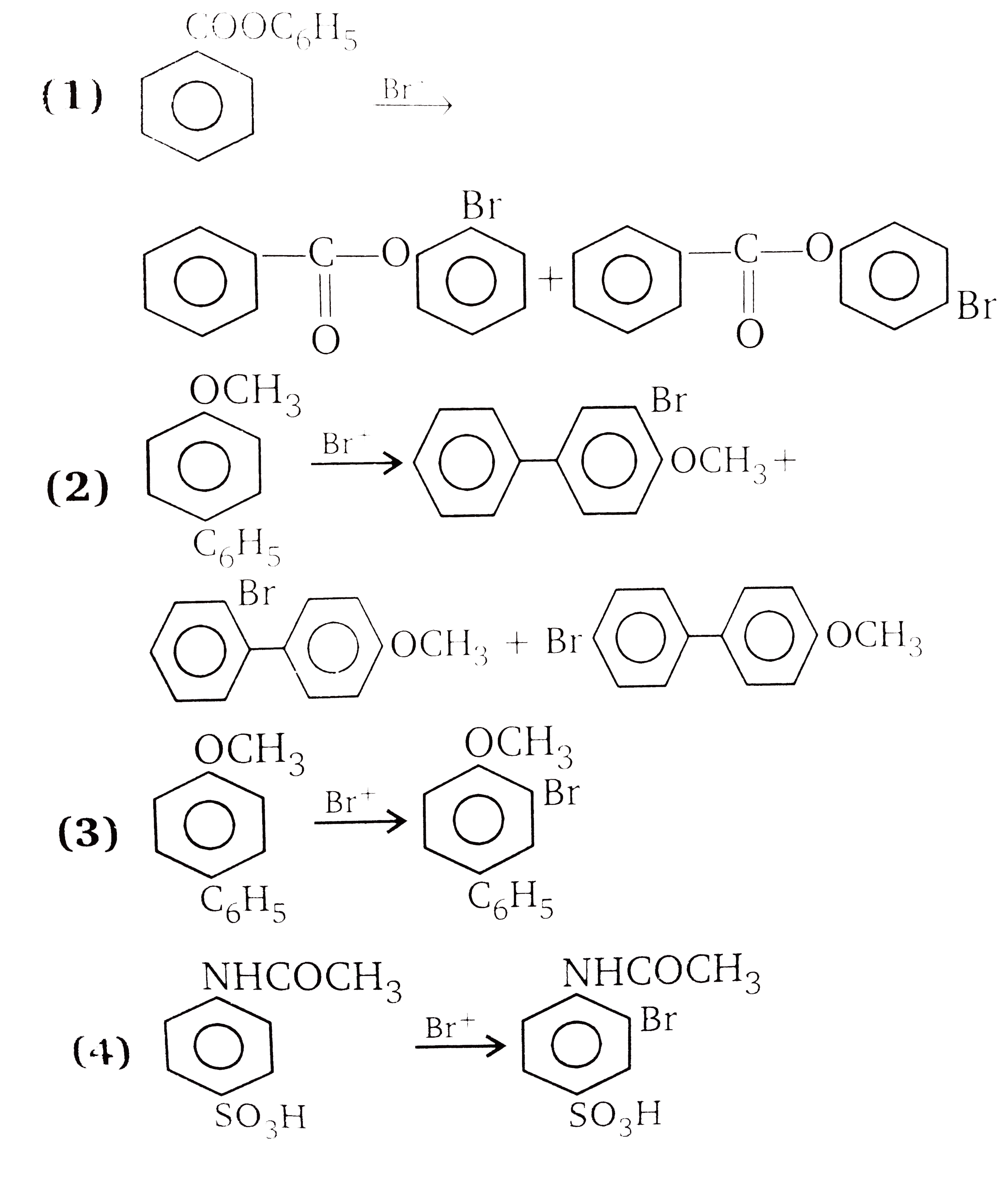
| 7. |
The compounds 1,2,3,4 given below are allowed in undergo electrophilic substitution by bromonium ions assuming only monobromo substitution the substitutes products are A,B,C,D respectively. The diamonds A is: |
|
Answer»
|
|
| 8. |
The compound ZnFe_(2)O_(4) is |
|
Answer» A NORMAL SPINEL COMPOUND |
|
| 9. |
The compound Y in the above sequence is |
|
Answer» 2-Methyl 1-2-phenyl-1-propanol |
|
| 10. |
the compound Y can not be prepared by using which of the reagents: |
|
Answer»
(phenol and ALIPHATIC ALCOHOL are considered as different functional groups). |
|
| 11. |
The compound X(C_5H_8) reacts with ammoniacal AgNO_3 to give a white precipitate and reacts with excess of KMnO_4 to give acid, (CH_3)_2CH-COOH. Therefore, X is: |
|
Answer» `CH_2=CH-CH=CH-CH_3` |
|
| 12. |
The compound (X) with M.F. C_(4)H_(11)N on treatment with HNO_(2) gives a tertiary alcohol with M.F.C_(4)H_(10)O. The compound (X) will give |
|
Answer» CARBYLAMINE reaction 
|
|
| 14. |
The compound X produces methane when treated with water. The compound X is |
|
Answer» calcium carbide |
|
| 15. |
The compound X on treatment with acidified K_2Cr_2O_7 gives compound Y which reacts with I_2 and Na_2CO_3 to form tri-iodomethane. The compound X is : |
|
Answer» `CH_3OH` |
|
| 16. |
The compound X on reaction with HgSO_4+H_2SO_4 gives Y which one oxidation gives acetic acid. X is |
|
Answer» `C_2H_2` |
|
| 17. |
The compound X on oxidation gives a mixture of acetic acid and formic acid, while Y on oxidation forms only propionic acid. X and Y are |
|
Answer» `CH_3 - underset(OH)underset(|)(CH) - CH_3, CH_3 - CO - CH_3` |
|
| 18. |
The compound X on heating gives a colourless gas. The residue is dissolved in water to obtain Y Excess CO_2 is bubbled through aqueous solution of Y, Z is formed. Z on gentle heating gives back X. The compound X is: |
|
Answer» `CaCO_3` |
|
| 20. |
The compound (X) is : |
|
Answer» `(CH_(3)CO)_(2)O` |
|
| 21. |
The compound X, in the reaction is |
|
Answer» `Xoverset(CH_(3)MgI)toYoverset("HYDROLYSIS")toMg(OH)I+CH_(3)COOH` 
|
|
| 22. |
The compound (X) is |
|
Answer» `CH_(3)COOH` |
|
| 23. |
The compound (X) has molecular formula C_(4)H_(7)Cl . Find out the number ofits cyclic isomers (structural and geometrical only excluding optical isomers). |
Answer» 
|
|
| 24. |
The compound X (C_(4)H_(11)N) on treatement with nitrous acid gives a tertiary alcohol (C_(4)H_(10)O). The compound X must also respond to |
|
Answer» Mustard oil reaction `{:(""CH_(3)""CH_(3)),("|""|"),(CH_(3)-C-NH_(2) overset(HNO_(2))(rarr) CH_(3)-C-OH),("|""|"),(""CH_(3)""CH_(3)),(" "(X) 1^(@)" Amine"3^(@)" Alcohol"):}` X being `1^(@)` amine should also RESPOND to mustard oil reaction and carbyl amine reaction. |
|
| 25. |
The compound X (C_9H_7CIO_2) is optically active.It mainly exists in enolic form.It racemises due to enolisation its structure is : |
|
Answer»
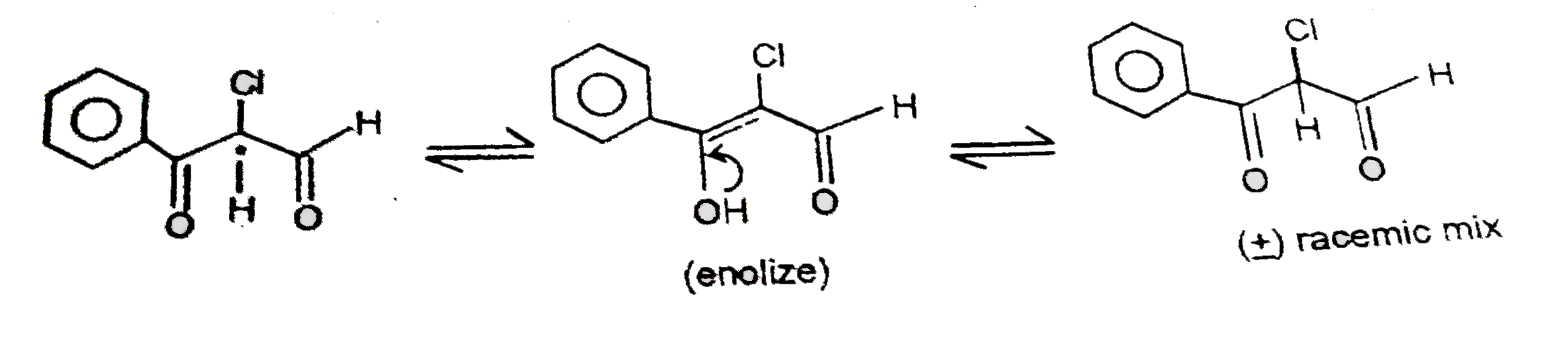
|
|
| 26. |
The compound X having -OH functional group, quickly reacts with Conc. HCl and anhydrousZnCl_(2) What is X? |
|
Answer» 3-methyl-2-butanol |
|
| 27. |
The compound X and Y in below reaction can be Ph-NH*NH_(2)+ (X) + (Y) overset(-H_(2)O)(to) |
|
Answer» `CH_(3)-CH_(2)-underset(OVERSET(||)(O))(C)-CH_(3) + CH_(3)-underset(overset(||)(O))C-Ph` |
|
| 28. |
The compound with two lone pairs of electrons on the central atom is (are) |
|
Answer» `BrF_(5)` |
|
| 29. |
The compound X and Y are |
|
Answer»

|
|
| 30. |
The compound with the maximum dipole moment among the following is : |
|
Answer» p-dichlorobenzene |
|
| 31. |
The compound with molecular formula C_(8)H_(10) which will give only two isomers on electrophilic substitution with Cl_(2)//FeCl_(3) or with HNO_(3)//H_(2)SO_(4) is |
|
Answer» NONE of these |
|
| 32. |
The compound with no dipole moment is |
|
Answer» METHYL chloride |
|
| 33. |
The compound with negative heat of formation are known as |
|
Answer» ENDOTHERMIC COMPOUND |
|
| 34. |
The compound with molecular formula C_8H_10 which will give two isomers on electrophilic substitution with Cl_2//FeCl_3 or with HNO_3//H_2SO_4 is |
|
Answer» p-dimethyl benzene |
|
| 35. |
The compound with molecular formula C_(4)H_(10)O does not reacts with sodium metal but with excess HI it gives one alkyl halide compound. Which is the compound ? |
|
Answer» METHOXY methane |
|
| 36. |
The compound with highest B.P. is |
|
Answer» n- pentylchloride |
|
| 37. |
The compound with highest boiling point is |
|
Answer» `CH_4` |
|
| 39. |
The compound with formula C_4H_(10)O yields a compound C_4H_8O on oxidation. The compound C_4H_(10)O is : |
|
Answer» An aldehyde |
|
| 40. |
The compound with alpha - hydrogen is _______ |
|
Answer» HCHO |
|
| 41. |
The compound with formula C_4H_10O yields a compound C_4H_8O on oxidation, the compound C_4H_10O is |
|
Answer» an ALDEHYDE |
|
| 42. |
The compound widely used in making reference electrode is: |
|
Answer» `ZnCl_2` |
|
| 43. |
The compound whose 0.1 M solution is basic is |
|
Answer» Ammonium acetate (b) Is acidic solution due to cationic hydrolysis, (C) Is acidic solution due to cationic hydrolysis, (d) Is basic solution due to anionic hydrolysis. |
|
| 44. |
The compound which will not reach with Hinsberg's reagent is |
|
Answer»
|
|
| 45. |
The compound which will not exist is |
|
Answer» `XeF_2` |
|
| 46. |
The compound which undergoes intramolecular dehydration with P_(2)O_(5) is ______ |
|
Answer» ACETIC acid |
|
| 47. |
The compound which will give negative iodoform test is |
|
Answer» `CH_(3)CHO` `C_(6)H_(5)-CH_(2)-OH overset(NaOH//I_(2))to ` No yellow ppt. |
|
| 48. |
The compound which retard the actaion of oxygen on food are called |
|
Answer» antioxidants |
|
| 49. |
The compound which reacts with Fehling solution is |
|
Answer» `C_(6)H_(5)COOH` |
|