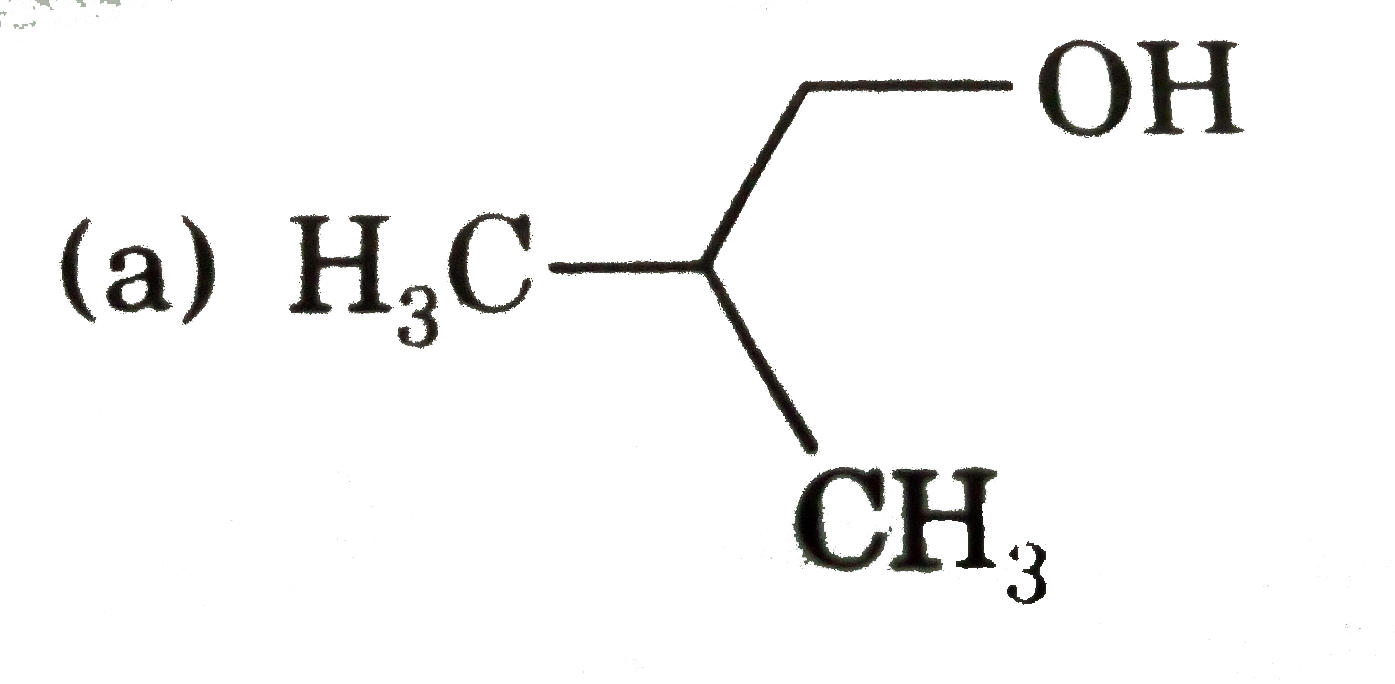Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The compound which reacts with HBr obeying Markownikoff's rule is |
|
Answer» `CH_2=CH_2` |
|
| 2. |
The compound which reacts fastest with Lucas reagent at room temperatureis |
|
Answer» butan-1-ol |
|
| 3. |
The compound which reacts fastest with Lucas reagent at room temperature is |
|
Answer» butan-1-ol |
|
| 4. |
The compound which reacts faster with Lucas reagent at room temperature is |
|
Answer» butan-1-ol |
|
| 5. |
The compound which reacts fastest with Lucas reagent at room temperature is : |
|
Answer» Butan-1-ol |
|
| 6. |
The compound which on reduction with LiAIH_4 gives two alcohols |
|
Answer» `CH_3 CHO` |
|
| 7. |
The compound which on reaction with cold HNO_2 gives only nitrosoamine is : |
|
Answer» `CH_3NH_2` |
|
| 8. |
The compound which on reduction with LiAlH_4 gives two alcohols: |
|
Answer» `CH_3COOCH_3` |
|
| 9. |
The compound which on reacting with aqueous nitrous acid at low temperature produces an oily nitrosamine is |
|
Answer» ETHYL amine |
|
| 10. |
The compound which on reaction with aqueous nitrous acid at low temperature produces an oil nitrosoamine is |
|
Answer» Methylamine |
|
| 11. |
The compound which on reaction with aqueous nitrous acid at low tremperature produces an oily nitrosoamine is : |
|
Answer» Methylamine ` Et_2 NH + NHO_2 rarr underset (N-"Nitroso N-ethyl ethanamine") (Et_2 N-N=O + H_2O)`. |
|
| 12. |
The compound which liberates H^2 gas with sodium metal is , |
|
Answer» aldehyde |
|
| 13. |
Which of the following is used as acetylating agent |
|
Answer» glacial acetic acid |
|
| 14. |
The compound which is insoluble in dil. HCl is: |
|
Answer» `BaSO_4` |
|
| 15. |
The compound which is not formed when a mixture of n-butyl bromide and ethyl bromide treated with sodium metal in the presence of dry ether is |
|
Answer» Butane |
|
| 16. |
The compound which is not isomeric with diethyl ether is : |
|
Answer» n-propyl METHYL ETHER |
|
| 17. |
The compound which is not formed on heating Be(NO_3)_2 is |
|
Answer» `N_2O` |
|
| 18. |
The compound which is not formed during the dry distillation of mixture of calcium formate and calcium acetate is |
|
Answer» PROPANAL `underset("Calcium acetate")((HCOO)_(2))Ca OVERSET(Delta)to underset("Methanal")(HCHO)+CaCO_(3)` `underset("Calcium acetate")((CH_(3)COO)_(2))Ca overset(Delta)tounderset("Propanone")(CH_(3)COCH_(2))+CaCO_(3)` `(HCOO)_(2)Ca+(CH_(3)COO)_(2)Ca overset(Delta)to underset("Ethanal")(2CH_(3)CHO)+CaCO_(3)` thus, only propanal is not formed. |
|
| 19. |
The compound which is not formed during the dry distillation of a mixture of calcium formate and calcium acetate is |
|
Answer» Propanal `underset("Calcium formate")((HCOO)_(2)Ca)overset(Delta)(to)underset("Methanal")(HCHO)+CaCO_(3)` `underset("Calcium acetate")((CH_(3)COO)_(2)Ca)overset(Delta)(to)underset("Propanone")(CH_(3)COCH_(3))+CaCO_(3)` `(HCOO)_(2)Ca+(CH_(3)COO)_(2)Ca overset(Delta)(to)underset("Ethanal")(2CH_(3)CHO+CaCO_(3)` Thus, only propanal is not formed. |
|
| 20. |
The compound which is mixed with alcohol to get a substitute for petrol is |
|
Answer» ethanol |
|
| 21. |
The compound which is least reactive among the following in a nucleophilic substitution reaction is |
|
Answer» `CH_2=CHCl` |
|
| 22. |
The compound which is insoluble in hot water and NH_3 is: |
|
Answer» `PbCl_2` |
|
| 23. |
The compound which is added to table salt for maintaining proper health is |
| Answer» ANSWER :C | |
| 24. |
The compound which is functional isomer of diethyl ether is, |
|
Answer» 1-methoxypropane |
|
| 25. |
The compound which has one isopropyl group is |
|
Answer» 2,2,3,3-tetramethyl PENTANE |
|
| 26. |
The compound which gives white ppt. with aqueous AgNO_3 and a green flame test is : |
|
Answer» `BaCl_2` `Ba^(2+)` GIVES GREEN colour in FLAME test . |
|
| 27. |
The compound that reacts fastest with Lucas reagent is |
|
Answer» butan -1-ol |
|
| 28. |
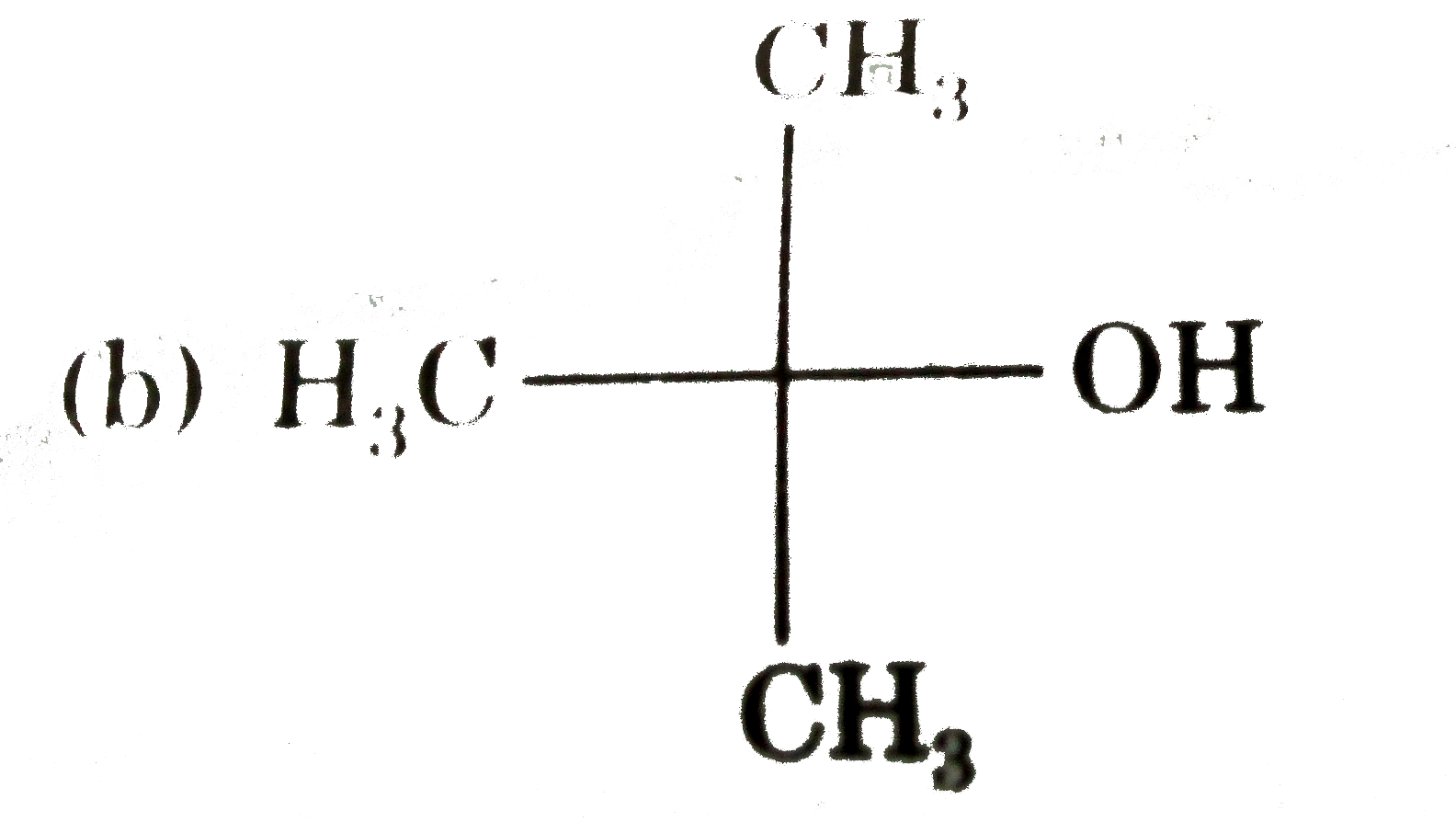
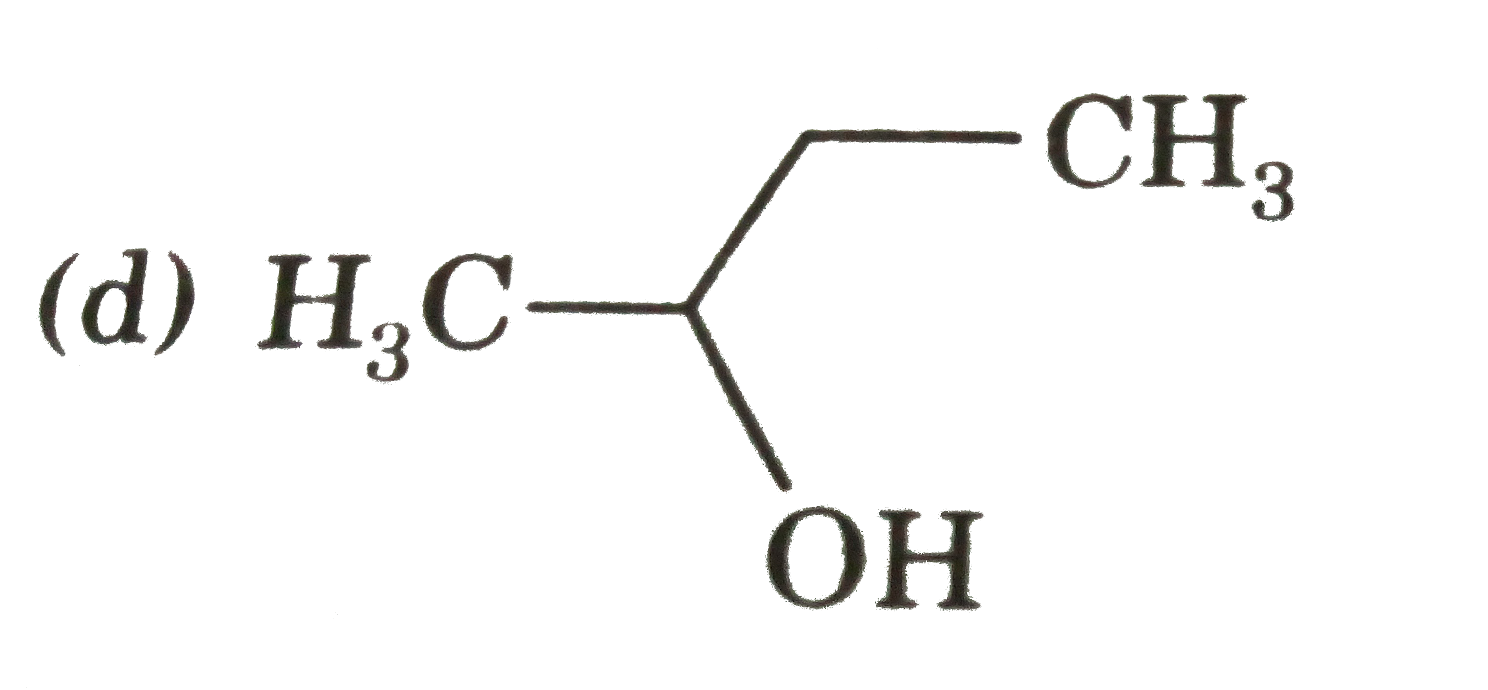
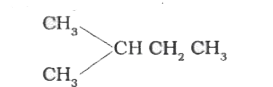
The compound which gives the most stable carbonium on de |
|
Answer»
|
|
| 29. |
The compound which gives the most stable carbonium ion on dehydration is |
|
Answer» `{:(CH_(3)-CH-CH_(2)OH),("|"),(""CH_(3)):}` |
|
| 30. |
The compound which gives the most stable carbonium ion and dehydration is : |
|
Answer»
|
|
| 31. |
The compound which gives positive iodoform test is : |
|
Answer» 1-pentanol |
|
| 32. |
The compound which gives propane on reduction with Zn and dilute hydrochloric acid is |
|
Answer» `CH_3CH_2CH_2OH` |
|
| 33. |
Which gives off oxygen on moderate heating: |
|
Answer» ZINC Oxide |
|
| 34. |
The compound which gives off oxygen on moderate heating is |
|
Answer» CUPRIC OXIDE |
|
| 35. |
The compound which gives off oxygen on moderate heating is : |
|
Answer» Cupric OXIDE |
|
| 36. |
The compound which gives negative iodoform test is : |
|
Answer» `CH_3CHO` |
|
| 37. |
The compound which gives an oily nitrosoamine on reaction with nitrous acid at low temperature is |
|
Answer» `CH_(3)NH_(2)` |
|
| 38. |
The compound which gives carbon with conc. H_(2)SO_(4) |
| Answer» Solution :Alcohol gives ether `2C_(2)H_(5)OH underset(-H_(2)O)overset("CONC. "H_(2)SO_(4))to H_(5)C_(2)-O-C_(2)H_(5)` | |
| 39. |
The compound which gives an oily nitrosomine on reaction with nitrous acid at low temperature is………. |
|
Answer» `C_2H_5NH_` |
|
| 40. |
The compound which gives carbon with con H_2SO_4 |
|
Answer» Sugar |
|
| 41. |
The compound which gives acetone on ozonolysis is ............ |
|
Answer» `CH_3 -CH = CH - CH_3` |
|
| 42. |
The compound, which give a positive ninhydrin test arid a negative Benedict's solution test, is |
|
Answer» a monosaccharide |
|
| 43. |
The compound which forms acetaldehyde when heated with dilute NaOH is |
|
Answer» 1, 1 Dichloroethane 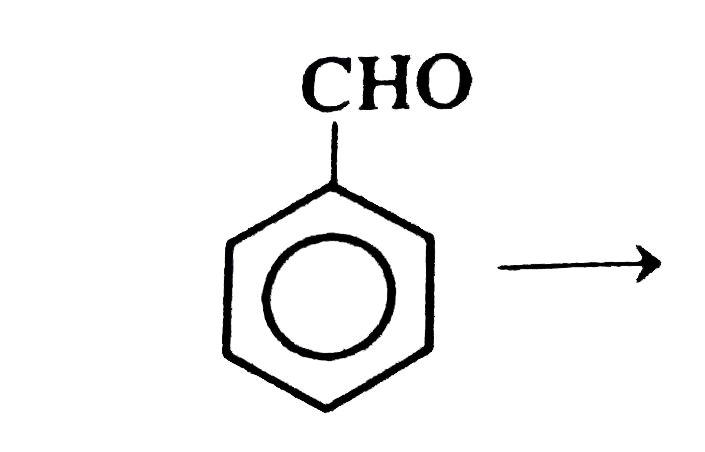 does not have an a hydrogen, so it will give Cannizzaro reaction does not have an a hydrogen, so it will give Cannizzaro reaction `{:("HCH"_(3)),("||"),(CH_(3)-C-C-CHOrarr),("||"),("HCH"_(3)):}` does not have an `alpha` hydrogen, it will give Cannizzaro reaction. 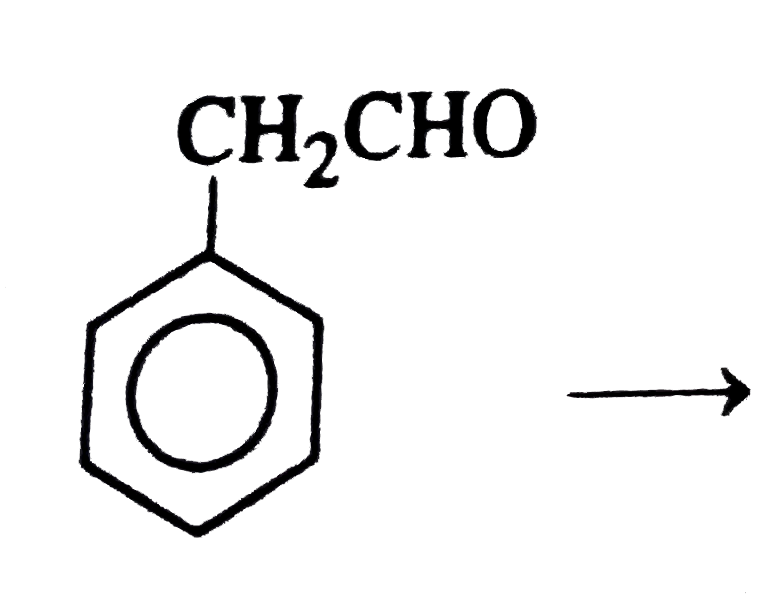 It has `alpha` hydrogen, hence will not give cannizzaro reaction. It has `alpha` hydrogen, hence will not give cannizzaro reaction.
|
|
| 44. |
The compound which exist as a dimer is |
|
Answer» LICL has HIGHER melting point than NaCl 
|
|
| 45. |
The compound, which evolves carbon dioxide on treatment with aqueous solution of sodium bicarbonate at 25^(@)C, is |
|
Answer» `C_(6)H_(5)OH` |
|
| 46. |
The compound which exhibits optical isomerism is: |
|
Answer» `CH_3CHOHCH_3` |
|
| 47. |
The compound which exhibits optical isomerism is : |
|
Answer» `CH_(3)CHOH-CH_(3)` |
|
| 48. |
The compound which will not react with sodium is : |
|
Answer» `CH_3CHOHCH_3` |
|
| 49. |
The compound which does not undergo hydrolysis by S_(N^(1)) mechanism is: |
|
Answer» `CH_(2)=CHCH_(2)Cl` |
|